Abstract
MicroRNAs plays important roles in the progression of diabetic nephropathy (DN) and podocyte injury. This study aimed to investigate the role and regulation mechanism of miR-1187 during the development of DN and podocyte injury. The content of miR-1187 in podocytes was up-regulated under high glucose (HG) treatment and increased in kidney tissue of db/db mice (DN model mice) compared with control db/m mice. The administration of miR-1187 inhibitor could decrease podocyte apoptosis induced by HG and attenuate the decline in renal function and reduce proteinuria as well as glomerular apoptosis in db/db mice. Mechanistically, miR-1187 could inhibit the autophagy level in HG-exposed podocytes and glomerulus of DN mice. Moreover, miR-1187 inhibitor could reduce HG-stimulated podocyte injury and autophagy flux inhibition. The mechanism may depend on autophagy. In conclusion, targeting miR-1187 may be a new therapeutic target for improving the high glucose damage of podocytes and the progression of DN.
Introduction
Diabetic nephropathy (DN) is a complication of diabetes and the main cause of end-stage renal disease.1–4 Currently, treatment mainly depends on blood glucose and hypertension control. 5 Despite the huge investment, many patients eventually developed diabetes nephropathy,6,7 suggesting the urgent need for new therapeutic strategies and therapies. Podocytes, the important constituent cells of glomerular filtration barrier, play important roles in DN progression. The loss of podocytes is one of the earliest glomerular morphological changes.8,9 Revealing the regulatory mechanism of podocyte injury and inhibiting it has become an important treatment strategy for improving DN.10–12
MicroRNA (miRNA) is a kind of noncoding single stranded RNA molecule with about 22 nucleotides long. Studies showed that compared with healthy people, miR-204 content in the kidney of patients with hypertension and DN decreased. 13 Anti-miR-204 injection would aggravate proteinuria and fibrosis. 14 miR-15b-5p induced mesangial cells apoptosis by targeting BCL-2 under HG. miR-15b-5p has the potential to predict kidney injury in DN. 15 miR-21 can also regulate the expression of PTEN and lead to the death of podocytes. 16 Inhibition of miR-21 16 and applying miR-93 mimics can improve proteinuria. 17 One study showed that compared with control, the expression of several miRNAs in DN mice changed, including miR-1187, miR-195, and so on. 18 Additionally, it was reported that miR-1187 content in hepatitis B infected mice is disturbed; 19 miR-1187 targets caspase 8 and increases apoptosis; 20 miR-1187 mimics inhibits osteoblast differentiation and mineralization. 21 However, miR-1187 is rarely reported in kidney and podocyte.
In this project, the role of miR-1187 in high glucose induced podocyte injury and in DN and the mechanisms were investigated. We report that miR-1187 inhibitor reduced apoptosis of podocytes and improved DN, likely through modulation of autophagy. The study suggests that inhibition of miR-1187 may be a new therapeutic strategy to improve DN.
Materials and methods
Cell culturing
Mouse podocyte MPC5 was purchased from ATCC, cultured with type I collagen (GIBCO, 17100-017), and 20 U/ml IFN-γ (Tonglihaiyuan, GMP-TL105) and RPMI-1640 medium (Hyclone, SH30809.01 B) with 10% FBS at 33°C for amplification. For inducing differentiation, they were cultured at 37°C in RPMI-1640 medium containing 5% FBS but without IFN-γ for 10–14 days. Thereafter, they were synchronized: being cultured in RPMI-1640 medium with 0.2% FBS for 24 h and they were used in the subsequent research.
CCK-8 assay
The differentiated MPC5 cells were inoculated in 96 well plates at 5 × 104/mL, and cultured overnight under 5% CO2 and37°C. Then, podocytes were transfected with miRNA inhibitor NC (genepharma) or miR-1187 inhibitor (genepharma), and then the medium was replaced with low sugar medium (5.5 mM D-glucose) or high sugar medium (30 mM D-glucose). After 72 h, CCK8 (MCE, HY-K0301) was added at 10 μL/well, and the plate was placed in the cell incubator. After 2 h, the OD450 nm was measured with microplate reader.
qRT-PCR
Total RNA was extracted with Trizol reagent. The first strand cDNA was synthesized with Entilink™ 1st cDNA synthesis Kit (ELK Biotechnology, EQ003). Enturbo™ SYBR Green PCR Supermix Kit (ELK Biotechnology, EQ001) was used for the qPCR detection on Stepone™ Real-Time PCR instrument (Life Technologies). Reaction conditions were: 1 cycle: 95°C (10 min), 40 cycles: 95°C (30 s), 60°C (30 s). The expression of miR-1187 was calculated by 2−ΔΔCT method with U6 as the internal reference. mmu-miR-1187 primer was 5′-GCCGAGTATGTGTGTGTGTA-3′, miRNAs universal downstream primer was 5′-CAGTGCGTGTCGTGGAGT-3′, U6 primers were 5′-CGCTTCGGCAGCACATATAC-3′ and 5′-AAATATGGAACGCTTCACGA-3′.
Flow cytometry assay
The cells were processed following the manual of Annexin V-FITC/PI (propidium iodide) apoptosis detection kit (Nanjing Kaiji Biology, KGA108). Cells were digested and collected with EDTA-free pancreatic enzyme (procell, PB180228). The cells were washed with PBS twice, then centrifuged at 800 g for 5 min, and 1 × 105 cells were collected. A 500 μL Binding Buffer was added and gently blown into the single-cell suspension. After the mixture was mixed with 5 μL Annexin V-FITC, 5 μL Propidium Iodide was added and mixed, and the reaction was carried out for 10 min at room temperature and avoided light. Then it was detected by flow cytometry (BECKMAN, CytoFLEX). Annexin V-FITC(Ex = 488 nm, Em = 530 nm) green fluorescence was detected by FITC channel. PI red fluorescence (Ex = 488 nm, Em ≥ 630 nm) was detected by PI channel.
Western blot
The operations followed the standard protocol with minor modifications. Briefly, podocytes or kidney tissue samples were processed to produced the protein extracts; the protein samples were separated by SDS-PAGE and transferred to PVDF membrane; the PVDF membrane was washed; the primary antibody was diluted with blocking solution (GAPDH, Hangzhou Xianzhi Biology Co., Ltd., AB-P-R 001, 1:1000; caspase3, Abcam, AB184787, 1:1000; p62, Affinity, DF2985, 1:1000; LC3, Cell signaling, 3868, 1:1000) was dropped on the PVDF membrane; the HRP labeled secondary antibody (HRP labeled Sheep anti rabbit secondary antibody, Wuhan Boshide Bioengineering Co., Ltd., BA1054, 1:10000) was then dropped on the PVDF membrane; the working droplets in ECL Kit (Beyotime, P0018) were dropped on the PVDF membrane; the membrane scanning was completed by scanner (Canon, K10486); the gray value was analyzed by BandScan software.
Immunofluorescence
The procedure followed the standard protocol. Briefly, mature podocytes were inoculated onto the coverslip, and fixed; 0.5% Triton X-100 (PBS) was added; normal goat serum was added; LC3 antibody (Abcam, AB192890, 1:200) was added; fluorescent secondary antibody (Goat Anti-Rabbit IgG (H + L) FITC-conjugated, Affinity, S0008, 1:200) was added; DAPI (Beyotime, C1002) was added; the coverslip was incubated in darkness and then observed under confocal microscope.
Construction of AAV-miR-1187 inhibitor (e.g. vector, sequence, and so on.)
The procedure followed the standard protocol. Briefly, the synthesized DNA fragments was dissolved; 2 μL DNA primer fragments (miR-1187 inhibitor-F: 5′-GATCCTTACACACATACACACACACATATTCAAGAGATTCCAGTATGGTCGGAGTCTTTTTTA-3′; miR-1187 inhibitor-R: 5′-AGCTTAAAAAATTACACACATACACACACACATATCTCTTGAATTCCAGTATGGTCGGAGTCG-3′) were added, annealed at 100°C and naturally cooled to room temperature; the annealed products were diluted and linked to the plasmid vector pAAV; the sequencing primer was U6-F (5′-TACGATACAAGGCTGTTAGAGAG-3′) for verification; the standard packaging procedure was followed, in brief, HEK-293T cells were inoculated into dishes; the medium was changed into 15 mL high glucose DMEM +10% FBS + P/S medium; two clean EP tubes were marked as A and B; 1 mL CPT buffer A was added to tube A; 20 μg plasmid, 20 μg pHelper, 10 μg pAAV RC, 100 μL CPT buffer B and other reagents were added into tube B; the solution of tube B were added dropwise to tube A; the solution was gently mixed by pipetting up and down with a 1 mL pipette for 10 times; the solution stand at room temperature for 30 min, then evenly added dropwise into a dish; the dish was cultured at 37°C, 5% CO2; the solution was changed into 20 mL high glucose DMEM +10% FBS + P/S medium; the cells were gently mixed by pipetting up and down with a 10 mL pipette; the cells and culture supernatants were collected into a new 50 mL centrifuge tube, freezed and thawed repeatedly at 80°C/37°C for 3 times, centrifuged at 3000 rpm for 10 min; the supernatant was collected, filtered with 0.45 μm PVDF filter, purified by virus purification column, and finally eluted with 1 mL buffer; the elute was the packaged virus.
Animal experiments
The male 12 weeks db/m mice and db/db mice in this study were purchased from Beijing Weitonglihua Experimental Animal Technology Co., Ltd. (Beijing, China). All mice were fed in a 12 h light/dark circulation chamber, and the temperature was controlled at 24 ± 0.5°C. All experimental protocols and procedures for this study were approved by the institutional Animal Care and Use Committee of Medical College of Soochow University and complied with the guidelines for the care and use of experimental animals of the National Institutes of Health. Mice were randomly divided into 4 groups with 5 mice in each group. The specific groups were as follows: (1) db/m mice injected with PBS from tail vein; (2) db/db mice injected with PBS from tail vein; (3) db/db mice injected with AAV-miRNA inhibitor NC from tail vein; (4) db/db mice injected with AAV-miR-1187 inhibitor from tail vein. The dosage of adeno-associated virus tail vein injection was 1.5 × 109 pfu per animal, and the mice were injected every 4 weeks. The experiment ended at week 20 and the materials were taken for related detection.
Detection of serum creatinine, urea nitrogen and 24 h urinary protein
Creatinine (serum) colorimetric assay kit (Cayman, 700460) was used to detect the serum creatinine content of mice in each group. The content of serum urea nitrogen of mice in each group was detected by urea nitrogen test box (mlbio, ml076479). After 24 h, the urine of mice in each group was collected and the 24 h urine protein content of mice in each group was detected by mouse urinary protein (UP) ELISA Kit (mlbio, ml037585).
Microscopic examination of renal tissue
The kidney tissues from db/m mice or db/db mice were fixed with formaldehyde, dehydrated and made transparent, and then embedded in paraffin. After that, the renal tissue was sliced into 5 μm thick pieces by Leica pathological slicer. Then, it was stained with hematoxylin and eosin (H and E). After the air drying and sealing with neutral glue, the slice was inspected under microscope.
TUNEL assay
TUNEL (terminal deoxynucleotidyl transferase-mediated dUTP-X nick end labeling) staining was performed using In Situ Cell Death Detection Kit as per manufacturer’s instructions (Roche, 12156792910). The slices were treated with protease K working solution (20 μg/mL) at 37°C for 20 min. 100 μL Equilibration Buffer and TdT enzyme reaction solution was added dropwise successively. After the incubation in the 37°C wet box for 60 min, the slices were put into the staining cylinder with 2× SSC solution to terminate the reaction, and placed at room temperature for 15 min. After PBS immersion at room temperature, 100 μL Streptavidin HRP (streptavidin horseradish peroxidase) solution was added to the slices, and then 100 μL DAB chromogenic solution was added. The slide was sealed after Mayer hematoxylin counterstaining. The slide was observed under a microscope and the images were taken.
Transmission electron microscope observation
The renal tissues of each group were placed in sufficient 2.5% glutaraldehyde fixing solution for more than 48 h. Then the samples were fixed with 1% osmium tetroxide at 4°C for 2 h. After the dehydration and embedding, ultra-thin sections with a thickness of 60 nm were sliced and stained with sodium acetate and lead citrate. Finally, the ultrastructural changes of glomerular podocytes in each group were observed under TEM.
Statistical analyses
The statistical software Prism 9.0 was used for data analyses. All data were expressed as mean ± standard deviation (mean ± SD). LSD method (least significant method) in one-way ANOVA was used for pairwise comparison between different groups. p < 0.05 was considered as statistically significant.
Results
The miR-1187 expression is up-regulated in DN model and high glucose podocyte injury model
In order to clarify the role of miR-1187 in podocyte injury in DN and its effect on DN, we constructed DN podocyte injury model and DN mouse model, and detected the changes of miR-1187 expression in cell model and kidney tissue in animal model. CCK-8 experiments showed that 72 h high glucose treatment significantly decreased MPC5 cell activity (Figure 1(a)), indicating that the high glucose induced DN podocyte injury model was successfully constructed. After that, we detected the content of miR-1187 by qRT-PCR. High glucose treatment significantly increased miR-1187 in podocytes (about 7 times, p < 0.01; Figure 1(b)). Subsequently, we constructed DN mouse model using db/db mice. Blood samples and 24 h urine samples of 20 week old db/m mice and db/db mice were collected. It was found that compared with db/m mice, the contents of creatinine and urea nitrogen in serum of db/db mice were significantly increased (creatinine increased by about 3 times, p < 0.01; urea nitrogen increased by about 4 times, p < 0.01; Figure 1(c)). In addition, compared with db/m mice, the protein content in 24 h urine of db/db mice was significantly up-regulated (increased by about 8 times, p < 0.01; Figure 1(d)). These results showed that compared with db/m mice, the renal function of db/db mice was damaged and accompanied by proteinuria. It indicated that db/db mice could be used as DN mouse models. Then we collected kidney tissue samples of 20 week old db/m mice and db/db mice, and detected the content of miR-1187 by qRT-PCR. It was found that compared with db/m mice, the content of miR-1187 in kidney tissue of db/db mice was significantly up-regulated (increased by about 2.5 times, p < 0.01; Figure 1(e)). Cell experiments and animal experiments showed that miR-1187 content in kidney tissues and podocyte high glucose injury models of DN mice were up-regulated. Changes of miR-1187 expression in podocyte high glucose injury model and diabetic nephropathy mice kidney tissue (a) The effect of high glucose stimulation on the activity of MPC5 cells detected by CCK-8 experiment (n = 3); (b) the effect of high glucose stimulation on the expression of miR-1187 in MPC5 cells detected by qRT-PCR, with U6 as the internal reference (n = 3); (c) the differences of serum creatinine and urea nitrogen between db/m mice and db/db mice detected by kits (n = 5); (d) the difference of 24 h urinary protein content between db/m mice and db/db mice detected by ELISA kit (n = 5); (e) the expression of miR-1187 in kidney tissue of db/m mice and db/db mice detected by qRT-PCR, U6 was used as internal reference (n = 5). ** Indicates HG versus NG or db/db versus db/m, p < .01. HG: high glucose, NG: normal glucose.
miR-1187 inhibitor can alleviate podocyte high glucose injury
In order to reveal the role of miR-1187 in podocyte high glucose injury, we constructed miR-1187 inhibitor and used it to transfect podocytes in order to change the expression level of miR-1187 in podocytes and clarify the effect of miR-1187 on podocyte high glucose injury. It was found that under low glucose treatment, miR-1187 inhibitor transfection could significantly reduce the content of miR-1187 in MPC5 cells (p < 0.01). Additionally, miR-1187 inhibitor transfection could abolish the up-regulation of miR-1187 content in MPC5 cells induced by high glucose (Figure 2(a)). The results showed that exogenous addition of miR-1187 inhibitor could reduce the content of miR1187 in podocytes. Effect of miR-1187 inhibitor on high glucose injury of podocytes (b) The effects of miR-1187 inhibitor and high glucose stimulation on the expression of miR-1187 in MPC5 cells detected by qRT-PCR (n = 3); (b) the effects of miR-1187 inhibitor and high glucose stimulation on the activity of MPC5 cells detected by CCK-8 experiment (n = 3); (c) the effects of miR-1187 inhibitor and high glucose stimulation on MPC5 cell apoptosis detected by flow cytometry (n = 3); (d) the effects of miR-1187 inhibitor and high glucose stimulation on caspase 3 shear of MPC5 cells detected by Western blot, with GAPDH as the internal reference (n = 3). The upper figure shows the experimental band diagram of Western blot, and the lower figure shows the shear quantification diagram of caspase 3 shear. ** Indicates p < .01. HG: high glucose, NG: normal glucose.
Then we used CCK-8 experiment to detect the effect of miR-1187 inhibitor on the down-regulation of podocyte activity induced by high glucose. The results showed that miR-1187 inhibitor could significantly up-regulate the activity of MPC5 cells under low glucose treatment (p < 0.01); compared with high glucose and miRNA inhibitor NC treatment group, podocyte activity in high glucose and miR-1187 inhibitor treatment group was significantly up-regulated (p < 0.01) (Figure 2(b)). Flow cytometry showed that high glucose treatment could increase the apoptosis level of MPC5 cells compared with low glucose treatment group (p < 0.01); under low glucose or high glucose treatment, miR-1187 inhibitor could reduce the apoptosis level of MPC5 cells (p < 0.01); miR-1187 inhibitor could reduce the up-regulation of apoptosis activity of MPC5 cells induced by high glucose (p < 0.01) (Figure 2(c)). The results of western blot were consistent with those of flow cytometry. It was found that high glucose treatment could increase the shear of caspase 3 in MPC5 cells (p < 0.01); miR-1187 inhibitor treatment could reduce the content of cleaved caspase 3 in MPC5 cells with low glucose or high glucose treatment (p < 0.01) (Figure 2(d)). In addition, it was also found that miR-1187 inhibitor could inhibit the increase of caspase 3 shear in MPC5 cells caused by high glucose (p < 0.01, Figure 2(d)). The results showed that the high glucose injury of podocytes was related to the increase of miR-1187 content. Inhibiting the expression of miR-1187 could reduce the level of podocyte apoptosis induced by high glucose.
miR-1187 inhibitor inhibits glomerular cell apoptosis and improves DN
In order to reveal the role of miR-1187 in the progression of DN, we constructed an adeno-associated virus expressing miRNA inhibitor NC (AAV-miRNA inhibitor NC) and miR-1187 inhibitor associated adeno-associated virus (AAV-miR-1187 inhibitor). Those two AAV vectors were injected into two groups of mice through the caudal vein, and the renal function and proteinuria of the two groups were observed. First, we obtained the kidney tissues of mice in each group and detected the content of miR-1187 in the kidney tissues of mice in each group by qRT-PCR. It was found that compared with AAV miRNA inhibitor NC injection treatment group, the content of miR-1187 in kidney tissue of db/db mice decreased significantly after AAV-miR1187 inhibitor injection treatment (p < 0.01) (Figure 3(a)). It indicated that AAV with miR-1187 inhibitor could significantly reduce the miR-1187 content in kidney tissue of DN mice. Effects of tail vein injection of AAV-miR-1187 inhibitor on proteinuria and glomerular cell apoptosis in db/db mice (a) The effect of tail vein injection of AAV-miR-1187 inhibitor on the expression of miR-1187 in kidney tissue of db/db mice detected by qRT-PCR, U6 was used as internal reference (n = 5); (b) the effects of tail vein injection of AAV-miR-1187 inhibitor on serum creatinine and urea nitrogen in db/db mice detected by kits (n = 5); (c) the effect of tail vein injection of AAV-miR-1187 inhibitor on 24 h urinary protein content of db/db mice detected by ELISA kit (n = 5); (d) the effect of tail vein injection of AAV-miR-1187 inhibitor on renal tissue lesions in db/db mice detected by HE staining (n = 5); red arrow indicates glomerular volume increases, black arrow indicates glomerular hyperplasia, and the ruler icon represents 20 μm; (e) the effect of tail vein injection of AAV-miR-1187 inhibitor on renal apoptosis in db/db mice detected by TUNEL test (n = 5); the left figure shows TUNEL staining under light microscope; the right figure shows the statistical chart of glomerular cell apoptosis level. ** Indicates p < .01. NC: negative control.
After that, serum and urine samples of mice in each group were collected. The effects of miR-1187 inhibitor on serum creatinine, urea nitrogen and 24 h urinary protein content of DN mice in each group were detected by kits and ELISA. The results showed that compared with AAV-miRNA inhibitor NC injection group, the contents of serum creatinine, urea nitrogen and 24h urinary protein in db/db mice decreased significantly after AAV-miR-1187 inhibitor injection (p < 0.01) (Figures 3(b) to (c)). It indicated that AAV with miR-1187 inhibitor can improve renal function and proteinuria in DN mice. In addition, we collected renal tissue for HE staining to observe the effect of miR-1187 inhibitor on glomerular structural lesions in DN mice. It was found that compared with db/m mice, the glomerular volume of db/db mice increased and accompanied with glomerular cell proliferation; compared with AAV-miRNA inhibitor NC injection treatment group, after AAV-miR-1187 inhibitor injection treatment, the glomerular volume and glomerular cell proliferation of db/db mice were improved (Figure 3(d)). At the same time, we detected the level of glomerular cell apoptosis in renal tissue of mice in each group by TUNEL experiment. It was found that the level of glomerular cell apoptosis in renal tissue of db/db mice was significantly up-regulated compared with db/m mice (p < 0.01); the level of glomerular cell apoptosis in db/db mice kidney decreased significantly after AAV-miR-1187 inhibitor injection (p < 0.01) (Figure 3(e)). AAV with miR-1187 inhibitor from tail vein injection can improve glomerular volume and glomerular cell apoptosis in DN mice.
miR-1187 inhibitor may increase the down-regulation of podocyte autophagy induced by high glucose
It has been found that autophagy plays an important role in the high glucose injury of podocyte in DN 32-34. We detected the effects of high glucose and miR-1187 inhibitor treatment on autophagy level in MPC5 cells by cellular immunofluorescence assay. The results showed that the number of LC3+ dot aggregates in MPC5 cells in high glucose treatment group was lower than that in low glucose treatment group; miR-1187 inhibitor transfection significantly abolished the decrease of LC3+ dot aggregates in MPC5 cells induced by high glucose (Figure 4(a)). In addition, the results of western blot showed that compared with the low glucose treatment group, high glucose stimulation could significantly reduce the ratio of LC3II/I and increase the content of p62 in MPC5 cells (p < 0.01); miR-1187 inhibitor could increase the ratio of LC3II/I and reduce the content of p62 in MPC5 cells under low glucose treatment and high glucose treatment (p < 0.01); miR-1187 inhibitor could significantly inhibit the decrease of LC3II/I ratio and the up-regulation of p62 content in MPC5 cells induced by high glucose (p < 0.01) (Figure 4(b)). The results showed that high glucose could reduce the autophagy level of podocyte, which could be reversed by miR-1187 inhibitor. Effects of miR-1187 inhibitor on autophagy level in podocyte high glucose injury model and kidney tissue of DN mice (b) (a) The effects of miR-1187 inhibitor and high glucose stimulation on the number of LC3+dot aggregates in MPC5 cells observed by IF experiment (n = 3); (b) the effects of miR-1187 inhibitor and high glucose stimulation on LC3II/I ratio and p62 content of MPC5 cells detected by Western blot (n = 3); the upper figure is the experimental band diagram of Western blot, and the lower figure is the quantitative diagram of LC3II/I ratio and p62 expression; (c) the effects of tail vein injection of AAV-miR-1187 inhibitor on the ultrastructural changes of renal podocyte in db/db mice observed under TEM (n = 5); the red arrow indicates autophagosome, the blue arrow indicates foot process fusion, and the red asterisk indicates basement membrane thickening; (d) the effects of tail vein injection of AAV-miR-1187 inhibitor on LC3II/I ratio and p62 content in kidney tissue of db/db mice detected by Western blot, GAPDH was used as internal reference (n = 5); the upper figure is the experimental band diagram of Western blot, and the lower figure is the quantitative diagram of LC3II/I ratio and p62 expression. ** Indicates p < .01. HG: high glucose, NG: normal glucose, NC: negative control.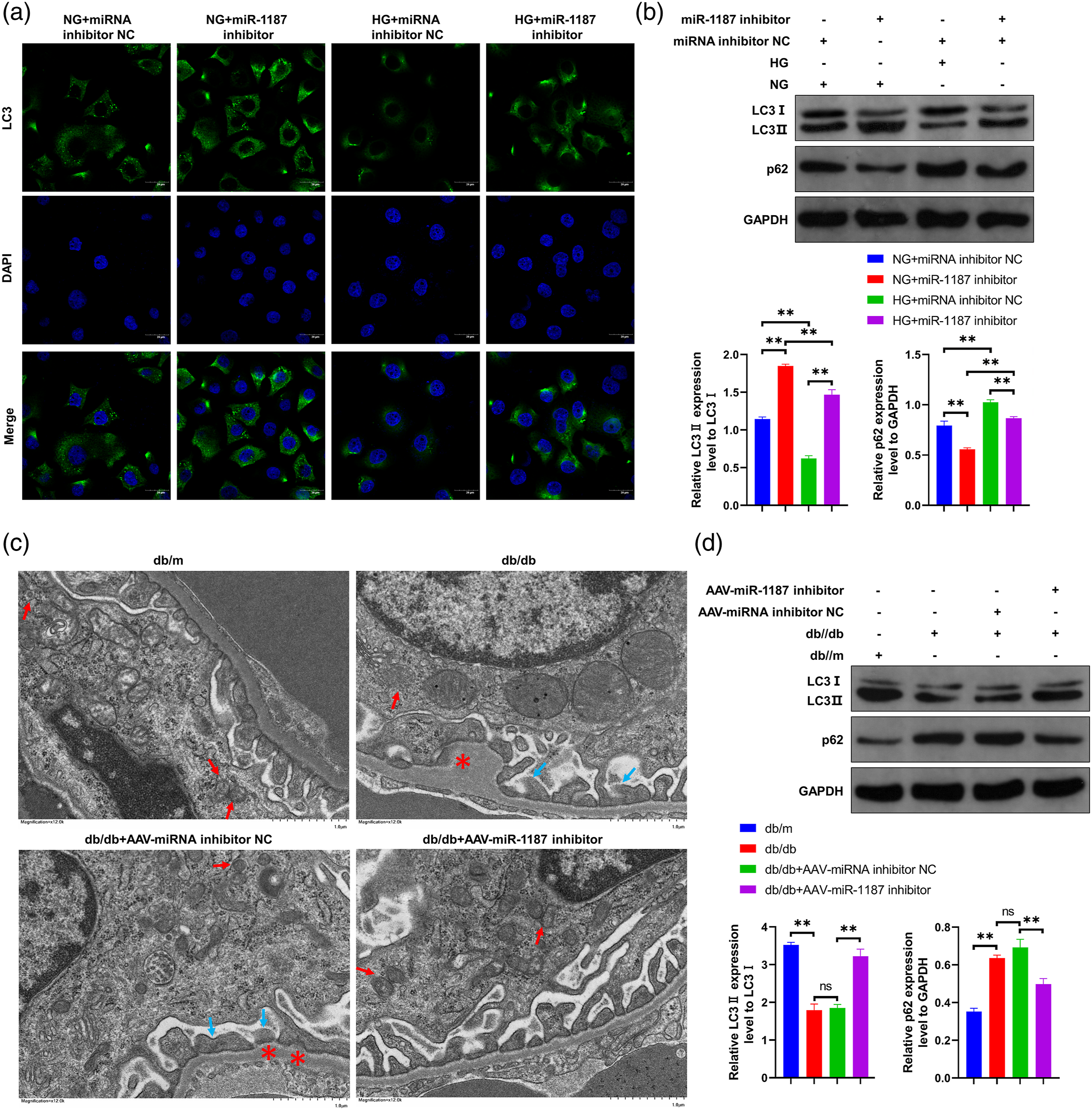
The results of TEM and the contents of LC3II/I and p62 in kidney tissue of DN mice were consistent with the results of cell experiment. Under TEM, it could be observed that compared with db/m mice, the number of autophagosomes in glomerular podocytes of db/db mice reduced (red arrow indicates autophagosome), accompanied by basement membrane thickening (* indicates thickened basement membrane) and foot process effacement (blue arrow indicates fused foot process); compared with AAV-miRNA inhibitor NC injection group, after AAV-miR-1187 inhibitor injection, the number of autophagosomes in glomerular podocytes of db/db mice kidney increased, and the thickness of basement membrane and foot process fusion were improved (Figure 4(c)). The results of western blot showed that compared with db/m mice, the proportion of LC3II/I decreased and the content of p62 increased in db/db mice (p < 0.01); compared with AAV-miRNA inhibitor NC injection treatment group, after AAV-miR-1187 inhibitor injection treatment, the proportion of LC3II/I in kidney tissue of db/db mice increased and the content of p62 decreased significantly (Figure 4(d)). It was suggested that miR-1187 inhibitor can increase the autophagy level of podocyte in DN mice and improve the podocyte fusion.
Discussion
Many studies have reported that miRNA plays an important role in DN. For example, injection of adeno-associated virus expressing miR-23a/27a can alleviate the decrease of skeletal muscle cross-sectional area and muscle function, so as to improve the renal function and fibrosis process of STZ induced DN mice. 22 Although some studies have reported abnormal expression of miR-1187 in glomerulus of STZ induced DN model mice, its regulatory role in DN is unknown. In this study, we found that the expression of miR-1187 was significantly up-regulated in the kidney of db/db mice, another DN model mouse. Tail vein injection of AAV expressing miR-1187 inhibitor could significantly reduce the content of miR-1187 in kidney tissue of db/db mice, and reduce the content of serum creatinine, urea nitrogen and 24 h urinary protein of db/db mice, so as to improve the renal function and proteinuria of DN model mice. In addition, AAV-miR-1187 inhibitor injection could improve basement membrane thickening and podocyte foot process fusion in db/db mice. It is suggested that the up-regulation of miR-1187 in renal tissue of DN plays a promotive role in the progression of DN. Inhibition of miR-1187 function, can improve the progression of DN, including renal function and proteinuria. In addition, the therapeutic potential of miR-1187 inhibitor in DN, including validation of therapeutic effects (different DN models, species), optimization of treatment methods, and side effects, still need further study.
Podocyte loss is one of the glomerular morphological changes in the early stage of DN,8,9 which can lead to impaired glomerular filtration barrier function, thereby mediating the production and aggravation of proteinuria. For example, Specific knockout of SIRT6 expression in podocytes can induce podocyte apoptosis to aggravate proteinuria in DN mice. 23 Podocyte injury in DN is closely related to the imbalance of miRNA expression. Reversing the imbalance of miRNA can improve podocyte injury and alleviate the progression of DN. For example, by constructing podocyte specific miR-93 mice or injecting miR-93 mimics, the miR-93 content in podocyte can be regulated to normal level, thereby improving the progression of DN in DN model mice. In this investigation, we found that the content of miR-1187 in podocytes was up-regulated after high glucose stimulation. It is suggested that the abnormal content of miR-1187 in renal tissue may be due to the abnormal expression of miR-1187 in podocyte cells, which needs to be further explored. In vitro experiments showed that miR-1187 inhibitor could reversibly change the content of miR-1187 in podocytes to normal level, and inhibit the down-regulation of podocyte activity and the increase of apoptosis induced by high glucose. Meanwhile, AAV-miR-1187 inhibitor could significantly improve the apoptosis level of glomerular cells in kidney tissue of db/db mice. These results suggested that miR-1187 inhibitor is likely to alleviate the progression of DN by reversing the imbalance of miR-1187 expression in podocytes and improving the damage of podocytes. Podocyte high glucose injury includes not only podocyte loss, but also podocyte EMT process and enhanced migration ability. 24 In addition, mesangial cell proliferation and fibrosis process play an important role in the progression of DN. 15 Whether miR-1187 can also regulate podocyte EMT process, migration ability, glomerular mesangial cell proliferation and fibrosis remains to be studied.
Previous studies have shown that autophagy plays an important role in the high glucose damage of podocyte in DN.23,25,26 For example, with the aggravation of proteinuria, the level of autophagy in glomerular podocytes decreased gradually; the specific knockdown of autophagy related gene Atg5 in podocytes can lead to autophagy of podocyte and loss of podocyte and aggravate proteinuria of DN. 26 Besides proteinuria was exacerbated in podocyte specific knockout SIRT6 mice, the level of podocyte autophagy decreased significantly. 23 In this study, we found that the level of autophagy decreased in podocytes stimulated with high glucose and in renal glomeruli of db/db mice. It was further confirmed that podocyte autophagy level was impaired in DN. Furthermore, it was found that miR-1187 inhibitor could increase the autophagy level of podocytes under high glucose treatment and kidney tissue of db/db mice. It suggested that miR-1187 inhibitor inhibits podocyte injury and improves the progression of DN, which is related to the reversal effect of podocyte autophagy level.
Conclusions
High glucose levels increase podocyte miR-1187 expression, which appears to inhibit autophagy, consequently leading to podocyte injury and development of proteinuria in DN. Inhibition of miR-1187 restores autophagy, limits podocyte injury and reduces progression of DN. Therefore, miR-1187 may represent an important therapeutic target for the protection of podocyte and reduction of DN risk in individuals with diabetes.
Footnotes
Author contributions
The experiments were designed by Qiang He. Bin Chen did the laboratory work. Qiang He drafted the manuscript. The final submitted manuscript was read and approved by all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Medical health Science and Technology project of Zhejiang Province (2021KY1062).
Ethical approval
All experiments were approved by the Ethics Committee of Medical College of Soochow University.
Data availability
All data generated or analyzed during this study are included in this published article and its additional files.
