Abstract
Objective
To report the long-term impact on cardiovascular (CV) outcomes and mortality of a 2-year hospital-based multi-interventional care programme as compared with general practitioner (GP)-provided standard care.
Methods
Patients with type 2 diabetes with ≥ 1 additional CV risk factor were randomized to 2 years of specialist-based, multi-intervention comprising lifestyle modification and specific pharmacological treatment, or GP-based standard care. After the 2-year intervention period, all participants returned to pre-study care, but were followed up for CV outcomes and mortality. The primary outcome was time to any first severe CV event or death.
Results
A total of 120 patients (31 women) were enrolled in the study. During the mean ± SD observational period of 8.7 ± 2.0 years, 27 patients (16 and 11 in the multi-intervention and standard care groups, respectively) experienced at least one primary outcome event, with a hazard ratio (HR) if allocated to the multi-intervention group of 1.73 (95% confidence interval (CI) 0.80, 3.75). The HR for total mortality was 1.82 (95% CI 0.66, 5.01).
Conclusions
Hospital-based multi-intervention in patients with type 2 diabetes mellitus improved long-term glycaemic control, but failed to reduce CV outcomes and deaths.
Clinical trials.gov id: NCT00133718.
Keywords
Introduction
In patients with type 2 diabetes, hyperglycaemia is associated with increased risk of cardiovascular (CV) morbidity and mortality, 1 and studies have shown that approximately 50% of all deaths among people with diabetes can be ascribed to CV disease.2,3 Stringent blood glucose control has been shown to decrease the risk for microvascular disease, but the effects on macrovascular outcomes remain controversial. 4 The ACCORD study even suggested increased mortality risk with intensive glucose lowering, 5 a finding that to date is not yet fully explained. 6
Since type 2 diabetes is often part of a cardiometabolic syndrome with hypertension, dyslipidaemia, and central obesity, a strategy of targeting multiple CV risk factors is considered necessary. This was also illustrated in the STENO-2 study, which observed a long-term CV benefit of an intensified, target-driven, multi-factorial approach in 160 type 2 diabetes patients with albuminuria and high CV risk;7,8 an effect largely mediated (>70%) by the effects of lipid modulation. 9
The randomized-controlled Asker and Bærum Cardiovascular Diabetes (ABCD) study showed that 2 years of structured, hospital-based multi-intervention significantly improved CV risk factors and reduced the estimated 10-year absolute risk for coronary heart disease, 10 driven by between-group differences in glycosylated haemoglobin (HbA1c), systolic blood pressure, low-density lipoprotein cholesterol (LDL-C) and triglycerides. At the conclusion of the 2-year study, all patients returned to pre-study care with no further scheduled clinical intervention by the study team. 10 The present pre-specified analysis assessed whether the shorter-term reduction in estimated CV risk translated into longer-term reductions in CV outcomes and death as evaluated at a mean ± SD of 8.7 ± 2.0 years following randomization.
Patients and methods
Study population and study design
The study design, intervention and intermediate results of the ABCD study (clinical trials.gov id: NCT00133718) have been reported previously.
10
In brief, 120 patients with type 2 diabetes and ≥1 additional CV risk factor were enrolled in an open, randomized controlled study and allocated to either 2 years of intensive, hospital-based, structured multi-intervention (n = 60) or standard care (n = 60) (Figure 1). All participants underwent a comprehensive diagnostic work-up at baseline including medical history, physical examination, 24-h ambulatory blood pressure monitoring (ABPM), laboratory assessment and urinary assessment of albumin excretion. A subpopulation of the cohort also underwent coronary angiography. All baseline procedures, except coronary angiography and 24-h ABPM, were repeated 2 years after randomization. Structured intensive multi-intervention comprised of 6 months of lifestyle intervention (i.e. advice on diet, exercise and smoking cessation and reimbursement of cost associated with exercise training), where medication was kept unchanged, followed by targeted, pharmacological treatment to reach pre-specified treatment goals (HbA1c ≤ 48 mmol/mol [6.5%]; total/LDL-C < 5.0/3.0 mmol/l; systolic/diastolic blood pressure [BP] < 130/80 mmHg) (for more details see Table 1). The participants were seen by a physician (diabetologist) at 3-monthly intervals at the out-patient clinic of Bærum Hospital, Vestre Viken Hospital Trust, Drammen, Norway. The standard care group remained under the care of their general practitioners (GPs) who were recommended to treat according to current guidelines with a recommended follow-up at 3-monthly intervals (Table 1).
10
Flowchart and outcomes during the Asker and Bærum Cardiovascular Diabetes study follow-up.
10
Lifestyle intervention algorithm, pharmacological treatment algorithm and treatment goals for both study groups in the Asker and Bærum Cardiovascular Diabetes study.
10
Or substitute for metformin if not tolerated. Or achieved treatment target or occurrence of side-effects. GP, general practitioner; ADA, American Diabetes Association; CV, cardiovascular; BP, blood pressure; ARB, angiotensin II receptor blocker; ACEI, angiotensin-converting enzyme inhibitor; NPH, neutral protamine Hagedorn; OD, once a day; BID, twice a day; MDI, multiple daily injections; glycosylated haemoglobin, HbA1c; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol.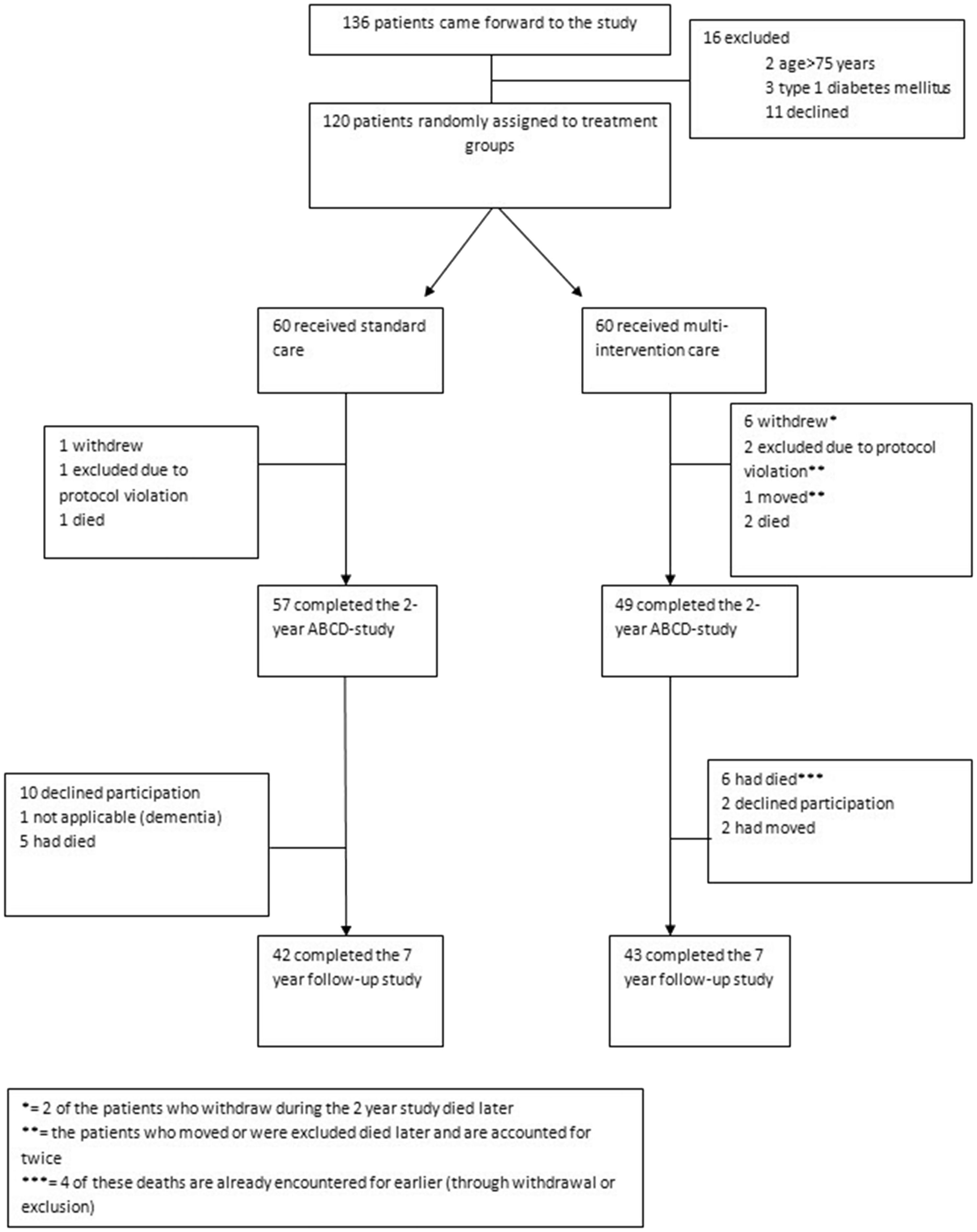

Patients who completed the 2-year study were returned to the standard of care they had prior to study enrolment. Patients were followed for CV events, hospitalizations and death and all patients alive were invited to participate in this current follow-up with clinical and laboratory assessments.
Informed consent to participate was obtained from all participants in this study, which was conducted in accordance with the Helsinki declaration and approved by the Regional Committees for Medical and Health Research Ethics.
Cardiovascular outcomes
The primary outcome was time to any first of the events of the composite outcome: non-fatal myocardial infarction (MI), non-fatal stroke, hospitalization for unstable angina pectoris (UAP), coronary revascularization, percutaneous transluminal angioplasty, amputation, hospitalization for heart failure, and death from all causes. Since coronary angiography was a study procedure, revascularization procedures occurring immediately during, and triggered by, the study procedure, were excluded. Secondary outcomes were total mortality as well as the overall cumulative event rates of the composite primary outcome and its components.
Information on events was collected from hospital records. In two cases, data on the cause of death were obtained from the Norwegian Cause of Death Registry. All outcome events were adjudicated by two of the authors (L.L.G. and K.I.B.) blinded to the treatment allocation.
Analyses of blood and urine
Laboratory parameters were assessed in fasting venous blood samples. Urinary albumin excretion was determined in timed overnight samples. Albuminuria was defined as urinary albumin excretion > 20 µg/min in two out of three samples. 11
Exercise testing
Exercise capacity was assessed at baseline, study-end, and at the 7-year follow-up by a modified conventional maximum stress test on a cycle ergometer as described previously. 12 Maximum oxygen consumption (ml/kg per min) was used to describe maximum exercise capacity.
Coronary angiography
Irrespective of the results from noninvasive tests, coronary angiography was performed in 91 patients at baseline according to standard procedures. 13 Significant coronary artery disease was defined as the presence of ≥ 50% luminal diameter narrowing of one or more of the epicardial arteries or its major branches.
Statistical analyses
Based on the STENO-2 study (approximately 715 patient-years of follow-up), 8 this present study targeted at least 1000 patient-years of follow-up to ensure that the effect of the multi-intervention could be assessed with a reasonable level of power. Analysis of outcomes was performed according to the intention-to-treat principle and each patient who did not have an event was censored on the last day of observation they were known to be free of the outcome.
Triglycerides and microalbuminuria were log-transformed due to their markedly skewed distributions. The between-group difference in change in outcome variables from baseline to study-end, and from study-end to the 7-year follow-up, were explored using linear regression analyses with measurements at either study-end or at 7 years as dependent variables and baseline measurements or measurements at study-end, respectively, and treatment group, as independent variables (analysis of covariance). Dichotomous variables were explored by the χ2-test and the between-group differences in change in categorical variables were estimated with the exact Wilcoxon Mann–Whitney U-test. Sensitivity analyses were performed with different imputation values (i.e. mean values, high values, low values). All regression models fulfilled the assumptions of normally distributed residuals and a Cook’s distance < 1.
The association between group allocation and outcome was explored with Cox regression analysis. Sensitivity analyses adjusting for baseline differences between the groups (body mass index [BMI] and microalbuminuria) as well as sulphonylurea (SU) use at baseline, were undertaken. The assumption of proportional hazards was tested using Schönfelds residuals.
All statistical analyses were performed using the SPSS® statistical package, version 23.0 (SPSS Inc., Chicago, IL, USA) for Windows®. A P-value < 0.05 was considered statistically significant.
Results
A total of 120 patients with type 2 diabetes were enrolled in the 2-year ABCD study. Of these, 106 patients (multi-intervention group: n = 49; standard care group: n = 57) completed the 2-year study (Figure 1), after which all participants returned to the care they had prior to study enrolment. Patients were followed for CV events, hospitalizations and death during a mean ± SD of 8.7 ± 2.0 years from randomization, and all patients alive were invited to participate in this current follow-up study with clinical and laboratory assessments that occurred at a mean ± SD of 7.2 ± 0.4 years after randomization.
Characteristics of the Asker and Bærum Cardiovascular Diabetes study population at baseline.

Continuous data presented as mean ± SD or median (interquartile range); categorical data presented as n of patients (%).
Myocardial infarction or coronary revascularization (i.e. percutaneous coronary intervention or coronary artery bypass grafting) performed prior to study inclusion.
Any known CAD, peripheral artery disease or cerebrovascular disease prior to study inclusion.
At baseline coronary angiography.
CAD, coronary artery disease; CVD, cardiovascular disease.
Clinical and laboratory findings, and the change in these, in the two treatment groups at baseline and at the end of the 2-year intervention in the Asker and Bærum Cardiovascular Diabetes study.

Data presented as mean ± SD.
Analysis of covariance.
CI, confidence interval; BMI, body mass index; BP, blood pressure; HbA1c, glycosylated haemoglobin; LDL-C, low-density lipoprotein cholesterol; eGFR, estimated glomerular filtration rate; MDRD, modification of diet in renal disease; NS, no significant between-group difference (P ≥ 0.05).
Overview of the number of events occurring in the two treatment groups during and after the 2-year intervention in the Asker and Bærum Cardiovascular Diabetes study.
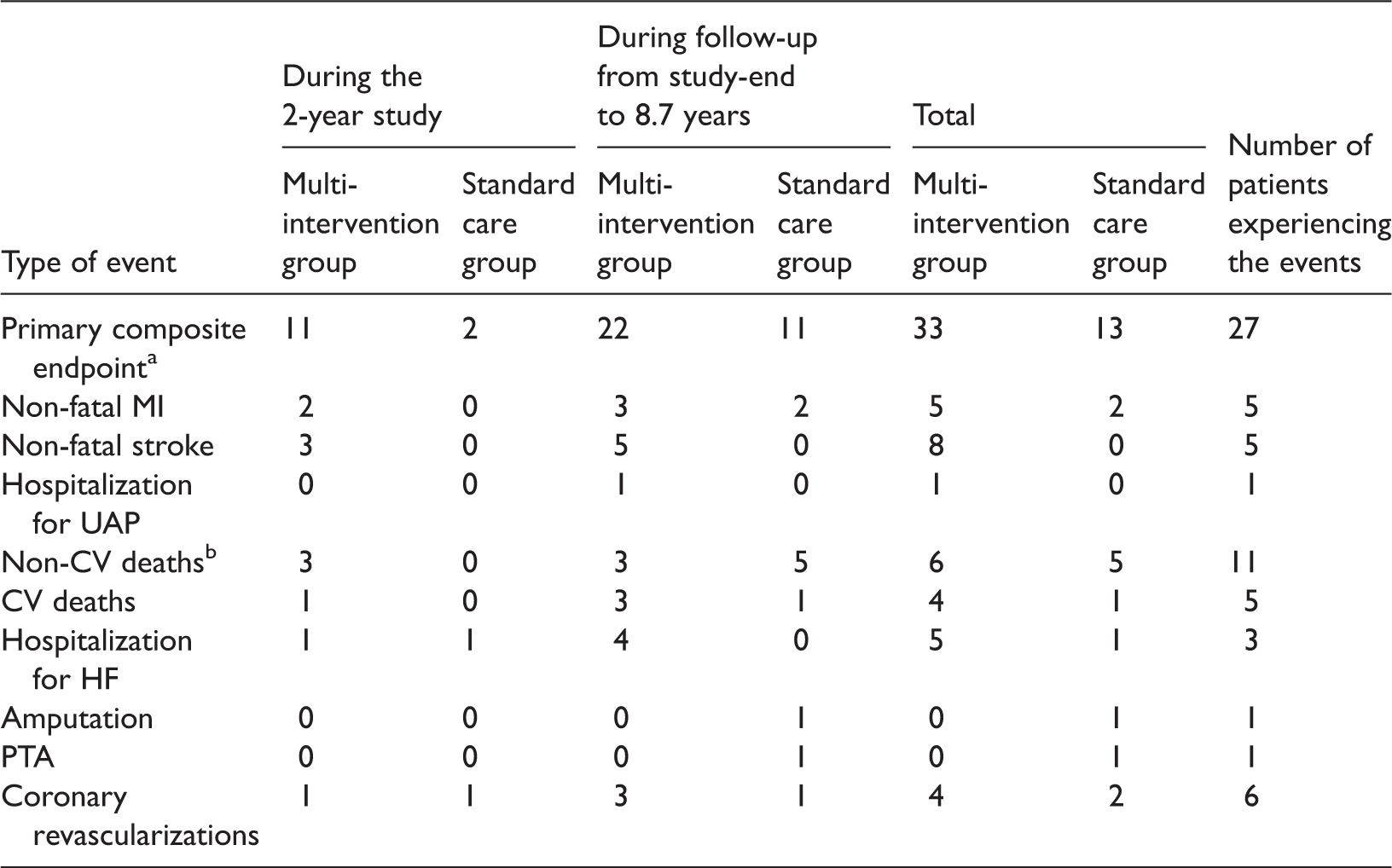
Non-fatal MI, non-fatal stroke, hospitalization for UAP, CV death, non-CV death, hospitalization for HF, amputation, PTA, coronary revascularization.
Two non-CV deaths, one in each group, occurred after the participants had completed the 7-year follow-up, but within the observational period of 8.7 years. These deaths are therefore not captured in the flow-chart in Figure 1.
MI, myocardial infarction; UAP, unstable angina pectoris; CV, cardiovascular; HF, heart failure; PTA, percutaneous transluminal angioplasty.

Hazard ratio (HR) and 95% confidence interval (CI) for the primary outcome and total mortality if allocated to multi-intervention as compared with standard care in patients in the Asker and Bærum Cardiovascular Diabetes study.
Microalbuminuria and medication use, and change in these, from study-end (2 years) to 7 years in the two treatment groups in the Asker and Bærum Cardiovascular Diabetes study.

Data presented as n of patients (%).
Wilcoxon Mann–Whitney U-test.
OAD, oral antidiabetic drug; SU, sulphonylurea; TZD, thiazolidinedione; ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; NS, no significant between-group difference (P ≥ 0.05).
Clinical and laboratory findings, and the change in these, in the two treatment groups from study-end (2 years) to 7 years in the Asker and Bærum Cardiovascular Diabetes study.

Data presented as mean ± SD.
Analysis of covariance.
CI, confidence interval; BMI, body mass index; BP, blood pressure; HbA1c, glycosylated haemoglobin; LDL-C, low-density lipoprotein cholesterol; eGFR, estimated glomerular filtration rate; MDRD, modification of diet in renal disease; NS, no significant between-group difference (P ≥ 0.05).
Microalbuminuria and medication use, and change in these, from baseline to study-end (2 years) in the two treatment groups in the Asker and Bærum Cardiovascular Diabetes study.

Data presented as n of patients (%).
Wilcoxon Mann–Whitney U-test.
OAD, oral antidiabetic drug; SU, sulphonylurea; TZD, thiazolidinedione; ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; NS, no significant between-group difference (P ≥ 0.05).
Adjustment for imbalances at baseline between the two treatment groups did not significantly alter the HR for the primary outcome (adjusted HR 1.65; 95% CI 0.69, 3.99; P = 0.26), neither did adjustment for SU use at baseline (HR 1.68; 95% CI 0.78, 3.65; P = 0.19). Also, applying different methods of imputation did not significantly alter the results (data not shown).
Discussion
In this present study, 2 years of specialist-based, targeted, multi-intervention with lifestyle intervention and intensification of pharmacological treatment did not translate into a reduced long-term risk for CV events and mortality as compared with standard care. While lipid and BP levels were similar in the two groups at 5 years after termination of the intervention period, the mean HbA1c level was still slightly lower in the multi-intervention group. Interestingly, a numerical imbalance in the primary outcome disfavouring intensified multi-intervention was observed. Although not significant, this finding may lead to speculation of an adverse effect of intensive glucose lowering, in line with what was reported from the ACCORD study. 5 Potential mechanisms for such an adverse outcome that have been proposed include the high use of glitazones in the intensively-treated group, and the low target for and rapid decline in HbA1c in this group. 6 In the present study, the lowest mean HbA1c level in the multi-intervention group of 50 mmol/mol (6.7%) was achieved slightly slower than in the ACCORD study, i.e. after approximately 12 to 18 months. 10 These present results showing no long-term CV benefit associated with improved glycaemic control are also in line with the follow-up study of the glucose arm in the ADVANCE study, 14 but in contrast to the UKPDS and VADT follow-up trials.15,16 This lack of benefit also occurred in spite of a clear beneficial impact on CV risk markers at the end of intervention, as previously reported: 10 the multi-intervention group had improved fasting blood glucose, HbA1c, total cholesterol, and LDL-C more than the standard group (Table 3), with a significantly greater proportion achieving treatment targets for HbA1c, fasting blood glucose and LDL-C. 10 As expected, the use of oral blood glucose lowering and antihypertensive medication, statins and acetylsalicylic acid had increased more in the multi-intervention group. One could speculate whether in particular the higher use of glitazones (7% to 35% in the multi-intervention group, 2% to 9% in the standard care group from baseline to the end of the 2-year intervention) and sulphonylureas (47% to 53% in the multi-intervention group and 37% to 27% in the standard care group from baseline to the end of the 2-year intervention), although small numbers, may potentially have contributed to the possible harm seen, as both these drug classes are reported to have adverse or uncertain CV effects (Table 7).17,18
These current findings are at variance with the STENO-2 study, where a significant reduction in the risk of CV events and mortality was associated with the use of a multifactorial intervention.7,8 Of note is that the STENO-2 study included high CV risk type 2 diabetes patients with microalbuminuria and hence higher baseline CV risk than in the present study. There could be several reasons for these diverging results, the major being the longer intervention period and follow-up used in the STENO-2 study and the different approach to lipid modulation.7,8 Furthermore, the minimum mean HbA1c level achieved in the current study was lower than in the STENO-2 study (50 mmol/mol [6.7%] versus 63 mmol/mol [7.9%]).7,8 It was speculated after the ACCORD study that too aggressive glucose lowering in patients at CV risk could be harmful.5,19 The use of glitazones was higher in the current study since these drugs were not a part of the treatment algorithm in the STENO-2 study and hence were not used.7,8 The use of other drugs with established CV effects also differed between the two studies: in STENO-2, the use of ACEIs in the intensive group was 15% at baseline, and 69% at the end of the intervention, and statins were only used by 2% at baseline and 33% at the end of the intervention. 20 The respective numbers in the multi-intervention group in the present study were 58% for ACEI/ARB use at baseline and 82% at study-end, and 48% for baseline statin use increasing to 88% at study-end. Thus, the present study population was receiving a different level of medication for CV protection, potentially making it more challenging to improve CV outcomes further. This assumption is supported by a mediation analysis indicating that > 70% of the CV risk reduction seen in the STENO-2 study was ascribed to lipid lowering. 9
Further studies supporting the STENO-2 study, with CV benefits being achievable also in populations with advanced type 2 diabetes, are the follow-up study of the VADT 16 as well as the EMPA-REG OUTCOME. 21 The latter study demonstrated that empagliflozin versus placebo, on top of standard care, in type 2 diabetes patients with established CV disease (i.e. at an advanced disease stage), significantly reduced CV death by 38% and hospitalization for heart failure by 35%. 21 The between-group difference in HbA1c was, as expected, modest (3–7 mmol/mol [0.3–0.6%]); and given the multi-modal effects of empagliflozin (reductions in BP, arterial stiffness, weight and visceral adiposity),22–24 it is likely that modulations of non-glycaemic pathways are more influential.
The present study had several limitations of which the most important were the relatively small number of participants and the relatively short intervention and follow-up periods. Another limitation relates to the slight imbalances at baseline with a higher prevalence of microalbuminuria and higher BMI in the multi-intervention group, which could influence the effects of the intervention. Furthermore, there was a lack data on the frequency of hypoglycaemic episodes in this study. Study strengths were the comprehensive characterization of the participants, a real-life setting with an intervention that was limited in time, however not too short, and the blinded adjudication of all clinical endpoints.
In conclusion, 2 years of structured, hospital-based multi-intervention in a population with type 2 diabetes and additional CV risk did not improve long-term CV outcomes or mortality despite a short-term improvement in estimated CV risk and sustained benefit on glycaemia. A numerical increased risk of CV events and death in the multi-intervention group may be related to the choice of drugs used.
Footnotes
Authors’ contributions
O.E.J., L.L.G., K.I.B and A.P.O. designed the study. O.E.J., G.R.U., E.O. and A.P.O. collected the data. A.P.O. performed the statistical analyses under guidance of our statistical expert M.W.F. All authors contributed to the writing of the manuscript.
Availability of data and material
The datasets generated during and/or analysed during the current study are not publicly available due to privacy regulations but are available from the corresponding author on reasonable request.
Declaration of conflicting interests
A.P.O. and O.E.J. are employed by Boehringer Ingelheim.
Funding
The study was funded by the South-Eastern Norway Regional Health Authority, and the funder had no involvement in the study design, data collection or publication decisions.
