Abstract
Cardiac autonomic neuropathy is a neglected diabetic chronic complication for which genetic predictors are rarely reported. Oxidative stress is implicated in the pathogenesis of microvascular complications, and glutathione peroxidase 4 is involved in the detoxification of peroxides and of reactive oxygen species. Thus, the association of a functional variant in the gene encoding glutathione peroxidase 4 (rs713041) with this diabetic complication was investigated in 341 individuals with type 1 diabetes evaluated for cardiac autonomic neuropathy status (61.7% women, 34 [27–42] years old; diabetes duration: 21 [15–27] years; HbA1c: 8.3% [7.4–9.4]; as median [interquartile interval]). Cardiac autonomic neuropathy was present in 29% of the participants. There was an inverse association of the minor T allele of rs713041 with cardiac autonomic neuropathy (odds ratio = 0.39; 95% confidence interval = 0.17–0.90; p = 0.0271) after adjustment for potential confounders. The functional glutathione peroxidase 4 variant rs713041 modulated the risk for cardiac autonomic neuropathy in the studied population with type 1 diabetes.
Cardiac autonomic neuropathy (CAN) is a neglected diabetic complication recognized as a cardiovascular risk factor. 1 Although the role of hyperglycaemia to its development is well established, genetic predictors are rarely reported. Thus, we investigated whether rs713041, a functional variant in the gene encoding glutathione peroxidase 4 (GPX4) 2 previously associated with several conditions, including diabetic kidney disease, 3 could explain genetic susceptibility to CAN in individuals with type 1 diabetes (T1D).
GPX4 is a selenoprotein that detoxifies compounds using glutathione as a cofactor. This functional variant was chosen because GPX4 is expressed in peripheral nerves 4 and because oxidative stress is implicated in the pathogenesis of chronic microvascular complications.
A total of 357 individuals with T1D was recruited from two university hospitals. Inclusion criteria were T1D for more than 10 years and HbA1c > 8% in any period of life. After exclusion of individuals with hypocortisolism and using medications which could interfere with heart rate (HR), 341 individuals were included. Participants were subjected to evaluation of spectral analysis of the HR variability, Ewing tests and measurement of systolic blood pressure after 3 min of standing; diagnosis of CAN was made in the presence of orthostatic hypotension or in the presence of ⩾3 abnormalities of HR variability and Ewing tests. Participants were also evaluated for clinical and biochemical characteristics, and for microvascular complications as previously described. 5
rs713041 (718C/T within the 3′ untranslated region) was genotyped by the TaqMan system (Applied Biosystems, Foster City, CA, USA) using the procedures recommended by the manufacturer. Genotyping success rate was >98%, and the distribution of genotypes was consistent with Hardy–Weinberg equilibrium. Statistical analysis was conducted using JMP 8.1. Results are expressed as median ± interquartile interval. Differences between groups were assessed by Pearson’s χ2 and by Wilcoxon’s tests. Continuous variables were log-transformed. Genetic association was assessed by logistic regression analysis adjusted for age, sex, diabetes duration, HbA1c, triglycerides concentrations, estimated glomerular filtration rate (eGFR) and status of albuminuria; odds ratio (OR) with 95% confidence interval (CI) was computed for the minor allele in a co-dominant model; a p < 0.05 was considered significant. The power of the study was ~70% to detect the association of the variant (co-dominant model) with a minor allele frequency ⩾0.4 and a relative risk ⩾1.5 (or protective ⩽0.65).
The characteristics of the 341 T1D individuals are shown in Table 1. There was an inverse association of the minor T allele of rs713041 with CAN (OR = 0.39; 95% CI = 0.17–0.90; p = 0.0271) after adjustment for potential confounders.
Characteristics of the type 1 diabetes individuals (overall and sorted by cardiac autonomic neuropathy status).
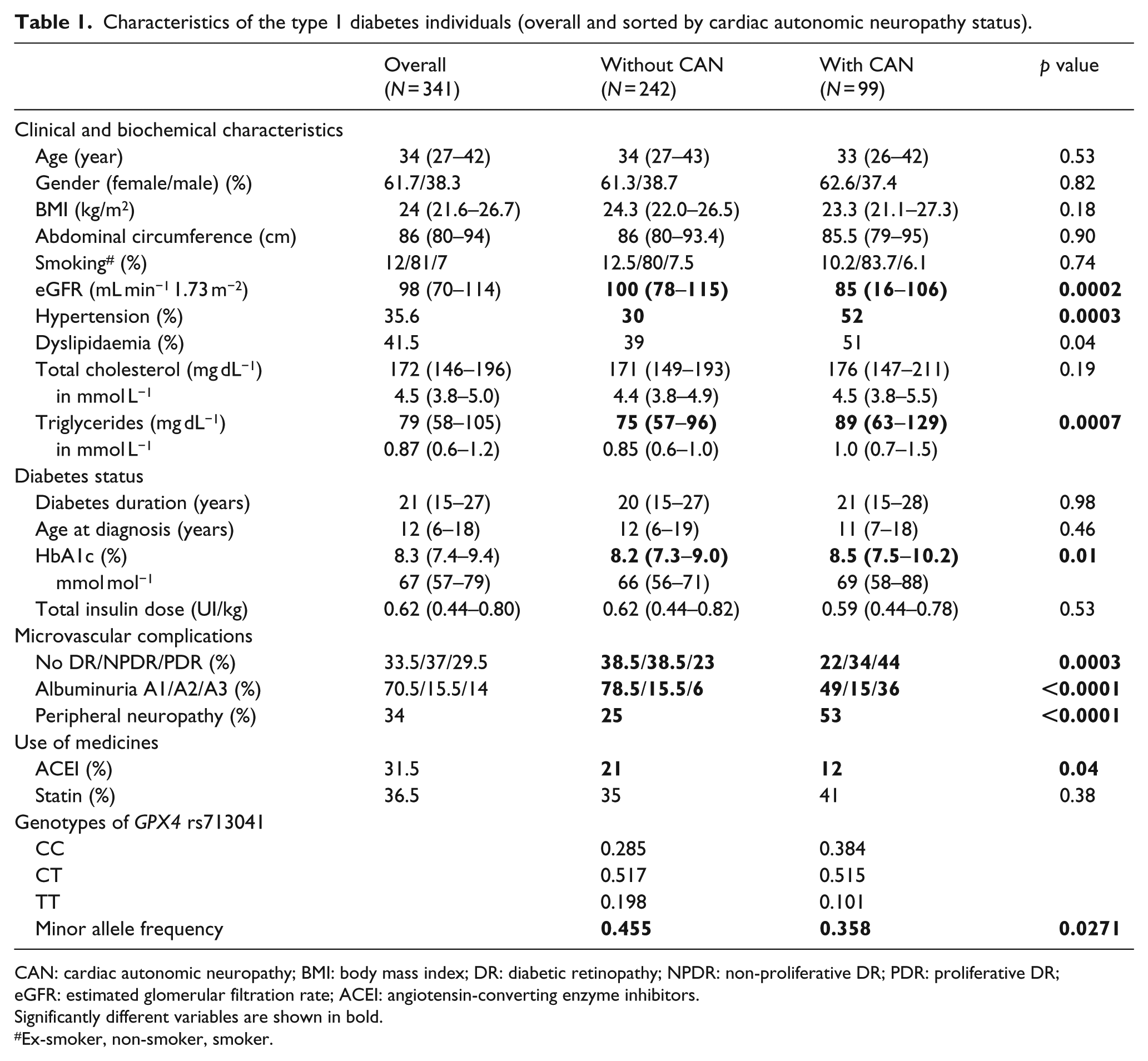
CAN: cardiac autonomic neuropathy; BMI: body mass index; DR: diabetic retinopathy; NPDR: non-proliferative DR; PDR: proliferative DR; eGFR: estimated glomerular filtration rate; ACEI: angiotensin-converting enzyme inhibitors.
Significantly different variables are shown in bold.
Ex-smoker, non-smoker, smoker.
The performance of GPX4 depends on factors such as the presence of functional variants in its coding gene; rs713041 was shown to be functional: 2 in selenium-deficient conditions, cells overexpressing the C variant presented higher contents of reactive oxygen species and lower GPX activity after an oxidative challenge in comparison to cells overexpressing the T variant. 6 As GPX activity was shown to be decreased in peripheral neurones from rats with diabetes neuropathy, it is plausible to speculate that carriers of the T allele of rs713041 are more protected against CAN because of their capacity of preserving a better degree of GPX activity in a situation of selenium deficiency secondary to hyperglycaemia-induced oxidative stress.
The present finding should be interpreted in the context of limitations of cross-sectional studies and needs validation in larger populations. Another limitation of this study is the absence of selenium plasma data – although it is possible that a punctual evaluation of plasmatic selenium could not reflect its chronic status. The study’s strengths are the detailed collected data that allowed adjustment for potential confounders and the systematic evaluation of CAN by a broad panel of tests.
To our knowledge, this is one of the few studies conducted in a sample of more than 300 individuals with T1D to report an association between CAN and a genetic variant. One potential implication of the present finding is the possibility of testing synthetic selenium compounds that mimic GPX activity in the treatment of CAN in its incipient phase.
Footnotes
Acknowledgements
S.N.A. and D.P.S.-B. have equal authorship to this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval and consent to participate
Study protocol was approved by the Ethics Committee of Hospital das Clinicas da Universidade de Sao Paulo (#07513013.8.1001.0068) and Hospital das Clinicas UNICAMP (#235.357). All participants gave written informed consent.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) to D.P.S.-B. (12/25490-8) and to M.L.C.-G. (09/09276-3); Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) to S.N.A.; and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) to M.L.C.-G.
