Abstract
Objective:
the objective of this article is to evaluate the role of siRNA-silenced TRPC6 on podocyte autophagy and apoptosis induced by AngII.
Methods:
mouse podocyte cell lines were cultured in vitro. The apoptosis rates of each group were detected using flow cytometry. The expression of LC3-II protein and changes in distribution were detected by confocal laser, and the western blot protocol was employed for detection of protein expression of LC3-II.
Results:
AngII-injured podocyte had a significant increase in apoptosis, while silencing TRPC6 could decrease the apoptosis induced by AngII. Autophagy remarkably increased after AngII injury. While silencing TRPC6 stabilized the autophagy expression, AngII could activate the autophagy of podocyte. Autophagy-associated protein LC3-II expression increased after AngII injury. The LC3-II mRNA and the protein level could be down regulated by 3-MA. The silencing of TRPC6 could stabilize the autophagy expression.
Conclusion:
the data suggest that AngII can lead to podocyte injury. Autophagy may have beneficial effects in preventing the progression of proteinuria. This study provides some new clues for further exploring the occurrence of podocyte injury and the development mechanism of proteinuria.
Introduction
Autophagy is a lysosome-dependent degradation pathway existing in eukaryotic cells, which run through the whole process of normal cell growth, development, and pathophysiology, and is an important way for biological degradation of intracellular proteins to complete organelle transformation and to maintain homeostasis.1–3 Autophagy plays a critical role in the growth and development, cell differentiation, and the environmental stress response of the organism, and its physiological function is involved in normal growth and the development process of cells and maintaining metabolic balance.4–6 In the kidney, there is a close relationship between autophagy and glomerular podocyte injury as well as proteinuria, and autophagy regulation may become a new measure for proteinuria prevention. TRPC6 was a newly found virulence gene which caused familial focal segmental glomerulosclerosis (FSGS). The product of this gene is a non-selective cation channel protein which is located in podocyte. The dysfunction of the podocyte structure and the function is essential for glomerular kidney diseases. TRPC6 plays a very important role in maintaining the structure and function of podocyte according to recent research. As the research of autophagy increases, new knowledge on the role of autophagy in podocyte impairment and proteinuria genesis has been partially revealed; the autophagy regulation could be a new mechanism for proteinuria prevention and treatment. In this study, we investigated the effects of TRPC6-silenced by siRNA on the podocyte autophagy induced by AngII. The molecular mechanism of this process was elucidated from the autophagy view.
Material and methods
Podocyte culture
Vitro immortalized mouse podocyte cell lines (MPC5) were authorized for the US Mundel Professor, Professor of Pediatrics, Peking University First Hospital, Ding Jie donation. Under the license conditions, podocyte recovery was at 33°C, contained 10 U/mL recombinant mouse γ-interferon (PEPRO Tech Company), and used a RPMI 1640 medium containing 10% FBS (Hyclone company) (Gibco company) culture proliferated. Then, it was inoculated into collagen coated I (BD Biosciences Inc.) flasks or six-well plates; 12 induced differentiations of the cells were cultured for 14 days at a mature podocyte at 37°C so they could be used for the experiments. All experiments were repeated three times.
Podocyte transfection
TRPC6 siRNA double strands were synthesized by Shanghai Jima Biotech and Engineering Co. Sequences were as follows: plussense:ATTGATCCTGGATCTAGAGTG and antisense:ATTGATCCTGGATCTAGAGTG. Transfection was conducted by oligofectamine equipment (Invitrogen). The culture medium was changed to a common RPMI 1640 medium after a transfection of approximately 6 h and cells were incubated at 33°C. siRNA transfection was monitored under a fluorescence microscope after 24 h and the efficiency was detected by flow cytometry. WB was taken out to measure TRPC6 protein expression 48 h after transfection. All cell groups in the experiments were designed with three replicates.
Western blotting
Cells in the 25 cm2 culture bottle were first washed twice by a pre-cooled PBS buffer, then they were incubated with a 100 µl cell disruption buffer (0.1% Triton X-100,75 mM NaCl, 5 mM EDTA, 50 mM Tris (pH 7.0), 1 mM PMSF, 1 μg/ml aprotinin, 1 μg/ml leupeptin, 1 μg/ml pepstatin) for 30 min. Then, they were centrifugalized at 12000 g and supernatant was taken for total protein determination by BCA method. The equivalent of 75 μg total protein was loaded for SDS PAGE and transferred to PVDF membrane. WB was then carried out using GAPDH as the interior reference. Quantitative analysis was done by Image J software and each independent experiment was repeated three times.
Laser scanning confocal microscopy measurement
FITC and TRITC fluorescence was monitored under laser scanning confocal microscopy (Olympus). Eye lens that were 10x and 60x (immersion objective) objective lens were used. Operations were carried out in a single blind situation where the operator did not know the detail of the sample grouping. Photography was taken with a SHARP-2000 and all experiments had three replicates.
FCM detection of podocyte apoptosis
According to the equipment operation instructions, cell cycle was synchronized for 24 h first. Nearly confluent cells were then grouped. Cells (5×105/ml) were collected and suspended with 200 µl of binding buffer, and then 10 µl of Annexin-v-FITC and 5 µl of PI were added for a 15 min light cycle avoiding incubation at room temperature. After incubation, 300 µl of binding buffer was added for FCM detection; the experiment had three replicates.
Statistical analysis
All data were analyzed by SPSS16.0 statistics software. Data were shown as mean ± SD. A t-test with p-value smaller than 0.05 was applied for differentiation with statistical significance.
Results
The optimization of podocyte transfection
Podocyte was transfected by GFP labeled NC-siRNA; cells with green fluorescence were counted under a fluorescence inverted microscope. The transfection efficiency was calculated by dividing the total cells in the same field of view (Figure 1(a) and (b)). Flow cytometry and the western blot protocol were also employed for detection of the transfection efficiency (Figure 1(c) and (d)).

(a) Podocyte after 48 h transfection (x200); (b) expression after 48 h transfection (x200); (c) cellular GFP expression after transfection; and (d) the expression of TRPC6 gene after siRNA transfection, 24 h.
Podocyte apoptosis monitored by FCM
The podocyte apoptosis ratio of the control group was 2.36 ± 0.50%, while podocyte apoptosis was significantly increased to 14.46 ± 0.40% after AngII treatment compared to the control group (p < 0.05). Mock vector transfection did not affect the apoptosis compared to the control group (p > 0.05). TRPC6-silenced podocyte had an apoptosis ratio of 4.33 ± 0.45% which was not statistically significant to the control group (p > 0.05). When TRPC6-silenced podocyte was treated with AngII, apoptosis was significantly decreased compared to the control group (p < 0.05) (Figure 2).

(a) FCM photograph of PI and AnnexinV-FITC double stained podocyte apoptosis and (b) the bar chart of PI and AnnexinV-FITC double stained podocyte apoptosis.
Autophagy detected by TEM
The ultrastructure showed that little autophagy expressed in podocyte. The structure and distribution of organelles, nucleus, and chromatosome were normal in the control group. AngII-impaired podocyte showed ultrastructural changes and the autophagy increased significantly. A great quantity of independent double membrane structures emerged, gradually extended, and bent. These included autophagosomes containing cytoplasm, lysosome, and other organelles. It is significantly different from the control group (p < 0.01) (Figure 3). While TRPC6-silenced podocyte was not sensitive to AngII, the autophagy was significantly lower than the AngII-treated group (p < 0.05).

(a) The TEM detection of autophagosome expression in different groups and (b) the bar chart of autophagosome expression transformed from TEM data.
Detection of expression of autophagy-associated protein LC3-II by western blotting
LC3-II expression in normal podocyte (control group) was very low as the immunoblotting band was slender. The same results were observed in the 3-MA treated group. When podocyte was impaired by AngII, the LC3-II expression increased remarkably compared to the control group (p < 0.01). TPRC6-silenced podocyte hardly changed the LC3-II level after AngII treatment; the same situation was observed in the 3-MA inhibited group. The expression difference was statistically significant compared to the control group (p < 0.05) (Figure 4).

LC3-II expression variation detected by western blotting.
LC3-II expression after TRPC6 silencing detection by laser confocal microscope
The expression of LC3-II was fairly weak in normal podocyte under a confocal microscope, while the AngII-injured podocyte had a remarkable increase in LC3-II expression compared to the control group (p < 0.01). When TRPC6 was silenced by siRNA, the expression of LC3-II became stable and statistically significant to the control group (p < 0.01) (Figure 5).
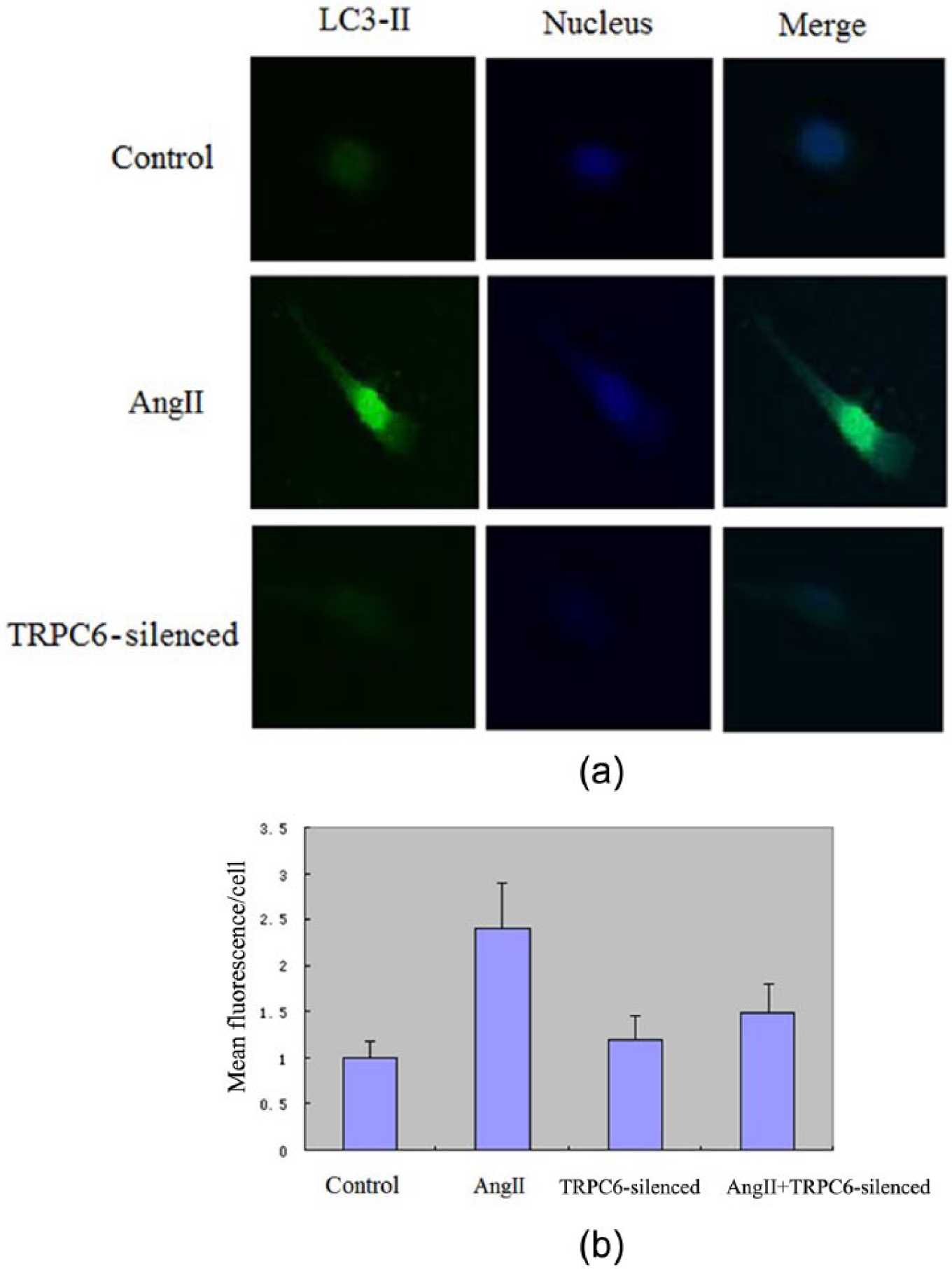
(a) Confocal images of LC3-II expression in different groups and (b) the bar chart of LC3-II expression.
Discussion
Autophagy is a lysosom-dependent degradation pathway widely existing within eukaryotic cells as a self-protection mechanism in response to pessimal stimulation, and the function of autophagy is not only involved in surrounding and isolating the damaged organelles, but more important in transporting the surrounded materials to lysosomes for degradation and producing amino acids, ATP, and other substances to maintain the energy metabolism cycle of the body. 7 Research demonstrated that a mouse lacking in autophagy-related gene 5 (Atg5) in podocyte cells would be more susceptible to glomerular diseases, and the decrease of autophagy within podocyte cells would delay the update of the cytoplasmic constituents, resulting in a mismatch protein accumulation, further leading to the formation of the inclusion bodies and the dysmorphic cells. The regulation of autophagy was becoming the new mechanism for proteinuria prevention and cure. TRPC6 was a newly discovered virulence gene which caused familial FSGS. It is a non-selective cation channel protein which is specifically located in glomerular podocyte. The dysfunction of the podocyte structure and the function is essential for glomerular kidney diseases. TRPC6 played a very important role in maintaining the structure and function of podocyte according to recent research.8, 9
Research has shown that cis-platinum can cause apoptosis and necrosis of renal tubular epithelial cells (RTEC) and that it was a dose and time dependent course. The cell death pattern differed when time and concentration were varied.10–13 Man Jiang et al. 13 observed that when RTEC was injured by hypoxia, the level of autophagy increased and finally caused cell apoptosis. Their research also suggested that autophagy had a protective effect on RTEC apoptosis. Pallet et al. 14 found that ciclosporin could impair RTEC through ER stress and autophagy had a protection. Yang et al. 15 found in the model of RTEC impaired by cis-platinum that LC3, Beclin1, and Atg5 were all increased when the autophagy formed. Autophagy inhibitor and autophagy gene Beclin1, Atg5-silenced could accelerate the activity of caspases and RTEC apoptosis. Conversely, improving the level of autophagy could inhibit RTEC apoptosis which suggested that autophagy could protect against poisoned kidney injury. Periyasamy-Thandavan et al. 16 found that an anti-apoptosis protein such as Bcl-2 could inhibit autophagy in a cis-platinum interfered mouse kidney. The same effect was found with 3-MA. Lau 17 observed that cis-platinum could induce RTEC necrosis with engorged kytoplasm and cause the membrane integrity to disappear. Man Jiang et al. 13 elucidated that 3-MA or Beclin-1, Atg5-silenced could induce the hypoxia impairment of podocyte leading to cell apoptosis. The research suggested that autophagy had a protective role in kidney ischemic and hypoxia injury. Suzuki et al. 18 studied the autophagy during human kidney transplant and rat testes ischemia-reperfusion. The autophagy-associated protein LC-3 expression increased. Thus, ischemia could induce autophagy and reperfusion could accelerate autophagysome formulation. Matsui et al. 19 found that autophagy mainly functioned as a protective factor during kidney ischemia when reperfusion was carrying on. Chien et al. 20 found that the transfection of the Bcl-xL gene could remarkably relieve RTEC autophagy and improve kidney failure in the ischemia-reperfusion mouse model. Sato et al. 21 defined two types of autophagy in the nephronia pathological section under TEM: Type I and Type II autophagysome. The study of IgA nephronia patients found eight cases of Type I autophagysome. Of these cases, 37.5% were FSGS for the first time pathologic diagnosis. The other eight cases were Type II autophagysome, of which 87.5% were FSGS for the first time pathologic diagnosis. The research suggested that the type of autophagysome was closely related to the prognosis. However, there were few reports about podocyte-associated molecules in autophagy.
TRPC6 was recently found as a virulence gene causing familial FSGS. 22,23 It is a six-transmembrane protein of which the cation channel was formed by the fifth and sixth transmembrane domains, while the NTD and CTD were located intracellularly. The TRPC family can be divided into four subgroups: TRPC1, TRPC2, TRPC4/5, and TRPC3/6/7. TRPC3/6/7 has 75% homogeneity which constitutively formed the functional channel. 24 TRPC6 had a low expression in rat glomcrulus and both the resistance vessel and arteria renalis expressed. TRPC6 expression in podocyte in the rat neohridial tissue.25, 26 The endothelial cell also had weak expression. Immune-gold labeling showed that TRPC6 was located in the podocyte primary and secondary foot process, and mainly distributed near the SD molecules. TRCP6, together with SD molecules such as podocin and nephrin, composed the signaling complex to maintain the structural and functional integrity of the foot process and SD molecules. TRPC6 played an important role in maintaining the glomerular filtration barrier. Moller et al. 27 showed that TRPC6 mRNA was remarkably lower in minimal lesion and MGN patients than in healthy adults. Reiser et al. 28 transfected podocyte with GFP-labeled TRPC6. These suggested that TRPC6 played a very important role in proteinuria genesis and development.
Our study showed that AngII-injured podocyte had a significant apoptosis increase, while the silencing of TRPC6 could decrease the apoptosis induced by AngII. Autophagy in normal podocyte was weak, but it remarkably increased after AngII injury. While the silencing of TRPC6 stabilized the autophagy expression even when faced with AngII injury, it suggested that the silencing of TRPC6 could inhibit the podocyte apoptosis when injured by AngII. AngII could activate the autophagy of the podocyte. Autophagy-associated protein LC3-II expression increased after AngII injury. The LC3-II mRNA and protein level could be down regulated by 3-MA. The silencing of TRPC6 could stabilize the autophagy expression. In short, the silencing of TRPC6 could inhibit the autophagy abnormal expression caused by AngII impairment and thus protect the autophagy and inhibit podocyte apoptosis to safeguard the podocyte.
Conclusions
TRPC6 participated in the molecular biological mechanism of podocyte autophagy induced by AngII. Autophagy played a very important role in podocyte impairment repair. Autophagy regulation is a complicated process which involved multiple mechanisms. Our study elucidated the podocyte impairment protection from the autophagy point of view which provided new insight and theory basis for kidney disease prevention and cure.
Footnotes
Acknowledgements
Yu Shengyou designed the study and performed the acquisition and analysis of the data. Yu Shengyou also helped to draft the paper and was involved in revising the paper. All authors read and approved the final paper.
Conflict of Interests
None declared.
Funding
This work was supported by grants from the National Natural Science Foundation of China (Grant 81273205).
