Abstract
Objective:
Type 2 diabetes is associated with cognitive dysfunction, but the mechanisms are unknown. We assessed the relationships of biomarkers of oxidation, endothelial function and inflammation with cognition in participants of the CAROLINA® trial (CARdiovascular Outcome Trial of LINAgliptin Versus Glimepiride in Type 2 Diabetes).
Methods:
Baseline circulating biomarkers of oxidation (8-iso-prostaglandin F2α), endothelial function (asymmetric dimethylarginine, endothelin-1) and inflammation (C-reactive protein, interleukin-6, tumour necrosis factor-α), based on linear regression, were related to cognition on five domains, as measured with an automated battery.
Results:
In 37 patients (mean age 66.7 ± 8.7 years, median HbA1c 6.9%/52 mmol/mol), 8-iso-prostaglandin F2α was associated with reduced mental flexibility and attention (standardised regression coefficients −0.47, −0.34), whereas asymmetric dimethylarginine was associated with reduced psychomotor speed and attention (standardised regression coefficients −0.39, −0.34). No significant associations were observed between biomarkers of inflammation and cognition.
Conclusion:
Elevated biomarkers of oxidation and endothelial function are associated and may play a role in reduced psychomotor speed, mental flexibility and attention in type 2 diabetes.
Keywords
Introduction
Type 2 diabetes is associated with modest decrements in cognitive functioning. 1 These decrements are predominantly found in the cognitive domains of psychomotor speed, mental flexibility and memory. 1 The underlying processes of cognitive dysfunction in type 2 diabetes are largely unknown. The present study explores the association between circulating biomarkers of oxidation, endothelial function and inflammation and cognition in type 2 diabetes, since these pathways have been postulated to play a role for diabetes-associated cardiovascular complications, 2 as well as for cognitive dysfunction outside the context of diabetes.3,4
Methods
Study population
For the present study, we selected a subset of participants from the CAROLINA® trial (CARdiovascular Outcome Trial of LINAgliptin Versus Glimepiride in Type 2 Diabetes). CAROLINA (NCT01243424) is a randomised, active comparator, double blind study involving 6041 participants from 43 countries with relatively early type 2 diabetes (median diabetes duration 6.2 years), with or without cardiovascular complications, with the primary goal to evaluate the cardiovascular safety and efficacy of the dipeptidyl peptidase-IV inhibitor linagliptin versus the sulfonylurea glimepiride. 5 CAROLINA also evaluates the cognitive effects of the study medication in a dedicated cognition substudy encompassing approximately 4500 participants, from 33 countries, all using a Latin alphabet 6 and all without the pre-defined exclusion criteria [mini-mental state examination (MMSE) <24 and a history of major neurological disease]. All participants in the CAROLINA cognition substudy underwent a brief cognitive assessment. 6 We also planned a smaller parallel study, specifically dedicated to evaluate the relation between cognition and various biomarkers. For this biomarker study, more detailed cognitive data were collected, using an automated cognitive assessment, prior to initiation of first study drug treatment. This automated comprehensive cognitive assessment was implemented in 13 centres from five countries. In total, 45 participants performed the automated comprehensive cognitive assessment. Six did not have biomarker data and two did not fulfil cognition-specific inclusion criteria (one with MMSE <24, and one with a major neurological disease), leaving 37 for the present analysis.
The study was carried out in compliance with the protocol, the principles laid down in the Declaration of Helsinki, version as of October 1996, in accordance with the ICH Harmonised Tripartite Guideline for Good Clinical Practice (GCP) and relevant BI Standard Operating Procedures. The study protocol has been reviewed and approved by the respective Institutional Review Board/Independent Ethics Committee and competent authority according to national and international regulations. Prior to patient participation in the trial, thorough study information was provided and written informed consent was obtained from each patient (or the patient’s legally accepted representative) according to ICH GCP and to the regulatory and legal requirements of the participating country.
Biomarker panel
Circulating biomarkers of oxidation [8-iso-prostaglandin F2α (8-iso PGF2α)], endothelial function [asymmetric dimethylarginine (ADMA), endothelin-1] and inflammation [C-reactive protein (CRP), interleukin-6 (IL-6) and tumour necrosis factor-α (TNF-α)] were drawn in blood sample at fasting state and analysed centrally (Quintiles laboratories Europe or North-America) using established methods [8-iso PGF2α in plasma (ACE 8-isoprostane EIA kit), ADMA in plasma (ADMA enzyme-linked immunosorbent assay [ELISA] Alpco), endothelin-1 in plasma (Biomedica Endothelin ELISA), CRP in serum (Tina-quant CRP), IL-6 in plasma (QuantiGlo® Human Immunoassays) and TNF-α in plasma (QuantiGlo Human Immunoassays)].
Cognitive assessment
Participants underwent an automated comprehensive cognition test battery [CAmbridge Neuropsychological Test Automated Battery (CANTAB)] assessing five domains (www.cantab.com). CANTAB has been validated and applied in several studies involving individuals with type 2 diabetes.
7
In order to be able to detect subtle cognitive differences, one subtest per domain was selected that was sensitive and best represented that domain.
8
For psychomotor speed, this was
Statistical analysis
Depending on the distribution of data, participant characteristics were described using mean ± standard deviation or medians and interquartile ranges for continuous variables and proportions for categorical variables. Biomarkers or cognitive variables that did not follow a normal distribution were log-transformed. Cognitive variables were converted into standardised z-scores prior to the analyses. Separate linear regressions were performed between each of the six biomarkers and each of the five cognitive variables. All analyses were adjusted for age, gender and years of formal education. When associations were observed between biomarkers and cognition, we explored if they were influenced by HbA1c, vascular risk factors (risk factors defined according to the modified NCEP-ATP-III criteria, 9 plus smoking), macrovascular disease, use of anti-inflammatory drugs or interrelations between biomarkers, in separate linear regression analyses. As all analyses were of exploratory nature, no corrections for multiple testing were made.
Results
Of the 37 participants (92% men, mean [±standard deviation] age 67 ± 9 years), 27% had a history of coronary artery disease and 22% a history of cerebrovascular disease. Mean years of formal education were 11.4 ± 4.4. Median (interquartile range) HbA1c was 6.9% (6.6%; 7.3%) [52 mmol/mol (49; 56)], diabetes duration was 8.3 (3.4; 11.1) years and MMSE was 29 (27; 30) (Supplemental Table S1).
Higher 8-iso PGF2α was significantly associated with reduced mental flexibility and attention (standardised regression coefficient −0.47,
Association between circulating biomarkers of oxidation, endothelial function and inflammation and cognition.
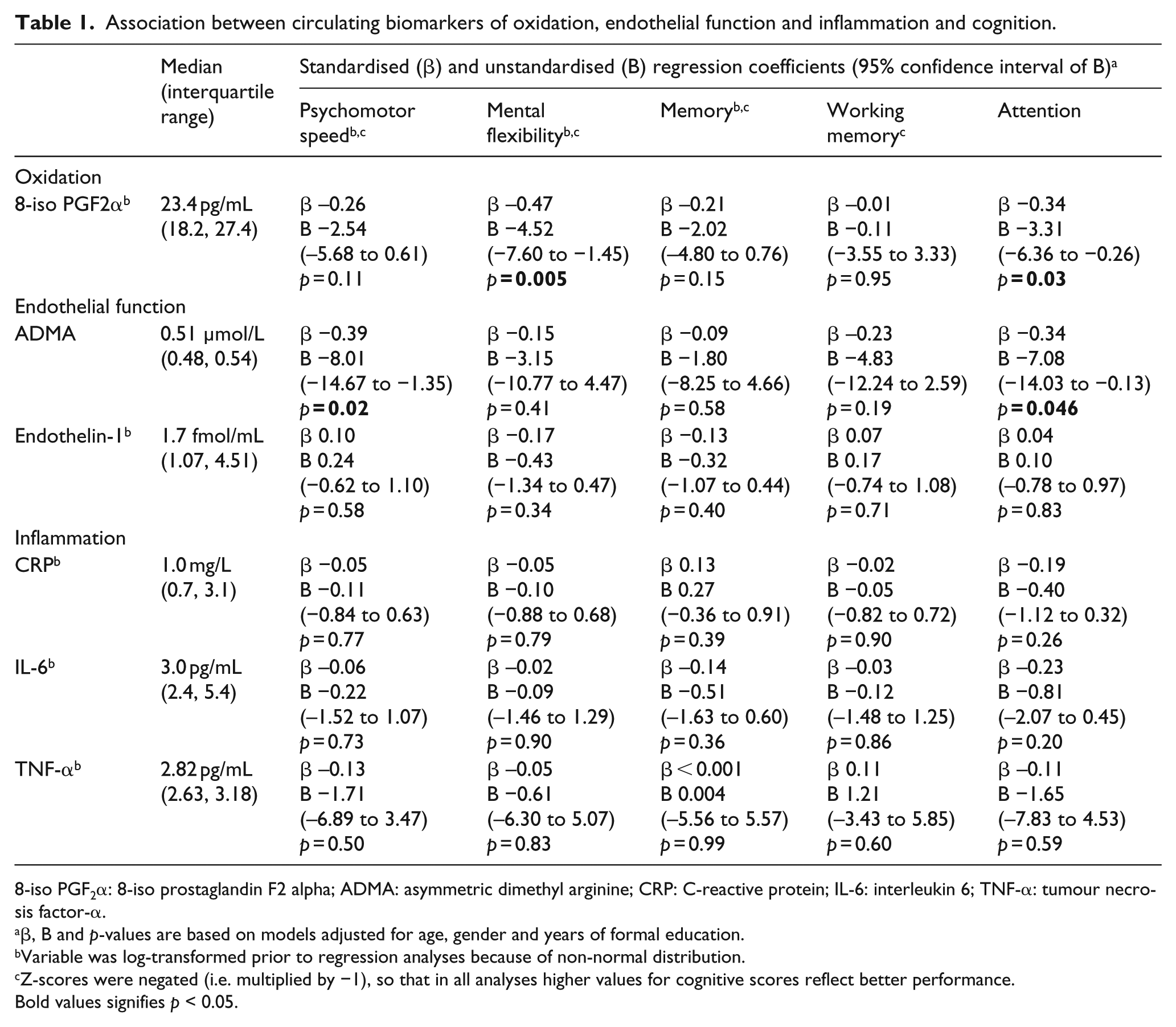
8-iso PGF2α: 8-iso prostaglandin F2 alpha; ADMA: asymmetric dimethyl arginine; CRP: C-reactive protein; IL-6: interleukin 6; TNF-α: tumour necrosis factor-α.
β, B and
Variable was log-transformed prior to regression analyses because of non-normal distribution.
Z-scores were negated (i.e. multiplied by −1), so that in all analyses higher values for cognitive scores reflect better performance.
Bold values signifies
Discussion
Elevated circulating biomarkers of oxidation (8-iso PGF2α) and endothelial function (ADMA) were associated with reduced psychomotor speed, mental flexibility and attention, in individuals with type 2 diabetes. No associations were observed between biomarkers of inflammation and cognition.
This appears to be the first study in type 2 diabetes to assess associations of biomarkers of oxidation and endothelial function with cognition. Long-standing type 2 diabetes is associated with oxidative stress. 2 Increased oxidative stress induces endothelial dysfunction via lipid peroxidation and nitric oxide inactivation in the endothelium. 10 Endothelial dysfunction is considered a key mechanism involved in the development of diabetes-associated vascular complications. 2 In the present study, oxidative stress and endothelial dysfunction were associated with worse cognitive performance for psychomotor speed, mental flexibility and attention. These cognitive functions are represented by multiple brain areas, which suggests that oxidative stress and endothelial dysfunction might be related to cognitive impairment in type 2 diabetes through a diffuse, global brain injury.
Previously, outside the context of type 2 diabetes, associations between increased oxidative stress and endothelial dysfunction and worse cognition have been reported in non-demented persons,3,4 although the literature is not extensive. One study demonstrated an association between elevated 8-iso PGF2α and reduced memory, reporting a medium effect size (Cohen’s
In the current study, biomarkers of inflammation were not associated with cognition. Limited by the sample size, the current study cannot detect small effects. Nonetheless, the observed effect sizes for inflammation were much smaller than those of the other biomarkers. Indeed, a previous cross-sectional study in non-demented patients with type 2 diabetes reported similar small standardised regression coefficients (−0.07 to −0.17) for IL-6 and TNF-α and cognition, but not for CRP. 11 Our findings on inflammation are consistent with a previous prospective study in type 2 diabetes which reported that CRP levels were not associated with cognitive decline within 6 years. 12
Our study has some additional limitations. The small sample size limited the number of adjustments that could be performed, thereby reducing the ability to discern the independence of the associations. Furthermore, as the analyses were of exploratory nature, no correction for multiple testing were made, which may have given rise to chance findings. However, the largest standardised regression coefficients were all negative, implying likely a true result. Moreover, the study had a cross-sectional design, which limits inferences about causality. Finally, the large proportion of men in our sample may affect the external validity.
In conclusion, elevated circulating biomarkers of oxidation and endothelial function were associated with worse performance on cognitive domains that depend on global information processing. Oxidative stress and endothelial dysfunction in type 2 diabetes may be a modifiable cause of cognitive impairment and deserves further investigation.
Supplemental Material
20181228_Supplemental_figure_1 – Supplemental material for Oxidative stress and endothelial dysfunction are associated with reduced cognition in type 2 diabetes
Supplemental material, 20181228_Supplemental_figure_1 for Oxidative stress and endothelial dysfunction are associated with reduced cognition in type 2 diabetes by Onno N Groeneveld, Esther van den Berg, Odd Erik Johansen, Sven Schnaidt, Karin Hermansson, Bernard Zinman, Mark A Espeland and Geert Jan Biessels in Diabetes & Vascular Disease Research
Supplemental Material
Supplemental_Table_S1_DVR – Supplemental material for Oxidative stress and endothelial dysfunction are associated with reduced cognition in type 2 diabetes
Supplemental material, Supplemental_Table_S1_DVR for Oxidative stress and endothelial dysfunction are associated with reduced cognition in type 2 diabetes by Onno N Groeneveld, Esther van den Berg, Odd Erik Johansen, Sven Schnaidt, Karin Hermansson, Bernard Zinman, Mark A Espeland and Geert Jan Biessels in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
O.N.G. wrote the first draft of the manuscript. O.E.J. and G.J.B. designed the study. O.N.G., E.v.d.B., S.S. and G.J.B. drafted the statistical analysis plan. O.E.J. and K.H. participated in the study coordination. The authors are fully responsible for all content, were involved at all stages of manuscript development and have approved the final version. G.J.B. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. The authors thank Anna Passera, Boehringer Ingelheim, for her work on validating the datasets and to Gudrun Wallenstein, Boehringer Ingelheim, for review.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: O.E.J. is a clinical programme leader, K.H. was previously a global clinical operation lead and S.S. a responsible trial statistician, all employed by Boehringer Ingelheim. B.Z., M.E. and O.E.J. are members of the CAROLINA executive steering committee. B.Z. and M.E. have received consultancy support from Boehringer Ingelheim. G.J.B. receives research support from Boehringer Ingelheim. O.N.G. and E.B. have no competing interests to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The CAROLINA-cognition biomarkers substudy is funded by the Boehringer Ingelheim & Eli Lilly and Company Diabetes Alliance. The study was carried out in compliance with the protocol, the principles laid down in the Declaration of Helsinki, version as of October 1996, in accordance with the ICH Harmonised Tripartite Guideline for Good Clinical Practice (GCP) and relevant BI Standard Operating Procedures. The study protocol has been reviewed and approved by the respective Institutional Review Board/Independent Ethics Committee and competent authority according to national and international regulations. Prior to patient participation in the trial, thorough study information was provided and written informed consent was obtained from each patient (or the patient’s legally accepted representative) according to ICH GCP and to the regulatory and legal requirements of the participating country.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
