Abstract
Objective:
We have previously reported insignificant changes in HbA1c after exercise in patients with both type 2 diabetes and coronary artery disease. In this study, we investigated the effect of exercise on endothelial function and possible associations between changes in endothelial function and HbA1c.
Methods:
Patients with type 2 diabetes and coronary artery disease (n = 137) were randomised to 12 months exercise or standard follow-up. Endothelial function was assessed by circulating biomarkers (E-selectin, intercellular adhesion molecule-1, vascular cell adhesion molecule-1, von Willebrand factor, tissue plasminogen activator antigen, asymmetric dimethylarginine and L-arginine/asymmetric dimethylarginine ratio). Differences between the randomised groups were analysed by analysis of covariance and correlations by Spearman’s rho or Pearson’s correlation.
Results:
No effect of exercise on endothelial function was demonstrated. The changes in HbA1c in the exercise group correlated with changes in E-selectin (r = 0.56, p < 0.001), intercellular adhesion molecule-1 (r = 0.27, p = 0.052), vascular cell adhesion molecule-1 (r = 0.32, p = 0.022) and tissue plasminogen activator antigen (r = 0.35, p = 0.011). HbA1c decreased significantly more in patients with versus without a concomitant reduction in E-selectin (p = 0.002), intercellular adhesion molecule-1 (p = 0.011), vascular cell adhesion molecule-1 (p = 0.028) and tissue plasminogen activator antigen (p = 0.009).
Conclusion:
Exercise did not affect biomarkers of endothelial function in patients with both type 2 diabetes and coronary artery disease. However, changes in biomarkers of endothelial activation correlated with changes in HbA1c, and reduced endothelial activation was associated with improved HbA1c after exercise.
Introduction
Type 2 diabetes is associated with endothelial dysfunction, a hallmark of atherosclerosis initiation and progression, characterised by activated endothelial cells with expression of adhesion molecules, inflammatory cytokines and procoagulant factors.1,2 Endothelial dysfunction shifts the vasoregulatory balance of the endothelium towards vasoconstriction, and this may negatively affect blood flow and organ perfusion. 3 Previous studies have demonstrated perfusion disturbances in patients with type 2 diabetes, which may be related to endothelial dysfunction. 4
Exercise training has numerous beneficial effects in type 2 diabetes, including improvement of glycaemic control and cardiovascular risk profile. 5 The mechanisms by which exercise benefits glucose control include enhanced insulin sensitivity and increased expression of GLUT4 transport molecules in the skeletal muscles. 5 Furthermore, exercise training may improve or even restore endothelial function in patients with type 2 diabetes, 6 and this may facilitate glucose uptake through increased skeletal muscle perfusion and thus delivery of insulin and glucose to the periphery. 7 Skeletal muscle blood flow is an important feature of whole body glucose disposal. 8
Patients with both type 2 diabetes and coronary artery disease (CAD) represent complicated diabetic patients with established atherosclerosis and further deteriorated vascular function. 9 These patients have been less investigated as a distinct group, and whether they experience comparable benefits from exercise training as diabetic patients without cardiovascular disease (CVD) is not clear. We have previously reported no overall significant effect of exercise training on HbA1c in this group of patients after 12 months of combined aerobic and strength training. 10 This may indicate reduced effect of exercise on glycaemic control, and it could be speculated whether diabetic patients with complicated vascular disease experience impaired glucose lowering effect of exercise, potentially through attenuated exercise-induced improvement of vascular function and skeletal muscle perfusion.
The aim of this study was to investigate the effect of 12 months exercise training on endothelial function, assessed by circulating biomarkers, in patients with both type 2 diabetes and CAD. Furthermore, we wanted to explore whether changes in these biomarkers were related to changes in HbA1c in the exercise group. We hypothesised that changes in the biomarkers, possibly reflecting changes in endothelial function, were associated with changes in glycaemic control.
Materials and methods
Study design and participants
This study is a part of a randomised clinical trial that investigated the effects of exercise training on glycaemic control, exercise capacity and measures of atherosclerosis in patients with type 2 diabetes and CAD (ClinicalTrials.gov: NCT01232608). Patients with known type 2 diabetes and verified CAD by coronary angiography (n = 137) were included at the Department of Cardiology, Oslo University Hospital, Ullevål, Oslo, Norway, between August 2010 and March 2012. The last follow-up was in March 2013. Exclusion criteria were presence of proliferative retinopathy, end stage renal disease, cancer, stroke or acute myocardial infarction within the last 3 months, unstable angina, uncompensated heart failure, serious arrhythmia, severe valvular disease, severe rheumatologic disease, chronic obstructive pulmonary disease stadium GOLD IV, thromboembolic disease, ongoing infections, severe musculoskeletal disorders and other disabilities limiting the ability for physical activity. Patients who were already engaged in exercise training that exceeded the training programme in the study protocol were not included. All patients gave informed, written consent to participate in the study. The study was approved by The Regional Ethics Committee (REK sør-øst) and was conducted according to the Declaration of Helsinki. At the time of inclusion, the patients were randomised to either the exercise group or the control group. Randomisation was performed using consecutively numbered, non-translucent envelopes containing allocation message to either of the randomised groups in a 1:1 ratio according to tables of random numbers, arranged by the Unit of Epidemiology and Biostatistics, Oslo University Hospital, Ullevål. The patients in the control group continued with standard follow-up by their general practitioner and were not discouraged from physical activity or exercise. Study design and methods have previously been reported in detail. 10
Exercise intervention
The study participants randomised to the exercise group underwent a 12 months combined aerobic and resistance training programme planned and conducted in collaboration with the Norwegian School of Sports Sciences, Oslo, Norway. The exercise programme consisted of group-based exercise sessions of 60-min duration twice a week under supervision of qualified instructors throughout the intervention period and a third weekly home-based individual exercise session. The total exercise volume was 150 min per week, of which approximately two-third was aerobic and one-third resistance exercises. The exercise sessions were guided by Borg’s scale of rated perceived exertion (RPE) and included parts of high-intensity interval training (RPE ≥ 15). 10
Laboratory methods
Blood samples were drawn at inclusion and within 1 week after the last exercise session by standard venipuncture between 08:00 and 10:00 a.m. after overnight fast, without medication taken since the preceding evening and without exercise training the last 24 h. Endothelial function was assessed by circulating levels of asymmetric dimethylarginine (ADMA), L-arginine/ADMA ratio, E-selectin, intercellular adhesion molecule-1 (ICAM-1), vascular cell adhesion molecule-1 (VCAM-1), von Willebrand factor (vWF) and tissue plasminogen activator antigen (t-PAag). E-selectin, ICAM-1 and VCAM-1 were measured in serum by enzyme-linked immunosorbent assay (ELISA) methods from R&D Systems Europe (Abingdon, UK), whereas vWF and t-PAag were measured in citrated plasma with commercial ELISA methods (Asserachrom vWF Ag; Stago Diagnostica, Asnieres, France and TintElize tPA; Biopool AB, Trinity Biotech plc, Bray, Ireland, respectively). Ethylenediaminetetraacetic acid (EDTA) plasma samples were used for analyses of
Statistical analysis
The power calculation in the main study was based on an expected 10% relative reduction in HbA1c in the exercise group while unchanged in the control group. Accounting for possible dropouts, the calculation indicated a minimum of 68 patients in each arm of the study. Statistical analyses were performed according to ‘per protocol’ principles, where patients with low exercise adherence were excluded from the calculations. 10
Demographic data are given as proportions, mean [±standard deviation (SD)] or median values (25th and 75th percentiles) with skewed data. Between-group differences in baseline characteristics were calculated by independent-sample Students t-test, Chi-square test or Mann–Whitney test as appropriate. Differences between the randomised groups in changes in the biomarkers from baseline to 12 months were calculated by one-way analysis of covariance (ANCOVA). Differences in changes in HbA1c between dichotomised strata in the exercise group were calculated by Mann–Whitney U test or independent-sample Students t-test as appropriate, whereas intragroup changes were analysed by Wilcoxon signed-rank test or paired-sample Students t-test. Associations between changes in HbA1c and the measured biomarkers were calculated by Spearman’s rho or Pearson’s correlation as appropriate. Adjustments for changes in body weight and
Results
Figure 1 shows the flow diagram of the patients. Baseline characteristics of the study population are shown in Table 1. No patients were in New York Heart Association (NYHA) class III or IV. There were no significant differences between the randomised groups at baseline.
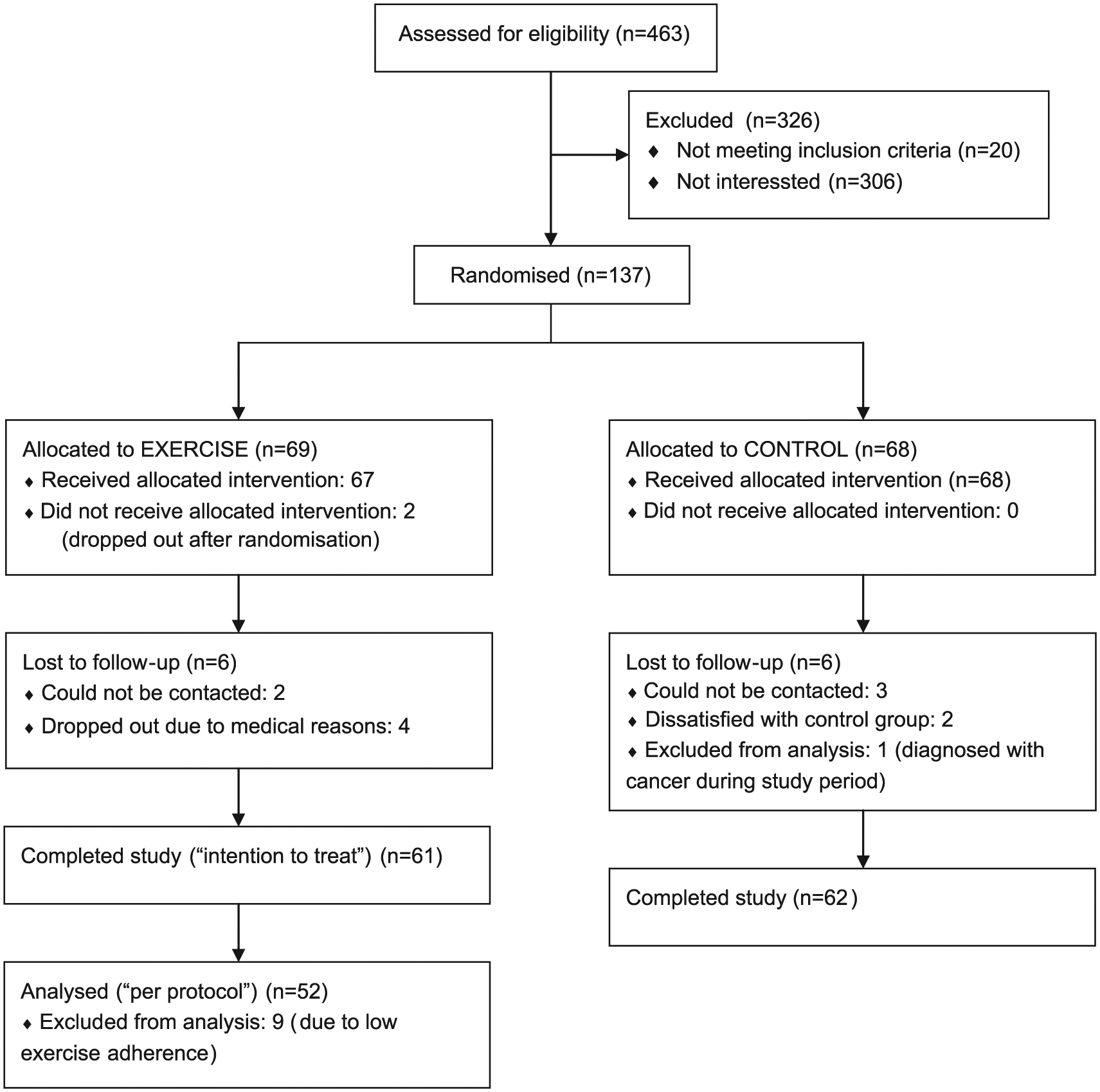
Flow diagram of the patients in the study.
Baseline characteristics of all included patients and patients completing the study according to the randomised groups.
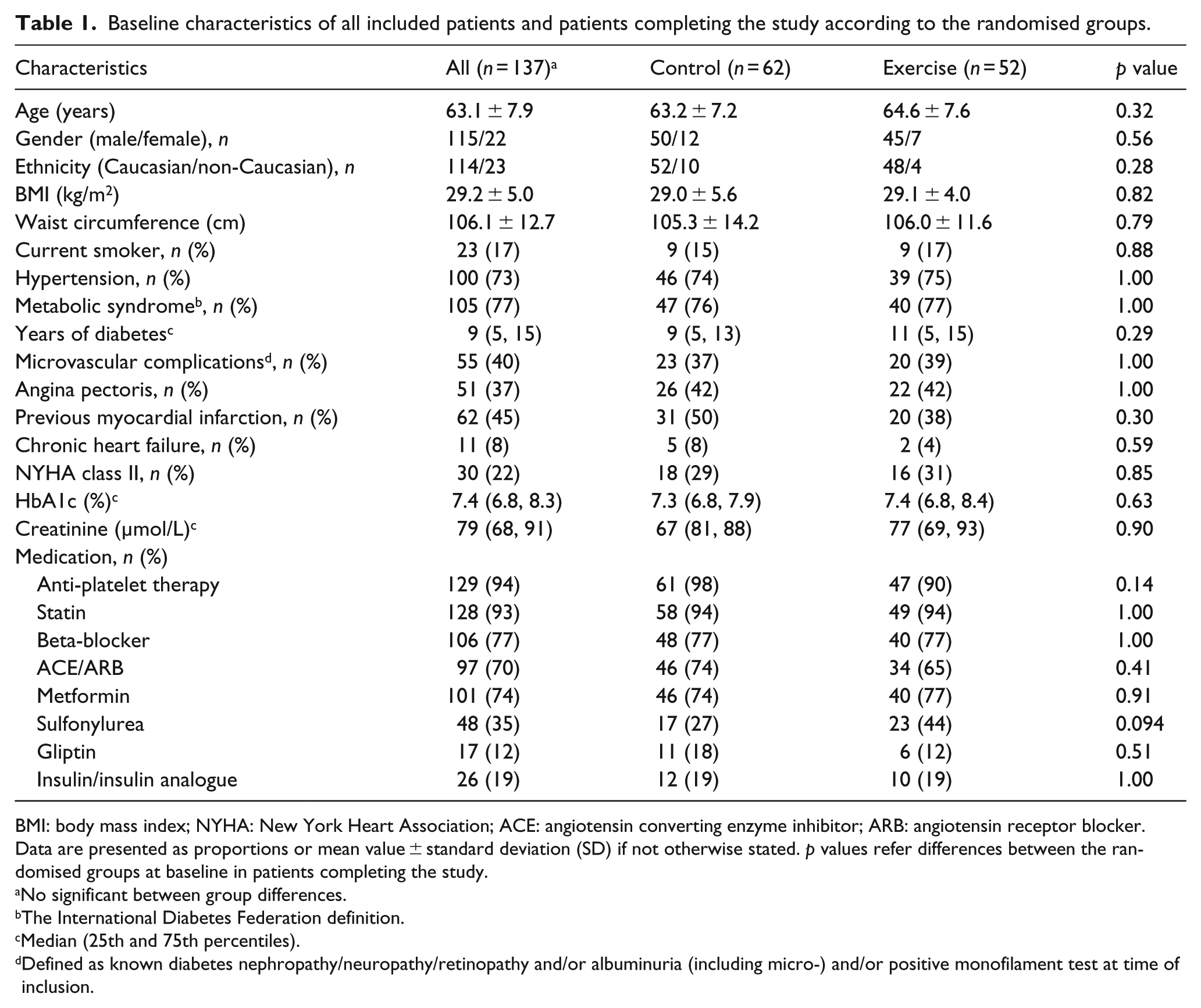
BMI: body mass index; NYHA: New York Heart Association; ACE: angiotensin converting enzyme inhibitor; ARB: angiotensin receptor blocker.
Data are presented as proportions or mean value ± standard deviation (SD) if not otherwise stated. p values refer differences between the randomised groups at baseline in patients completing the study.
No significant between group differences.
The International Diabetes Federation definition.
Median (25th and 75th percentiles).
Defined as known diabetes nephropathy/neuropathy/retinopathy and/or albuminuria (including micro-) and/or positive monofilament test at time of inclusion.
In the main study, as previously reported, we did not observe an overall significant change in HbA1c from baseline to 12 months in the exercise group compared to the control group [−0.22% (−0.49 to 0.04) vs −0.01% (−0.25 to 0.24), p = 0.24]. 10 Progression of atherosclerosis, evaluated by changes in carotid intima-media thickness, showed also no difference between the randomised groups (p = 0.57). 11 However, exercise performance improved with significant increase in ventilatory (anaerobic) threshold and time to exhaustion in the exercise group compared to controls (p = 0.046 and p = 0.034, respectively), whereas a trend for increase in VO2peak was demonstrated (p = 0.077). 10
Effects of exercise on biomarkers of endothelial function
In Table 2, we present the biomarkers of endothelial function at baseline and after 12 months. We could not demonstrate significant differences between the randomised groups in changes in the biomarkers during the study.
Biomarkers of endothelial function at baseline and 12 months according to the randomised groups.
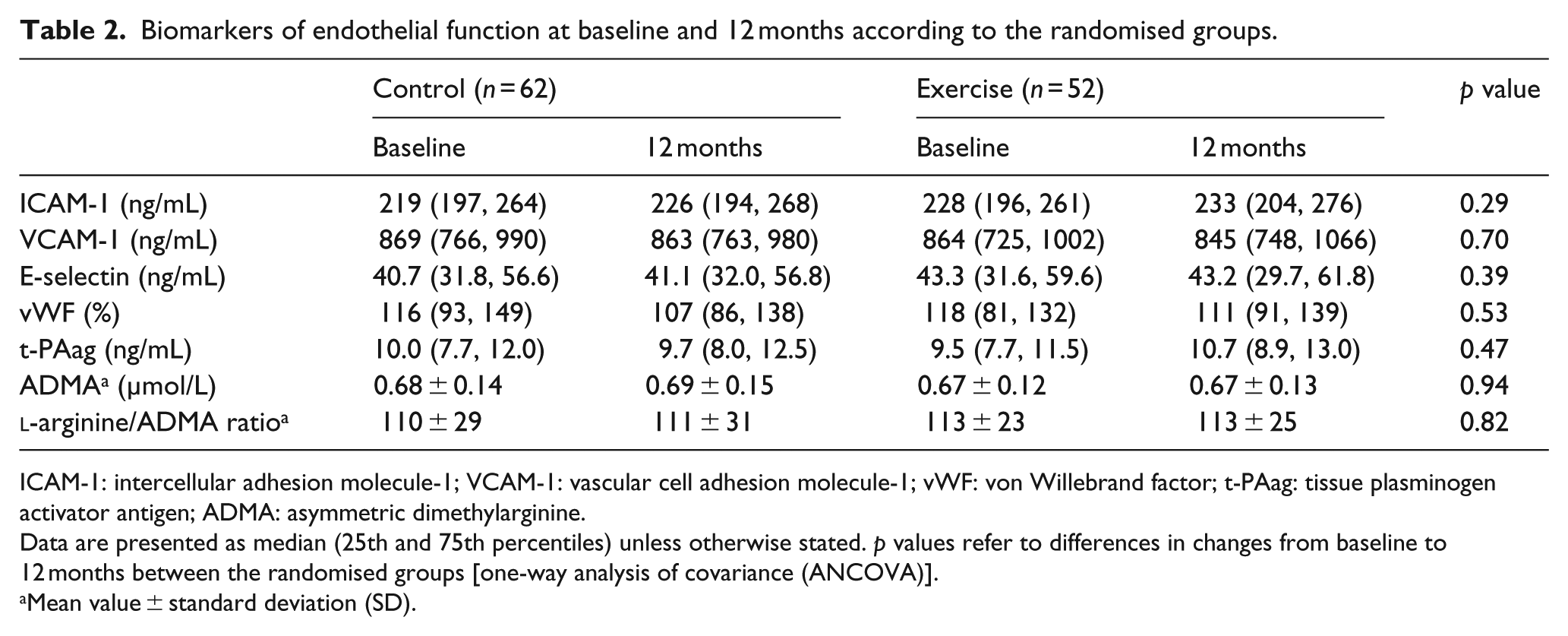
ICAM-1: intercellular adhesion molecule-1; VCAM-1: vascular cell adhesion molecule-1; vWF: von Willebrand factor; t-PAag: tissue plasminogen activator antigen; ADMA: asymmetric dimethylarginine.
Data are presented as median (25th and 75th percentiles) unless otherwise stated. p values refer to differences in changes from baseline to 12 months between the randomised groups [one-way analysis of covariance (ANCOVA)].
Mean value ± standard deviation (SD).
Associations between changes in biomarkers of endothelial function and HbA1c
Changes in the biomarkers of endothelial activation (i.e. E-selectin, ICAM-1, VCAM-1, t-PAag and vWF) during the study period correlated with changes in HbA1c (Table 3). The strongest correlation was observed between changes in E-selectin and HbA1c (Table 3, Figure 2), highly significant also after adjustment for body weight changes during the intervention period (β = 0.43, p = 0.005). There were no significant correlations between changes in HbA1c and changes in ADMA or
Correlations between changes in the biomarkers of endothelial function and HbA1c during the study in the exercise group (EX) (n = 52) and the control group (CO) (n = 62).
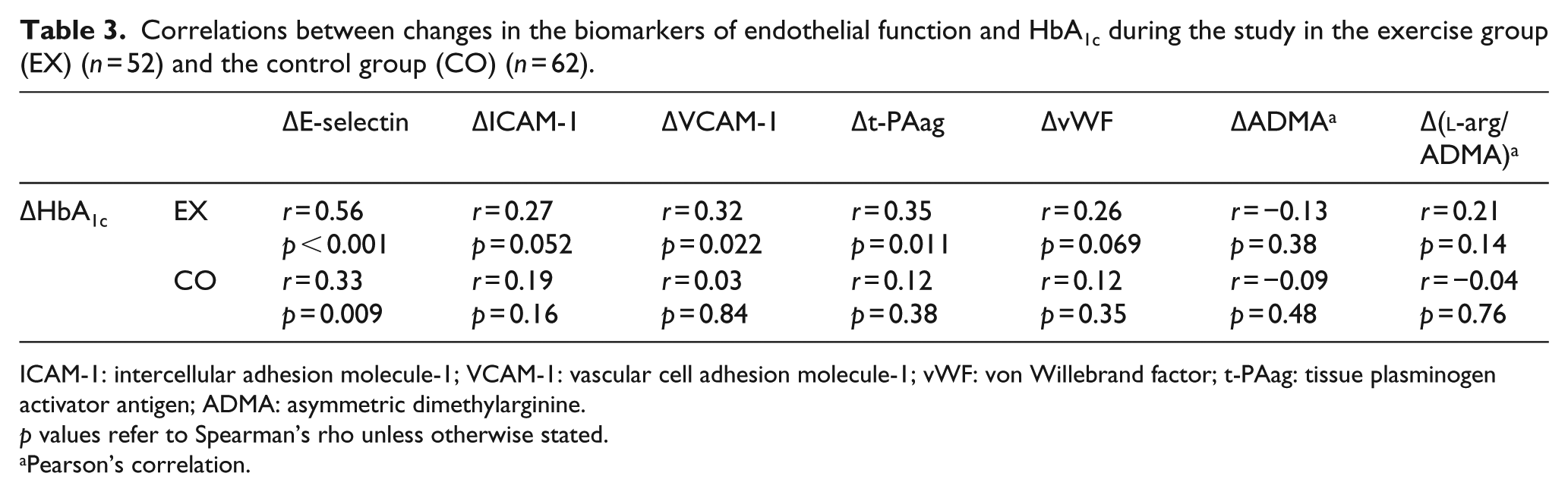
ICAM-1: intercellular adhesion molecule-1; VCAM-1: vascular cell adhesion molecule-1; vWF: von Willebrand factor; t-PAag: tissue plasminogen activator antigen; ADMA: asymmetric dimethylarginine.
p values refer to Spearman’s rho unless otherwise stated.
Pearson’s correlation.
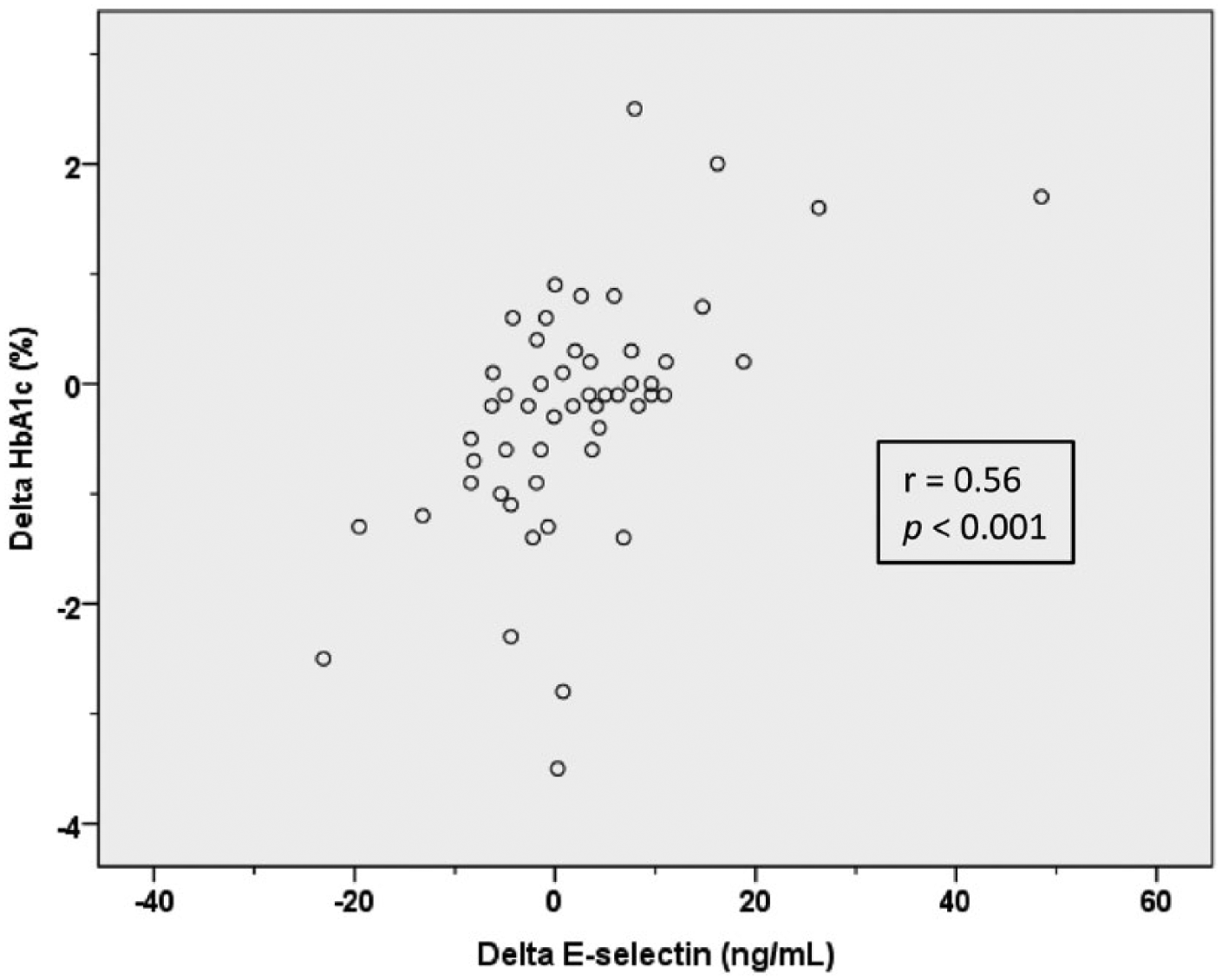
Scatterplot of absolute changes in HbA1c (%) and E-selectin (ng/mL) in the exercise group (n = 52). p value refers to Spearman’s rho.
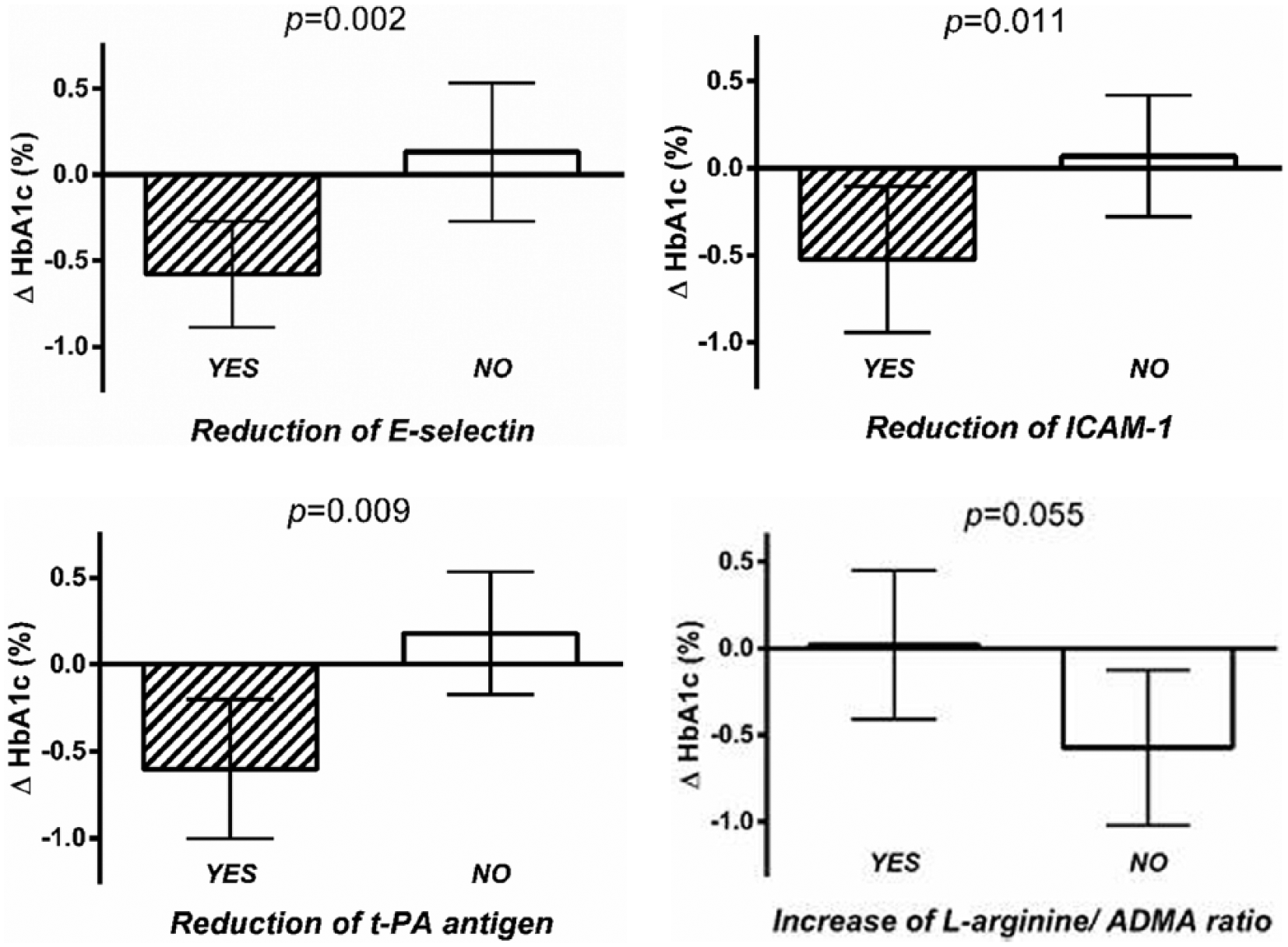
Absolute changes in HbA1c (95% confidence interval) in the exercise group (n = 52) in patients with reduction in E-selectin (n = 23), ICAM-1 (n = 21) and t-PA antigen (n = 23) or increase in
There was no difference in change in HbA1c between patients with or without reduction in ADMA (p = 0.27). However, HbA1c improved more in patients who did not increase
The association between changes in the endothelial biomarkers and HbA1c was less pronounced in the control group as compared to the exercise group (Table 3).
There were no significant differences in the baseline characteristics between patients in the exercise group with or without reduction in E-selectin during the study, although the use of metformin tended to be more prevalent among patients with reduced E-selectin (91% vs 66%, p = 0.063). However, patients with decrease in E-selectin experienced significantly larger weight reduction during the study compared to patients without (−3.1 ± 3.1 kg vs 0.7 ± 4.2 kg, p < 0.005). There were no differences between these subgroups in changes in carotid intima-media thickness or exercise capacity.
Discussion
In this study, we could not demonstrate significant effects of 12 months of combined aerobic and strength training on biomarkers of endothelial function in patients with both type 2 diabetes and CAD. However, changes in biomarkers of endothelial activation during the study correlated with the change in HbA1c, and reduced endothelial activation was associated with improved HbA1c after exercise. This may imply that the limited decrease in HbA1c observed in these patients was related to an attenuated effect of exercise on endothelial function.
Effect of exercise on endothelial function in patients with type 2 diabetes and CAD
This study reports ancillary results from a relative large randomised controlled exercise trial in patients with both type 2 diabetes and CAD. Despite an increased awareness of the relationship between glucometabolic disorders and CVD in recent years, 12 complicated diabetic patients with CVD have not been extensively investigated, and whether they experience comparable benefits from different treatment options as patients with uncomplicated diabetes is not clear. Our previous results indicate that exercise may have reduced influence on glycaemic control in diabetic patients with macrovascular disease. 10
The exercise trial was of 12 months duration and aimed at investigating sustainable effects of a manageable exercise intervention in a diseased group of patients. The exercise programme was arranged in collaboration with the Norwegian School of Sports Sciences and included two supervised group-based exercise sessions per week throughout the intervention period. Furthermore, the intervention was ‘exercise-only’, and there were no changes in dietary habits or medication during the study. 10
Endothelial dysfunction was assessed by circulating biomarkers, and realising the limitation of evaluating endothelial function with biomarkers, the chosen biomarkers are widely accepted as surrogate measures of endothelial function/dysfunction.13,14 E-selectin, which is expressed on the surface of activated endothelial cells and triggers leukocyte adhesion and migration to the subendothelial space, is probably the most specific biomarker for endothelial activation and has also been associated with cardiovascular risk factors and adverse cardiovascular prognosis.13,14 Studies have also shown relationship between E-selectin and type 2 diabetes and adiposity.15,16
Like exercise trials in general, the volume and intensity of the training programme may have influenced the results. We aimed at training the patients according to recommended guidelines in type 2 diabetes, 5 and this was achieved for most of the intervention time. However, we experienced that our patients, with two chronic diseases and prevalent additional comorbidities, were susceptible for intercurrent illnesses and physical complains, and some patients had periods during the exercise year where they were unable to follow the prescribed exercise programme. 10 This influenced the average exercise adherence and thus, possibly, also our results.
The relative long duration of our study may have had a more direct influence on the outcome. It has been discussed by others that endothelial function measured by flow-mediated dilation (FMD) may have an initial, transient beneficial response to exercise training that subsides as the exercise stimulus continues and a structural adaptation of the vessels occurs. 17 In line with this, a meta-analysis of patients with type 2 diabetes concluded that exercise training not only improved arterial endothelial function assessed by FMD but also revealed a tendency towards lesser improvement of FMD with increasing number of weeks of the training programme. 6 A short 4-week exercise programme also demonstrated beneficial effects on levels of E-selectin, ICAM-1 and VCAM-1 in patients with type 2 diabetes or impaired glucose tolerance, 18 whereas a 6-month exercise trial in patients with type 2 diabetes showed no effect on FMD and biomarkers of endothelial function. 19 Although changes in FMD occurring along with vascular adaptation to exercise may not be applicable for endothelial biomarkers, any structural adaptation of the vasculature may influence endothelial function and thus our results.
Sixt et al. 20 investigated patients with both type 2 diabetes and CAD, and they observed a strong trend for a decrease in ICAM-1 at 4 weeks after multifactorial lifestyle intervention (p = 0.051), which returned to baseline levels at 6 months. The dilatory response to acetylcholine in the coronary arteries examined in this study remained unchanged after 4 weeks but improved after 6 months. 20 This study, however, had a comprehensive intervention with daily exercise training and a hypocaloric diet the first months 20 making it not comparable to our study. In another trial of older patients with type 2 diabetes without CVD, 6 months of aerobic exercise twice a week significantly decreased P-selectin and ICAM-1; however, no control group was included. 21 In patients with CAD, Hambrecht et al. 22 showed improved endothelium-dependent vasodilation in coronary vessels after 4 weeks of exercise training, and a meta-analysis of patients with risk factors for or established CVD has shown reduced ICAM-1 and VCAM-1 after exercise training for 3 weeks–6 months. 23
ADMA is a competitive inhibitor of nitric oxide (NO) synthase, thereby reducing NO bioavailability and contributing to endothelial dysfunction. 13 Elevated levels of ADMA have been associated with cardiovascular risk factors, type 2 diabetes and CVD. 24 The effect of exercise training on ADMA has not been extensively investigated, but there are studies indicating beneficial effect in patients with atherosclerotic disease25,26 and unchanged levels in patients with heart failure. 27
The patients in this study all had type 2 diabetes and CAD, and in addition, a majority of the patients met the definition of having the metabolic syndrome. Many also presented with microvascular complications. Patients with atherosclerosis may have severely impaired vascular response to exercise, 28 and type 2 diabetes and other cardiovascular risk factors may further deteriorate vascular function. 3 One could therefore speculate that the patients in our study were more resistant to the beneficial effects of exercise on the vasculature compared to uncomplicated diabetic patients, possibly due to attenuated exercise-induced NO-mediated responses in the diseased vessels. The results of this study are in accordance with our resent publication reporting neutral findings on exercise effect on carotid intima-media thickness in the same population. 11
Association between exercise-induced changes in HbA1c and endothelial function
There is an established association between glycaemic control and endothelial function;3,13 however, the relationship between changes in these parameters is less clear. Trøseid et al. 29 demonstrated significant correlation between changes in HbA1c and E-selectin in patients with the metabolic syndrome, irrespective of the intervention (exercise, pravastatin or control). Barone Gibbs et al. 19 showed correlation between reduced levels of ICAM-1 and HbA1c after exercise in patients with type 2 diabetes, although change in E-selectin unexpectedly was inversely correlated with change in HbA1c. Furthermore, in a small exercise study of patients with type 2 diabetes (n = 16), a correlation coefficient of −0.32 was observed between changes in FMD and HbA1c, however, not significant (p > 0.1). 30 No associations were observed between improved acetylcholine response in coronary arteries and changes in endothelial biomarkers in patients with type 2 diabetes and CAD. 20
In this study, we demonstrated significant correlations in the exercise group between changes in E-selectin, t-PAag, VCAM-1 and ICAM-1 and the changes in HbA1c. This indicates an association between exercise-induced changes in endothelial activation and glycaemic control. In type 2 diabetes, hyperglycaemia may lead to accumulation of glucose and its metabolites in endothelial cells, activating alternative metabolic pathways that increase inflammation and oxidative stress, form advanced glycation end products and modify endothelial nitric oxide synthase (eNOS) activity, resulting in endothelial activation and dysfunction.3,13 The observed association may therefore be caused by a change in glucose levels that subsequently affects endothelial activation. A change in insulin resistance may also influence endothelial function, directly through the vasodilatory properties of insulin 7 or indirectly by change in glucose uptake.
Changes in HbA1c and endothelial function after exercise may also be associated through a primary variation in endothelial activation. Exercise training induces translocation of the GLUT4 molecule from intracellular storage depots to the plasma membrane and T-tubules in skeletal muscle cells, making more transport molecules accessible for peripheral glucose uptake. 8 However, increased blood flow and glucose delivery to the skeletal muscles is also important for enhanced whole body glucose disposal to occur. 8 The observed association may therefore be explained by exercise-induced eNOS activation and NO release, which may reduce endothelial activation and further induce vasodilation and increased skeletal muscle perfusion. This may increase peripheral glucose delivery and uptake. 7 Our finding that the associations between changes in endothelial biomarkers and HbA1c were most pronounced in the exercise group indicates that this relation primarily may be of importance during and after exercise.
Changes in HbA1c did not significantly correlate with changes in ADMA or
Is reduced endothelial activation after exercise important for improved HbA1c?
In this study, we demonstrated that patients with reduced endothelial activation after exercise experienced beneficial effects on HbA1c. Interestingly, we could not observe any differences in changes in the endothelial biomarkers between patients with or without improved HbA1c. Although our results are based on post hoc analyses and may only serve as hypothesis generating, a possible interpretation of these findings is that reduced endothelial activation is important for reduction in HbA1c after exercise in this group of patients. This would be in accordance with the theory that adequate vascular function and skeletal muscle perfusion are of importance for whole body glucose uptake. 7 It could be speculated that diabetic patients with attenuated vascular response to exercise experience smaller improvements of glycaemic control compared to patients with preserved exercise effects on the vasculature. We observed a limited reduction in HbA1c in patients with type 2 diabetes and established macrovascular disease, and a possible explanation could be that a majority of the patients in the exercise group did not improve endothelial function and skeletal muscle perfusion. Nevertheless, changes in endothelial activation and HbA1c after exercise were associated in our patients, and despite no overall effect of exercise on HbA1c, improvements were observed in patients with a concomitant reduction in endothelial activation.
Conclusion
We did not observe significant change in biomarkers of endothelial function after 12 months of combined aerobic and strength training in this group of patients with both type 2 diabetes and CAD. However, changes in the biomarkers of endothelial activation during the study correlated with changes in HbA1c, and reduced endothelial activation was associated with improved HbA1c after exercise. This may imply that the limited decrease in HbA1c observed in these patients was related to an attenuated effect of exercise on endothelial function and thus, possibly, skeletal muscle perfusion.
Footnotes
Acknowledgements
The authors want to thank Sigmund Anderssen, Marthe Aulie, Lars Amund Toftegaard, Bente Neraal, Bente Vestad, Sissel Åkra and Vibeke Bratseth for their valuable contributions to the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
