Abstract
Objective:
In this study, we investigated the molecule mechanisms of podocyte injury and proteinuria and the protective effects of losartan.
Methods:
This study set up three groups: a control group; an Ang II group (Ang II 10–6 mol/l, Sigma); and a losartan group (losartan 10–6 mol/l, Sigma). We used RT-PCR assay to detect TRPC6 mRNA expression, and Western blot to detect TRPC6 protein expression.
Results:
TRPC6 overexpression was the basic change of podocyte injury and proteinuria occurrence. Losartan can treat podocyte injury and proteinuria induced by Ang II via downregulation of TRPC6 in podocytes.
Conclusion:
These findings maybe provide an ideal drug target for the diagnosis and treatment of acquired glomerular diseases.
Introduction
TRPC6 is a slit diaphragm (SD)-associated protein in podocytes involved in regulating glomerular filter function, which is closely related to proteinuria and positioned in the structure protein molecules of SDs. Proteinuria is a common feature of kidney dysfunction of glomerular origin and is itself a risk factor for renal disease. 1 There is a growing body of experimental and clinical literature showing that podocyte number is a critical determinant for the development of proteinuria and glomerulosclerosis.2,3 Angiotensin II (Ang II) is a key contributor to glomerular disease and may regulate TRPC6 expression in nonrenal cells. We demonstrate that Ang II regulates TRPC6 messenger RNA (mRNA) and protein levels in cultured podocytes and that Ang II infusion enhances glomerular TRPC6 expression. 4 Losartan is widely used for the treatment of a variety of glomerular diseases characterized by podocyte injury and proteinuria, including membranous nephropathy, minimal change disease, focal segmental glomerulosclerosis (FSGS) and lupus nephritis. However, the signaling mechanisms underlying the antiproteinuria effects of losartan have not been well defined.
The aims of this study were to determine whether losartan regulates TRPC6 expression in podocytes, to gain insight into the downstream effectors of losartan/TRPC6-mediated signaling, and to evaluate the molecular mechanism of experimental proteinuric disease and the special beneficial effects of losartan on podocyte injury.
Materials and methods
Cells in culture
Podocytes were cultured at 33°C in Roswell Park Memorial Institute (RPMI)-1640 containing 10% fetal bovine serum (FBS) (Gibco, Gaithersburg, MD, USA), 100 U/ml penicillin/streptomycin, and 10 U/ml of mouse recombinant γ-interferon (PEPRO Tech, London, UK) and then shifted to 37°C for differentiation by removal of γ-interferon which had typical character of mature podocyte after two weeks. In the studies described below, all experiments were performed in growth-restricted podocytes.
Experimental design
In order to examine the effect of losartan on Ang II-induced podocyte injury, podocytes grown under growth-restrictive conditions for 14 days were incubated with media containing 10% FBS in the presence of 10–6 mol/l losartan (Sigma Chemical Co.). Losartan was not removed until the end of each experiment. To determine if losartan reduced a range of injuries, the following experiments were undertaken after one hour of losartan incubation. To determine the mechanisms of podocyte injury induced by Ang II, we exposed podocytes to Ang II (Sigma Chemical Co.) at a concentration of 10–6 mol/l. Following a 48-hour incubation with Ang II in the presence or absence of losartan, we observed and harvested at eight, 24 and 48 hours, respectively. The experiments were all repeated three times.
Reverse transcription-polymerase chain reaction (RT-PCR) analysis
Total RNA was extracted from podocytes using Trizol reagent according to the manufacturer’s instructions, and the RNA concentration was determined after the sample was dissolved in diethylpyrocarbonate-treated water. Isolated RNA (1 μg) of each sample was subjected to RT by using Rever Tra Ace (TOYOBO Co.) according to the manufacturer’s protocol. The resulting cDNA (3 μl) was used for PCR amplification. The sequence-specific primers were designed and synthesized by Shanghai Invitrogen Biotechnology Co, Ltd. Primers used and TRPC6 upstream and downstream primers respectively were as follows. Forward: 5-GTTAATTGCGATGATCAATAGTT-3. Reverse:5-GACTTGGTACAAGATTGAAGG-3, product length being 143 bp. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) upstream and downstream primers were as follows. Forward: 5-GGTGAAGGTCGGTGTGAACGGAT-3. Reverse: 5-CCACTTTGCCACTGCAAATGGCAG-3, product length being 118 bp. The PCR amplification was started with two minutes of denaturation at 94ºC, which was followed by 34 cycles (for GAPDH, 30 cycles) of denaturation at 94ºC for 30 seconds, annealing at 55ºC for 30 seconds (GAPDH, 55ºC for 30 seconds) and polymerization at 72ºC for 75 seconds (GAPDH, 30 seconds). The final extension lasted seven minutes at 72ºC and then ended at 4ºC. PCR products (5 μl) were separated on 1% ethidium bromide-stained agarose gels and later scanned with a gel imaging system. Each independent experiment was repeated three times.
Western blot analysis
Podocytes were lyzed in buffer containing 1% Tritonx-100, 150 mM NaCl, 1 mM ethylenediaminetetraacetic acid (EDTA), 50 mM Tris-HCl (pH 7.7), 1 mM NaF, 1 mM NaVO3, and a protease inhibitor cocktail (Sigma Chemical Co.). Seventy-five micrograms of total protein was loaded to run 8% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and the gel was set up for transfer protein to nitrocellulose membranes (Sigma Chemical Co.). Then, the membranes were rinsed in Tris-buffered saline with 0.02% Tween-20 (TTBS), followed by immersion in 5% low-fat milk. Subsequently, the membranes were incubated with rabbit anti-TRPC6 antibody (Sigma Chemical Co.) and mouse anti-GAPDH antibody (Sigma Chemical Co.). After rinsing three times with TTBS, the membranes were incubated with horseradish peroxidase (HRP)-conjugated goat anti-rabbit or mouse immunoglobulin G (IgG) (Sigma Chemical Co.) for 45 minutes at room temperature and then developed using ECL chemiluminescence reagent (Sigma Chemical Co.). The specific protein bands were scanned and quantitated using densitometry in relation to the GAPDH by the Western Blotting Detection System (GE Healthcare, Chalfont St Giles, UK). We repeated each Western blot analysis using protein from three different and separate experiments.
Immunostaining
TRPC6 was fixed with 4% paraformaldehyde, then permeabilized and blocked with 0.3% TritonX-100 and 5% bovine serum albumin. The primary antibody, rabbit anti-TRPC6 antibody (Sigma Chemical Co.), was applied for overnight at 4ºC. Fluorescein isothiocyanate (FITC)-conjugated goat antirabbit or mouse IgG (Sigma Chemical Co.) and the nuclei dye Hoechst were used for 45 minutes at room temperature. Finally, the coverslips were mounted and images were taken by an immunofluorescence microscope (Zeiss, Germany). To determine the percentage of the cells in which RPC6 is localized in nuclei, we counted at least 200 nuclei in triplicate in each experiment.
Statistical analysis
Data were reported as mean±SD with n equal to the number of experiments. Statistical evaluation was performed using a one-way analysis of variance (ANOVA) (two-sided test), followed by least significant difference (LSD) (equal variances assumed) or Dunnett’s T3 (equal variances not assumed) for post hoc test between two groups, and also using the nonparametric tests (Mann-Whitney U-test) as a post-test. Values of p< 0.05 were considered statistically significant.
Ethics considerations
All of the details of the study were approval by the institutional review board of Guangzhou First People’s Hospital and Guangzhou Medical University, therefore our ethics committee waived the need for consent in China.
Results
Effect of losartan on Ang II-induced podocyte changes
Podocytes were observed and photographed under inverted microscope. The relative area of podocytes was calculated by Image J and SPSS 13.0. The cell bodies and nucleus of Ang II-induced podocytes were significantly decreased. The cell bodies, which connected to each other between cells, stretched out like branches. Foot processes appeared to show retraction, and the area of Ang II-induced podocytes was significantly reduced to 75% at eight hours (p < 0.05); foot processes were obviously retracted, and the cell bodies were reduced to 46% at 24 hours (p < 0.01); part of foot processes disappeared or were lost at 48 hours. However, after losartan treatment, the area of podocytes was significantly greater at eight, 24 and 48 hours (Figure 1); the difference was significant (p < 0.05). Based on these findings, we developed the hypothesis that Ang II-induced injury may be prevented by losartan.

Ang II-induced podocytes changes at different time points (inverted-phase contrast microscope ×200). (a), (d), (g) The control group at eight, 24 and 48 hours, respectively (foot processes and the connection between podocytes are intact); (b) Ang II-induced at eight hours; (c) losartan-treated at eight hours; (e) Ang II-induced at 24 hours; (f) losartan-treated at 24 hours; (h) Ang II-induced at 48 hours (foot process retracted and lost, cell interconnectivity disappeared) and (i) losartan-treated at 48 hours (foot processes and the connection between podocytes are still preserved). Ang II: angiotensin II.
Effect of losartan on Ang II-induced TRPC6 mRNA expression
Our studies showed that TRPC6 mRNA expression is very low under normal circumstances. TRPC6 mRNA expression did not change significantly at Ang II-induced eight hours. But TRPC6 mRNA expression significantly increased at 24 and 48 hours (*p < 0.05); TRPC6 mRNA expression did not show significant changes at losartan-treated eight hours (Table 1 and Figure 2); the TRPC6 mRNA expression was significantly decreased at 24 and 48 hours (*p < 0.05).
TRPC6 mRNA levels at different times.

mRNA: messenger RNA; Ang II: angiotensin II. Compared with the control group. ap < 0.05.

Compared with the control group, incubation of the podocytes with Ang II (10–6 mol/l) markedly increased TRPC6 mRNA levels at 24 and 48 hours. Losartan prevented the TRPC6 mRNA increase induced by Ang II and the difference was statistically significant (p < 0.01). Ang II: angiotensin II; mRNA: messenger RNA.
Effect of losartan on Ang II-induced TRPC6 protein expression
Western blot analysis showed that TRPC6 and GAPDH, respectively, have a specific band at 36 kd and 106 kd. Under normal circumstances, podocytes have a certain amount of TRPC6 protein expression (Table 2). Compared with the control, TRPC6 protein expression did not change significantly at Ang II-induced eight hours, but was higher at 24 and 48 hours (*p < 0.05); TRPC6 protein expression did not significantly change at losartan-treated eight hours; decreased at 24 hours (Figure 3); and became normal (*p < 0.05). Protein expression significantly decreased at 48 hours (*p < 0.05).
TRPC6 protein levels at different times.

p < 0.05. Ang II: angiotensin II.
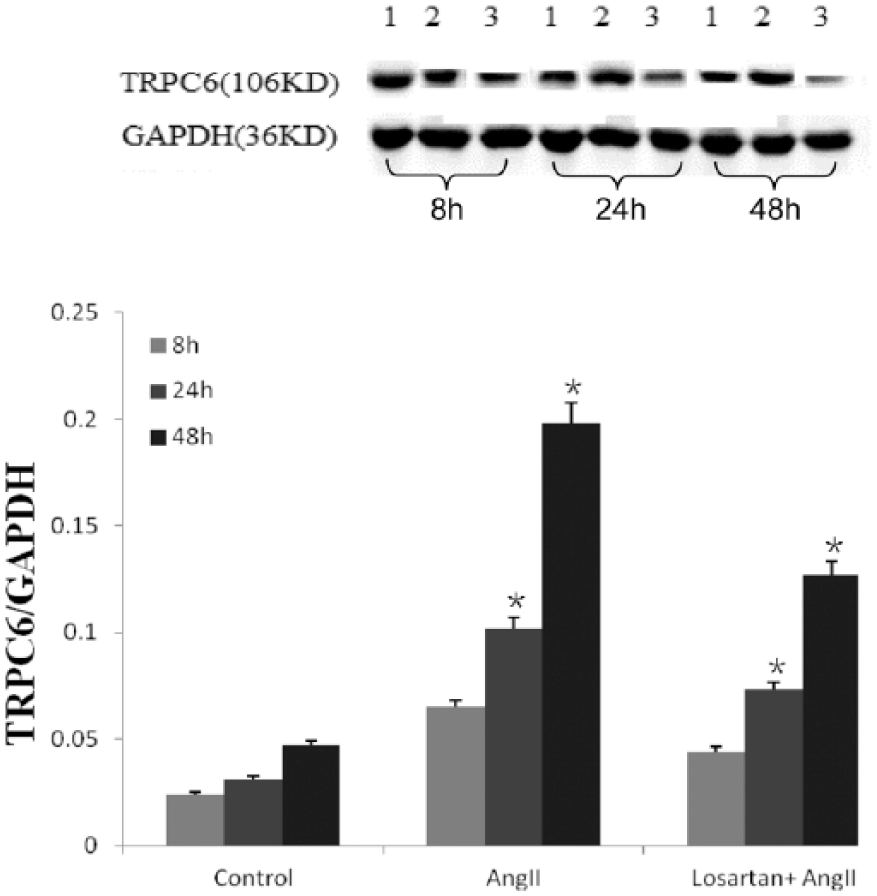
The Western blot band of TRPC6 and GAPDH at different time points.
Effect of losartan on Ang II-induced TRPC6 distribution changes in podocytes
TRPC6 was linear and evenly distributed in the control, little in the cytoplasm. At Ang II-induced 24 hours, TRPC6 was not continuously distributed along the cell membrane and increased in cytoplasm; at 48 hours, TRPC6 distribution increased in the cell membrane, and part of the cell membrane lost or gathered into a granular, widely distributed area in the cytoplasm. But, after losartan treatment, TRPC6, which significantly improved at different time points, is more homogeneously distributed in the plasma membrane and became normal (Figure 4).
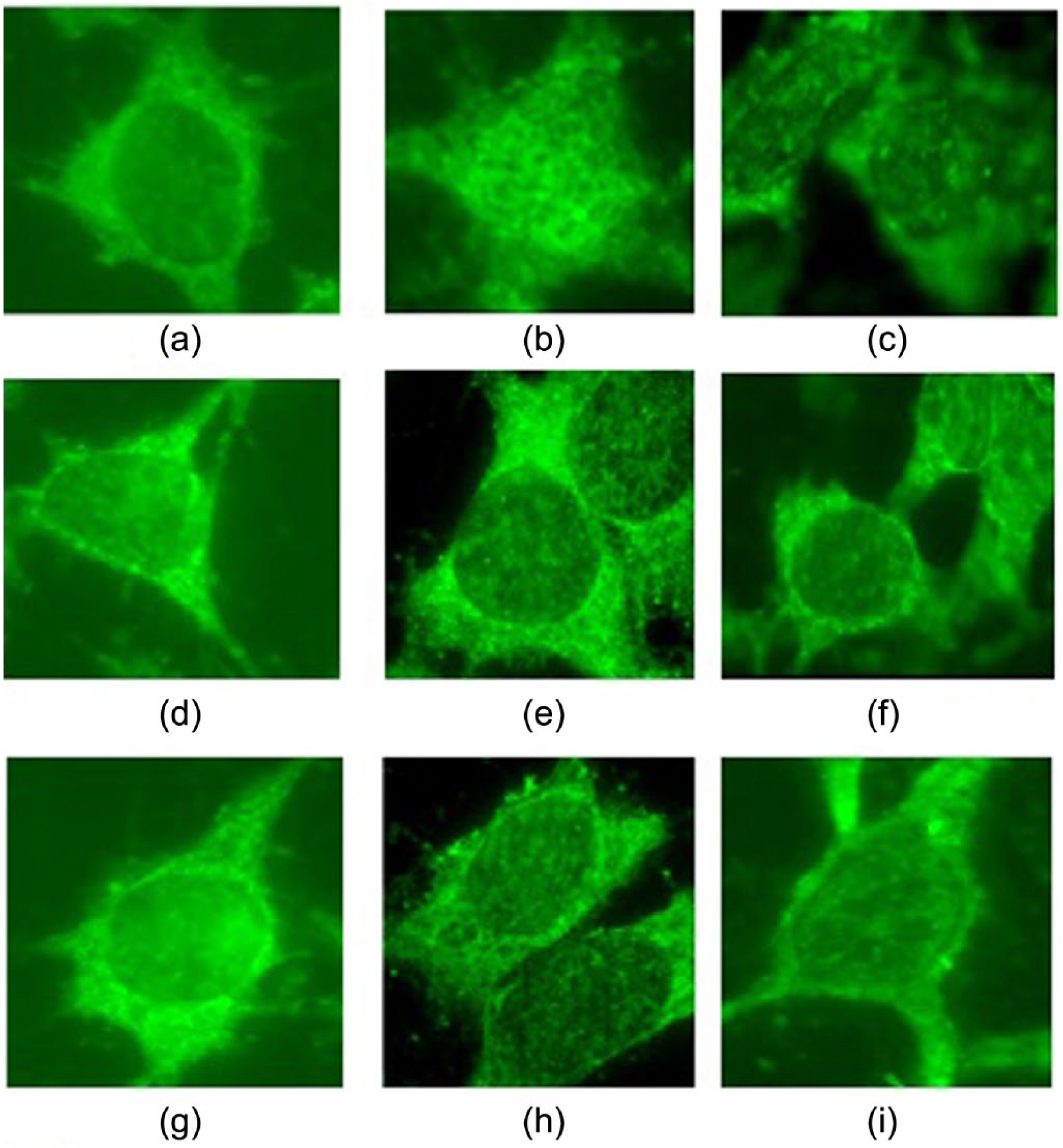
Effects of losartan and Ang II on the distribution and protein expression of TRPC6 at different time points (fluorescence microscope ×400).
Discussion
The glomerular capillary filtration barrier consists of endothelial cells, glomerular basement membrane (GBM), and podocytes linked by the SD. The SD is a complex of interconnected proteins that connect podocyte foot processes, which provide both physical linkage and a signaling unit that regulates podocyte behavior. 1 Damage to the podocyte is of crucial importance in the pathophysiology of proteinuria. 5 TRPC6 is a member of the large transient receptor potential superfamily of non-selective cation channels. Further studies showed that there was interaction between TRPC6 protein and podocyte SD molecules. Together they constitute the diaphragm hole complex; 6 this finding links podocyte structural proteins and ion channels.
TRPC6 is a receptor-operated cation channel that can be activated by Ang II through stimulation of the angiotensin type 1 receptor (AT1R) and secondary generation of diacylglycerol.6–8 Ang II is a key contributor to the pathogenesis of glomerular disease, and the antiproteinuric effects of angiotensin-converting enzyme (ACE) inhibition and AT1R blockade are undisputed.9,10 In nonrenal cells, Ang II activates TRPC6 currents and enhances TRPC6 transcription.11,12 TRPC6 overexpression appears crucially involved in the pathogenesis of podocyte and glomerular damage, leading to glomerulosclerosis and proteinuria in FSGS. Currently, TRPC6 was less reported in the kidney tissue, and the results were not consistent. Facemire et al. 13 found that TRPC6 has a weak expression in glomerular, and is also expressed in the kidney artery and renal resistance vessels. TRPC6 mainly expresses in the glomerular and tubular cells of human renal specimens. 6 TRPC6 has a little expression in the endothelial cells, and is localized in the podocyte primary and secondary foot processes, especially the majority distribution in the vicinity of the SD.4,14 TRPC6 located in the SD of the glomerular podocyte TRPC6, nephrin, podocin and CD2AP were co-localized in podocytes. They together form a signaling translation complex to maintain the integrity of the structure and function of foot process and SD. In podocyte molecules, TRPC6 played an important role in maintaining the glomerular filtration barrier, but the specific mechanism is not very clear, pending further study.
Based on our results, TRPC6 had no significant changes in its protein expression after Ang II stimulation at eight hours and losartan intervention at eight hours. Its protein expression was significantly higher after Ang II stimulation at 24 and 48 hours; TRPC6 protein expression were significantly decreased after losartan treatment at 24 and 48 hours. Immunofluorescence staining results showed that TRPC6 showed a linear, uniform distribution in the podocyte cell membrane surface in the control group. A little was also distributed in the cytoplasm; it was mainly a dotted line-like distribution along the membrane after Ang II stimulation at 24 hours, and cytoplasmic distribution increased. At 48 hours, TRPC6 distribution increased in the cell membrane. Part of the cell membrane distribution missing, gathered into a granular, widely distributed area within the cytoplasm; and after losartan treatment, TRPC6 was more homogeneously distributed in the cell membrane at different time points. The distribution in the cytoplasm was significantly weaker than in the stimulation group. The difference with other podocyte molecules was that TRPC6 expression strengthened in the podocytes of Ang II damage, the distribution increased, and expression decreased after losartan treatment, and the distribution reduced. This is the special position of TRPC6 as a cation channel protein.
The use of angiotensin receptor blockers (ARBs) has been promoted to slow the progression of chronic renal disease and chronic nondiabetic renal disease.15,16 The data supporting this mechanism mainly come from the remnant kidney model in rats.17,18 Ang II is the major effector molecule of the renin-angiotensin system (RAS). Activation of the RAS exacerbates progression to end-stage renal disease, and interruption of the RAS markedly retards proteinuria and advancement to nephrosclerosis.19,20 Ang II injures podocytes through various channels in the early stages of diabetic nephropathy (DN), for instance, regulation of hiatus membrane-associated molecules of nephrin expression resulting in the occurrence of proteinuria. At present, our study suggests that TRPC6 feels the damage signal after Ang II-induced podocyte injury, and then activates downstream molecules, causing the stimulation signal transduction in cells; cells respond to retraction and disappearance of the foot process, abnormal expression and distribution of TRPC6, which leads to imbalance of TRPC6 channels and function. Then changes in the podocyte filtration rate cause the occurrence of proteinuria, and the losartan may make downstream molecules that have been activated inactive and podocytes restored their original structure by blocking TRPC6 signal channels, thus maintaining the structural and functional integrity of the SD by stabilizing TRPC6 expression, and playing a protective role of podocytes and antiproteinuria.
Our results have important potential clinical implications. TRPC6 overexpression may be one of the fundamental changes in podocytes leading to proteinuria and impairment of renal function. Perhaps blocking the TRPC6 channel may be a candidate for diagnosis and therapeutic targeting of renal diseases. Our study suggests that TRPC6 participated in the signal transduction mechanisms of losartan treating Ang II-induced podocyte injury. Losartan stabilized podocyte TRPC6 expression through binding to its receptor, and blocked the TRPC6 signaling pathway. Our finding underlines the crucial role of TRPC6 in the pathogenesis of podocyte injury and proteinuria, and provides an ideal drug target for the diagnosis and treatment of acquired glomerular diseases.
Footnotes
Acknowledgements
The authors thank the Central Laboratory of the First Hospital Affiliated to Ji’Nan University for technical help.
Author contributions
Meng-Yu and SY Yu designed the study and performed the acquisition and analysis of data, helped to draft the paper and was involved in drafting and revising the paper. All authors read and approved the final paper.
Conflict of interest
None declared.
Ethical considerations
All of the details of the study were approved by institutional review board of the First Hospital Affiliated to Ji’Nan University. So,our ethics committee waived the need for consent in China.
Funding
This study was supported by grants from the Science and Technology Planning Project of Guangdong Province, China (#2013B040404005).
