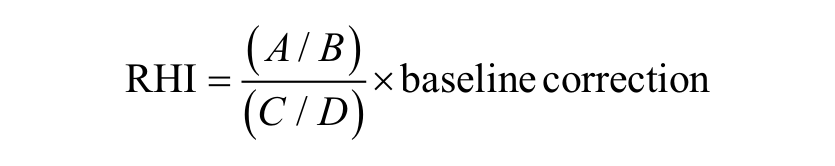Abstract
This single visit study examined whether endothelial function, in addition to cardiovascular (CV) risk factors and plasma microparticle content, was normalised in 15 patients with type 2 diabetes + acute coronary syndrome (ACS) (6 weeks–6 months post cardiac event) undergoing standard clinical care compared to 16 sex- and age-matched healthy controls.
Results:
While total and low-density lipoprotein (LDL) cholesterol levels were well controlled in the patients with type 2 diabetes + ACS, residual CV risk profiles such as increased body mass index (BMI), systolic blood pressure, glucose levels and triglycerides and lower high-density lipoprotein (HDL) levels were still apparent. Endothelium-dependent responses to acetylcholine (ACh) were significantly lower in type 2 diabetes + ACS patients compared to controls. Correspondingly, the reactive hyperaemic index (RHI) was lower in the patient cohort. Endothelial microparticle (EMP) levels (CD31+, CD41−) were 40% lower in the patient cohort. Simultaneous analysis of platelet microparticle (PMP) levels (CD41+) showed no difference between cohorts.
Conclusions:
Patients with type 2 diabetes suffering from recent ACS exhibit residual CV risk factors despite being on standard clinical care. In addition, these patients continue to present with endothelial dysfunction despite having lower levels of EMPs.
Keywords
Background
Acute coronary syndrome (ACS) presents as myocardial infarction (MI) or unstable angina usually associated with the rupture of an atherosclerotic plaque and partial or complete thrombosis of the related artery. Type 2 diabetes, now one of the most common ailments globally, with 285 million people worldwide diagnosed in 2010, is on the rise and expected to increase by 2030 to 439 million. 1 Patients with type 2 diabetes have up to a fourfold increased risk of a cardiovascular (CV) event, 2 –5 and the co-morbidity of diabetes in patients who have suffered an ACS increases by twofold the risk of short-term (30 days) and long-term (1 year) mortality independent of all other risk factors. 6 The underlying mechanisms are not fully elucidated, appear to be multifactorial and are thought to include increased cardiac risk factors, atherosclerotic burden, hyperglycaemia, inflammation and platelet aggregation. 7
In patients with ACS, endothelium-dependent vasoreactivity has been shown to be predictive of recurrent instability and CV event rates. 8 In addition, recovery of endothelium-dependent reactivity 8 weeks post ACS was reported to be predictive of event-free survival in this cohort. Interestingly, with the exception of type 2 diabetes, patients who had a recurrent major CV event after surviving an episode of ACS were not different from patients who were event free with regard to classic risk factors for coronary artery disease (CAD). Diabetes, therefore, was the single classic risk factor predictive of a recurrence.
Objectives and hypothesis
In this study, we explore whether standard clinical care of patients who had suffered ACS and have type 2 diabetes demonstrate residual endothelial dysfunction. Current treatment for patients with diabetes after ACS focuses on blood glucose, blood pressure and lipid control together with anti-platelet therapy. Some of these therapies have been shown to reduce endothelial dysfunction, 9,10 while other treatments, such as lipid lowering, which do not concurrently improve endothelial function, do not reduce the risk of CV events. 11,12 We hypothesise that residual endothelial dysfunction in ACS patients with diabetes, despite standard clinical care, may be one reason why these patients demonstrate a higher chance of CV event recurrence.
In addition, we explored the role of endothelial microparticles (EMPs) and platelet microparticles (PMPs), which are protein containing microvesicles of 0.1–1 µm shed from the endothelial or platelet cell membrane during cellular activation, damage or apoptosis. 13 Increasingly, evidence shows that both are proatherogenic, and in the clinical setting of type 2 diabetes and atherosclerosis, EMP levels are known to be augmented. 14 –16 Indeed, it has been suggested that EMP levels may be more a predictor of coronary atherosclerosis than lipid levels, the period of diabetes or the incidence of hypertension. 17 In addition, microparticles from patients with type 2 diabetes and metabolic syndrome have been shown to be capable of inducing endo-thelial dysfunction and decreasing nitric oxide (NO) bioavailability. 18,19
Research design and methods
Participants
In total, 14 men and 1 woman with diabetes mellitus (DM) (type 2 diabetes) and 16 age-matched normal men and women were studied in 2010–2011. The diabetes group, recruited from the Heart Centre, The Alfred Hospital, Melbourne, Australia, had all previously exhibited ACS (confirmed by angiogram and/or electrocardiogram (ECG) indicating ischaemia and a rise in serum troponin) occurring between 6 weeks and 6 months before the study visit day. Exclusion criteria included thiazolidinediones, more than two anti-diabetes drug treatments and other serious illness or disease. Inclusion criteria for normal participants were as follows: non-smoking status (determined by self-report), systolic pressure < 140 mmHg, body mass index (BMI) < 30 kg/m2, no serious illness or disease and not currently medicated with drugs known to affect the CV system. The study was approved by the Alfred Hospital Human Research Ethics Committee, and all participants provided informed written consent. Participants were instructed to fast for a minimum of 6 h prior to arrival. Upon arrival to the research clinic they were provided a light meal of approximately 250 kcal and rested for approximately 30 min before commencement of the experimental protocol and maintained at supine position in a quiet environment, at a constant temperature of 22°C.
Reactive hyperaemia peripheral arterial tonometry
Reactive hyperaemia peripheral arterial tonometry (RH-PAT) index was measured using Endo-PAT2000 (Itamar Medical, Caesarea, Israel), a device that records digital pulse volumes of peripheral arterial beds. A blood pressure cuff was positioned proximally (above elbow) on the dominant arm while the contralateral arm served as a control. PAT probes were placed on the index finger of each hand. After a 5-min equilibration period, the cuff was inflated for 5 min to 60 mmHg above systolic pressure (but no less than 200 mmHg) and then deflated to induce reactive hyperaemia. The reactive hyperaemic index (RHI) was calculated automatically from the inbuilt software of the Endo-PAT2000 device using the following equation
where (A) is the mean digital pulse volume during the reactive hyperaemia, (B) is the baseline mean digital pulse volume and (C) and (D) are the respective values obtained in the control arm.
Forearm venous occlusion plethysmography
Forearm blood flow (FBF) was measured using a double-strand mercury-filled silastic strain-gauge plethysmograph (D.E. Hokanson, Inc., Bellevue, WA, USA) fitted to the patient as described previously. 20 The brachial artery was cannulated for intra-arterial pressure recordings as well as for local, sequential infusions of Miochol-E [acetylcholine (ACh); Novartis Pharmaceuticals, North Ryde, NSW, Australia; 9.25, 18.5 and 37 µg/mL/min] and sodium nitroprusside (SNP; Hospira Inc, Lake Forest, IL, USA; 2, 4 and 8 µg/mL/min). Each concentration was infused at 2 mL/min for 2 min. Rest periods of 5 min between concentrations and 15 min between drugs were observed. Physiological saline (0.9%) was infused during baseline measurements. Heart rate and arterial blood pressure were constantly recorded via an ECG lead (Spacelabs Inc, Issaquah, Wash, USA) and an AE 840 physiological pressure transducer (connected to the arterial line), respectively, throughout drug infusion to check for non-local effects for SNP and ACh even though these are highly unlikely due to venous occlusion of the upper arm. A drug infusion pump (Terumo Medical, Somerset, NJ, USA) was used to deliver accurate amounts of the study agonist to each participant.
The mean FBF values of three measurements obtained before and after drug infusion were used for analysis. The response to each concentration of drug was expressed as a percentage of the basal FBF.
Lipid and glucose profile
After the vascular function studies, 10 mL of arterial blood was collected from all participants into an ethylenediaminetetraacetic acid (EDTA) vacutainer tube for measurement of serum triglycerides (Tg), total cholesterol, high-density lipoproteins (HDLs) and low-density lipoproteins (LDLs) and glucose content using the automated Cholestech LDX System (Inverness Medical, San Diego, CA, USA).
EMP and PMP numbers by fluorescence-activated cell sorter
A volume of 3 mL of blood was collected into a sodium citrate vacutainer for EMP and PMP identification. The blood was centrifuged at 25°C for 10 min at 400g, and the supernatant was re-centrifuged for 10 min at 600g to collect platelet-poor plasma, which was then stored at −80°C until further analysis. EMPs and PMPs are defined as CD31+/CD41− and CD41+ particles, respectively, sized between 0.1 and 1 µm in platelet-depleted plasma. They were determined by the analysis for the expression of surface antigens by flow cytometry. In brief, 500 µL of thawed plasma was centrifuged at 16,000g for 5 min at 4°C to deplete platelets or debris. The top 450 µL of plasma was then transferred into a fresh tube, which was centrifuged at 16,000g for 30 min at 4°C. The top 250 µL of plasma was carefully removed, and the remaining 200 µL of plasma was used for fluorescence-activated cell sorter (FACS) analysis. Following 15 min incubation with 50 µL of Fc-γ receptor blocking agent (Miltenyi Biotec, Auburn, CA USA) at room temperature, half of the treated plasma was incubated with antibodies against human CD31 (Alexa647-conjugated; BD Biosciences, Franklin Lakes, NJ, USA) and CD41 (phycoerythrin (PE)–conjugated; BD Biosciences, USA). The other half incubated with Alexa647- and PE-conjugated isotype control antibodies from the same manufacturer served as controls. At the end of 20 min incubation, 300 µL of double-filtered 1% formaldehyde/0.2% fetal bovine serum (FBS)/phosphate buffered saline (PBS) (filtered through a 0.2-µm and then a 0.1-µm membrane filter) was added for fixation, and 50 µL of diluted calibration beads (BD Biosciences) was added for EMP calculation and size reference. Each sample and its corresponding control were counted on BD FACSCalibur™ (BD Biosciences) for 5 min. EMP gate was predefined using commercial beads sized at 0.1 and 1 µm (Sigma–Aldrich, St. Louis, MO, USA). Only events included within this gate were further analysed for fluorescence signal. For microparticle enumeration, a formula was used based on the concentration of the added calibration beads, which discriminated themselves from the EMP population on the forward scatter (FSC)/side scatter (SSC) cytogram. 21 All counting data were then processed with a blinded randomised approach using BD CellQuest Pro (BD Biosciences). Results are presented as number of CD31+/CD41− EMP or CD41+ PMP per microlitres of plasma.
Statistical methodologies
Data are presented as mean ± standard error of the mean (SEM) unless otherwise specified. Data sets were tested for statistical significance by Student’s t-test (unpaired) or Mann–Whitney U test dependent on data distribution as determined by D’Agostino–Pearson omnibus normality test except for the comparison of drug responses between groups, which was tested by two-way repeated-measures (RM) analysis of variance (ANOVA). All tests were performed using SigmaStat (Jandel Scientific Software, San Rafael, CA, USA); p < 0.05 was used as a measure of statistical significance.
Results
Study participants
A total of 31 study participants were recruited (16 controls; 15 type 2 diabetes + ACS; Table 1), and the study groups were well matched for age and gender. The supplementary table presents the details of the study medications and supplements, with the majority of the study cohort medicated with statins, β blockers and anti-diabetes agents. Systolic blood pressure, but not diastolic blood pressure, of the type 2 diabetes + ACS patients was significantly higher than the control group even though all were taking blood pressure medication with 47% taking two or more. Table 1 also charts the CV risk profiles for these subjects. While the total and plasma LDL cholesterol levels of the type 2 diabetes + ACS cohort were well controlled and indeed statistically significantly lower than the healthy cohort, they demonstrated significantly higher BMI, triglyceride and glucose levels and significantly lower HDL cholesterol levels.
Participant baseline characteristics and plasma lipid and glucose profiles.
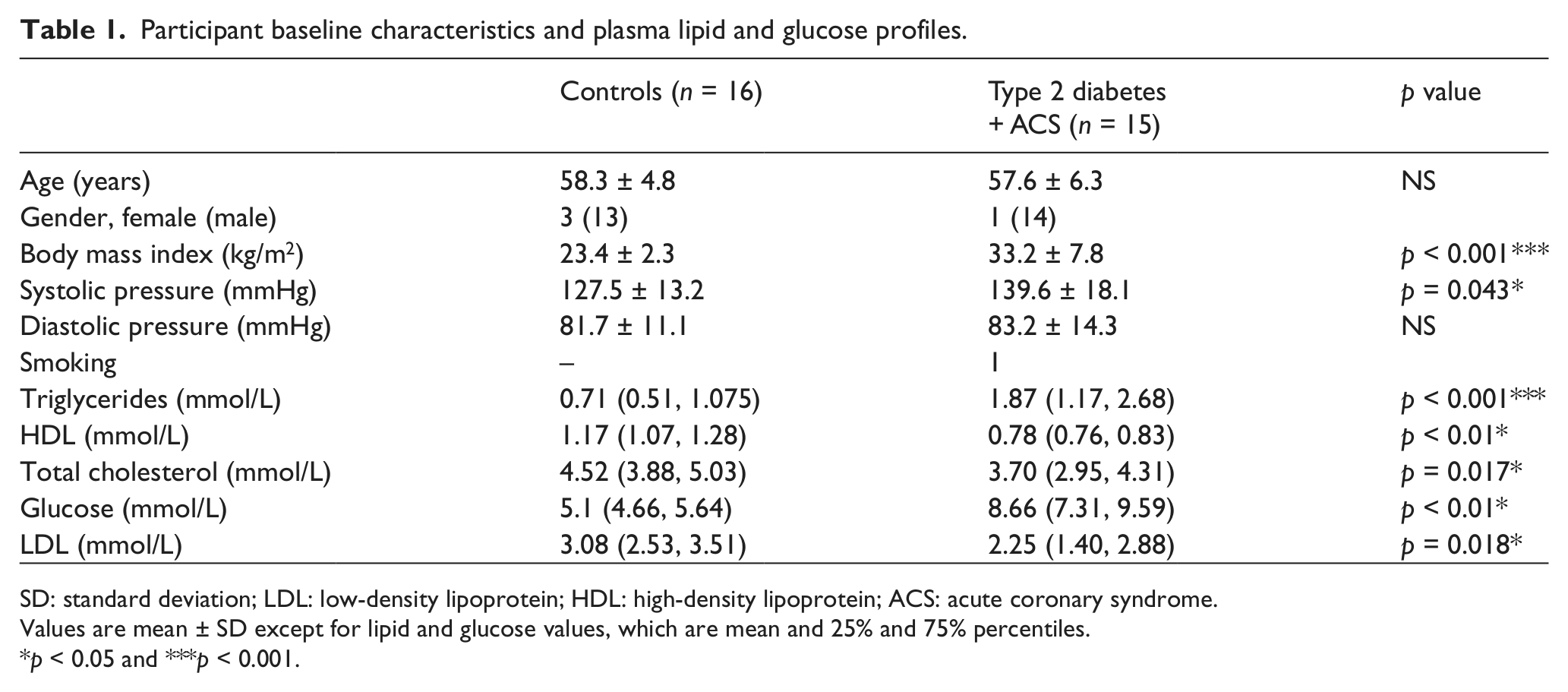
SD: standard deviation; LDL: low-density lipoprotein; HDL: high-density lipoprotein; ACS: acute coronary syndrome.
Values are mean ± SD except for lipid and glucose values, which are mean and 25% and 75% percentiles.
p < 0.05 and ***p < 0.001.
Endothelial function
Endothelial function was assessed using both the Food and Drug Administration (FDA)-approved non-invasive EndoPAT technology and the more traditional measure of FBF response to infusions of the endothelium-dependent agonist ACh and the non-endothelium-dependent agonist SNP using forearm venous occlusion plethysmography.
Figure 1 shows that the RHI obtained with EndoPAT was significantly lower in the type 2 diabetes + ACS cohort compared with their healthy counterpart. An RHI of less than 1.67 has been referenced by the manufacturer as the point below which a diagnosis of ‘endothelial dysfunction’ can be made; 8/15 type 2 diabetes + ACS patients scored an RHI below 1.67, while only 2/16 of the healthy cohort registered an RHI below 1.67.
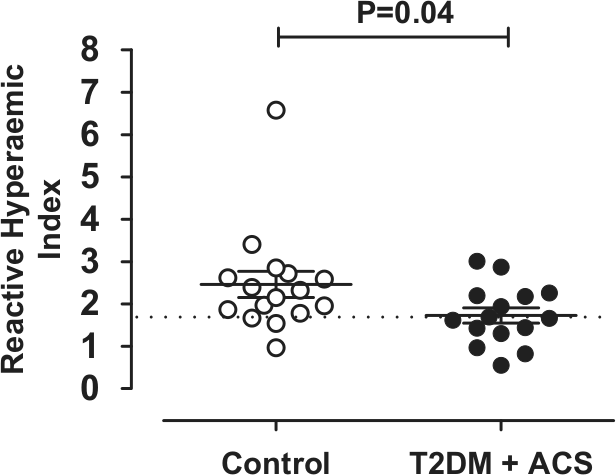
Reactive hyperaemic index determined with EndoPAT of the control and the type 2 diabetes + ACS cohort. Values are mean ± SEM and were compared using the Mann–Whitney U test.
Baseline FBF was not different between the two groups [healthy vs type 2 diabetes + ACS (mL/100 mL/min): 2.5 ± 0.4 vs 3.3 ± 0.6; p > 0.05; Student’s unpaired t-test]. Responses to ACh were significantly blunted in the type 2 diabetes + ACS cohort (n = 14–15; p < 0.05; two-way RM ANOVA; Figure 2(a)). While there appeared to be a similar trend in responses to SNP (Figure 2(b)), this did not reach statistical significance (n = 14–15; p = 0.142; two-way RM ANOVA).
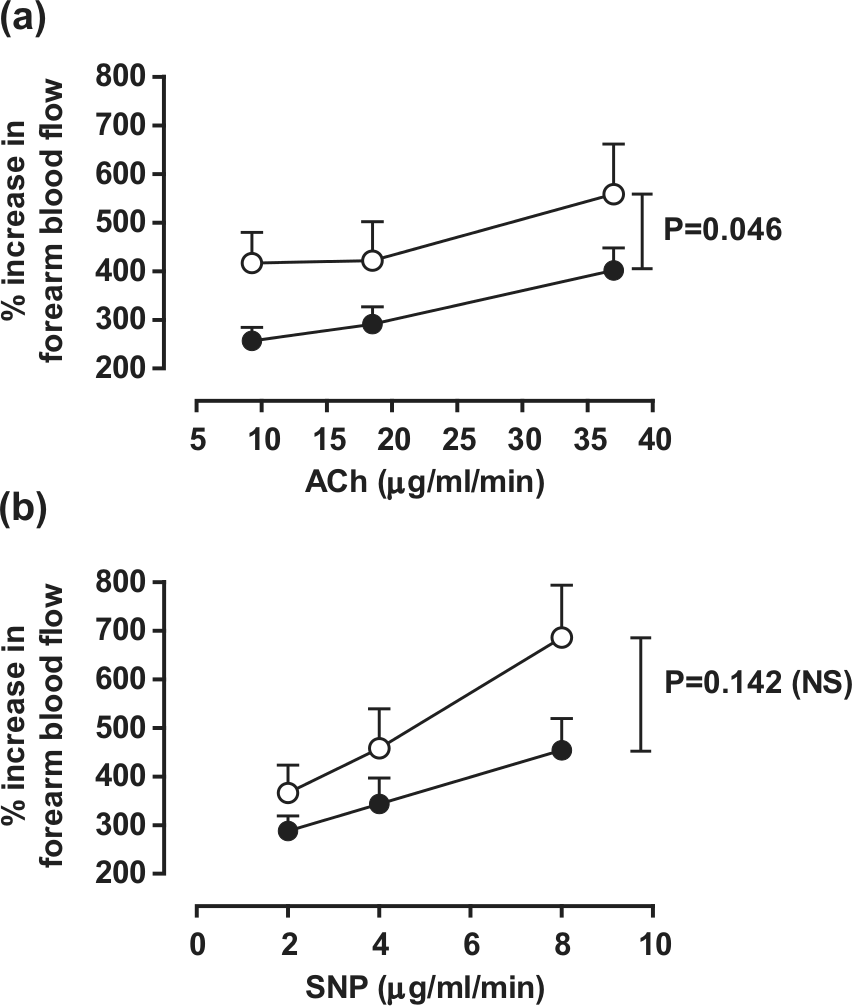
Forearm blood flow responses to infusions of (a) acetylcholine (ACh) and (b) sodium nitroprusside (SNP) measured via forearm venous occlusion plethysmography in control and type 2 diabetes + ACS cohorts.
Microparticles
CD41+ PMP numbers isolated from peripheral blood were not different between the study cohort and controls (n = 14–15; p > 0.05; Student’s unpaired t-test; Figure 3(a)). In contrast, the levels of circulating CD31+/CD41− EMPs in patients in the type 2 diabetes + ACS group were significantly less than in control subjects (type 2 diabetes + ACS: 942 ± 279 µL of plasma vs healthy: 2349 ± 503 µL of plasma; p < 0.05; Student’s unpaired t-test; Figure 3(b)).
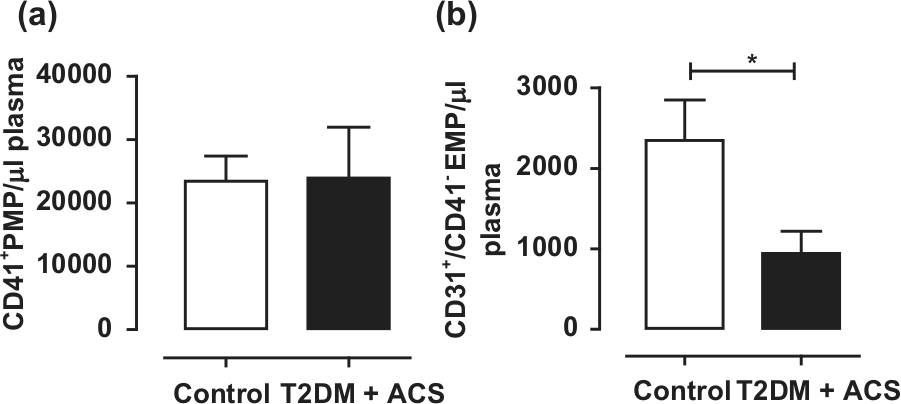
(a) CD41+ platelet microparticle (PMP) and (b) CD31+/CD41− endothelial microparticle (EMP) numbers isolated from peripheral blood from the control and type 2 diabetes + ACS cohorts.
Discussion
Our findings show that patients less than 6 months post ACS with type 2 diabetes on standard clinical care with well-controlled cholesterol levels continue to present with increased triglycerides and glucose levels, higher systolic blood pressure and reduced HDL levels when compared to age- and sex-matched controls. They also demonstrate endothelial dysfunction and decreased EMP levels.
Increased BMI, systolic hypertension, triglycerides, glucose and, conversely, lower HDL levels have all been independently associated with increased risk of a CV event and many are associated with endothelial dysfunction. The finding of endothelial dysfunction in the current cohort of subjects given this context is thus not surprising despite total cholesterol levels being well controlled.
Endothelial dysfunction has been shown to be an independent predictor of CV events. 8,22 Therefore, to improve the prognosis of patients with classic high-risk CV conditions, therapeutic interventions, which also improve endothelial function, are likely to have additional benefit. The potential positive health outcomes of this approach is highlighted by the combination therapy of calcium channel blockers and angiotensin-converting enzyme (ACE) inhibitors employed to normalise systolic blood pressure where the demonstrated effect of restoring endothelial function 23 significantly improved prognosis independent of the effect on blood pressure reduction. 24 Most of the major classes of CV therapies, including aspirin, statins, ACE inhibitors and some of the new-generation β blockers, have now been shown to improve endothelial function 9,25 albeit recent evidence suggesting that statins can worsen endothelial dysfunction has also been reported. 26 The latter highlights the need for further study into this additional, potentially therapeutic effect and its contribution towards improved clinical outcomes.
The use of digital PAT is becoming an increasingly common method of measuring peripheral microvascular endothelial function via its ability to measure pulse wave amplitude during reactive hyperaemia non-invasively. 27 Indeed, the RHI has been shown to correlate with CV risk factors in the Framingham Heart Study, 28 is increased by treatments that reduce endothelial dysfunction 29 and is NO-dependent, 30 and low RHI has been shown to be predictive of future CV events. 30 Furthermore, low RHI has been reported for patients with diabetes, hypertension and CAD. 15,28 In this study, we show for the first time in the type 2 diabetes + ACS study cohort, a significantly reduced RHI compared to controls suggesting reduced microvascular endothelial function.
Similar to reports in the literature for subjects with diabetes, 31 –33 there was no difference between baseline FBF when compared to the control group. The responses to the endothelium-dependent vasodilator, ACh, were reduced in this study indicating endothelial dysfunction, which has also been shown in other studies. 31 –33 Although not statistically significant in this study, the response to the endothelium-independent dilator appeared to trend towards a decrease, and perhaps with a larger sample size, it may have been possible to observe this decrease. Unchanged 32 or reduced 31,33 responses to SNP have been shown previously in type 2 diabetes patients. However, only decreases have been reported in DM patients post ACS. 8
It has recently been suggested that microparticles are important in vascular homeostasis and that the increases seen in disease promote host defence mechanisms. Yet increase in circulating microparticle numbers have been reported in many patients with CV disease and were found to be an independent risk factor for CV events in patients with CAD. 15
Interestingly, EMP numbers were significantly reduced and PMP numbers unchanged in the DM + ACS cohort on medications, findings that were somewhat unexpected as numbers of both types of microparticles are typically increased in patients with DM or MI. 14,34 –36 However, there is some evidence that medical treatment can affect microparticle levels, for instance, diabetes patients treated post MI with vitamin C for 5 days had reduced microparticle numbers. 37 Calcium channel antagonists have also been shown to decrease EMP numbers in patients with DM. 38,39 In contrast, statins have been shown to both increase 40 and decrease 16 microparticle numbers in vitro. Thus, our findings are consistent with reports that clinical care can induce a reduction in circulating EMPs and PMPs. Of interest in this study is the demonstration that endothelial function, defined as a decrease in response to the NO inducing agonist ACh, remains dampened in a patient cohort on treatment despite a reduction in EMP levels. This suggests that these two markers of endothelial health are independent, revealing different mechanisms contributing to cellular function.
While the results of this study provide evidence of vascular dysfunction in patients with type 2 diabetes and ACS despite standard clinical care, further work is required. It remains to be determined whether the results collected from the modest sample size can be extrapolated to a larger population. Future studies should include an ACS control group to ascertain the contribution diabetes has on the observed outcome measures.
In summary, despite standard clinical care, patients with type 2 diabetes post ACS still exhibit endothelial dysfunction, measured via PAT and forearm venous occlusive plethysmography.
Conclusion
We conclude that there is a treatment gap in this patient cohort for future therapies that can improve endothelial function, beyond what is currently prescribed for these patients.
Footnotes
Acknowledgements
Natalie G Lumsden and Karen L Andrews contributed equally to this study. All authors contributed to the study conception and design. Anthony M Dart, David M Kaye and James A Shaw conducted the brachial artery cannulations. Natalie G Lumsden, Xiao L Moore, Amanda K Sampson, James A Shaw, David M Kaye, Anthony M Dart and Karen L Andrews analysed data. Karen L Andrews, Natalie G Lumsden and Jaye PF Chin-Dusting wrote the first draft of this article. All authors commented on and took part in the revision of this article and approved the final version. The authors acknowledge and thank Jenny Starr and Donna Vizi (Heart Centre, The Alfred Hospital, Melbourne, VIC 3004, Australia) for their technical support and assistance during the course of this study.
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This study was partly funded by the Victorian Government’s Operational Infrastructure Support Programme and by a grant-in-aid from F. Hoffmann-La Roche, Basel, which contributed to study design and manuscript preparation. Professors David M Kaye, Anthony M Dart and Jaye PF Chin-Dusting are Research Fellows funded by the National Health Medical Research Council of Australia.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.