Abstract
Aims
Rheopheresis is an extracorporeal haematotherapy that improves haemorheological status by filtering proteins that enhance blood viscosity. It also has anti-inflammatory effects by removing inflammatory cytokines. Our study aims to examine the effects of rheopheresis on the endothelial status in diabetic lower extremity ulceration.
Methods
In vitro experiments were performed in a HUVEC model to mimic hyperglycaemic stress. We determined the changes in gene expression levels of IL-6, IL-8, TNF-alpha, endothelin convertase enzyme, ET-1, and NO synthase, as well as the ROS and intracellular GSH levels upon hyperglycaemia. In in vivo studies, two rheopheresis procedures were performed on seven patients with diabetic lower extremity ulceration with hyperviscosity, and we measured the changes in plasma concentrations of ET-1, TXB2, SOD enzyme activity, and extracellular components of the glutathione pool depending on treatments.
Results
Our results showed that hyperglycaemia increases endothelial expression of inflammatory cytokines, ET-1, and endothelin convertase enzyme, while NO synthase was decreased. As a result of rheopheresis, we observed decreased ET-1 and TXB2 concentrations in the plasma and beneficial changes in the parameters of the glutathione pool.
Conclusion
To summarize our results, hyperglycaemia-induced oxidative stress and endothelial inflammation can be moderated by rheopheresis in diabetic lower extremity ulceration with hyperviscosity.
Introduction
Rheopheresis is an extracorporeal, selective double cascade membrane filtration procedure. It is one of the five currently available haemorheotherapies in addition to specific immunoadsorption, heparin-induced extracorporeal lipoprotein (LDL-) precipitation (HELP), dextran sulfate adsorption, and direct lipoprotein adsorption (DALI). 1 After securing extracorporeal treatments with continuous blood volume monitoring, membrane differential filtration, which is practically synonymous with double filtration plasmapheresis, was introduced by Agishi et al. in the 1980s. 2 Since the 1950s, the explicit development of various filtration technologies has allowed extracorporeal procedures to become increasingly specific and selective, thus ensuring the broad applicability of apheresis therapies. 3 The rheofilter used in rheopheresis can remove plasma components that increase blood viscosity and have a molecular weight greater than 250–300 kDa. The MONET filter (Membrane filtration Optimized Novel Extracorporeal Treatment, Fresenius Medical Care AG & Co. KgaA) made of polysulfone can normalize the haemorheological status by removing LDL, Lp(a), triglyceride, cholesterol, fibrinogen, α2 macroglobulin, vWF, and IgM, thereby improving microcirculation. Based on data from the literature, rheopheresis alone can reduce LDL levels by 70%, total cholesterol levels by approximately 50%, triglyceride levels by an average of 35–40%, and fibrinogen levels by 50%. It is also an advantage of the treatment that rheopheresis only slightly changes the level of total protein, HDL, which is protective against atherosclerosis, and albumin, which is responsible for maintaining oncotic pressure. In addition, rheopheresis reduces platelet aggregation and improves erythrocyte deformability and endothelial status.1,4,5 As recommended by the American Society for Apheresis, indications for treatment include age-related macular degeneration and sensorineural hearing loss. 6 According to the international recommendation, the first treatments were performed successfully in dry macular degeneration. Convinced of the benefits of modality, we started to use the treatment for diabetic foot syndrome based on the logical conclusion that rheopheresis can be used successfully in other microcirculatory diseases with hyperviscosity. Since 2014, we have treated 18 patients with rheopheresis who suffered from diabetic lower extremity ulcers and/or polyneuropathy associated with microcirculatory damage. Twelve patients had lower extremity ulcers, 10 had lower extremity neuropathy, and 4 had both conditions present. Patients received an average of 3 treatments. Our clinical results have shown that rheopheresis treatment effectively reduces whole blood (by an average of 24.78%) and plasma viscosity (by an average of 21.53%), thereby normalizing haemorheological status. In addition, we observed subjective improvements in neuropathic complaints supported by electroneurography (ENG) studies, and in all ulcer patients, previously unhealed ulcers had healed. 7 Based on these findings, the clinical results went far beyond the normalization of haemorheological parameters. To clarify this response, we began to investigate the effects of rheopheresis treatment on the plasma concentrations of diabetes-induced proinflammatory cytokines (IL-6, IL-8, and TNF-alpha). The results of our previous studies in 6 patients showed that IL-6 levels decreased by an average of 38.69%, IL-8 levels by 61.76%, and TNF-alpha plasma concentrations by 78.56% after two rheopheresis treatments. 8 Due to the novelty of rheopheresis, there is very little literature information available on treatment. Therefore, our further studies aimed to identify and explore the biochemical processes through which rheopheresis treatment can improve endothelial status by filtering inflammatory cytokines and viscosity-enhancing macromolecules, thus facilitating the treatment of complications associated with diabetic foot syndrome. The importance of our detailed research is justified by the fact that the symptoms of diabetic foot syndrome are extremely challenging to treat, and treatment options are limited. Neuropathy and ulcers in the lower extremity often lead to amputation. In addition, the number of patients with diabetes is growing year by year, so it is essential that effective and explored treatment alternatives appear in the therapy of complications. 9 To this end, depending on rheopheresis, we examined the changes in serum levels of vasoactive endothelin-1 and the changes in plasma concentrations of thromboxane-B2, which indicate thrombotic processes, as well as the changes in SOD enzyme activity, and we also performed a complex analysis of the changes in the extracellular components of the glutathione pool. In parallel with the treatments, we studied the cell damage caused by hyperglycaemia in an in vitro model system of endothelial cells (HUVECs) isolated from human umbilical cord vein. The effects of rheopheresis treatment on endothelial inflammation have not been previously studied. Our experiments in the HUVEC model aimed to determine the biochemical parameters that objectively describe endothelial inflammation by simulating inflammatory-inducing intravascular hyperglycemia. Our experiments in the HUVEC model are justified by the fact that in vivo endothelial changes after rheopheresis could not be studied cellularly. Therefore, the treatment effects can only be inferred from the changes in the levels of biomarkers in the blood plasma. Using an in vitro experiment, hyperglycemia-induced inflammation, consequent deterioration of antioxidant status, and release of pathological vasoactive substances were validated in a single, independent model.
Materials and methods
In vitro investigations
Cell culture conditions
Modelling of pathological endothelial lesions caused by diabetes mellitus was performed on HUVEC cultures with ethical approval (reference number: RKEB/IKEB 3712-2012). Human umbilical cord vein endothelial cells (HUVECs) were isolated from human umbilical cords. The umbilical vein was incubated at 37 °C for 15 min after loading with collagenase solution. After enzymatic digestion, the cell suspension was centrifuged, and after aspiration of the supernatant, the cells were resuspended in M199 medium and plated in cell culture flasks. When adequate confluence was achieved, phenotyping was performed by flow cytometric analysis to confirm that endothelial cells were propagated during cell culture. For identification, endothelial cells were labelled with fluorophore-conjugated mouse monoclonal antibodies (FITC-CD31 (+), PE-CD54 (+), APC-CD106 (-), PerCP-Cy5.5-CD45 (-)). Based on flow cytometry, approximately 97% of the cells expressed positive markers and did not express negative markers, demonstrating isolation efficiency and accuracy and providing standardized conditions for further in vitro studies. The cells were then treated with a 30 mM glucose solution for 24 h to simulate intravascular hyperglycaemia in patients with diabetes.
Gene expression studies in the HUVEC model (IL-6, IL-8, TNF-alpha, endothelin convertase enzyme, ET-1, and NO synthase)
Gene expression assays were performed by qRT–PCR analysis. One microgram of total RNA was reverse-transcribed into cDNA using a LunaScript RT SuperMix kit (PCR Biosystems, London, UK). The number of cycles required for the detection threshold was determined by the fluorescence of the reporter molecule cleaved by TaqMan polymerase (Luna Universal Probe qPCR Master Mix - PCR Biosystems, London, UK). As an internal control, glyceraldehyde 3-phosphate dehydrogenase was used. The results were expressed relative to 100% of the control group.
ROS study in the HUVEC model
A reactive oxygen radical study was performed with 2′,7′-dichlorofluorescein diacetate (DCDFA) staining (Sigma–Aldrich, St Louis, MO, USA). The cells were incubated with 100 μM DCDFA for 60 min to label the intracellular reactive oxygen species, and then the fluorescence intensity was measured using a Clariostar microplate reader (BMG Labtech, Ortenberg, Germany) (excitation = 485 nm; emission = 530 nm). The results were compared to an untreated control group.
Investigation of the antioxidant status in the HUVEC model
The reduced glutathione concentration in endothelial cells was determined using the Glutathione Assay Kit from Cayman Chemical (Ann Arbor, Michigan, USA). Endothelial cells were treated with 30 mM glucose solution, and after 0, 6, 12, 24, 48, and 96 h, the cells were deproteinized to measure the concentration of reduced glutathione in the cells compared to the control group treated with a normoglycaemic solution. The absorbance spectrum of the sample was examined at 414 nm by spectrophotometry (Clariostar microplate reader). The results were expressed relative to 100% of the control group.
Investigations of the effects of rheopheresis treatment
Rheopheresis treatment and patients
Rheopheresis treatments were performed with ethical approval (IVU/10026-4/2020/EKU, Hungarian Medical Research Council). Eligibility criteria for treatment included type II diabetes mellitus, high whole blood and/or plasma viscosity, lower extremity ulcer without macroangiopathy requiring revascularization, and/or ENG-confirmed diabetic polyneuropathy. A detailed angiological examination was performed during patient selection (Duplex ultrasound, angiography if necessary) to exclude all patients with a diagnosis of macroangiopathy requiring intervention. During the complex angiological examination, the superficial venous system was also reviewed by performing a Duplex ultrasound. Despite the atypical locations, diabetes was presumed to be the primary etiological factor in the background of leg ulcers when saphenous vein reflux, superficial varicose veins, and inadequately functioning perforator veins were not detectable. However, dilatation at the level of postcapillary venules and superficial veins below the detection threshold cannot be excluded by ultrasound, so the possibility of initial stage of superficial varicosity as an additional etiological factor cannot be ruled out. The immunological background of ulcers is refuted by the fact that the patients were negative for antibodies of antiphospholipid syndrome (Anti-beta-2-glycoprotein-I antibodies, anti-cardiolipin antibodies, Lupus anticoagulant). Cryglobulin-positivity and anti-neutrophil cytoplasmic antibody positivity were also excluded. Polyneuropathy was diagnosed with ENG. 9031A006401 Keypoint Clinical system was used for ENG examinations. Patients' pain was recorded on Numerical Pain Rating Scale (0–10). Seven patients with diabetic foot syndrome were enrolled in the study. Due to the strict criteria, there were five males and two females. Six patients suffered from lower extremity diabetic ulcers. Smart wound dressings were used for conservative treatment of ulcers. Diabetic polyneuropathy occurred in 4 patients. The mean age of the patients was 63.3 years. After peripheral venous cannulation, preliminary plasma separation was performed with an Art Universal system. We used MONET for rheopheresis to filter 40 mL/kg of plasma per patient, with an average volume of 2500–3000 mL. 10 All filters were only used once according to the manufacturer’s instructions. Peripheral blood samples were taken from patients in sterile tubes containing K-3 EDTA before the first and after the second treatment. Blood samples were centrifuged at 3000 RPM at 4 °C for 15 min.
Effect of rheopheresis treatment on endothelin-1 plasma concentrations
The Abcam ab133030 Endothelin-1 ELISA Kit (Abcam PLC, Cambridge, UK) was used to examine endothelin-1 concentrations in plasma samples. After setting the measurement standard, a dilution series was established and washed five times. Next, 100 μL of endothelin-1 antibody was added to the samples and incubated for 30 min. The samples were then treated with 100 μL of TMB substrate solution and tested for absorbance at 450 nm after an additional 30 min of incubation.
Effect of rheopheresis treatment on thromboxane B2 plasma concentrations
The Abcam ab133022 Thromboxane B2 ELISA Kit (Abcam PLC, Cambridge, UK) was used to determine thromboxane B2 plasma concentrations. A measurement standard was set, a dilution series was made, and three washes were performed. After adding 5 μL of TXB2 Alkaline Phosphatase Conjugatum followed by 200 μL of pNpp substrate solution, the samples were incubated for 45 min at room temperature. After adding the STOP solution, the absorbance was examined at 405 nm.
Effect of rheopheresis treatment on the SOD enzyme activity of red blood cells
To measure the antioxidant capacity of the SOD enzyme, we used erythrocytes from patient blood samples. The enzyme activity was examined with a Photochem instrument using the ACW kit (Analytik Jena, Jena, Germany). Then, 1.5 mL of Reagent 1, 1 mL of Reagent 2, and 25 μL of Reagent 3 (photosensitizer) were added to 10 μL of red blood cells. SOD enzyme (Superoxide Dismutase, Sigma Aldrich, Germany) was used as a standard.
Effect of rheopheresis treatment on the changes in the plasma glutathione pool
Samples were prepared for the total thiol and disulfide measurements by the method of Fu et al. 11
Preparation of standard solutions
A standard mixture (concentration: 10 μg/mL for each compound) was prepared from the standards cysteine, homocysteine, cysteineylglycine, γ-glutamylcysteine, cystine, and glutathione (168149-25 mg, 69453-10 mg, C01666-25 mg, G0903-25 mg, C8630-1 g, and PHR1359-500 mg) from Merck Life Science Ltd. (Budapest, Hungary). N-Acetylcysteine (A15409.14) was obtained from VWR International Ltd. (Debrecen, Hungary). Briefly, 150 μL of standard mixture was combined with 300 μL of N-ethylmaleimide (NEM) solution (concentration: 100 μg/mL), 1020 μL of water and 30 μL of formic acid solution (0.01%, V/V). The reaction mixture was thermostated at 37 °C for 30 min. After cooling, standard solutions were prepared at the seven concentrations of 0.1, 1, 5, 10, 25, 50, and 100 ng/mL by dilution with water.
LC–MS analysis
The analyses were performed using the Dionex Ultimate 3000RS UHPLC system (Thermo Fisher, Waltham, MA, USA) coupled to a Thermo Q Exactive Orbitrap hybrid mass spectrometer equipped with an Acclaim Mixed-Mode HILIC-1 (2.1 × 150 mm, 3-μm particle size) analytical column. The flow rate was maintained at 0.3 mL/min. The column oven and postcolumn cooler temperatures were set to 25 °C ± 1 °C. Samples were thermostated at 25 °C ± 1 °C. The mobile phase consisted of water (A) and methanol (B) (both acidified with 0.1% formic acid). The gradient program was as follows: 0–1 min, 95% A; 1–6 min, →0% A; 6–10 min, 0% A; 10–10.5 min, →95% A; and 10.5–20 min, 95% A. The injection volume was 5 μL. The Thermo Q Exactive Orbitrap hybrid mass spectrometer (Thermo Fisher, Waltham, MA, USA) was equipped with a HESI source. The samples were measured in positive ion mode using the Selected Ion Monitoring (SIM) technique with the following inclusion list ([M + H]+): cysteine-NEM (C9H15N2O4S), 247.07525; N-acetylcysteine-NEM (C11H17N2O5S), 289.08582; homocysteine-NEM (C11H17N2O5S), 261.09090; cysteineylglycine-NEM (C11H18N3O5S), 304.09672; γ-glutamylcysteine-NEM (C14H22N3O7S), 376.11785; cystine (C6H13N2O4S2), 241.03167; glutathione-NEM (C16H25N4O8S), 433.13931; and glutathione disulfide (C20H33N6O12S2), 613.15979. The capillary temperature was set to 320 °C, and the spray voltage was 4.0 kV. The resolution was 35,000. The sheath gas flow rate and Aux gas flow rate were 32 AU and 7 AU, respectively. The differences between the measured and calculated monoisotopic molecular masses were less than 5 ppm in each case. The data were acquired and processed using Thermo Xcalibur 4.0 software (Thermo Fisher, Waltham, MA, USA).
Statistical analysis
Statistical analysis of results (in vitro results, ET-1 plasma concentrations, TXB2 plasma concentrations, SOD enzyme activity)
Each experiment was performed in triplicate. Data are expressed as the mean ± SEM of three individual experiments. Statistical Analysis for multiple comparisons, results were analyzed by ANOVA followed by a modified t-test for repeated measures according to Bonferroni’s method. p < .05 was considered statistically significant.
Statistical analysis of total thiol and disulfide studies
The Shapiro–Wilk test was used to determine whether the measured values were normally distributed. In cases of normal distribution, Welch’s t-test was used for statistical significance testing. Otherwise, the Kruskal–Wallis and Wilcoxon rank-sum nonparametric tests were applied. p values were corrected using the Benjamini–Hochberg method. The significance threshold was set to p < .05.
Results
Results of in vitro investigations
Results of gene expression investigations
Our results on the expression of inflammatory cytokines showed that incubation with a 30 mM glucose solution for 24 h significantly increased the expression of the TNF-alpha, IL-6, and IL-8 genes compared to expression in the control group. Hyperglycaemia also significantly increased the expression of the endothelin-1 and endothelin-converting enzyme genes. In contrast, NO synthase enzyme expression was significantly inhibited by stress caused by extremely high extracellular glucose concentrations (Figure 1). Changes in the expression of TNF-alpha (1./a) IL-6, (1./b) IL-8 (1./c), ET-1 (1./d), ECE (1./e), and NOS (1./f) genes in endothelial cells in a hyperglycaemia in an in vitro model system of endothelial cell model after treatment with 30 mM glucose for 24 h. Each experiment was performed in triplicate. Data are expressed as the mean ± SEM of three individual experiments. *, and *** mark significant (p < .05, .0005, respectively) differences compared with control (modified t-test).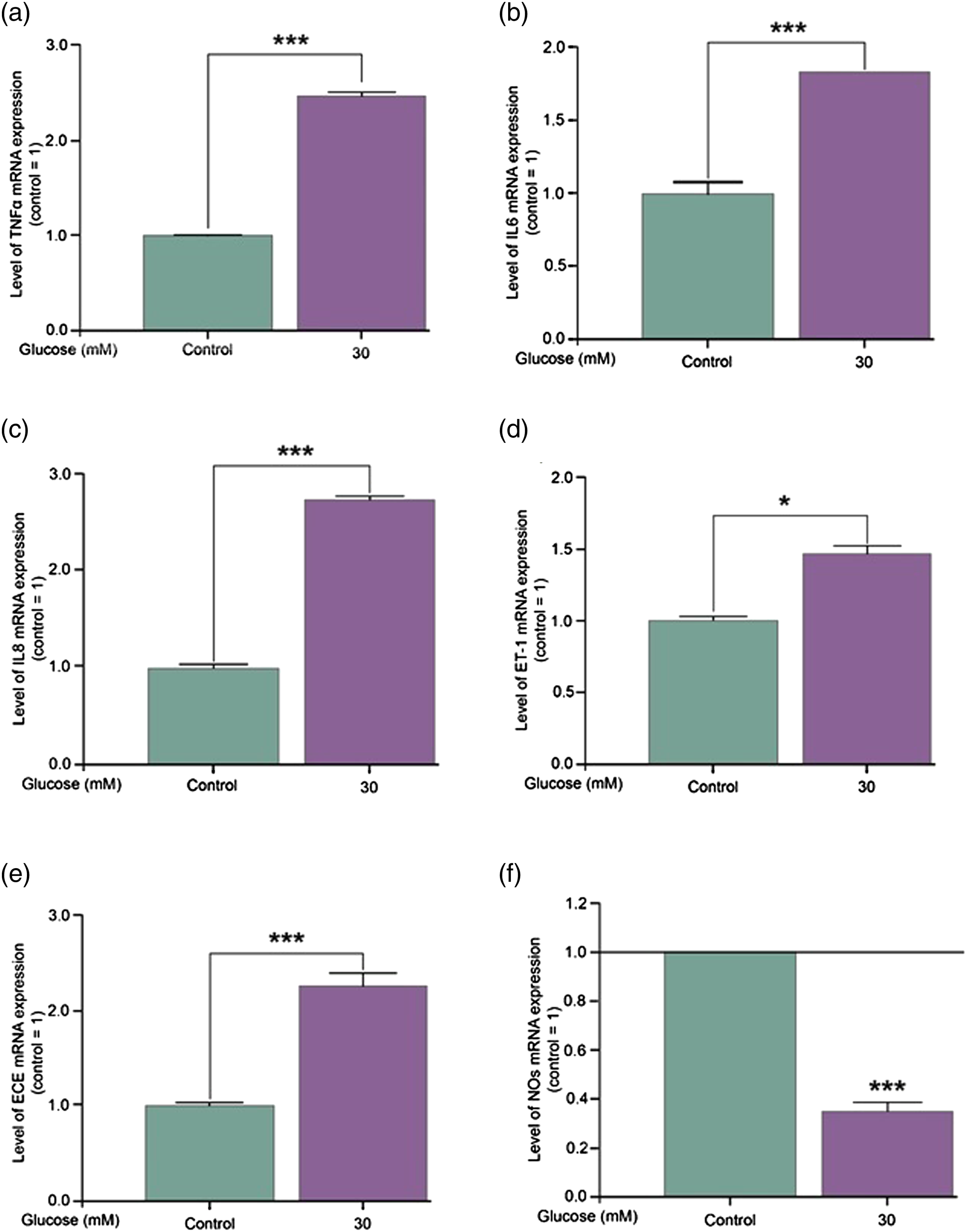
Changes in ROS levels due to hyperglycaemia
Our results on the study of free radicals showed that hyperglycaemia promotes the production of reactive oxygen species by endothelial cells. Persistent hyperglycaemia for 24 h heavily increases the ROS content of cells (Figure 2). Changes in the ROS levels in a hyperglycaemia in an in vitro model system of endothelial cell model after treatment with 30 mM glucose for 24 h. The experiment was performed in triplicate. Data are expressed as the mean ± SEM of three individual experiments. * marks significant (p < .05) differences compared with control (modified t-test).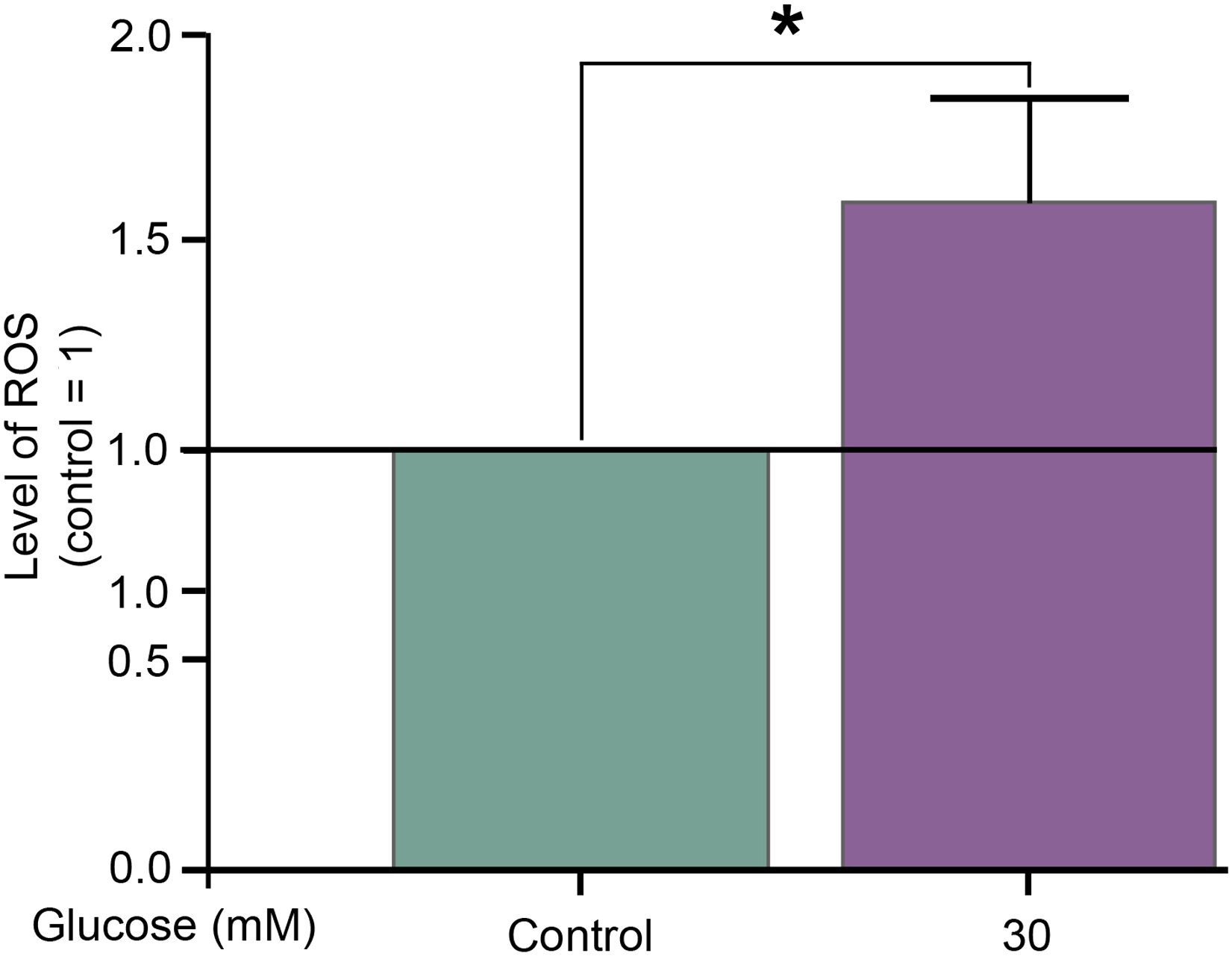
Changes in the reduced glutathione concentration of endothelial cells in the HUVEC model
Our results showed that the concentration of reduced glutathione in the hyperglycaemic samples was far lower than that in the control samples. Depending on time, the difference in the GSH concentration between the samples with extremely high glucose concentrations and the control group gradually increased over the first 24 h. After 24 h, when the hyperglycaemic samples had the lowest GSH concentration, the difference seemed to gradually narrow, although it was still notable (Figure 3). Changes in the glutathione concentration of cells in response to 30 mM glucose at 6, 12, 24, 48, and 96 h after initiation of treatment. The experiment was performed in triplicate. Data are expressed as the mean ± SEM of three individual experiments. *, and *** mark significant (p < .05, 0.0005, respectively) differences compared with control (modified t-test).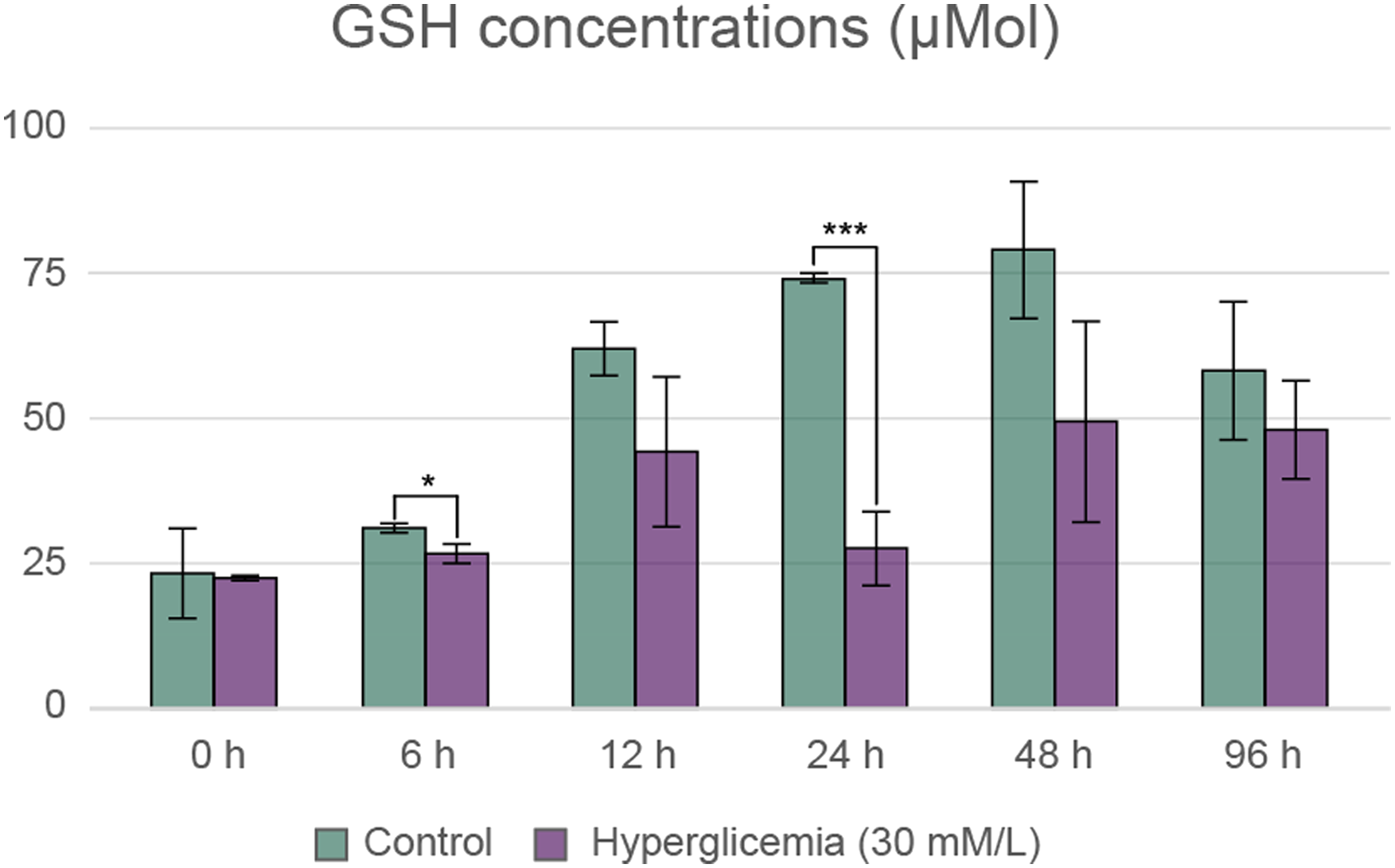
Results of rheopheresis treatment
Clinical results of rheopheresis treatment
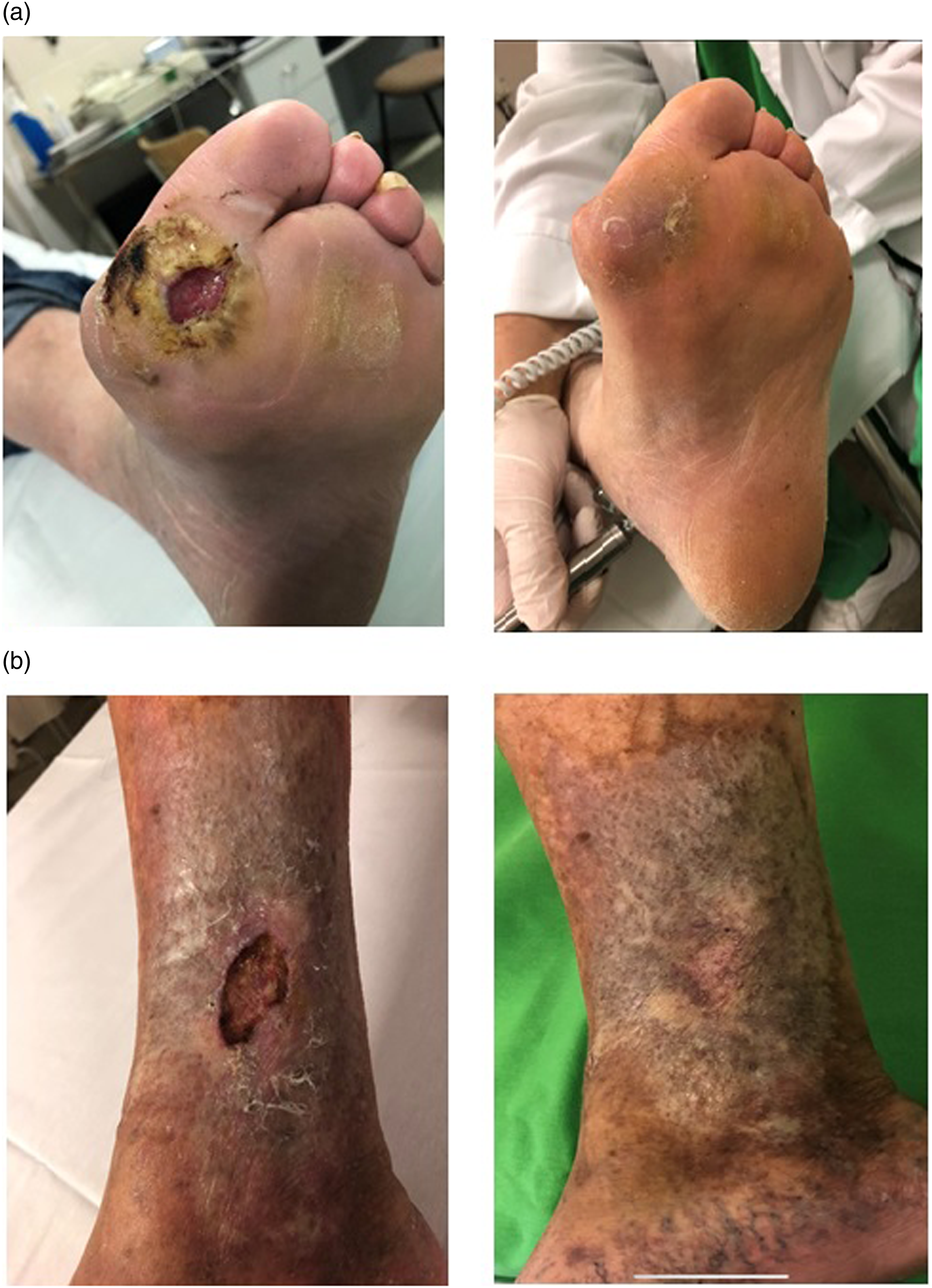
Healing of ulcers of patient No. 5 (4./a) and patient No. 7 (4./b) after 2 consecutive rheopheresis treatments.
Effect of rheopheresis on the plasma concentrations of endothelin-1
Comparing the endothelin-1 concentration of the blood samples taken before and after the treatments, we found that after 2 rheopheresis procedures, the endothelin-1 concentration in the blood samples was markedly lower in 6 of the 7 patients compared to the pretreatment condition (Figure 5.) Changes in the endothelin-1 plasma concentrations in patients after 2 consecutive rheopheresis treatments. The experiment was performed in triplicate. Data are expressed as the mean ± SEM of three individual experiments. *, and *** mark significant (p < .05, .0005, respectively) differences between the before treatment blood plasma and after treatment blood plasma (modified t-test).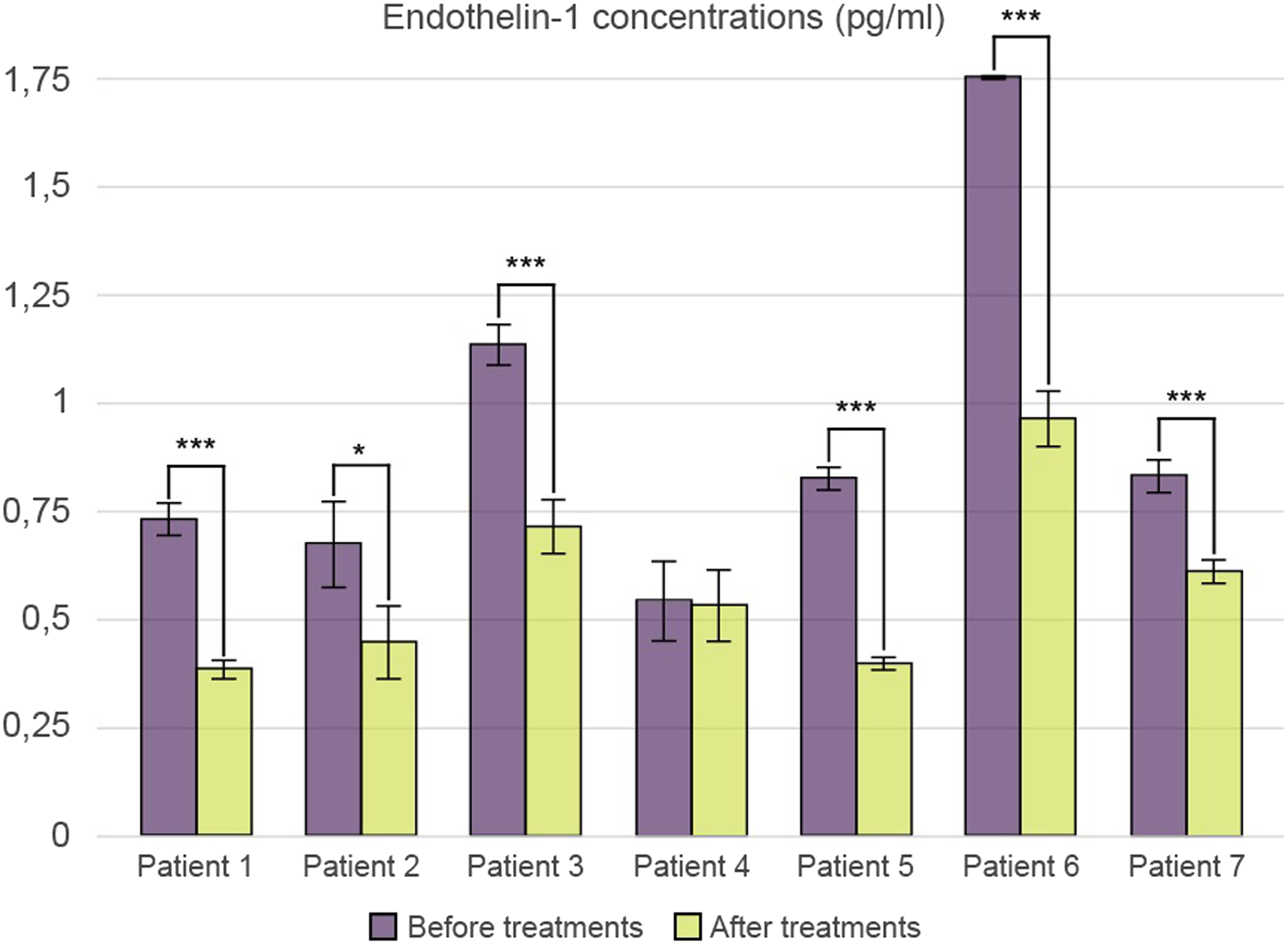
Effect of rheopheresis on the plasma concentrations of thromboxane-B2
ELISA studies on blood samples from 7 patients showed a slight decrease in plasma thromboxane-B2 levels in all 7 patients after 2 rheopheresis treatments (Figure 6). Changes in the thromboxane-B2 plasma concentrations in patients after 2 consecutive rheopheresis treatments. The experiment was performed in triplicate. Data are expressed as the mean ± SEM of three individual experiments *, **, and *** mark significant (p < .05, p < .005, and p < .0005) differences between the before treatment blood plasma and after treatment blood plasma (modified t-test).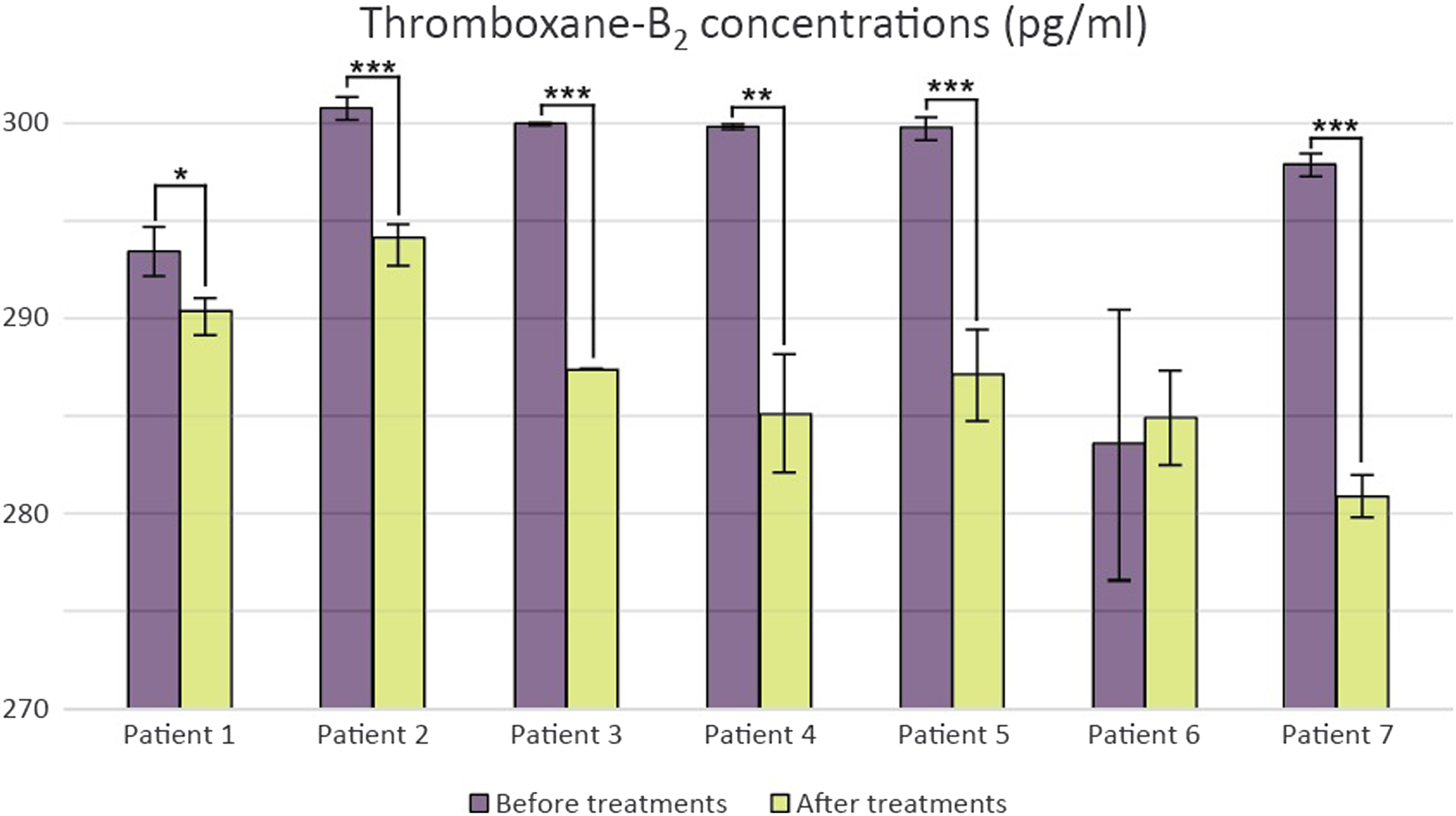
Effect of rheopheresis treatment on SOD enzyme activity
Our measurements did not show precise results for determining the SOD enzyme activity in the blood samples of seven patients. However, of the 7 patients, 5 showed a considerable reduction, averaging 18.06%. In the case of patient No. 2, the SOD enzyme activity increased by 16%, while in the case of patient No. 3, the enzyme activity after the treatment was almost the same as before. In this case, we only saw an increase of 3.13% (Figure 7). Change in superoxid dismutase enzyme activity in patients after 2 consecutive rheopheresis treatments. The experiment was performed in triplicate. Data are expressed as the mean ± SEM of three individual experiments.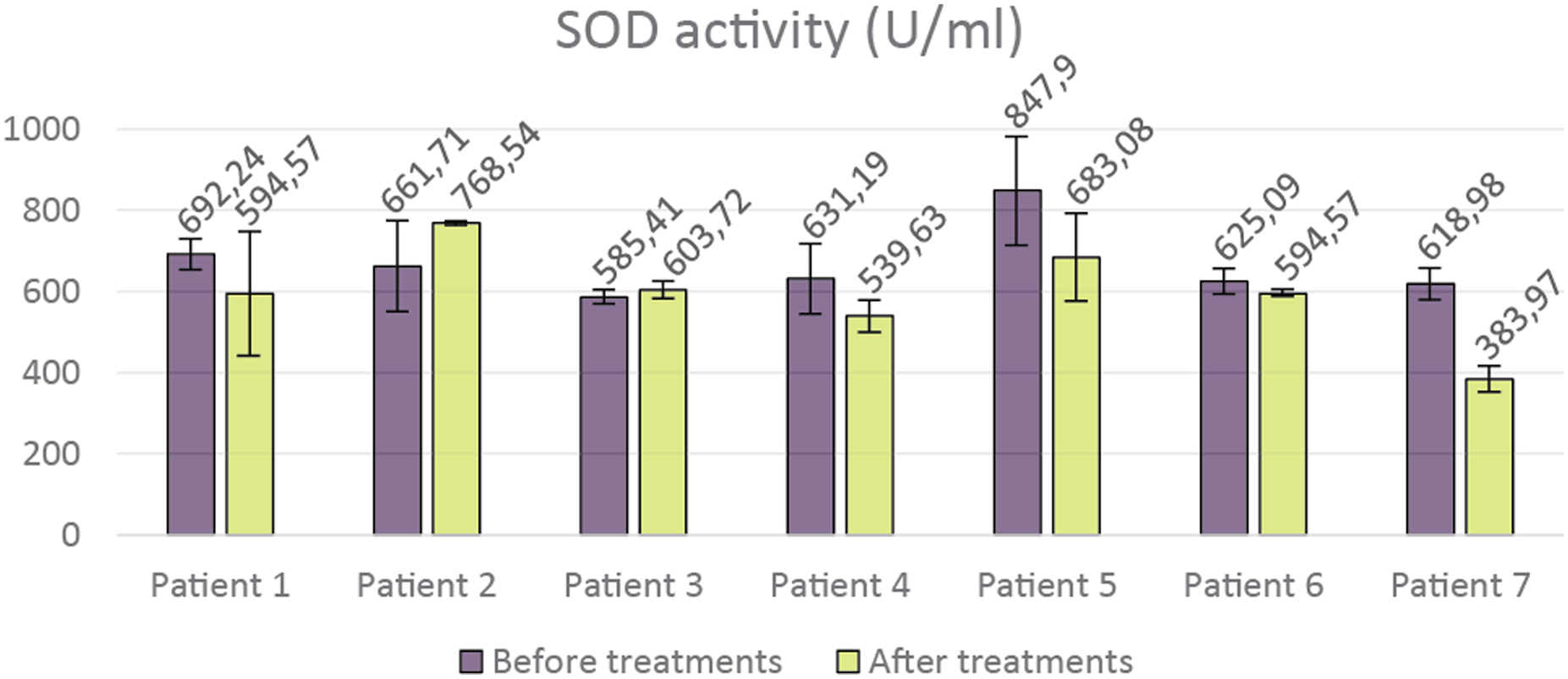
Effect of rheopheresis treatment on the serum levels of total thiol and disulfide components
Examination of the thiol components provided a complex picture of the changes in serum levels of precursors that play a key role in the glutathione pool. The results of the studies showed that the total and protein-bound forms of cysteine were significantly reduced and that the free cysteine was markedly reduced by treatment. Plasma levels of free homocysteine also decreased. On the other hand, serum levels of the direct precursor of glutathione, free-form gamma-glutamylcysteine, significantly increased after rheopheresis treatment in all patients. With the exception of one patient, an inverse proportionality can be detected in the changes in plasma levels of individual small molecule forms of cystine and the protein-bound form of cysteinyl glycine. A clear trend cannot be realized based on the studies of 7 patients, considering the results of the GSH levels in the plasma. Changes in GSH levels did not yield precise results (Figure 8). Changes in the glutathione pool after 2 consecutive rheopheresis treatments.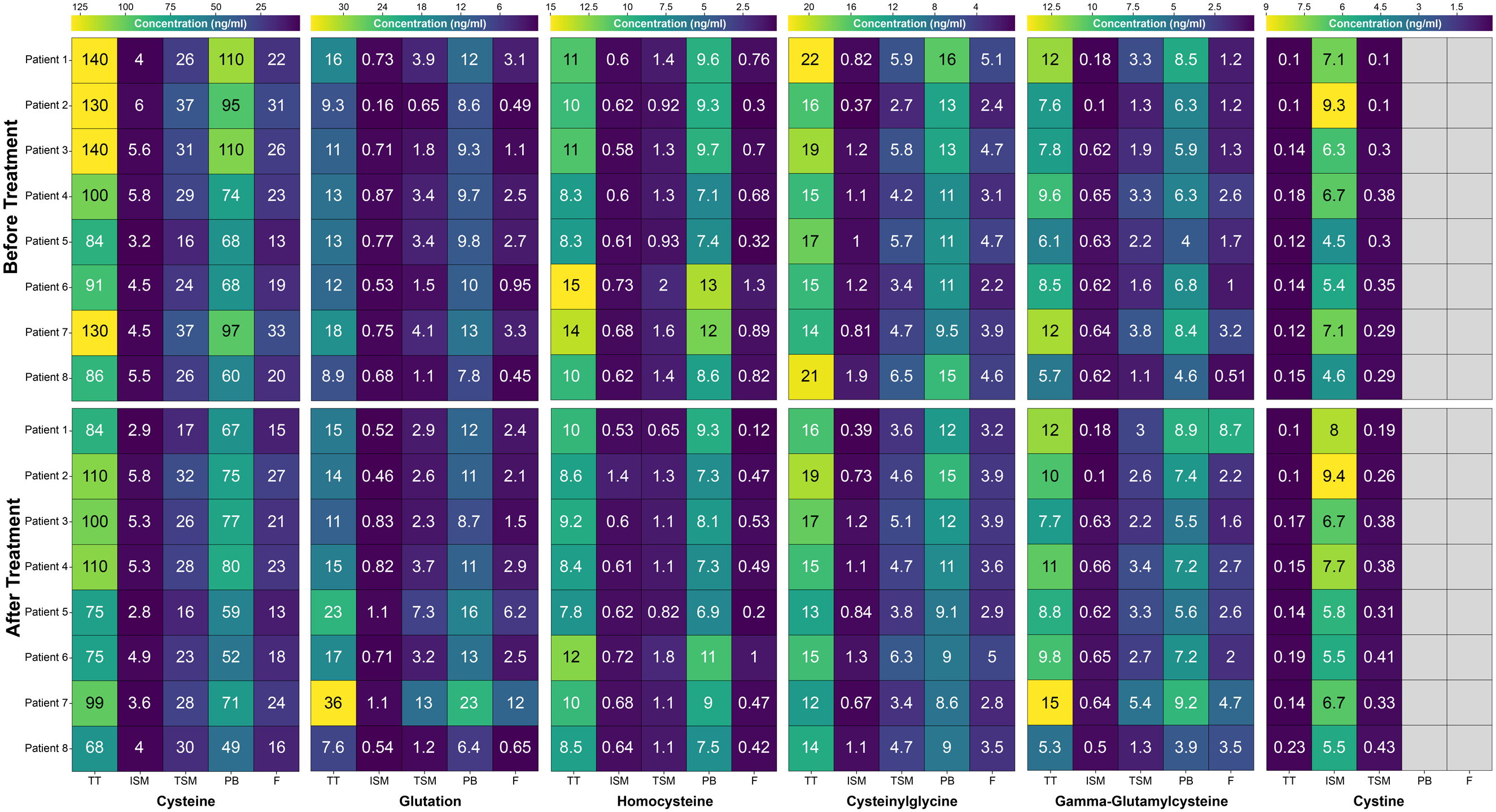
Statistical analysis of the total thiol and disulfide studies
Total cysteine (p = .04) and protein-bound cysteine (p = .03807) showed a significant decrease, and the free form of gamma-glutamylcysteine significantly increased (p = .044) after 2 rheopheresis treatments (Figure 9). Statistical analysis of the changes in total cysteine (9./a), protein-bound cysteine (9./b), and free gamma-glutamyl cysteine (9./c). The experiment was performed in triplicate. * marks significant (p < 0.05) differences between the before treatment blood plasma and after treatment blood plasma (Welch’s t-test; Kruskal–Wallis and Wilcoxon rank-sum nonparametric tests).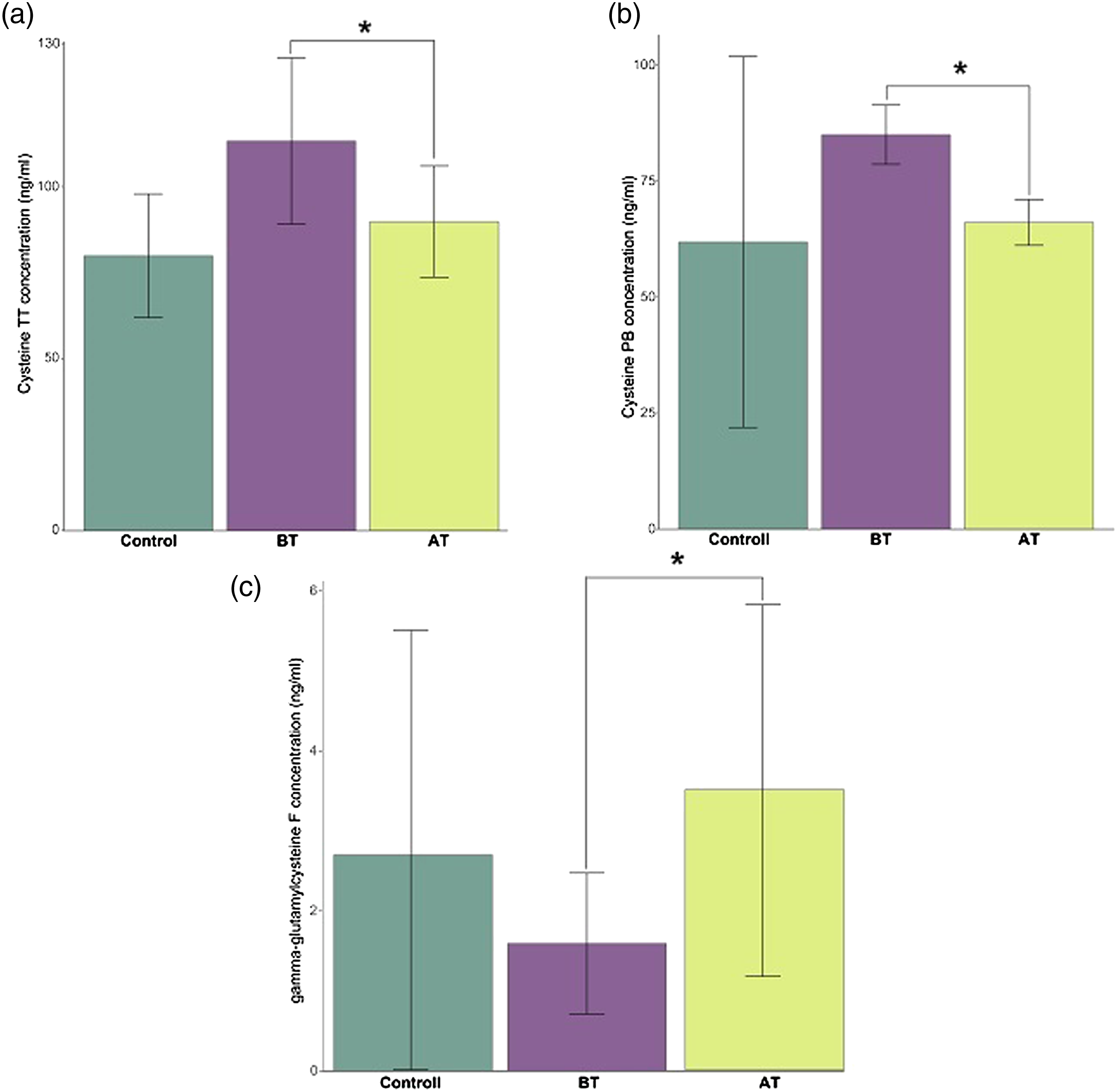
Discussion
It is well known that microvascular damage to the precapillary arterioles and capillaries (<500 μm) plays a cardinal role in the pathomechanism of diabetic foot syndrome.12,13
Vascular destruction results from oxidative stress caused by persistent hyperglycaemia and permanent impairment of antioxidant status.
14
In patients with diabetes, normal metabolism of intravascular glucose cannot occur, and cells containing insulin-independent GLUT can increase intracellular glucose levels in parallel with blood glucose levels. Thus, alternative metabolic pathways are activated due to extreme hyperglycaemia in endothelial cells.
15
By disrupting the mitochondrial electron transport chain, hyperglycaemia blocks the respiratory transport chain and oxidative phosphorylation, resulting in significant superoxide anion release. The released superoxide anion can be converted to reactive hydrogen peroxide and hydroxyl radicals. It can also react with NO produced in endothelial cells and form peroxynitrite. The peroxynitrite that forms is harmful to protein and DNA, inhibits NO release, and has lipid peroxidation effects.
16
In addition, by inhibiting the enzyme GAPDH, the superoxide anion shifts sugar metabolism towards 4 alternative pathways: polyol, hexosamine, PKC, and AGE.
15
As a result of the four alternative pathways, a large amount of vascular mediator is produced, NO monoxide levels are reduced, the endothelium is severely damaged, and the vessel wall structure is altered.
17
All of these factors induce subclinical inflammation with increased production of inflammatory cytokines, acute-phase proteins, and chemokines, resulting in a high degree of damage to the microcirculation.
18
In addition to all of these effects, the haemorheological status of patients with diabetes is frequently impaired. Red blood cell deformability decreases, and the level of viscosity-enhancing plasma proteins greatly increases, resulting in abnormally increased whole blood and plasma viscosity in patients. Deteriorated haemorheological conditions increase blood flow resistance, which damages the blood supply to the tissues, causing further microcirculatory abnormalities (Figure 10).19–21 In 2014, our research team introduced rheopheresis in Hungary for the treatment of diabetic foot syndrome with hyperviscosity that does not require macrocirculatory restoration.
7
We hypothesized about the additional beneficial effects of rheopheresis treatment based on the clinical results, and thus, we performed further studies focusing on diabetes-induced endothelial inflammation. As a first step in our research, we set up an in vitro endothelial cell model to map the mechanisms of hyperglycaemia-induced endothelial inflammation and to identify the inflammatory biomarkers that can be used to realize the beneficial effects of rheopheresis on endothelial status. Although not directly related to in vivo studies, the model is also relevant to them. Regarding that the effects of rheopheresis treatment could not be studied cellularly, we considered it essential to determine the extracellular biochemical markers with which we could monitor endothelial inflammation and antioxidant status before and after rheopheresis. It is important to note that although the protein concentration in the supernatant could have been determined, this would have provided information on the amount of cytokine delivered to the extracellular space by short-term vesicular transport during the treatment. The study of the long-term elemental response of the cells justified the study of the gene expression of the parameters. The signalling pathways of diabetes-induced subclinical inflammation are well elucidated. The literature primarily identifies white blood cells as the source of inflammatory cytokines.22–24 TNF-alpha has a significant MMP-activating effect and therefore severely impairs vascular remodelling and neovascularization by inhibiting VEGF synthesis. In addition to these inhibitory functions, TNF-alpha directly contributes to the apoptosis of endothelial cells, fibroblasts, and keratinocytes.
25
Furthermore, TNF-alpha increases ROS levels via the NF-κB pathway, enhancing COX-2-dependent prostanoid production, which dramatically aggravates pain in patients with diabetic neuropathy.26,27 IL-6 is a major acute-phase protein and, as a major regulator of fibrinogen synthesis, it can maintain the inflammatory state and enhance the proliferation of neutrophils and B lymphocytes.
22
IL-8 is a neutrophil chemotactic factor with a ROS-inducing effect. Therefore, it impairs the body’s antioxidant status by maintaining inflammation.
28
The results of our studies confirmed that the expression of cytokines in endothelial cells is also increased due to the extremely high extracellular glucose concentration, with a significant accumulation of reactive oxygen species in the cells. In addition, the expression of vasoactive substances also changes in a pathological direction. The expression of the potent vasoconstrictor ET-1 and its synthesizing enzyme, ET-1 convertase, is markedly increased, while the expression of NO synthase is significantly reduced. Pathological changes in vasoactive substances play an important role in the progression of microcirculatory complications of diabetic foot syndrome.29,30 Oxidative stress caused by endothelial inflammation and the accumulation of reactive oxygen radicals depletes the reduced glutathione content in cells. The most important cofactor of the glutathione peroxidase enzyme is reduced glutathione, so the GSH/GSSG system has a very significant effect on the body’s antioxidant balance. Reduced glutathione (GSH) is a tripeptide composed of glycine, cysteine, and glutamic acid. It is a potent reducing agent due to its free thiol group. Therefore, it can neutralize reactive free radicals and hydrogen peroxide. During the reaction of reduced glutathione, it binds together through two glutathione disulfide bridges to form oxidized glutathione (GSSG). There is a delicate balance of the GSH:GSSG ratio in a healthy human body. Pathological diversion of the GSH:GSSG ratio and decreased reduced glutathione concentrations may indicate that the body is exposed to high levels of oxidative stress.
31
As the results show, the concentration of reduced glutathione decreased gradually during the hyperglycaemic treatment period compared to the control group. When hyperglycaemic stress was interrupted after 24 h, the difference was gradually decreased, and the reduced glutathione started to regenerate in the cells. Summarizing our in vitro results, the extremely high hyperglycaemia in diabetes mellitus indicates significant inflammation in endothelial cells, leading to the release of free radicals and resulting in a pathological change in antioxidant status. Endothelial impairment due to hyperglycaemia. ETC (electron transport chain), GAPDH (Glyceraldehyde-3-phosphate dehydrogenase), AGE (advanced glycation end), PKC (protein kinase C), NO (Nitrogen monoxide) SOD (Superoxid dismutase).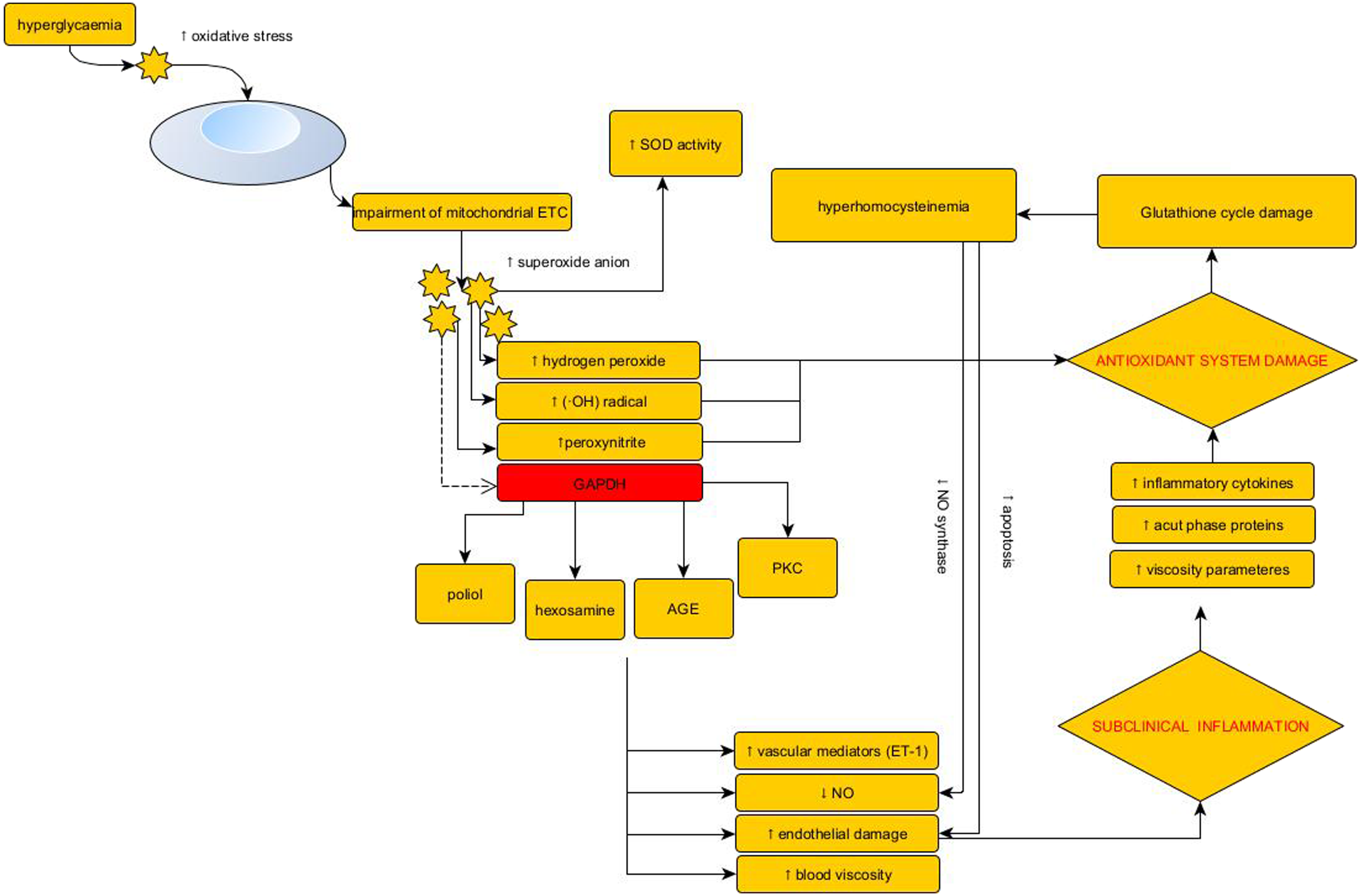
In our previous publication, we demonstrated in 6 patients that 2 consecutive rheopheresis treatments could significantly reduce the plasma concentrations of these inflammatory parameters. 8 Diabetes mellitus also has a significant prothrombotic effect, so we extended our studies to a marker describing the prothrombotic state. 32 Endothelin-1 plasma concentrations showed a slight decrease in all patients. In addition to vasoconstriction, abnormally high levels of endothelial-produced endothelin-1 potentiate oxidative stress in a vicious cycle; thus, rheopheresis treatment has a beneficial effect on plasma endothelin-1 concentrations. 30 Decreases in thromboxane B2 plasma concentrations also indicate beneficial effects of rheopheresis. The active thromboxane A2 produced by platelets has prothrombotic and vasoconstrictor effects, with a short half-life (30 min), making it very difficult to measure. However, its degradation product, thromboxane B2, is a much more stable formula that is easier to measure and provides a comprehensive picture of the patient’s thromboxane A2 supply. Decreased TXB2 plasma levels after treatment provide information on the improvement in endothelial status.33,34 An essential element of the study was the monitoring of treatment-emergent antioxidant changes. One of the most important parameters that provides essential information about antioxidant status is the enzyme activity of superoxide dismutase. The SOD enzyme is able to convert superoxide free radicals in the body to H2O2 and molecular oxygen. Highly oxidative H2O2 is eliminated from the body by reaction with catalase or glutathione peroxidase. 35 However, the change in SOD activity did not yield precise results, which was further limited by the small patient population. The results suggest that SOD activity in patients decreases in parallel with the amount of reactive oxygen species. The effects of rheopheresis on the glutathione pool provide more detailed information about the antioxidant supply. However, reduced glutathione can be found only in very small amounts extracellularly and can only perform its antioxidant function intracellularly. Thus, in our studies, we investigated the glutathione precursors and degradation products that are essential components of the glutathione cycle, and we can deduce the complete glutathione pool from their plasma concentrations. Cysteine is a nonessential amino acid and an important component of glutathione. Its free thiol group plays a key role as a structural and functional element of proteins. However, in its free form, it is highly reactive in the extracellular space, forming a free radical on its own that is extremely unstable and therefore potentially toxic. Plasma levels of free cysteine were markedly reduced in all patients. Protein-bound cysteine by itself is a dominant indicator of antioxidant status. 11 Significantly decreased protein-bound and total cysteine levels in the extracellular space indicate a positive change in antioxidant status. No clear change was observed regarding the extracellular form of reduced glutathione, although it does not provide meaningful information about the body’s antioxidant supply. 31 The cysteine analogue homocysteine is formed from methionine and can be converted to cysteine by the transsulfuration pathway or converted back to methionine by remethylation. Homocysteine is an independent cofactor of atherosclerosis. Hyperhomocysteinaemia damages the endothelium, and the resulting chronic inflammation provokes atherogenesis. Total homocysteine levels in plasma are important in the development of pathological conditions.36,37 Rheopheresis treatment reduced total homocysteine levels in all patients. Decreased homocysteine levels are clearly associated with reduced inflammation and indicate a positive change in the glutathione pool. The decomposition of reduced glutathione forms cysteinylglycine, while cystine is an oxidized dimer of cysteine. We found an interesting correlation in their changes in plasma. Apart from one patient, our results showed that the plasma levels of individual small molecule forms of cystine and protein-bound forms of cysteinylglycine changed in inverse proportion to each other upon rheopheresis treatment. These forms function as extremely valuable cysteine donors during the gamma-glutamyl cycle; however, they are much more stable than free cysteine, and changes in their plasma levels did not adversely affect the amount of free and protein-bound cysteine forms, so their change can be considered positive.38,39 The direct intracellular precursor of reduced glutathione is gamma-glutamylcysteine, which is also directly related to glutathione peroxidase. Although recent literature data suggest that gamma-glutamylcysteine cannot be transported directly into the cell, in vivo experiments have shown that orally administered gamma-glutamylcysteine significantly increased the amount of intracellularly reduced glutathione.40–42 Rheopheresis treatments resulted in a significant increase in the free form of gamma-glutamylcysteine in the plasma of all patients. It can be considered an indicator of an evident positive change in antioxidant status. In conclusion, the endothelial inflammation demonstrated in the HUVEC model, which severely impairs the microcirculation in patients with diabetic foot syndrome, can be reduced by rheopheresis treatment. Our clinical and biochemical results show that the treatment works even in exceptional cases where the location of diabetic ulcer is atypical in terms of classical terminology, and venous insufficiency as an associated etiological factor cannot be ruled out up to 100%. We hypothesize that even when etiological factors are mixed, diabetes-induced endothelial inflammation and hyperviscosity at the level of postcapillary venules lead to further impairment of microcirculation and worsening wound healing. Thus, diabetes should also be treated as a critical etiological factor in these cases. However, our results are limited by the small patient population due to the underfunding of treatment in Hungary thus far and the limited treatment options due to the COVID pandemic. Despite the limiting factors, our findings may inspire further research in the treatment of diabetic foot syndromes and the widespread use of selective apheresis techniques.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

