Abstract
Purpose:
Associations of semicarbazide-sensitive amine oxidase (SSAO) activity with renal and vascular function, oxidative stress, glycaemia and diabetes complications were determined.
Methods:
Plasma SSAO activity in 94 type 1 diabetes (T1DM) patients, including 34 with microvascular complications T1DM CX[+], and in 96 healthy subjects (CON) was measured by production of benzaldehyde using high-performance liquid chromatography (HPLC).
Results:
SSAO activity (mean ± SD) was greater in T1DM than in CON (1049 ± 294 vs 749 ± 204 mU/L; p < 0.00001) and was higher in T1DM CX[+] vs complication-free DM subjects (1148 ± 313 mU/L vs 982 ± 269 mU/L; p = 0.01). In T1DM, SSAO activity correlated with renal dysfunction [estimated glomerular filtration rate (eGFR): r = −0.44; p = 0.0001; cystatin C: r = 0.47; p = 0.0001] and markers of inflammation [soluble vascular cell adhesion molecule-1 (sVCAM-1): r = 0.41, p = 0.0001; soluble intercellular adhesion molecule-1 (sICAM-1): r = 0.33, p = 0.002] and was inversely related to small artery elasticity (SAE) (r = −0.23, p = 0.03). In CON, SSAO activity correlated with HbA1c (r = 0.26; p = 0.02).
Conclusion:
In T1DM, SSAO activity correlates with renal dysfunction, but not with glycaemia, and may promote vascular inflammation and be a therapeutic target.
Keywords
Introduction
Semicarbazide-sensitive amine oxidases (SSAOs) are an enzyme group located in cell membranes and in the circulation 1 that have roles in inflammation 2 and adipocyte glucose uptake.3,4 SSAO deaminates methylamine and aminoacetone 5 to produce formaldehyde and methylglyoxal, which are also found in cigarette smoke, 6 and ammonia and hydrogen peroxide, all of which are implicated in vascular damage. SSAO is present in a soluble form in plasma and in many tissues relevant to diabetes complications, including the retina, 7 vascular endothelial 8 and smooth muscle cells, 9 arteries, 10 adipocytes 11 and in areas of inflammation.12,13 Enzymatic sources of SSAO activity include lysyl oxidase and three copper-containing amine oxidases (AOC). Levels of AOC3, also known as vascular adhesion protein-1 (VAP-1), correlate well with circulating SSAO activity, and AOC3 is regarded as the main source of SSAO activity in assays utilising benzylamine as the substrate, such as the assay used in this study. 14
The physiological role of SSAO is thought to relate to the production of hydrogen peroxide, which acts as a signal-transducing molecule, affecting GLUT1 and GLUT4 translocation to the plasma membrane in an insulin-mimicking manner.15,16 However, in diabetic animal studies, modulating SSAO has divergent effects, some adverse and some beneficial. Administration of methylamine (another SSAO substrate) to animals improves glycaemia, 17 but promotes hypertension, atherosclerosis and glomerulosclerosis, 18 thought to relate to reactive aldehydes from methylamine oxidation, which has been averted by administration of different SSAO substrates.13,19,20 Most human data regarding SSAO activity in diabetes relate to type 2 diabetes (T2DM), with few studies in type 1 diabetes (T1DM).21,22 The levels of SSAO activity, its clinical correlates, regulatory factors and role of SSAO/VAP-1 activity in diabetes complications require further elucidation.7,22–24
Circulating SSAO activity is increased in diabetes, both in humans 22 and animals, 25 in renal disease, 26 cardiovascular disease (CVD), 12 inflammation 27 and in disseminated cancer. 28 Elevated adrenaline output, 29 which is associated with increased CVD-related morbidity and mortality, 30 may also upregulate SSAO activity. Higher SSAO activity has been shown to be an independent predictor of mortality in patients with heart failure. 31 Because there is a lack of detailed studies of SSAO activity in well-characterised patients with T1DM, we conducted a cross-sectional study of T1DM patients, both with and without microvascular complications, and subjects without diabetes (CON) to evaluate SSAO activity levels and their associations with vascular function and traditional and novel vascular risk factors.
Subjects
The study was approved by the St Vincent’s Hospital Melbourne Human Research Ethics Committee, and each subject gave written informed consent. T1DM subjects were recruited from hospital diabetes clinics and CON subjects, matched for age, gender and ethnicity, were recruited from the community. Exclusion criteria were end-stage renal disease (ESRD), inflammatory conditions, recent (<3 months) surgery, infective illness, myocardial infarction, stroke, cardiac arrhythmia, diabetic ketoacidosis, cancer or anti-oxidant vitamin supplement intake. A medical history and examination were performed and diabetic complication status verified by treating clinicians. Retinopathy was defined as pre-proliferative or proliferative retinopathy requiring pan-retinal laser treatment. Nephropathy was defined as albuminuria (>20 µg/min) on repeated timed (12 or 24 h) urine collections in the absence of infection. Even if albuminuria subsequently regressed to normal range with treatment (such as with angiotensin-converting enzyme inhibitors), subjects were still categorised as having nephropathy. CVD was defined as a documented myocardial infarction or angina with electrocardiography (ECG) changes and/or positive cardiac imaging study, a transient ischaemic attack (TIA) or stroke, amputation, angioplasty or vascular bypass surgery.
Methods
Non-invasive measures of vascular health
Pulse wave analysis, including derivation of large and small artery elasticity (LAE and SAE) and systemic vascular resistance (SVR), which correlate with pulse wave velocity and brachial artery flow-mediated dilation, respectively, 32 was performed on rested supine subjects (PulseWave™ CR-2000; Hypertension Diagnostics Inc., Eagan, MN, USA), as previously described, 32 and the mean of three measures over 5–10 min was used in data analysis. Inter-measurement coefficients of variation (CVs) for LAE and SAE were 7.1% and 4.9%, respectively. The mean of three measures of blood pressure (BP) and pulse pressure, used in data analyses, was taken from the CR-2000. Subjects were regarded as hypertensive if the mean BP was >139/89 mmHg or they were taking any anti-hypertensive therapy.
Clinical chemistry
All subjects were evaluated after an overnight fast and prior to medication. Fresh blood and urine samples were analysed by the St Vincent’s Hospital Melbourne Clinical Chemistry Laboratory (blood glucose, HbA1c, creatinine, lipids and urine albumin and creatinine). Within 30 min of venepuncture, the blood (kept on ice, after clotting of serum) was centrifuged (2000 g, 15 min), and plasma and serum aliquots were stored (−80 °C) until analysis in the research laboratory [SSAO activity, 1,5-anhydroglucitol (1,5-AG), homocysteine, cystatin C, oxidised low-density lipoprotein (Ox-LDL), high-sensitivity C-reactive protein (hsCRP), cell adhesion molecules (CAMs)]. HbA1c, fasting glucose and lipid levels, serum creatinine and urine albumin and creatinine were measured using standard analytical methods. Estimated glomerular filtration rate (eGFR) was calculated using the modified Cockcroft–Gault formula. Serum cystatin C and homocysteine levels were measured by nephelometry using a BN II System (Dade Behring, Marburg, Germany) with intra- and inter-assay CVs of 2.3% and 2.7% (cystatin C), and 3.9% and 5.3% (homocysteine), respectively. As an assessment of glycaemia and glycaemic variability 1,5-AG was measured by the GlycoMark assay (GlycoMark, Inc., Winston-Salem, NC, USA) implemented on a Roche COBAS INTEGRA autoanalyser (Roche Diagnostic, Basel, Switzerland) with intra- and inter-assay CVs of 2.6% and 3.7%, respectively.
SSAO activity
Plasma SSAO activity was assayed as described by Van Dijk et al. 33 Briefly, 50 µL of heparinised plasma was added to a reaction mixture containing 350 µL 100 mM sodium phosphate buffer (pH 7.8), 50 µL 9.07 mM chlorgyline in 10 mM hydrochloric acid and 50 µL 26.11 mM benzylamine (the substrate for SSAO) in 10 mM hydrochloric acid and incubated (37 °C, 60 min) in a water bath. The reaction was stopped by addition of 50 µL 40% trichloroacetic acid, and the sample was cooled on ice and centrifuged (10,000 g, 3 min). Supernatant (400 µL) was then derivatised by the addition of 200 µL dimedone solution and 50 µL 9 M sulphuric acid and incubation (95 °C, 45 min). After cooling, 20 µL of sample was injected into a high-performance liquid chromatography (HPLC) system (comprising a Waters 510 isocratic pump, 717plus auto-sampler coupled with a 474 fluorescence detector). Samples were separated on a Spherisorb 3 µm ODS2 (150 mm × 4.6 mm) column using mobile phase of water/acetonitrile (50:50, v/v) at a flow rate of 1.0 mL/min. The reaction product benzaldehyde was detected fluorometrically at excitation/emission wavelengths of 386 and 451 nm, respectively. Data represent the mean of triplicate analyses. Intra- and inter-run CVs were 9.0% and 11.2%, respectively.
Inflammation/oxidative stress markers
Serum levels of CAMs soluble intercellular adhesion molecule-1 (sICAM-1), soluble vascular cell adhesion molecule-1 (sVCAM-1) and sE-selectin were measured by enzyme-linked immunosorbent assay (ELISA) (R&D Systems, Inc., Minneapolis, MN, USA) with intra- and inter-assay CVs of 2.2% and 8.4%, 2.3% and 8.7%, and 3.8% and 10.3%, respectively. Serum CRP was measured using a high-sensitivity, immunonephelometric method (BN II System; Dade Behring) with intra- and inter-assay CVs of 2.1% and 2.5%, respectively. Ox-LDL in serum was quantified by ELISA (Mercodia, Uppsala, Sweden) with intra- and inter-assay CVs of 4.3% and 5.6%, respectively, and was expressed as the Ox-LDL/LDL-cholesterol ratio.
Statistics
Data were analysed by STATISTICA (data analysis software system), version 8.0 (StatSoft, Inc., 2008). Differences between groups in mean values for continuous variables were tested using Student’s t-test or by the Mann–Whitney U-test for non-normally distributed variables [levels of triglycerides, hsCRP, 1,5-AG, serum creatinine, cystatin C, eGFR, urinary albumin/creatinine ratio (ACR) and Ox-LDL/LDL-cholesterol ratio]. Differences in prevalence were tested using Chi-square tests. Univariate associations between variables were tested by Pearson correlation (or Spearman for non-normally distributed variables) coefficients. For principal component analysis, 19 variables reflecting inflammation (hsCRP), endothelial cell activation (sE-selectin, sICAM-1 and sVCAM-1), vascular function [mean arterial pressure (MAP), LAE, SAE], dyslipidaemia [triglycerides, high-density lipoprotein (HDL)-cholesterol], glycaemia (fasting blood glucose and HbA1c), renal function (ACR, cystatin C, eGFR), oxidative stress (homocysteine, Ox-LDL/LDL-cholesterol ratio), body mass index (BMI) and age were chosen. Based on a scree plot representing changes in eigenvalue vs number of factors, four factors were analysed as determinants of SSAO activity in T1DM and in control subjects. After initial factor extraction, varimax rotation was used and factor loadings (correlations) calculated. Values ≥0.5 of factor loadings were arbitrarily set to confirm that independent variables identified a priori are represented by a particular factor, on the rationale that the 0.5 level corresponds to 25% of the variance in the indicator being explained by the factor. Statistical significance was taken at p < 0.05.
Results
Clinical characteristics
Clinical chemistry results of the 96 CON and 94 T1DM subjects [including 34 patients with micro- and/or macrovascular complications of diabetes (T1DM CX[+])] recruited for the study are shown in Table 1. Average diabetes duration was 21 ± 13 years [mean ± standard deviation (SD)], and age ranged from 18 to 70 years. Compared to CON, T1DM had higher levels of fasting glucose, HbA1c, BP, serum creatinine, ACR, Ox-LDL/LDL, hsCRP and CAMs and lower levels of 1,5-AG, SAE and LAE. Of the 34 T1DM CX[+], 20 had both retinopathy and nephropathy, 9 had nephropathy only and 5 had retinopathy only. Five patients also had macrovascular complications as well as a microvascular complications. Compared to T1DM without complications of diabetes (T1DM CX[−]), the T1DM CX[+] group had higher BMI and BP, longer T1DM duration, higher HbA1c, serum creatinine, homocysteine, cystatin C, triglycerides and sVCAM-1 levels and lower eGFR and SAE.
Clinical characteristics of patients with type 1 diabetes and controls.
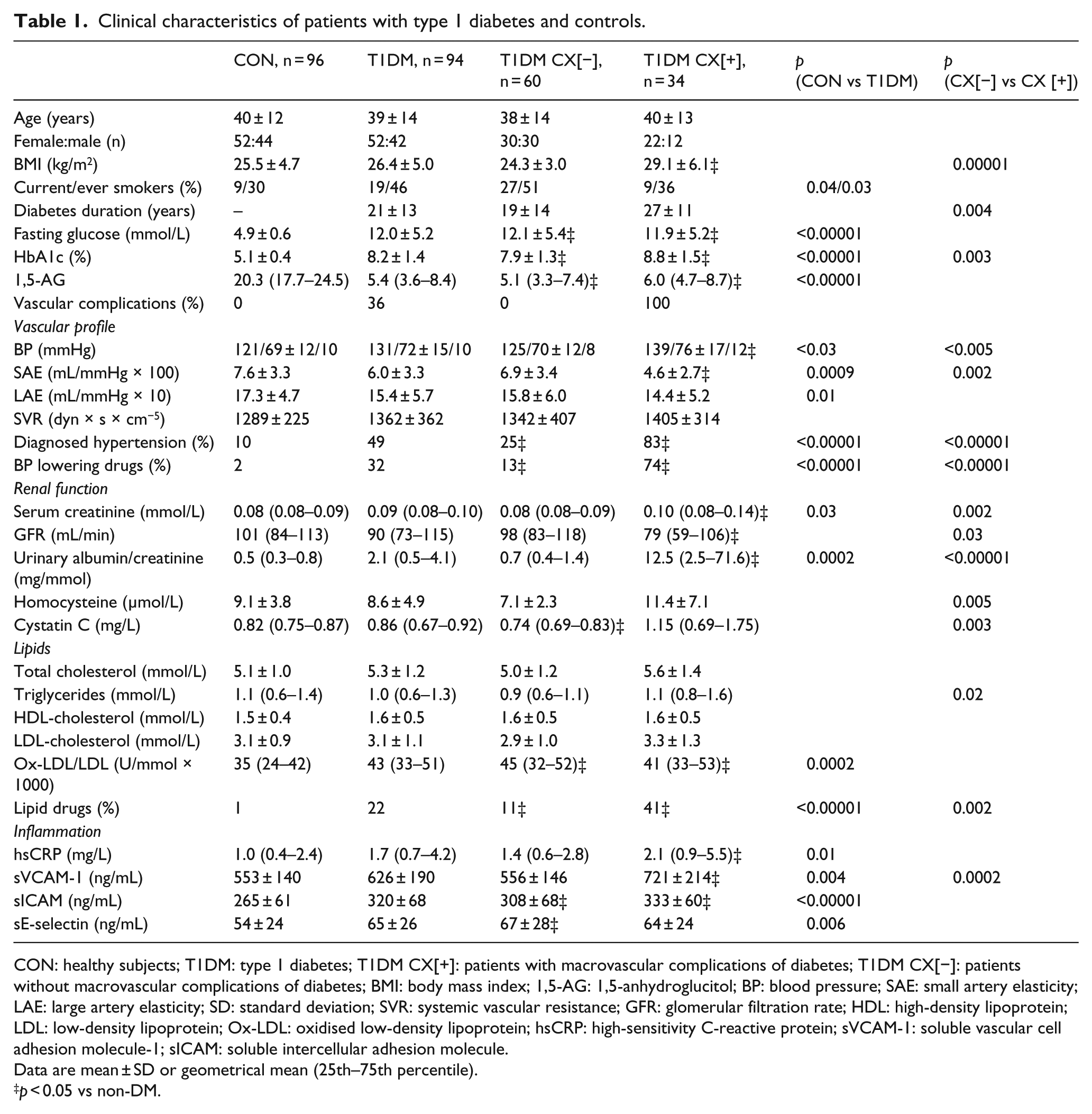
CON: healthy subjects; T1DM: type 1 diabetes; T1DM CX[+]: patients with macrovascular complications of diabetes; T1DM CX[−]: patients without macrovascular complications of diabetes; BMI: body mass index; 1,5-AG: 1,5-anhydroglucitol; BP: blood pressure; SAE: small artery elasticity; LAE: large artery elasticity; SD: standard deviation; SVR: systemic vascular resistance; GFR: glomerular filtration rate; HDL: high-density lipoprotein; LDL: low-density lipoprotein; Ox-LDL: oxidised low-density lipoprotein; hsCRP: high-sensitivity C-reactive protein; sVCAM-1: soluble vascular cell adhesion molecule-1; sICAM: soluble intercellular adhesion molecule.
Data are mean ± SD or geometrical mean (25th–75th percentile).
p < 0.05 vs non-DM.
Plasma SSAO activity
As shown in Figure 1, plasma SSAO activity was significantly greater in T1DM relative to CON (1049 ± 294 vs 749 ± 204 mU/L; p < 0.00001) and was also significantly (17%) higher in T1DM with vs without vascular complications: T1DM CX[+] vs T1DM CX[−] (1148 ± 313 mU/L vs 982 ± 269 mU/L; p = 0.01).
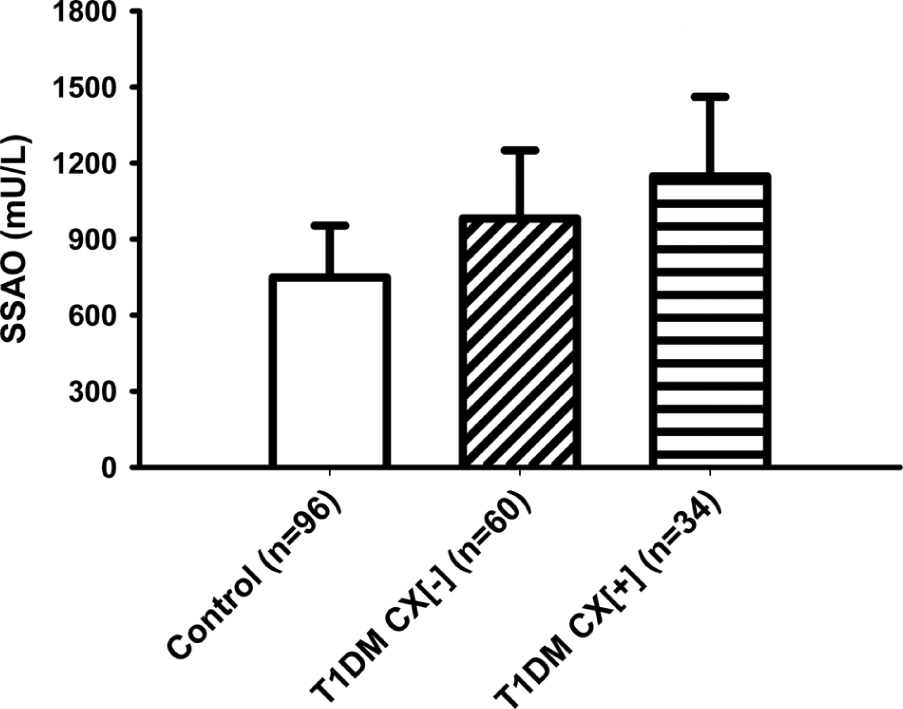
SSAO activity (mean ± SD) in healthy subjects (control), patients with type 1 diabetes (T1DM) and patients with diabetes and complications (T1DM CX[+]).
Correlates of SSAO activity in T1DM
In univariate correlation analyses, plasma SSAO activity correlated significantly with diabetes duration (r = 0.27, p = 0.01), vascular inflammation marker levels (sVCAM-1: r = 0.37, p = 0.001; sICAM-1: r = 0.34, p = 0.001) and impaired vascular function (SAE: r = −0.23, p = 0.04). Plasma SSAO activity increased with declining renal function (eGFR: rho = −0.40, p = 0.0002; cystatin C: rho = 0.36, p = 0.0001; serum creatinine: rho = 0.26, p = 0.02) and was positively correlated with homocysteine levels (r = 0.53, p = 0.0001). SSAO activity was not significantly correlated with BMI, glycaemia (fasting glucose, HbA1c, 1,5-AG), lipid Levels or BP.
Correlates of SSAO activity in subjects without diabetes
Of the factors listed in Table 1, plasma SSAO activity was only statistically significantly correlated with HbA1c levels (r = 0.26, p = 0.02).
Determinants of SSAO activity–principal component analysis
Factor loadings are shown in Table 2. Furthermore, regression analysis revealed that only factors no 1 and 4, with highest loadings for age, MAP, LAE, SAE, eGFR and serum cystatin C, sVCAM-1 and homocysteine, were independent predictors of plasma SSAO activity in T1DM, with overall r = −0.38, p = 0.0001 and r = −0.23, p = 0.02, respectively. In the CON group, no factor was a statistically significant predictor of SSAO activity.
Principal component analysis – factor loadings in type 1 diabetes.
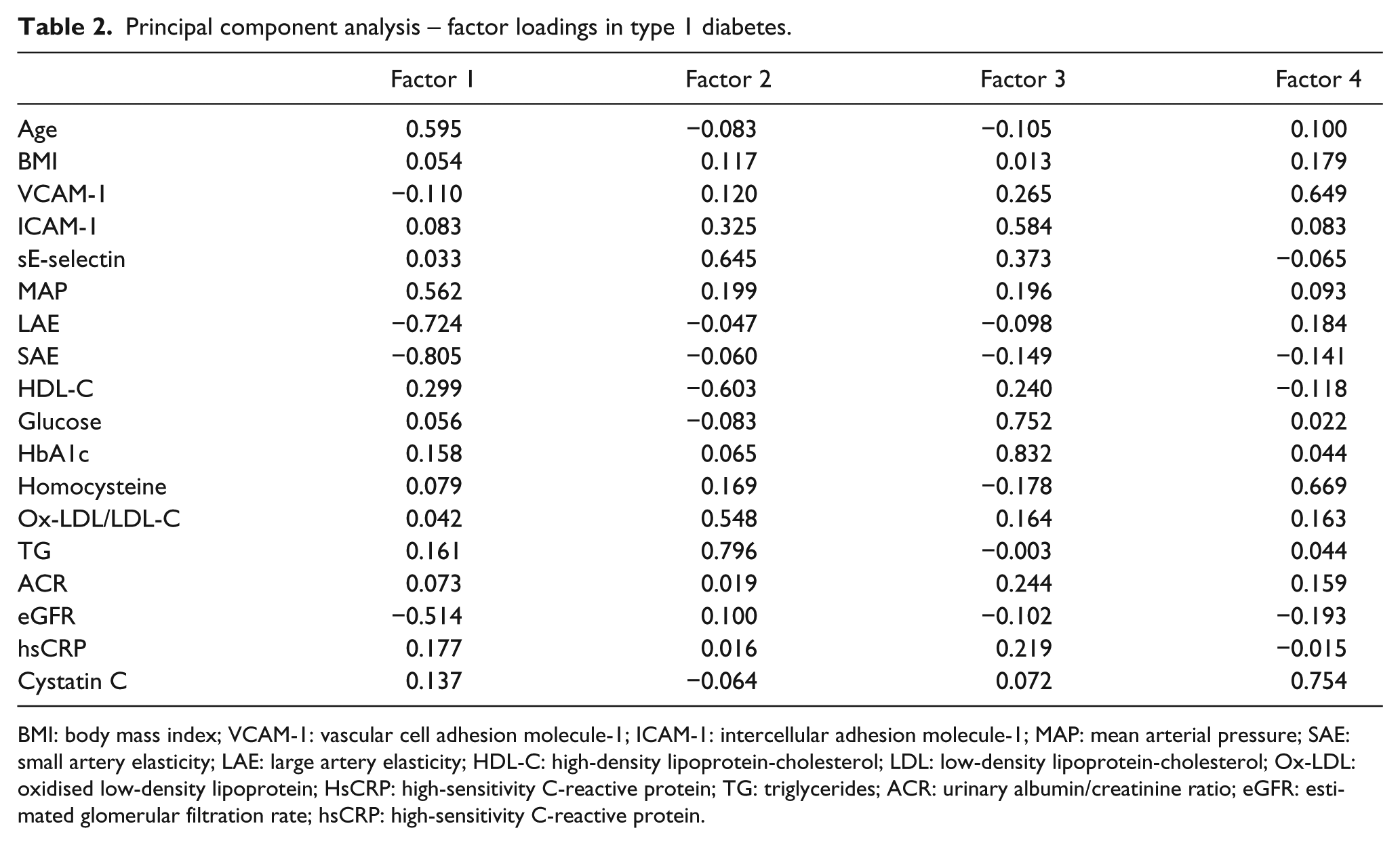
BMI: body mass index; VCAM-1: vascular cell adhesion molecule-1; ICAM-1: intercellular adhesion molecule-1; MAP: mean arterial pressure; SAE: small artery elasticity; LAE: large artery elasticity; HDL-C: high-density lipoprotein-cholesterol; LDL: low-density lipoprotein-cholesterol; Ox-LDL: oxidised low-density lipoprotein; HsCRP: high-sensitivity C-reactive protein; TG: triglycerides; ACR: urinary albumin/creatinine ratio; eGFR: estimated glomerular filtration rate; hsCRP: high-sensitivity C-reactive protein.
Discussion
In a cross-sectional study, we have demonstrated that T1DM is associated with higher plasma SSAO activity, particularly in patients with microvascular complications. Given the functional connections between SSAO activity, inflammation and vascular damage, this elevated activity in T1DM may contribute to the pathogenesis of microvascular complications; however, we recognise that measures of membrane-bound SSAO in tissues would be desirable. We demonstrated increased plasma SSAO activity in T1DM but no correlation with measures of glycaemia including HbA1c, 1,5-AG and fasting glucose. Our findings are in agreement with a cross-sectional study by Boomsma et al., 22 in which SSAO activity was higher in newly diagnosed T1DM (n = 100), compared to their siblings (n = 76), but did not correlate with HbA1c levels. In another article by Boomsma et al., 21 plasma SSAO activity in T1DM (n = 104) with microvascular complications was increased relative to controls and correlated with renal function and with HbA1c. However, the control group consists of subjects with borderline hypertension (n = 51) with unknown treatment and healthy laboratory workers (n = 16), rather than a more representative and better characterised control group, as in our study. In another cross-sectional study of T1DM, Boomsma et al. 26 reported a positive correlation between plasma SSAO activity and HbA1c and with angiotensin-converting enzyme activity. In our study, plasma SSAO activity correlated with HbA1c in CON but was unrelated to any measure of glycaemia (HbA1c, 1,5-AG or fasting blood glucose) in T1DM. Although we noted a correlation between 1,5-AG, a marker of glycaemia and glycaemic variability over days to weeks, and plasma SSAO activity in the combined T1DM and CON (data not shown), it was driven by the different levels of 1,5-AG in these groups, as there was no statistically significant correlation with SSAO activity within each of the groups. Associations of membrane-bound SSAO activity, which we did not measure, with measures of glycaemia would be of interest.
Similarly in T2DM, circulating SSAO activity has been reported to be increased relative to control subjects21–23 and has been found to be higher in T2DM subjects with proliferative diabetic retinopathy and/or clinically significant macular oedema. 23 Positive correlations have been described with HbA1c, 14 adiposity and dyslipidaemia. 22 Variation in glycaemia (measured as mean amplitude of glucose excursions, MAGE) in T2DM has been linked with increased oxidative stress and microvascular complications. 34 In contrast, plasma SSAO activity in our study was not linked with glycaemia, BMI or lipid levels in T1DM or in CON. Apart from Ox-LDL levels, we did not perform any measures of oxidative stress. We demonstrated a correlation between increased plasma SSAO activity and impairment of vascular function (lower SAE), as well as with levels of biomarkers for vascular inflammation and atherosclerosis (CAMs). These correlations between plasma SSAO activity in T1DM and both novel vascular risk factors and vascular dysfunction suggest a possible role for SSAO in the pathogenesis of vascular complications and a potential target for therapeutic intervention.
In our cross-sectional study, by both univariate analysis and principal component analysis, renal dysfunction was a correlate of SSAO activity in T1DM but this association could operate causally in either direction. As Poortmans et al.’s 35 experiment showed, dietary creatine (creatinine substrate) supplementation caused increased methylamine (SSAO substrate) formation and excretion; thus, the correlation observed in our study is not unexpected. In transgenic animals overexpressing SSAO, glomerulosclerosis was increased; 18 thus, increased SSAO in T1DM could promote microvascular damage and impaired renal function. In patients with uraemia, Nemcsik et al. 36 demonstrated reduced serum SSAO activity (measured by radiochemical assay) in haemodialysis patients (compared to healthy people), but serum SSAO activity measured by hydrogen peroxide production was increased. They suggested that this method-based disparity in serum SSAO activity in uraemia was due to the increased levels of methylamine in uraemic patients competing with the radiolabelled exogenous SSAO substrate leading to falsely low values in the radiochemical assay. In keeping with the results of our study, Boomsma et al. 21 showed increased plasma SSAO activity in T1DM subjects with retinopathy and nephropathy when compared to complication-free T1DM subjects and also found correlations between heparinised plasma SSAO activity and renal dysfunction reflected by urinary albumin excretion. In our study, in T1DM, plasma SSAO activity was increased with multiple measures of renal dysfunction, including eGFR, cystatin C, serum creatinine and homocysteine. Levels of homocysteine, a recognised risk factor for vascular dysfunction and for CVD,32,37 generally increase with renal function decline. In our study, homocysteine levels in T1DM were inversely correlated with renal function measured by serum creatinine, cystatin C levels and eGFR.
It is not clear whether elevated plasma or serum SSAO activity precedes and causes renal damage or whether there is reduced renal clearance of SSAO resulting in greater circulating levels and activity. Regardless, increased SSAO activity may be one of the mechanisms by which renal disease accelerates CVD and mediates vascular dysfunction 38 associated with diabetic renal disease, 39 through the pathways identified above. The correlation between plasma SSAO activity and vascular dysfunction measured by SAE was observed in T1DM, but not in the control subjects. To our knowledge, this is the first report of a correlation between circulating SSAO activity and vascular function (such as reflected by pulse wave analysis) in human T1DM. Li et al. 40 reported that the acute increase in serum SSAO activity during an oral glucose tolerance test in subjects without diabetes correlated with carotid intima-medial thickness (IMT), a structural measure of vascular health. In an animal model overexpressing tissue SSAO, Göktürk et al. 10 reported reduced elasticity of the aorta and renal arteries attributed to misfolding of elastic laminae. Furthermore, in SSAO null animals, increased arterial diameter but normal mechanical properties have been reported. 41 Other predictors of CVD events 42 and indicators of vascular endothelial dysfunction 43 are circulating levels of CAMs such as sVCAM, sICAM and sE-selectin, which promote leukocyte vascular adhesion and ingress, 44 and we found positive correlations between levels of VCAM-1 and ICAM-1 and plasma SSAO activity. We also demonstrated increased levels of hsCRP, which is produced predominantly by the liver but may also be produced by human arteries, 45 and increased circulating CAMs in T1DM vs CON, but levels of hsCRP were not significantly correlated with plasma SSAO activity. Correlations with tissue SSAO, which we did not measure, would be of interest.
Smoking is also a pro-inflammatory stimulus, but in our study, plasma SSAO activity did not differ significantly between smokers and non-smokers (in T1DM, CON or the combined groups). Other inflammatory diseases such as liver disease have also been associated with increased serum SSAO activity. 46 Overall, the human data are supportive of a positive correlation between inflammation and circulating SSAO activity in diabetes. Longitudinal studies of inflammation and of SSAO activity and of the effects of anti-inflammatory drugs on plasma and membrane-bound SSAO activities would elucidate associations between inflammation and SSAO activity.
A variable-reduction data approach through principal component analysis revealed that factors combining age, vascular health (MAP, SAE, LAE), eGFR and serum cystatin C, sVCAM-1 and homocysteine levels were the independent correlates of plasma SSAO activity in T1DM but not in CON subjects. Although consistent with our univariate analysis findings and with other publications,21,36,43 it remains to be determined whether SSAO activity changes over time in people with diabetes, and if so, what factors, such as glycaemia, renal function and inflammation, are associated with these changes, and whether circulating and membrane-bound SSAO activities in various tissues change in parallel. Longitudinal studies of the predictive power of SSAO activity for microvascular and macrovascular complications in diabetes would be more informative, and mechanistic studies and intervention studies to reduce SSAO activity would be more definitive in determining the role (or lack thereof) of SSAO in the vascular complications of diabetes. Ideally both plasma and membrane-bound SSAO activity measures are desirable. We acknowledge that correlations and associations in a clinical study, as we have observed, or lack thereof, do not prove cause and effect. Mechanistic studies, such as with molecular models and tools, are desirable.
In conclusion, plasma SSAO activity is increased in T1DM, more so in the presence of vascular complications. The association of SSAO activity in plasma and in tissues for future vascular outcomes merits further research, as SSAO has been suggested as a therapeutic target, particularly in relationship to its inflammatory and anti-angiogenic effects. 47
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research was supported by grants from National Heart Foundation of Australia and VicHealth.
