Abstract
Objective:
To investigate the association between plasma Dipeptidyl peptidase-4 (DPP4) activities and diabetic nephropathy in type 2 diabetes.
Research design and methods:
A total of 1193 newly diagnosed type 2 diabetic subjects were studied. Plasma DPP4 activity, mannose 6-phosphate receptor, inflammatory markers and oxidative stress parameters were measured in all participants. Diabetic nephropathy was defined as the presence of albuminuria or an estimated glomerular filtration rate < 60 mL/min/1.73 m2.
Results:
Participants in the highest quartile of DPP4 activity had higher HbA1c, homeostatic model assessment of insulin resistance, nitrotyrosine, 8-iso-PGF2a, interleukin-6, high-sensitivity C-reactive protein, mannose 6-phosphate receptor, urinary albumin-to-creatinine ratio and lower estimated glomerular filtration rate compared with participants in the lowest quartile (all p < 0.001). DPP4 activities were associated positively with HbA1c, homeostatic model assessment of insulin resistance, nitrotyrosine, 8-iso-PGF2a, interleukin-6, high-sensitivity C-reactive protein, mannose 6-phosphate receptor, urinary albumin-to-creatinine ratio and negatively with estimated glomerular filtration rate (all p < 0.001). In the highest DPP4 quartile, diabetic nephropathy risk was significantly higher (odds ratio: 3.77; 95% confidence interval: 2.34–6.07) than in the lowest quartile after adjustment for potential confounders. This association remained strong (2.85; 1.74–4.68) after further controlling for HbA1c, homeostatic model assessment of insulin resistance, nitrotyrosine and high-sensitivity C-reactive protein.
Conclusion:
This study shows that increased DPP4 activities are strongly and independently associated with diabetic nephropathy in type 2 diabetes. The associations between DPP4 and diabetic nephropathy, although strong, do not imply causality. There are however plausible mechanisms which could explain such a link.
Introduction
Diabetic nephropathy (DN) is a common complication of type 2 diabetes and the leading cause of end-stage renal disease (ESRD) worldwide. 1 In addition, DN is also a major risk factor for the development of macrovascular complications such as heart attacks and strokes. 2 Consequently, an understanding of the clinical characteristics and risk factors associated with DN is useful to establish effective diagnostic and therapeutic strategies to prevent DN progression and the onset of cardiovascular complications.
Dipeptidyl peptidase 4 (DPP4), is a serine protease found on the apical surface of diverse cells, with a catalytic activity that cleaves X-proline dipeptides from the N terminus of polypeptides such as chemokines and peptide hormones. In addition to its membrane form, DPP4 exists in plasma as a soluble form (sCD26), which is the extracellular domain of the molecule thought to be cleaved from the cell surface. 3 Recent studies have demonstrated that DPP4 is also a newly identified adipokine that has been shown to contribute to hyperglycaemia, insulin resistance, oxidative stress and inflammation,4–10 all of which have been suggested to be involved in the pathogenesis of DN. Furthermore, several studies 11 have shown that DPP4 inhibition treatment might provide a protective effect on microalbuminuria (MAU) via reduction of oxidative stress and inflammation in addition to glycaemia lowering in type 2 diabetic patients.
Therefore, it is reasonable to speculate that plasma DPP4 activity may be positively correlated with DN; however, no study has evaluated whether DPP4 may serve as a risk marker for DN and to what extent it is associated with DN. Consequently, we aimed to evaluate the association between plasma DPP4 activities and DN in a cross-sectional population study of 1193 Chinese patients with newly diagnosed type 2 diabetes.
Research design and methods
Study subjects
The study population consisted of males and females, aged 18–76 years, who participated in the Guangxi Diabetes and Metabolic Disorders study, a cohort study that aimed to evaluate the cross-sectional and prospective association between psychosocioeconomic factors, biomarkers and the incidence of type 2 diabetes, diabetic complications and metabolic disorder. The subjects were volunteers with newly diagnosed type 2 diabetes recruited from the medical examination centre of affiliated hospital of Guilin medical university in Guangxi province between 2013 and 2014. The study was approved by the Drugs/Medical Apparatus and Instruments Ethics Committee at Affiliated Hospital of Guilin Medical University, and consent has been obtained from each patient or subject after full explanation of the purpose and nature of all procedures used. This study was registered on the Chinese clinical trial registry (ChiCTR-EPC-14005273).
The final sample size for the present analysis was 1193 participants (691 men and 502 women) with newly diagnosed type 2 diabetes. The diagnostic criteria of type 2 diabetes were based on the criteria recommended by the World Health Organization. 12 Inclusion criteria: (1) newly diagnosed type 2 diabetes, age between 18 and 76 years old; (2) long-term residing (⩾5 years) in China’s Guangxi province; (3) being able to give informed consent. Exclusion criteria: (1) patients with prediabetes diagnosed by an oral glucose tolerance test (OGTT), gestational diabetes, type 1 diabetes or diabetes induced by steroid use or other endocrinological diseases; (2) using varieties of drugs to control blood glucose, blood pressure, blood lipid and other drugs used in preventing the natural process of DN; (3) subjects deprived of personal safety and presence of any of the diseases including nonalcoholic fatty liver disease (NAFLD), pancreatic cancer, acute inflammatory diseases, stroke, myocardial infarction, other heart, liver and respiratory dysfunction were excluded as progression of these in any stage may hinder our study; (4) subjects with malignancy and pregnant subjects; (5) subjects who were on dialysis or had received a kidney transplant; (6) subjects with incomplete data (study population data which were not completely determined mainly refer to the study population who failed to complete the questionnaire or the absence of any physical indicators).
DN definitions
DN was defined as the presence of albuminuria (micro or macro) or an estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2. 13 According to the 2013 Kidney Disease: Improving Global Outcomes guidelines, 14 the estimation of GFR was considered to be more accurate using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation compared with the Modification of Diet in Renal Disease (MDRD) formula, particularly for values >60 mL/min/1.73 m2. More importantly, the CKD-EPI equation has been validated in Chinese CKD patients, 15 and we therefore calculated eGFR using the CKD-EPI equation. The CKD-EPI equation was expressed as GFR = 141 × min(SCr/k,1)α × max(SCr/k,1)−1.209 × 0.993age × 1.018 (if female), where k is 0.7 for females and 0.9 for male, α is −0.329 for females and −0.411 for males – min indicates the minimum of SCr/k or 1, and max indicates the maximum of SCr/k or 1. 16 Albuminuria was defined by a urine albumin-to-creatinine ratio (ACR) of 30 mg/g creatinine or above on spot urine analysis in at least two samples on two separate occasions within 6 months. 17
Clinical measurements
A standard questionnaire was administered by trained staff to the participants to record demographic characteristics and lifestyle risk factors. 18 Measurements of body weight and height, waist and hip circumference, body mass index (BMI), waist/hip ratio (WHR) and blood pressure have been described previously. 19 Subjects were instructed to maintain their usual physical activity and diet for at least 3 days before undergoing an OGTT. After an overnight fast of ⩾10 h, venous blood samples were collected to measure HbA1c, fasting plasma glucose (FPG), fasting insulin, blood lipids [including total cholesterol (TC), triglycerides (TG), low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C)], interleukin-6 (IL-6), high-sensitivity C-reactive protein (hs-CRP), mannose 6-phosphate receptor (M6P-R), nitrotyrosine, 8-iso-PGF2a and DPP4 activity. Blood samples were also drawn at 30 and 120 min after a 75 g glucose load to measure glucose and insulin concentrations.
Laboratory measurements
Plasma glucose levels, insulin, TC, TG, HDL-C, LDL-C, IL-6, hs-CRP, M6P-R, nitrotyrosine, 8-iso-PGF2a and urine ACR were measured as previously described.5,7,8 Plasma DPP4 activity was determined as the rate of cleavage of 7-amino-4-methylcoumarin (AMC) from the synthetic substrate H-glycyl-prolyl-AMC (H-Gly-Pro-AMC; Biovision, San Francisco, CA, USA). DPP4 activity is expressed as the amount of cleaved AMC per minute per mL (nmol/min/mL). DPP4 activity was measured in the absence or the presence of sitagliptin, a specific DPP4 inhibitor, to test the specificity of the enzymatic assay. In our samples, sitagliptin inhibited the assayed DPP4 activity by >95%. The intra- and inter-assay coefficients of variation were 4.58% and 9.85%, respectively. Blood samples were stored at −80°C and subsequently all parameters were measured within 6 months of sample collection. All samples were analysed in duplicate, random order, blinded to the clinical status of the participants. The homeostasis model assessment of insulin resistance (HOMA-IR) was calculated as previously reported. 5
Statistical analysis
All of the statistical analyses were performed using the SPSS 16.0 software (SPSS Inc., Chicago, IL). Normally distributed data were expressed as means ± standard deviation (SD), whereas variables with a skewed distribution were reported as median (interquartile range) and log transformed to approximate normality before analysis. Categorical variables were represented by frequency and percentage. We divided the study population into quartiles of plasma DPP4 activity with cut-off points of 17.71, 24.28 and 30.64. Clinical and biochemical characteristics were compared by an analysis of covariance, χ2 or t-test. Associations between continuous variables were tested by partial correlation analyses. Multivariate logistic regression models were used to estimate the odds ratios (ORs) for elevated HOMA-IR, nitrotyrosine, 8-iso-PGF2a, hs-CRP, IL-6 and DN. Owing to a lack of current global guidelines regarding the normal range of HOMA-IR, nitrotyrosine, 8-iso-PGF2a, hs-CRP and IL-6, the upper quartiles of these parameters were defined as elevated. Potential confounding variables including age, gender, smoking, alcohol consumption, leisure-time physical activity, family history of diabetes, BMI, systolic blood pressure (SBP), TG, HDL-C, HbA1c, HOMA-IR, nitrotyrosine and hs-CRP were controlled in the regression models. The variables included in the multivariate regression models were those that were statistically significant in univariate analyses or were biologically relevant. In all statistical tests, two-sided p values < 0.05 were considered significant.
Results
Clinical and laboratory characteristics
Among the 1193 participants included in this study, 272 participants (22.80%) had DN. The prevalence of DN according to DPP4 quartiles was 12.4%, 14.4%, 26.1% and 38.4%, respectively. The subjects with higher DPP4 activity were more likely to be older, current smokers and to have a lower leisure-time physical activity level (all p < 0.05). With respect to metabolic parameters, the subjects in the higher DPP4 quartiles exhibited higher levels of BMI, WHR, SBP, diastolic blood pressure (DBP), TG, TC, LDL-C, FPG, 2-hour plasma glucose (2h-PG), fasting insulin, 2h-insulin, HOMA-IR, HbA1c, nitrotyrosine, 8-iso-PGF2a, IL-6, hs-CRP, M6P-R, urinary ACR and lower eGFR compared with subjects in the lowest quartile (all p < 0.01). Gender, alcohol consumption, family history of diabetes and HDL-C did not differ significantly across DPP4 activity categories (Table 1). Compared with subjects without DN, the subjects with DN had higher levels of HOMA-IR, HbA1c, nitrotyrosine, 8-iso-PGF2a, IL-6, hs-CRP, M6P-R, urinary ACR, DPP4 activity and lower eGFR (all p < 0.001) (Supplemental Table 1).
Characteristics of study participants according to DPP4 quartiles.
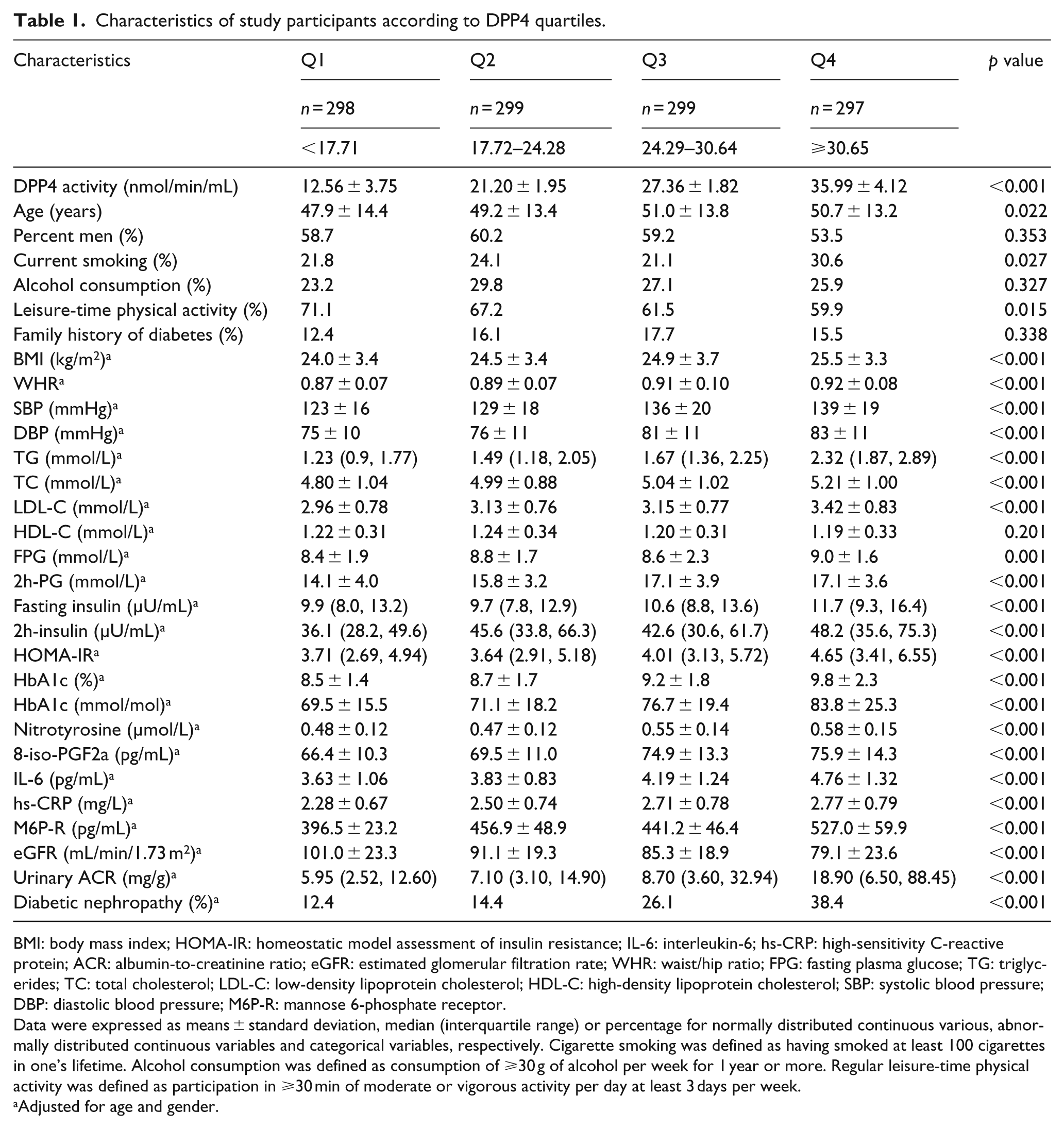
BMI: body mass index; HOMA-IR: homeostatic model assessment of insulin resistance; IL-6: interleukin-6; hs-CRP: high-sensitivity C-reactive protein; ACR: albumin-to-creatinine ratio; eGFR: estimated glomerular filtration rate; WHR: waist/hip ratio; FPG: fasting plasma glucose; TG: triglycerides; TC: total cholesterol; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; SBP: systolic blood pressure; DBP: diastolic blood pressure; M6P-R: mannose 6-phosphate receptor.
Data were expressed as means ± standard deviation, median (interquartile range) or percentage for normally distributed continuous various, abnormally distributed continuous variables and categorical variables, respectively. Cigarette smoking was defined as having smoked at least 100 cigarettes in one’s lifetime. Alcohol consumption was defined as consumption of ⩾30 g of alcohol per week for 1 year or more. Regular leisure-time physical activity was defined as participation in ⩾30 min of moderate or vigorous activity per day at least 3 days per week.
Adjusted for age and gender.
Correlation analysis between DPP4 activity and other variables
Partial correlation analysis demonstrated that DPP4 activities was positively associated with BMI, WHR, SBP, DBP, TG, TC, LDL-C, FPG, 2h-PG, fasting insulin, 2h-insulin, HOMA-IR, HbA1c, nitrotyrosine, 8-iso-PGF2a, IL-6, hs-CRP, M6P-R, urinary ACR and was negatively associated with eGFR in DN participants, non-DN participants and all participants after adjustment for age, gender, current smoking, alcohol consumption, leisure-time physical activity and family history of diabetes (all p < 0.001) (Table 2). Remarkably, the relationships (r value) between DPP4 activity and HbA1c increased gradually with increasing quartiles of HbA1c. According to HbA1c quartiles, the r values between DPP4 activity and HbA1c were 0.118, 0.156, 0.276 and 0.338, respectively (all p < 0.05) (Supplemental Table 2).
Partial correlation coefficients among DPP4 activities, anthropometric indicators and metabolic features a .
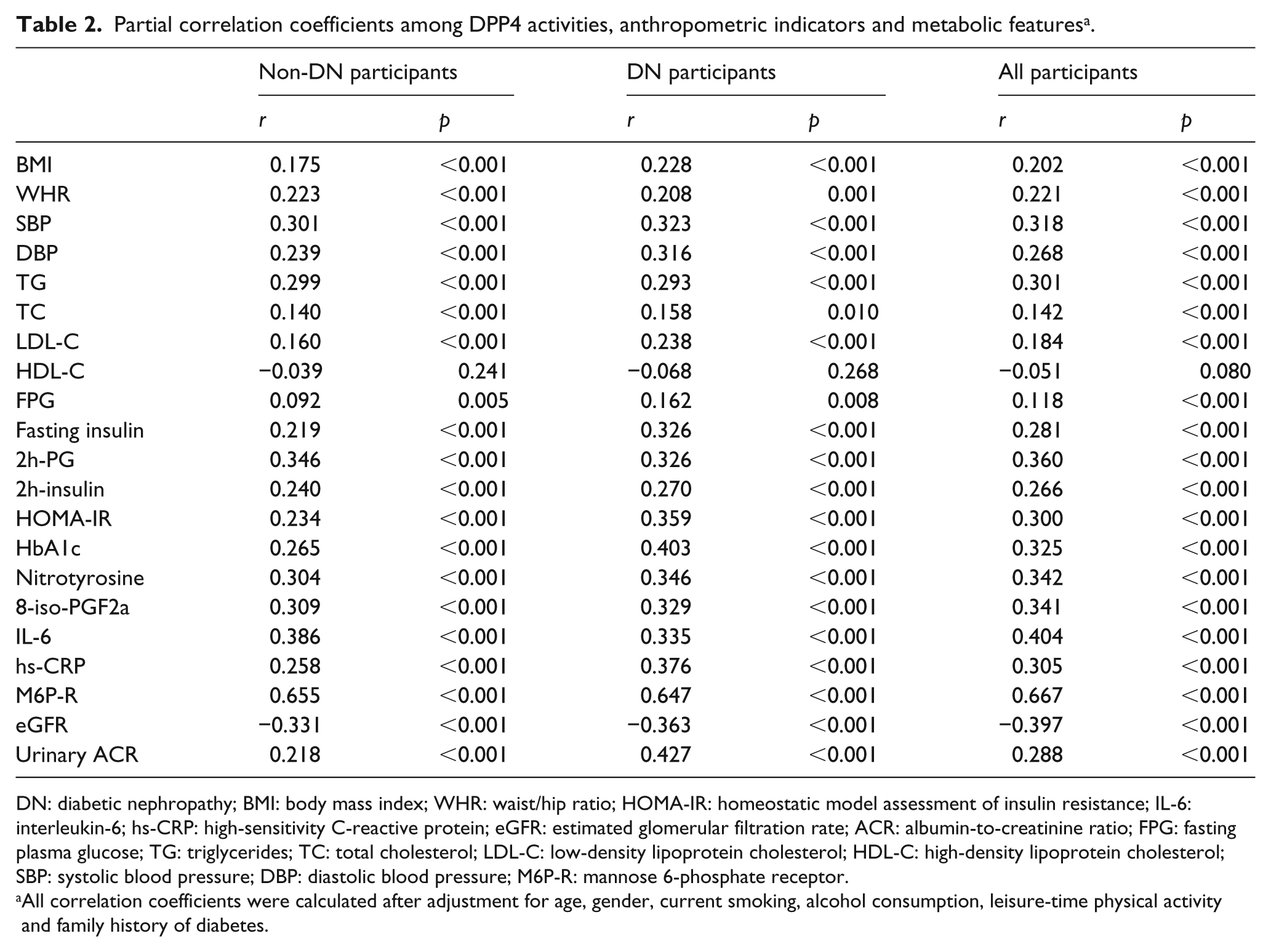
DN: diabetic nephropathy; BMI: body mass index; WHR: waist/hip ratio; HOMA-IR: homeostatic model assessment of insulin resistance; IL-6: interleukin-6; hs-CRP: high-sensitivity C-reactive protein; eGFR: estimated glomerular filtration rate; ACR: albumin-to-creatinine ratio; FPG: fasting plasma glucose; TG: triglycerides; TC: total cholesterol; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; SBP: systolic blood pressure; DBP: diastolic blood pressure; M6P-R: mannose 6-phosphate receptor.
All correlation coefficients were calculated after adjustment for age, gender, current smoking, alcohol consumption, leisure-time physical activity and family history of diabetes.
Associations between DPP4 activity and DN
As presented in Table 3, the ORs for increased oxidative stress, inflammation, insulin resistance and DN were higher with increasing DPP4 quartiles (p < 0.001 for trend). In the highest DPP4 quartiles, the ORs were 2.08 [95% confidence interval (CI) 1.34–3.22] for elevated HOMA-IR, 2.82 (1.80, 4.40) for elevated nitrotyrosine, 4.45 (2.80, 7.09) for elevated 8-iso-PGF2a, 2.02 (1.32, 3.10) for elevated hs-CRP, 2.86 (1.87, 4.37) for elevated IL-6 and 3.77 (2.34, 6.07) for DN after adjusting for age, gender, current smoking, alcohol consumption, leisure-time physical activity, family history of diabetes, BMI, SBP, TG, HDL-C and HbA1c (model 2). Interestingly, further adjustment for HOMA-IR, nitrotyrosine and hs-CRP (model 5) only reduced the magnitude of the ORs for DN.
Adjusted ORs and 95% CIs for diabetic nephrology and high levels of oxidative stress, inflammation and insulin resistance according to DPP4 quartiles.
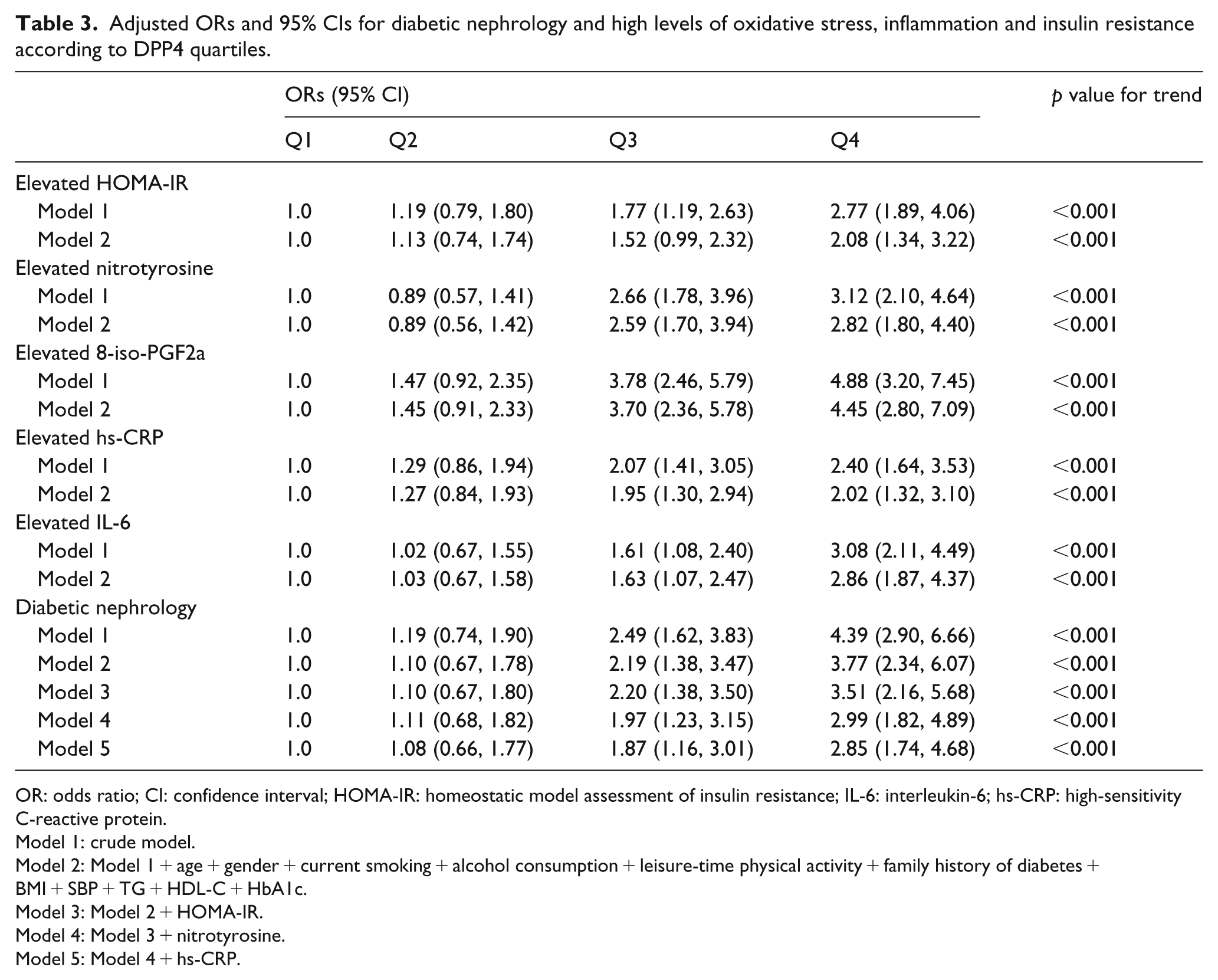
OR: odds ratio; CI: confidence interval; HOMA-IR: homeostatic model assessment of insulin resistance; IL-6: interleukin-6; hs-CRP: high-sensitivity C-reactive protein.
Model 1: crude model.
Model 2: Model 1 + age + gender + current smoking + alcohol consumption + leisure-time physical activity + family history of diabetes + BMI + SBP + TG + HDL-C + HbA1c.
Model 3: Model 2 + HOMA-IR.
Model 4: Model 3 + nitrotyrosine.
Model 5: Model 4 + hs-CRP.
As shown in Figure 1, the risk for DN increased markedly with rising levels of DPP4, higher levels of HOMA-IR [Figure 1(a)], higher levels of nitrotyrosine [Figure 1(b)], higher levels of 8-iso-PGF2a [Figure 1(c)], higher levels of hs-CRP [Figure 1(d)] and higher levels of IL-6 [Figure 1(e)]. Moreover, even in the lowest quartile of HOMA-IR, nitrotyrosine, 8-iso-PGF2a, hs-CRP and IL-6, the risks for DN were one- to five-fold higher in the highest DPP4 quartile than in the lowest quartile.
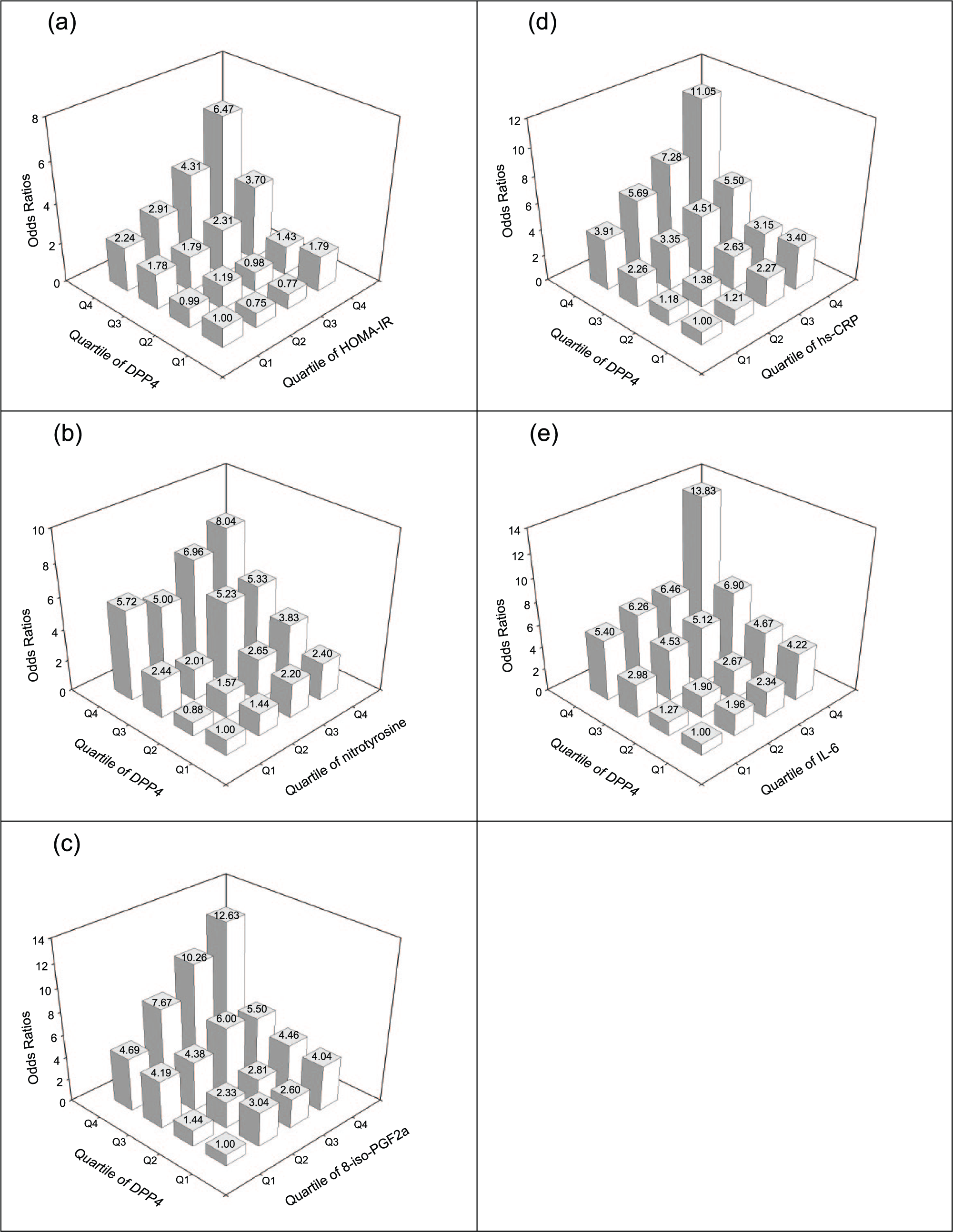
Adjusted ORs for diabetic nephrology according to the quartiles of (a) DPP4 and HOMA-IR, (b) DPP4 and nitrotyrosine, (c) DPP4 and 8-iso-PGF2a, (d) DPP4 and hs-CRP and (e) DPP4 and IL-6. Adjusted for age, gender, current smoking, alcohol consumption, leisure-time physical activity and family history of diabetes.
Discussion
In this study, we found a strong association between DPP4 activity and the risk of DN and its pathogenic factors such as hyperglycaemia, insulin resistance, oxidative stress and inflammation in type 2 diabetes in a large-scale cross-sectional study. Moreover, the association between increased plasma DPP4 activities and DN in type 2 diabetes is independent of lifestyle factors, family history of diabetes, BMI, SBP, blood lipids and, remarkably, blood glucose, insulin resistance, oxidative stress and inflammation. Although we cannot evaluate the causal relationship between DPP4 activity and DN, we still tried to explain this association by asking whether the elevation in DPP4 activity leads to an increased risk of DN or vice versa.
The chronic elevation in blood sugar, termed ‘hyperglycaemia’, is considered as the master switch for promoting the development of DN, the most effective way to reduce the risk for DN in type 2 diabetes is to achieve optimal glycaemic control with the goal of reaching normoglycaemia as early as possible in the course of the disease. 20 The mutual effect between DPP4 and hyperglycaemia has been investigated in previous studies, Ryskjær et al. 21 found a significant positive correlation between plasma DPP4 activity and HbA1c levels in type 2 diabetes; moreover, research done by Mannucci et al. 22 indicated that chronic hyperglycaemia induced a significant increase in DPP4 activity in type 2 diabetes. Interestingly, recent studies4,6,23 further revealed that DPP4 was a newly identified adipokine and its activity was found to be elevated even in normal glucose tolerance (NGT) subjects with increasing levels of BMI. Similarly, our previous prospective study 5 showed that DPP4 activity was also higher in subjects with NGT who developed prediabetes or type 2 diabetes 4 years later than in subjects with NGT who remained normoglycaemic. Although a significant relationship between DPP4 activity and HbA1c was not found in both groups at baseline, Cox proportional hazards models revealed that increased DPP4 activity was an important predictor of the onset of hyperglycaemia in apparently NGT Chinese individuals. In this cross-sectional study, our data further indicated that (1) newly diagnosed diabetic subjects in the higher DPP4 quartiles exhibited higher levels of HbA1c; (2) DPP4 activity and HbA1c were positively correlated in type 2 diabetes and this correlation increased gradually with increasing quartiles of HbA1c. Based on these findings in our study and others, we extrapolate that the elevation in circulating DPP4 activity could occur at a very early stage well before the deterioration of glucose homeostasis and promote the development of hyperglycaemia; when the deterioration of glucose homeostasis reaches a certain degree, hyperglycaemia might in turn results in an further increase in DPP4 activity. Increased DPP4 activity might play a causal role in this possible vicious circle.
Considering the possible vicious circle between DPP4 activity and hyperglycaemia, DPP4-induced hyperglycaemia might, at least in part, contribute to DN development. Interestingly, another prospective study done by us showed that increased DPP4 activity was also a good candidate for predicting new-onset MAU in Chinese with NGT; furthermore, although high levels of HbA1c were closely associated with an increased risk of DN, increased DPP4 activity per se was an independent risk factor for the DN, even within the lowest HbA1c quartile. Therefore, it is possible that DPP4 may promote the DN risk through a pathway not fully overlapping with hyperglycaemia.
Several cross-sectional and prospective studies have shown that there is a positive relationship between DPP4 activity and insulin resistance in both diabetic and nondiabetic subjects; some data suggest that DPP4 may impair insulin sensitivity in an autocrine and paracrine fashion.4–6 Consistent with these publications, we also found that increased DPP4 activities were positively associated with insulin resistance assessed by HOMA-IR in type 2 diabetes. More importantly, some studies demonstrated that insulin resistance induced deficient insulin signalling was identified not only in conventional insulin-target tissues such as skeletal muscle and liver but also at sites of DN, 20 and a deficiency in insulin receptor signalling in podocytes of the kidney can induce a disease state reminiscent of DN even in the setting of normoglycaemia. 24 Since DPP4 may be a pathogenic factor for insulin resistance,4,5 it may also promote DN development by exerting some influence on the deficiency in insulin receptor signalling in podocytes of the kidney; however, because of the nature of our study, this speculation remains to be clarified by further clinical and basic studies. In addition, despite the fact that insulin resistance has been considered a contributor to DN, here, we found that the ORs for DN according to DPP4 quartiles were not substantially attenuated by additional adjustment for HOMA-IR; even within the lowest HOMA-IR quartile, the risks for DN were 2.24 fold higher in the highest DPP4 quartile than in the lowest quartile, suggesting that an elevated DPP4 activity may enhance the DN development via mechanisms other than insulin resistance.
Recent studies 10 have shown that DPP4 dose dependently increased reactive oxygen species generation in endothelial cells, the mechanism of which appears to be related, at least in part, to endothelial cell surface-anchored M6P-R. Research by Jeyaratnaganthan et al. 25 further indicated that serum M6P-R originating from cleavage of the membrane-associated receptor was increased in type 2 diabetic patients. In this study, we found that diabetic subjects in higher quartiles of DPP4 activity or with DN had higher levels of oxidative stress and M6P-R than diabetic subjects in the lower quartiles of DPP4 activity or without DN; a positive and independent relationship was also found between oxidative stress, M6P-R and DPP4 activity in type 2 diabetes; in addition, the ORs for elevated oxidative stress were higher with increasing DPP4 quartiles. These results of our study are consistent with previous researches mentioned above. Since clinical and basic studies have both proved the critical role played by DPP4 in oxidative stress, we speculate that DPP4-induced oxidative stress might, at least in part, promote DN development via its interaction with M6P-R.
Numerous studies had provided evidence of an important role for chronic low-grade inflammation in the development of diabetic complications such as DN. 20 Moreover, the proinflammatory effects of DPP4 had been well addressed by clinical and basic researches.9,26–28 Similarly, in this study, we found an increase in inflammatory markers such as IL-6 and hs-CRP in subjects with higher quartiles of DPP4 activity or with DN; a positive correlation between DPP4 activity and inflammation was also found in type 2 diabetic subjects; in addition, the ORs for increased inflammation were higher with increasing DPP4 quartiles. Together, the association between DPP4 activity and inflammatory markers may provide new insights into the potential involvement of proinflammatory effect of DPP4 in the pathogenesis of DN.
Oxidative stress and chronic low-grade inflammation are well-documented risk factors for DN, and DPP4 activities have been reported to be highly correlated with oxidative stress parameters and inflammatory markers, 20 which was confirmed in our study; more importantly, several studies 11 have shown that DPP4 inhibition treatment might provide a protective effect on MAU via reduction of oxidative stress and inflammation in addition to glycaemia lowering in type 2 diabetic patients. However, further adjustment for nitrotyrosine and hs-CRP yielded only a reduction of the DN risk across the DPP4 quartiles. Thus, increased DPP4 activities in the individuals with high DN risk may not be merely a consequence of enhanced oxidative stress and inflammation. Considering the multiple functions of DPP4 activity and its high expression in the kidney proximal tubular cells and endothelial cells,3,29 the mutual effects between DPP4 activities and other unknown factors might also exert some influence on the development of DN.
Since our study is cross-sectional, we cannot draw a causal conclusion that increased DPP4 activities promote the development of DN. The parallel increase in DPP4 activity and DN risk could also be interpreted in an opposite way. Impaired renal function with the accumulation of proinflammatory toxins could promote leukocyte activation and result in an increased DPP4 expression with higher activity. In addition, as DN deteriorated, increased DPP4 activity might also occur as a result of reduced DPP4 clearance and decreased uptake and/or turnover by the cells of the reticular endothelial system. 30 Previous study 30 reported that DPP4 expression and soluble DPP4 concentration were significantly higher in subjects with end-stage kidney disease than in control subjects, and the DPP4 enzyme activity in these patients was also found to be higher. More importantly, DPP4 concentrations and its activities were found to be significantly decreased after kidney transplantation. Taken together, these findings suggest the possibility that increased DPP4 activities might partly be the result of DN.
To our knowledge, this is the first cross-sectional study to evaluate the relationship between DPP4 activities and DN in newly diagnosed type 2 diabetes. Most potential confounders were carefully controlled, which limited the possibility of residual confounding effects. However, whether increased DPP4 activities actually predict DN development in type 2 diabetes cannot be determined through the present study. Nevertheless, given the close association between DPP4 activities and risk factors for DN such as hyperglycaemia, insulin resistance, oxidative stress and inflammation, together with the findings that increased DPP4 activities are strongly and independently associated with DN after adjustment for related risk factors, it is plausible to consider DPP4 as a promising candidate for DN risk assessment. In addition, if the speculation that increased DPP4 activities promote the development of DN can be proved by further studies, DPP4 activity might also be able to serve as a potential therapeutic target for the prevention and treatment of DN at clinical practice. Further researches are still needed in this regard.
Some limitations of our study should also be considered. First, this study is an epidemiological cross-sectional study and somehow it fails to address the causal role of DPP4 in the pathogenesis of DN which is needed to be elucidated by further prospective and basic investigation. Second, some other risk factors related to DPP4 activity such as glucagon-like peptide-1 (GLP-1) and advanced glycation end products (AGEs),31,10 which could also promote the development of DN as well, were not evaluated in this study. Finally, the lack of a control group without diabetes is an important weakness of the study; although most common confounders have been adjusted in our analysis, additional unmeasured confounders in type 2 diabetes might also have had some impact on the independent relationship between DPP4 activity and DN.
In conclusion, we found that increased DPP4 activities are strongly and independently associated with DN in type 2 diabetes. Although longitudinal studies are needed, our findings might suggest of potential role of DPP4 in the pathogenesis of DN and in the prevention and management of this complication.
Footnotes
Acknowledgements
The authors are indebted to all subjects for agreeing to participate in this study. Trial Registration Number: ChiCTR-EPC-14005273.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This study was supported by grants from the scientific research and technology development projects of Science and Technology Department of Guangxi Province [Grant No.1598012-13], National Natural Science Foundation of China [Grant No.81560144/H0713], Natural Science Foundation of Guangxi Province [Grant No. 2015GXNSFBA139119], and Health and Family Planning Commission of Guangxi Province [Z2015415].
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
