Abstract
Type 2 diabetes mellitus (T2DM) is associated with increased morbidity and mortality in patients with acute coronary syndromes (ACS). Cardiometabolic risk factors, including hyperglycaemia, insulin resistance, atherogenic dyslipidaemia, increased visceral fat and inflammation, are associated with increased risk in this population and represent potential targets for treatment. In this review, management strategies for patients with T2DM post-ACS, both in the acute-care setting and in the long-term, are discussed. Although the benefits of long-term, aggressive, multifactorial risk factor modification are well established, a significant burden of recurrent events remains and the search for novel strategies continues. Several studies are assessing the potential cardiovascular (CV) benefits and safety of various classes of newer agents. Of these, AleCardio (aleglitazar), Examination of Cardiovascular Outcomes With Alogliptin versus Standard of Care in Patients With Type 2 Diabetes Mellitus and Acute Coronary Syndrome (EXAMINE; alogliptin) and Evaluation of LIXisenatide in Acute Coronary Syndrome (ELIXA; lixisenatide) specifically address patients with type 2 diabetes post-ACS. The mechanisms of action of these new therapies and aims of the CV outcome studies are briefly reviewed. The prevalence of type 2 diabetes continues to increase worldwide highlighting the need for new strategies that address the complex underlying processes that drive atherosclerosis and CV events in this high-risk patient population.
Keywords
Introduction
Despite major breakthroughs in management,1,2 cardiovascular disease (CVD) – and more specifically acute coronary syndromes (ACS) – remains a leading cause of mortality worldwide.3–5 Large population-based studies indicate that the typical CV risk factor profile of patients has changed over the past few decades, reflected by substantial reductions in the incidence of acute myocardial infarction (MI).2,6 The overall reduction in event rates is likely due to the increasing proportion of patients meeting blood pressure and low-density lipoprotein cholesterol (LDL-C) targets, as well as the declining prevalence of smoking. However, there have also been notable negative trends in the risk factor profile of MI patients, which today is characterized by higher body mass index, elevated triglyceride (TG) levels and increased prevalence of type 2 diabetes mellitus (T2DM) compared with previous decades.2,6
There is a strong association between T2DM and CVD mortality and morbidity. A meta-analysis, conducted by the Emerging Risk Factors Collaboration, that included almost 700,000 subjects with no history of MI, angina or stroke at baseline, revealed that diabetes confers an approximately twofold excess risk for coronary heart disease (CHD), major stroke and deaths attributed to other vascular causes. 7 Furthermore, patients with prior MI and T2DM are at significantly greater risk of subsequent MI compared to those without T2DM (45% vs 19%; p < 0.001), 8 and the combination of T2DM and a coronary event increases the risk of subsequent MI and mortality, regardless of the severity of the index event. 9 This poor prognosis also extends to patients with diabetes following a revascularization procedure. 10
The International Diabetes Federation estimates that, in 2012, 4.8 million deaths worldwide were caused by diabetes and its complications, the majority of which were in patients with T2DM and CV causes.11,12 Indeed, the burden of CVD is increased not only in patients with T2DM but also among people with a modestly elevated plasma glucose, including those with impaired fasting glucose (IFG) and impaired glucose tolerance (IGT).13,14 In absolute terms, mortality is likely to increase further in dysglycaemic populations as the number of people with IFG, IGT and T2DM continues to increase. 11
Several factors are associated with a heightened risk of subsequent events, morbidity and mortality in patients with T2DM post-ACS. 15 Improving the management of these patients is a challenge that requires urgent attention. 15 This review will focus on the short-term cardiometabolic treatment of patients with T2DM and ACS and will also briefly review strategies for their long-term risk reduction. We will also summarize recent and ongoing clinical trials examining the efficacy and safety of newer glucose-lowering and cardiometabolic agents on CV risk in patients with type 2 diabetes, in particular post-ACS.
Pathophysiology of atherosclerosis in patients with T2DM post-ACS
Although T2DM is defined based on hyperglycaemia, its underlying pathophysiology is that of a prothrombotic milieu in which a raft of risk factors, including insulin resistance, high TGs, low high-density lipoprotein cholesterol (HDL-C), increased visceral fat and a proinflammatory environment, exacerbates the deleterious effects of hyperglycaemia on the entire vascular system 16 (Figure 1).
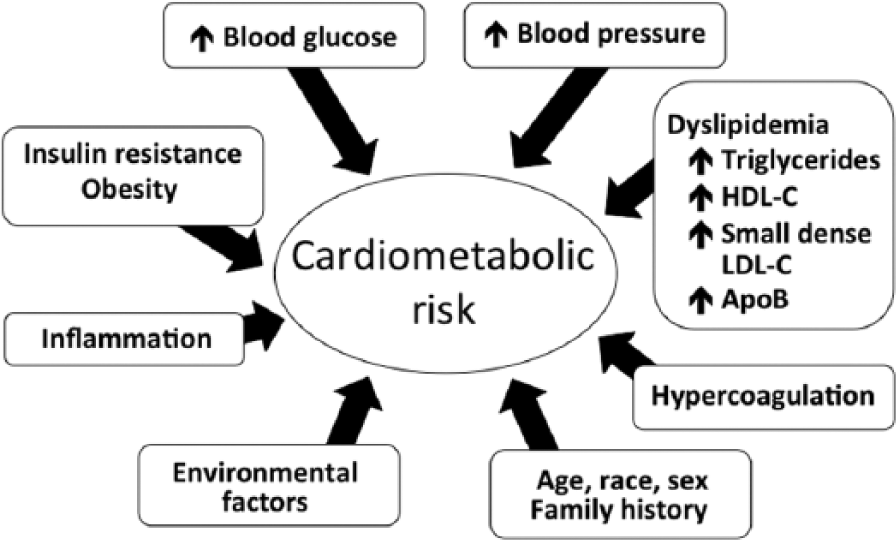
Traditional and emerging cardiovascular disease risk markers contributing to cardiometabolic risk.
The detrimental effects of hyperglycaemia
Elevations in glucose levels, resulting from impaired response to insulin because of insulin resistance and/or relative insulin deficiency, can lead to detrimental CV effects through several pathways. The combination of multiple risk factors is likely to create a particularly unstable environment for the patient with T2DM post-ACS. For example, chronic hyperglycaemia leads to enhanced formation of advanced glycation end-products (AGEs), which directly alter tissue function and mechanical properties through intracellular binding. In addition, binding of AGEs to cell-surface receptors (RAGEs) can induce signalling pathways, leading to inflammation and vasoconstriction. 17 In the vulnerable plaque, AGE/RAGE expression correlates with the expression of matrix metalloproteinase-9, an enzyme responsible for plaque destabilization and rupture.18,19 Hyperglycaemia-induced reactive oxygen species (ROS) also activate many pathways of tissue damage and increase expression of RAGE and its proinflammatory endogenous ligands. 20
Platelet hyperactivity is also a characteristic of hyperglycaemia and results in intensified adhesion, activation and aggregation of platelets, thus contributing to the prothrombotic environment in diabetes. 21 Increased platelet activation leads to the expression of surface adhesion molecules, such as P-selectin and consequently soluble P-selectin, 21 which mediates leukocyte and platelet rolling and recruitment of leucocytes. 22
Insulin resistance
Insulin resistance, which may have direct proatherogenic effects at the level of the arterial wall, promotes plaque progression and exacerbates atherothrombotic vascular disease. 23 Preclinical studies have demonstrated that downregulation of the insulin signalling pathway in macrophages causes accelerated macrophage apoptosis and impaired phagocytic clearance, which contribute to plaque necrosis. Indeed, plaque necrosis is a prominent feature in T2DM lesions, and advanced atherosclerotic lesions in T2DM are associated with larger necrotic cores than similar-sized lesions in non-diabetic subjects. Thus, insulin resistance could partially explain the accelerated thrombotic disease and susceptibility to recurrent MI in patients with T2DM.16,23
In addition, high visceral adipose tissue mass, resulting from insulin resistance and obesity, is a rich source of proinflammatory adipokines that may contribute to coagulation and fibrinolysis, both directly and indirectly. Direct effects include production of tissue factor, plasminogen activator inhibitor-1 and possibly thrombin-activated inhibitors of fibrinolysis. Indirect effects include the secretion of proinflammatory cytokines, such as tumour necrosis factor-α, interleukin-6 and macrophage chemoattractant protein-1. 24
Diabetic dyslipidaemia
The profile of dyslipidaemia is particularly atherogenic in patients with T2DM. While LDL-C levels are often in the ‘normal’ range, there is a shift of the LDL-C pool towards small, dense atherogenic LDL-C particles that are cholesteryl ester–depleted and prone to oxidization by ROS. 16 Oxidized LDL-C is implicated throughout the atherosclerotic process through internalization by macrophages and foam cell formation. Changes in macrophage physiology trigger a cascade of inflammatory events, including release of chemokines, cytokines and ROS, which contribute to plaque instability and increased risk of rupture. 25 In patients with T2DM, there is also an increase in glycated LDL-C, which is more atherogenic than the natural form. 26 Small, dense LDL-C particles are more preferentially glycated than buoyant LDL-C particles, and, even in the absence of oxidation, glycation of LDL-C results in its rapid uptake by macrophages. 26
In T2DM, dyslipidaemia is also characterized by high levels of TGs and low levels of HDL-C, which contribute to chronic inflammation and oxidative stress, driving the atherosclerotic process.16,27 Also in T2DM, there are increases in apolipoprotein B–carrying lipoproteins and decreases in apolipoprotein A–carrying lipoproteins. 16 It has been suggested that the concentration of apolipoprotein B is a better measure of atherogenicity than LDL-C level since it quantifies the concentration of lipoproteins rather than their cholesterol content. 28
Screening for dysglycaemia post-ACS
Because patients with T2DM are at such a heightened risk of a post-ACS event, it is vital to ensure that a patient’s glycaemic state is correctly identified early after an event so that it can be addressed appropriately. Many patients with ACS (20%–30%) 29 have an already established diagnosis of T2DM on hospital admission. Among those without such history, the prevalence of undiagnosed diabetes and prediabetes (IFG or IGT) is high, up to 60%–65%, according to several studies.29–31
This highlights the need to screen all ACS patients for glucose perturbations if their glycaemic status is unknown. European guidelines recommend the oral glucose tolerance test (OGTT) for the detection of glycaemic abnormalities; however, this is a time-consuming procedure. For this reason, the American Diabetes Association (ADA) recommends glycated haemoglobin (HbA1c) testing 32 in the stable outpatient setting. HbA1c is a more convenient tool suitable for screening in the general population, although there are some limitations, including the effects of perturbations in red blood cell turnover, which can falsely raise or lower readings. Although not specifically recommended in the ADA guidelines, HbA1c can be used for screening in patients with ACS in whom an elevated HbA1c (>6.5%) indicates diabetes. If, however, HbA1c is normal or only slightly elevated, then the test may be too insensitive for screening, and thus, an OGTT should be performed.
Indeed, the superiority of the OGTT over HbA1c to detect diabetes and glucose dysregulation in patients with CVD has been demonstrated in a number of studies,29,30,33,34 with reported failure rates as high as 96% by HbA1c in the detection of previously hidden T2DM compared with the OGTT. 34 Given the high post-ACS mortality rates in patients with T2DM, high-risk patients should be identified using the most relevant tools to ensure that correct decisions are made for their management. In summary, a fasting glucose and/or an HbA1c can be used as the first and easiest screening tool. Since a normal result does not eliminate glucose perturbations, including T2DM, such patients should be followed up as required with an OGTT to firmly establish the true glycaemic state.
Short-term management of ACS in patients with diabetes
Use of acute insulin infusion to improve metabolic control post-ACS
Hyperglycaemia on hospital admission is a risk factor for death and in-hospital complications in patients with ACS.35,36 In the acute post-ACS setting, tight control of blood glucose levels with acute insulin infusion is particularly important if hyperglycaemia is considerable. 35 The Diabetes Mellitus, Insulin Glucose Infusion in Acute Myocardial Infarction (DIGAMI) trial in patients with T2DM demonstrated that intensive insulin therapy, initiated within 24 h of an MI, reduced long-term (3.4 years) mortality by 11% compared with control. 37 Subsequently, the DIGAMI 2 trial found no difference in mortality rates with the use of insulin compared with oral glucose-lowering agents. However, updated blood glucose (as recorded during follow-up) was shown to be a significant and independent predictor of mortality. 38 A possible explanation of the different outcomes between the two DIGAMI trials is the lack of difference in glucose control between the treatment arms in the DIGAMI 2 trial, while HbA1c was >1% lower in the insulin arm in the first DIGAMI trial. A recently published 20-year follow-up of the DIGAMI 1 trial showed that intensive insulin-based glycaemic control for 3 months after an initial acute MI in patients with type 2 diabetes increased survival by 2.3 years compared with standard glycaemic control, with the mortality benefit most apparent within the first 8 years. 39
A number of studies have investigated the effect of glucose–insulin–potassium (GIK) infusion in the acute post-ACS setting. The concept of metabolic modulation by GIK was based on the ability of insulin to suppress free fatty acid levels and stimulate potassium uptake, thereby facilitating glucose transport into cells. 40 Recently, the Immediate Myocardial Metabolic Enhancement During Initial Assessment and Treatment in Emergency care (IMMEDIATE) trial, which included approximately 30% of patients with diabetes, investigated the effect of GIK on MI in the out-of-hospital setting, enabling treatment soon after ischaemic symptom onset. 41 Although IMMEDIATE did not meet its primary endpoint with no difference in the rate of progression to MI, there was a significant reduction in the secondary composite endpoint of cardiac arrest or in-hospital mortality for the GIK group compared with placebo (6.1% vs 14.4%; p = 0.01), and, in the small subgroup that underwent imaging at 30 days, infarct size was up to fivefold smaller in the GIK group compared with placebo. 41 These results warrant further investigation of the use of GIK in the out-of-hospital setting with a larger population and more robust endpoints, such as mortality and final infarct size. 42
Antiplatelet agents
Prompt and sustained reperfusion of the artery, which is essential in the acute post-ACS phase, may be aided pharmacologically by early, aggressive intervention with antiplatelet and antithrombotic agents. ACS guidelines recommend that acetylsalycilic acid (ASA) is given to all patients without a contraindication as early as possible following diagnosis of ACS and continued indefinitely in order to reduce the risk of both acute ischaemic complications and recurrent atherothrombotic events.43–46 Regarding individuals with T2DM, a meta-analysis from the Antiplatelet Trialists’ Collaboration clearly demonstrated the benefits of antiplatelet therapy (predominantly ASA) in patients with diabetes who are at high risk of a CV event, for example, those with unstable angina, acute MI, prior MI, stroke or transient ischaemic attack. 47 Indeed, guidelines recommend that patients with diabetes and acute MI should be considered for antiplatelet therapy (e.g. ASA) on the same grounds as their non-diabetic counterparts.43,48
Patients with T2DM may have an inadequate response to platelet inhibitors compared with normoglycaemic individuals, possibly because of their hyperactive platelet phenotype, which could account for higher rates of recurrent events. 21 Studies with clopidogrel in this population have revealed that this agent has limited benefits, even at higher doses, possibly due to increased resistance. 49 The introduction of more potent platelet inhibitors appears to be particularly beneficial for patients with T2DM; for example, while more intensive antiplatelet therapy with prasugrel reduced CV events in all post-ACS patients compared with clopidogrel, absolute reductions in these events were greater for patients with diabetes than for those without diabetes. 50 Perioperative pharmacologic management for post-ACS patients is, however, challenging because of bleeding risks associated with potent antiplatelet and antithrombotic therapies. 51 Specific recommendations on antiplatelet therapy in post-ACS patients with diabetes are beginning to appear in guidelines.43,44,52
Revascularization procedures
In terms of mechanical revascularization, the choice of intervention is either to perform coronary artery bypass grafting (CABG) or a percutaneous coronary intervention (PCI) using stents. 10 Management strategies must be at least as aggressive for patients with T2DM compared with normoglycaemic individuals because of their poor prognosis post-ACS. 10 Decisions on timing and type of coronary revascularization procedure should be based on multiple factors, for example, clinical presentation, coronary anatomy, ischaemic burden, left ventricular function and comorbidities, and the threshold for CABG should be lower in patients with diabetes than for individuals without glucose perturbations. 53 The recently reported Future Revascularization Evaluation in Patients with Diabetes Mellitus: Optimal Management of Multivessel Disease (FREEDOM) trial was conducted in T2DM patients with multivessel (at least three vessels) disease, one-third of whom had a recent ACS. The study found a significantly lower rate of the 5-year primary composite outcome in patients undergoing CABG compared with PCI using drug-eluting stents (DESs) (18.7% vs 26.6%; p = 0.005), with a twofold higher risk of MI in the PCI group (99 vs 48 patients; p < 0.001), although the reverse effect was seen for stroke (5.2% CABG vs 2.4 PCI; p = 0.03). 54 These benefits may be, in part, due to the fact that CABG provides flow not only at the primary lesion but also in distal disease, which is not the case with PCI. 55 However, the less invasive nature of PCI and the high reduction in restenosis rates in patients with diabetes with DESs often make the technique more attractive to cardiologists.
In terms of choice of DESs, a meta-analysis of the effect of sirolimus-eluting stents and paclitaxel-eluting stents in patients with T2DM, using data from randomized controlled trials and patient registries, found both DESs to be highly effective in reducing revascularization rates and major CV event rates to similar extents. 56 However, comparison of the second-generation zotarolimus-eluting stent with the sirolimus-eluting stents showed that use of the zotarolimus-eluting stent was associated with worse outcomes for patients with T2DM compared with the sirolimus-eluting stents, both in increased risk of revascularization and in major CV event rates. 57 Further research is needed to clarify the role of PCI in treatment of patients with T2DM post-ACS.
Long-term cardiometabolic strategies for post-ACS patients with T2DM
The Steno-2 study evaluated the effects of intensive multifactorial intervention in patients with T2DM and persistent microalbuminuria, of whom 19.5% had electrocardiographic evidence of ischaemia at baseline. 58 Steno-2 showed that targeting hyperglycaemia, hypertension and dyslipidaemia, combined with lifestyle modification, substantially reduced the risk of a primary composite endpoint, which included MI, CABG and PCI intervention. 59 The outcome of the Steno-2 study has strongly influenced the overall standard of care for patients with T2DM. Comprehensive CV risk reduction is an essential focus of therapy in these patients. Current management guidelines strongly underline the importance of achieving recommended treatment targets in patients with diabetes (Table 1).32,48,52,60–63
Treatment targets for people with and without diabetes and with varying risk for cardiovascular disease manifestations according to European, Canadian and American management guidelines issued 2012–2013.
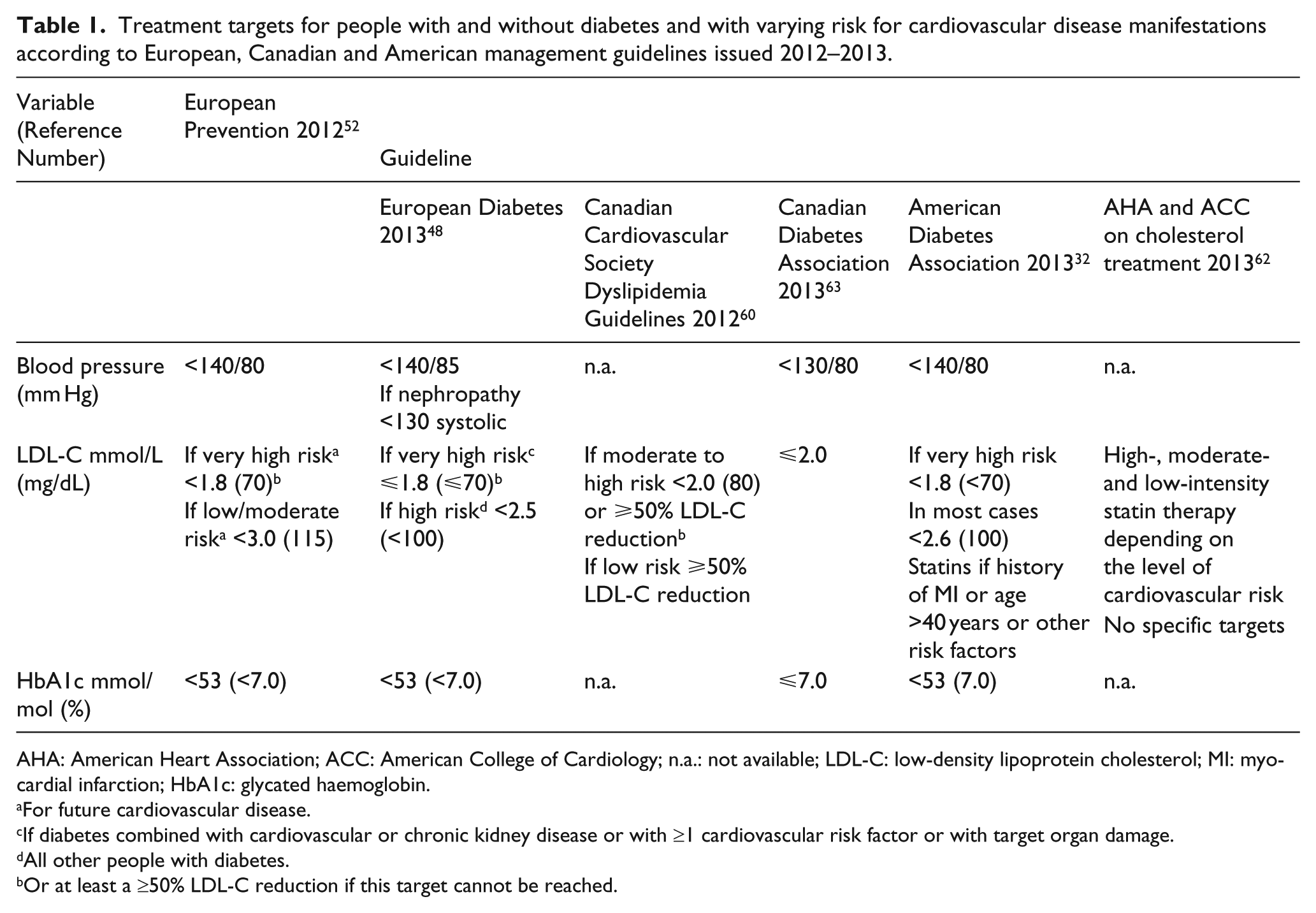
AHA: American Heart Association; ACC: American College of Cardiology; n.a.: not available; LDL-C: low-density lipoprotein cholesterol; MI: myocardial infarction; HbA1c: glycated haemoglobin.
For future cardiovascular disease.
If diabetes combined with cardiovascular or chronic kidney disease or with ≥1 cardiovascular risk factor or with target organ damage.
All other people with diabetes.
Or at least a ≥50% LDL-C reduction if this target cannot be reached.
Glycaemic control
The ADA and the European Association for the Study of Diabetes (EASD) recommend lowering HbA1c to <7.0% in most patients to reduce the incidence of microvascular disease. 64 However, glycaemic targets should be individualized, and less stringent goals may be appropriate for patients with a history of severe hypoglycaemia, advanced complications, extensive comorbid conditions including established vascular complications and those with limited life expectancy. 64
A complete review of the relationship between glucose control and CVD is beyond the scope of this article. While none of the landmark trials individually have shown a benefit of intensive glycaemic control on macrovascular complications, a meta-analysis of the four studies (Action to Control Cardiovascular Risk in Diabetes (ACCORD), Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE), Veterans Administration Diabetes Trial (VADT) and United Kingdom Prospective Diabetes Study (UKPDS)) did show a modest, 9% reduction (hazard ratio (HR) 0.91; 95% confidence interval (CI) 0.84–0.99)) in the risk of major CV events with intensive glycaemic control, although rates of hypoglycaemic events were increased (HR 2.48; 95% CI 1.91–3.21). 65 Furthermore, in the extended follow-up of the UKPDS of intensive glycaemic control in patients with newly diagnosed T2DM, the risk of MI was significantly reduced by 15% in patients in the originally intensive arm compared with the previously conventional arm, although HbA1c rapidly converged between the two groups, hence demonstrating a ‘legacy’ effect for early intensive intervention. 66
Several studies have examined the potential CV benefit of individual glucose-lowering agents in patients with diabetes, many of whom with established CVD, although not specifically in the immediate post-ACS setting. Data from clinical trials that specifically focus on patients with type 2 diabetes in the post-ACS setting are limited; therefore, some studies have been included in this section that were not conducted in a post-ACS population since they still provide valuable insights into the long-term management of this high-risk patient population.
The Outcome Reduction with an Initial Glargine Intervention (ORIGIN) trial investigated whether a strategy based on the early institution of insulin replacement therapy targeting fasting normoglycaemia with insulin glargine would reduce the risk of CV events more than standard approaches to dysglycaemia in high-risk subjects with IFG, IGT and T2DM. 67 Upon entry into the trial, 59% of patients were classified as having had a prior CV event (MI, stroke or revascularization), with 35% having had a prior MI. The study reported no significant difference in CV outcomes between insulin-based therapy and standard care over a median follow-up of 6.2 years; however, the insulin glargine group was associated with a modest increase in hypoglycaemia and weight gain. 67
The PROspective PioglitAzone Clinical Trial in MacroVascular Events (PROactive) 68 investigated whether the pleiotropic (antihyperglycaemic, antiinflammatory and antiatherogenic) effects of the peroxisome proliferator–activated receptor-γ (PPAR-γ) agent pioglitazone translated into CV benefit in patients with T2DM. Of note, PROactive was conducted in a population with evidence of extensive CVD at entry, for example, MI, stroke, PCI or CABG at least 6 months prior to recruitment. Although pioglitazone significantly decreased the secondary composite endpoint of all-cause mortality, non-fatal MI and stroke, the agent had no significant effect on the very broad primary composite endpoint that also included leg amputation, and coronary or leg revascularization. 68 In addition, pioglitazone significantly lowered the rate of progression of coronary atherosclerosis 69 and slowed the progression of carotid intima-media thickness compared with glimepiride,70,71 but no other outcome data for pioglitazone have been reported.
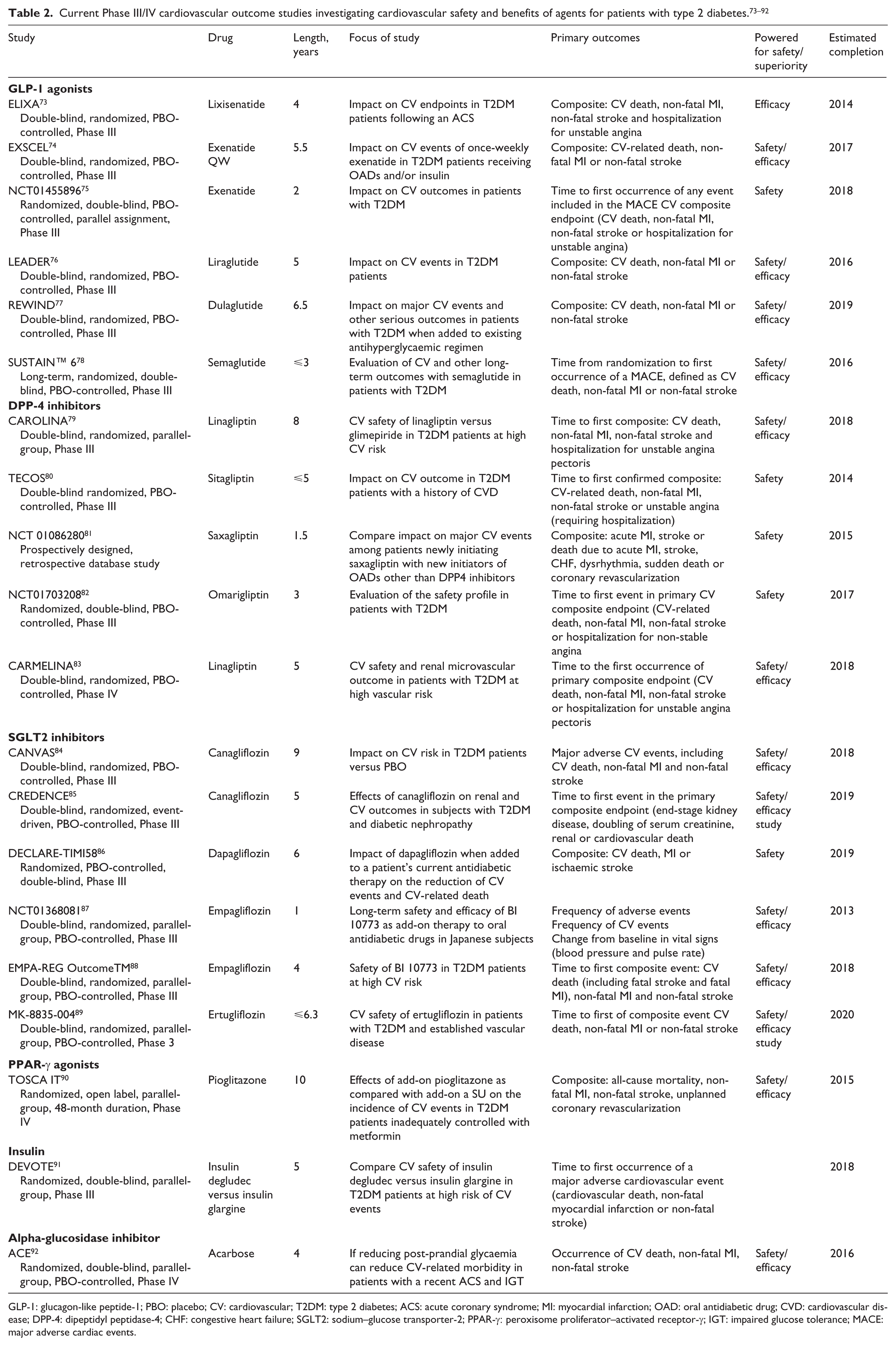
In 2008, the US Food and Drug Administration (FDA) 72 published a Guidance for Industry, recommending investigation of the CV effects of therapies in development for treatment of hyperglycaemia. To this end, a number of ongoing studies are assessing the CV safety of various agents with different mechanisms of action, some of which exhibit properties beyond glucose-lowering effects, for example, on dyslipidaemia, insulin resistance and vascular inflammation. In addition to evaluating the CV safety of these agents, these outcome studies should identify whether any specific glucose-lowering agent can reduce CV events (Table 2).73–92
GLP-1: glucagon-like peptide-1; PBO: placebo; CV: cardiovascular; T2DM: type 2 diabetes; ACS: acute coronary syndrome; MI: myocardial infarction; OAD: oral antidiabetic drug; CVD: cardiovascular disease; DPP-4: dipeptidyl peptidase-4; CHF: congestive heart failure; SGLT2: sodium–glucose transporter-2; PPAR-γ: peroxisome proliferator–activated receptor-γ; IGT: impaired glucose tolerance; MACE: major adverse cardiac events.
Glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase inhibitors
The native human incretin hormone glucagon-like peptide-1 (GLP-1), which is secreted in response to food intake, stimulates insulin secretion in a glucose-dependent fashion and decreases glucagon secretion, thus reducing hepatic glucose release. GLP-1 also decreases gastric emptying, producing a feeling of satiety and thereby reducing food consumption. The incretin effect is diminished in patients with T2DM; however, the short half-life of native GLP-1 (around 2 min) makes administration of the native hormone impractical. 93 The GLP-1 receptor agonists exenatide, liraglutide and albiglutide are approved for market use and are injectable agents with longer half-lives than native GLP-1, producing greater than or equal to fivefold higher stimulation of the GLP-1 receptor than the native hormone. They are associated with robust reductions in HbA1c and weight loss and may have beneficial pleiotropic effects on β-cell function and the vasculature. 93 Their impact on CV events in patients with T2DM is currently under investigation (Table 2).73–78
Dipeptidyl peptidase-4 (DPP-4) inhibitors are oral agents that decrease degradation of native GLP-1 by the enzyme DPP-4, resulting in an approximately twofold increase in the native hormone. Glycaemic effects with DPP-4 inhibitors are somewhat more modest than with GLP-1 receptor agonists, and, since these agents do not reduce gastric emptying or increase satiety to the same extent, they are generally regarded as weight neutral.
The results from Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus (SAVOR) – TIMI 53 with the DPP-4 inhibitor saxagliptin were recently reported. 94 In this study, 16,492 patients with type 2 diabetes, who had a history of, or were at risk for CV events were randomly assigned to receive saxagliptin or placebo. The primary composite endpoint of CV death, MI or ischaemic stroke occurred in 613 (7.3%) patients in the saxagliptin group and 609 (7.2%) patients in the placebo group (HR 1.00; 95% CI 0.91–1.17), which met criteria for non-inferiority (p < 0.001) but not superiority (p = 0.99). Although saxagliptin did not increase or decrease the rate of ischaemic events, the rate of hospitalization for heart failure was increased (3.5% vs 2.8%; HR 1.27; 95% CI 1.07–1.51; p = 0.007).
The Examination of Cardiovascular Outcomes With Alogliptin versus Standard of Care in Patients With Type 2 Diabetes Mellitus and Acute Coronary Syndrome (EXAMINE) trial investigated the effect of the DPP-4 inhibitor alogliptin in patients with T2DM and a recent ACS. 95 EXAMINE met its primary objective to demonstrate CV safety based on the FDA requirements. The rate of the composite primary endpoint of CV death, non-fatal MI and non-fatal stroke neither increased nor decreased as compared to placebo (11.3 vs 11.8%; HR 0.96; upper boundary of the one-sided repeated CI 1.16; p < 0.001 for non-inferiority; p = 0.32 for superiority) in this very high-risk population with well-managed conventional risk factors. The HR for heart failure was 1.19 (0.89–1.58) (p = non-significant).
Similar to EXAMINE, the Evaluation of LIXisenatide in Acute Coronary Syndrome (ELIXA) study was also designed to specifically examine the safety and efficacy of a glucose-lowering agent, this time a GLP-1 agonist, lixisenatide, on CV outcomes in patients with T2DM post-ACS (Table 2).73,95 The 4-year ELIXA trial is expected to be completed in 2015 and will evaluate the GLP-1 receptor agonist for its effect on the composite endpoint of CV death, non-fatal MI, non-fatal stroke and hospitalization for unstable angina. 73 As GLP-1 analogues and DPP-4 inhibitors have somewhat different effects on the incretin system and other CV risk factors, it will be interesting to compare the outcome of ELIXA with that of EXAMINE.
Dual PPAR-α/γ agonists
Activation of PPAR pathways has a number of favourable physiologic effects. 96 In order to simultaneously improve glycaemic control and CV outcomes, research attention was given to the clinical development of PPAR-α/γ agonists, which have potentially beneficial effects on lipids (PPAR-α effect), insulin sensitivity and glucose uptake (PPAR-γ effect). The most recent agent in clinical development was aleglitazar.96,97 In a dose-finding Phase II study, aleglitazar demonstrated significant reductions in HbA1c, which were accompanied by reductions in TGs and LDL-C and increases in HDL-C. 98 The drug was also tested in patients with stage 3 chronic kidney disease. No new toxicities or major safety concerns were identified in this 52-week Phase IIb safety study (AleNephro). 99 However, following a regular safety review of the AleCardio Phase III trial investigating aleglitazar in patients with a recent ACS event and T2DM, 100 the aleglitazar development programme was halted due to safety concerns and lack of efficacy. While there was no significant difference in the primary endpoint (time to CV death, non-fatal MI or non-fatal stroke) occurring in 344 patients (9.5%) in the aleglitazar group and 360 patients (10.0%) in the placebo group, the rates of serious adverse events, including heart failure (3.4% for aleglitazar vs 2.8% for placebo), gastrointestinal haemorrhages (2.4% for aleglitazar vs 1.7% for placebo) and renal dysfunction (7.4% for aleglitazar vs 2.7% for placebo), were increased. 100 The unexpected outcome of the AleCardio trial, based on early clinical experiences with aleglitazar,98,99 will likely induce a debate on the continued use of PPAR agonists as a drug class and demonstrate that, although early investigations may indicate a number of beneficial effects for patients with diabetes, large clinical trials are needed to conclusively establish the role of promising drugs.
Sodium–glucose cotransporter-2 inhibitors
Sodium–glucose cotransporter-2 (SGLT2) inhibitors are another recently introduced class of oral agents that reduce plasma glucose concentrations by inhibition of the renal sodium–glucose cotransporter, resulting in glucosuria. 101 They also result in some weight loss and blood pressure reduction. 102 The effects of several SGLT2 inhibitors on CV outcomes are currently under investigation (Table 2).84–89
Dyslipidaemia management
The specific benefits of intensive lipid lowering with statins in the early period after an ACS were well established by trials such as the Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering (MIRACL) Study, Pravastatin or Atorvastatin Evaluation and Infection Therapy (PROVE-IT) Study and A to Z,103–105 in which up to one-quarter of patients had diabetes. The Cholesterol Treatment Trialists’ Collaborators (CTTC) meta-analysis showed that statins reduce major vascular events by approximately one-fifth per mmol/L reduction in LDL-C. 106 This benefit is seen in patients with diabetes, with similar reductions in those with vascular disease (relative risk 0.80; 95% CI 0.74–0.88) and those without (relative risk 0.73; 95% CI 0.66–0.82). Indeed, there is a quantitative relationship between reduced LDL-C levels and reduced CV risk and the Treat to New Targets (TNT) trial, conducted in patients with clinically evident CHD, 15% of which had diabetes, showed that this relationship persisted at lower levels of LDL-C. In this study, intensive lowering of LDL-C levels to a mean of 77 mg/dL (2.0 mmol/L) with high-dose atorvastatin significantly reduced the primary endpoint – occurrence of a major CV event – by 22%, with similar reductions observed in those patients with diabetes compared to those without.107,108
Even with the considerable benefits that have been observed in the era of statin therapy, a substantial residual CV risk remains,109,110 perhaps due to other components that are not adequately addressed by statins. 111 However, to date, addition of other agents to effective statin therapy has not been shown to further reduce CV risk. The ongoing IMProved Reduction of Outcomes: Vytorin Efficacy International (IMPROVE-IT) Trial, which has enrolled over 18,000 patients including >20% with diabetes, was designed to evaluate the clinical benefit on the primary endpoint, defined as the occurrence of the composite endpoint of CV death, major coronary events and stroke, of the combination of simvastatin with the cholesterol absorption inhibitor ezetimibe, compared with simvastatin alone, in patients with stabilized high-risk ACS. 112 In the ACCORD LIPID trial, which was carried out in a large cohort of high-risk patients with T2DM, 37% of whom had experienced a previous CV event, the addition of fenofibrate to statin therapy provided a greater reduction in TG levels compared with statin plus placebo. However, this did not translate into improvement in CV event rates, and there was no significant difference in the fenofibrate group on primary endpoint of fatal or non-fatal CV events. 113 Similarly, to date, CV benefits have not yet been seen using agents that raise HDL-C levels. For example, in the Atherothrombosis Intervention in Metabolic Syndrome with Low HDL/High Triglycerides: Impact on Global Health Outcomes (AIM HIGH) study conducted in patients with atherosclerotic CVD, 34% of whom had diabetes, niacin in combination with a statin resulted in significant improvements in lipid levels compared with statin alone; however, this did not translate into clinical benefit, and the trial was terminated for futility after 3 years. 114 A lack of benefit of niacin/laropiprant added to statin therapy has also been reported recently for the large Heart Protection Study 2 Treatment of HDL to Reduce the Incidence of Vascular Events (HPS2-THRIVE) study.115,116 Cholesteryl ester transfer protein (CETP) inhibitors provide more robust elevations in HDL-C, 117 and studies are ongoing evaluating their effects on CV outcomes. Although the dal-OUTCOMES Phase III trial, 118 which was conducted in patients with recent ACS, did not demonstrate clinically meaningful efficacy for dalcetrapib, leading to the cessation of the dal-HEART program, 119 it cannot be used to draw any definitive conclusions about the effect of raising HDL-C levels on CV outcomes.
Ongoing studies with other investigational CETP inhibitors, anacetrapib and evacetrapib,120–123 which raise HDL-C to a greater degree and also lower LDL-C, will provide further information on the impact of this strategy on major coronary events. The increased mortality and CV morbidity related to treatment with torcetrapib, causing the early termination of the Investigation of Lipid Level Management to Understand its Impact in Atherosclerotic Events (ILLUMINATE) trial, 124 has raised concerns. In the dal-OUTCOMES study, 125 however, the use of dalcetrapib was associated with a neutral effect on CV outcomes.
Conclusion
The burden of illness in patients with T2DM in the post-ACS setting is high, and the toll of this morbidity that is aggravated by underlying cardiometabolic risk factors is likely to rise with the inexorable increase in cases of T2DM and prediabetes. While the need for a multifactorial approach to treating patients with T2DM and CVD is now well recognized, there is a heightened need for optimal intervention in these patients in the post-ACS setting. One way forward is to focus more on therapeutic strategies to, in a comprehensive way, address the multiple cardiometabolic risk factors characteristic for patient with T2DM and CVD. Meanwhile, further research should be directed to better understand the key pathophysiological background to the increased CV morbidity and mortality, hopefully identifiable through new bio-markers, allowing the development of pharmacological tools targeting the complex mechanisms that drive atherosclerosis and CV events in this high-risk patient population.
Footnotes
Acknowledgements
P.K., L.A.L., L.M. and L.R. provided the concept for the article, contributed substantially to interpretation of data, drafting and critically revising for important intellectual content and provided final approval of the version to be submitted for publication. Editorial support, from concept through to an earlier submission, was provided by Ann Parkin, PhD, MediTech Media, UK, funded by F. Hoffmann-La Roche AG.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
P.K. has received a speakers’ honorarium from Merck Canada Inc. L.A.L. has received research funding from, has provided CME on behalf of and/or has acted as a consultant to Amgen, AstraZeneca, BI, BMS, Eli Lilly, GSK, Janssen, Merck, Novo Nordisk, Roche, Sanofi, Servier and Takeda. L.M. has received research grants from MSD and Sanofi-Aventis and speakers’ honoraria from MSD, Sanofi-Aventis, Novartis, Bayer-Schering, AstraZeneca and Lilly. L.R. has received research grants from the Swedish Heart Lung Foundation, Karolinska Institute, Stockholm County Council, AFA Insurance and Roche and honoraria as a consultant or lecturer from AstraZeneca, Roche, BMS and Sanofi.