Abstract
Objective:
This study investigated the protective effect of FK506 in podocytes and the correlation of TRPC6 with podocyte injury repair.
Methods:
MPC5 were cultured in vitro, parallel with the control group, an Ang II stimulation group, and an FK506 intervention group. The apoptosis rate with flow cytometry also detected TRPC6 mRNA and protein expression by RT-PCR and Western blot, and then observed the distribution of TRPC6 with indirect immunofluorescence labeling under confocal microscope.
Results:
The activities of podocytes significantly increased after FK506 intervention for 24 hours and 48 hours when compared with the Ang II stimulation group, and the apoptosis rate was markedly lower than that of the Ang II stimulation group, while in contrast to TRPC6 mRNA and protein expression.
Conclusion:
FK506 can directly act on the podocytes to inhibit Ang II-induced damage on podocyte structures and reduce the apoptosis rate of podocytes, which may be related to stabilizing TRPC6 expression and distribution in podocytes by FK506, thus maintaining the structure and function integrity of the slit diaphragm and playing a role in protecting podocytes and antiproteinuria.
Introduction
Glomerular disease is one of the main causes of chronic kidney disease (CKD) and either primary or secondary glomerular diseases, the common features of which are proteinuria occurrence and foot processes fusion or loss. Therefore, podocytes play a key role in the pathogenesis of glomerular disease. 1 In recent years, TRPC6 has been determined to be the structural protein molecule positioned in the slit diaphragm (SD) and closely related to the occurrence of proteinuria. The discovery of TRPC6 opens up a new field for us to investigate podocyte injury, repair and prevention at the molecular level. TRPC6 combines with nephrin, podocin and CD2AP molecules to form a signaling complex that maintains the structure and function integrity of the SD, while playing an important role in the mechanisms of podocyte injury and repair. FK506 is a new generation of calcineurin inhibitors (CNI) used in the treatment of kidney diseases and graft rejection by inhibiting the activation of T cells, the proliferation of dependent B cells on Th helper cells, and the expression of lymphokines such as interleukin (IL)-2, IL-3 and γ-interferon, and IL-2R, to induce immune inhibitory effects. 2 A large number of clinical studies demonstrated that FK506 significantly reduced renal tissue damage and proteinuria in various kidney diseases, and the in vitro studies3,4 also confirmed that common distribution existed between FK506 ligand FKBP and TRPC6, and bound with TRPC channel protein by the Leu-Pro (LP) dipeptide, thus we inferred that FK506 may interact with TRPC6. The application of FK506 to observe its effect on TRPC6 expression and distribution in the damaged glomerular podocytes has not been reported. In this study, an angiotensin II (Ang II)-induced podocyte injury model was used to observe the effect of TRPC6 in the process of Ang II-induced apoptosis of mouse podocytes and the effect of FK506 intervention on TRPC6 expression and distribution, to investigate the protective effect of FK506 on podocytes and the correlation of TRPC6 with podocyte injury repair.
Materials and methods
A mouse podocyte cell line (MPC5) was donated by Professor Jie Ding in the Pediatric Department of Peking University First Hospital and authorized by Professor Peter Mundel of the United States. Podocytes were cultured at 37°C in Roswell Park Memorial Institute (RPMI) 1640 medium (Gibco Company) containing 10% fetal bovine serum (FBS) (Hyclone Company) in a 5% CO2 incubation box. The experiments were divided into three groups: the control group: culture with RPMI 1640 medium containing 0.02% dimethyl sulfoxide (DMSO); the Ang II stimulation group: Ang II (10−6 mol/l) was added to stimulate the podocytes; the FK506 intervention group: Ang II and FK506 (5 μg /ml) was added and dissolved in 0.02% DMSO to interact with the podocytes at the same time.
Flow cytometry
The cells were digested with ethylenediaminetetraacetic acid (EDTA)-free trypsin, cells were collected, centrifuged and 500 μl buffer was added, then the suspended cells were transferred into a streaming tube, and 5 μl propidium iodide (PI) and 5 μl Annexin V-fluorescein isothiocyanate (FITC) were simultaneously added to keep a light-resistant reaction for 10 minutes, and further analyzed by flow cytometry.
Real-time polymerase chain reaction (PCR)
According to the operating instructions of RNAgents Total RNA Isolation System (Promega Corporation), total cellular RNA was extracted, subjected to reverse transcription (RT) and real-time PCR reaction as required by the 7500 Fast Real-Time PCR System operation, in which the melting curve chart was used for quantitative PCR, the specificity of the PCR product was tested by agarose gel electrophoresis, and the objective gene was quantitatively analyzed by the double standard curve method.
Western blot
The internal control glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and TRPC6 protein expression were respectively detected, then each objective protein expression was divided by the internal control (GAPDH) then the obtained value represented the relative objective protein content in each sample after the calibration by internal control (GAPDH). This value was further used for the comparison between the samples.
Immunofluorescence staining to check TRPC6 distribution in podocytes
The cells were seeded nearly full in a cover glass, fixed with ice cold acetone, and incubated by primary and secondary antibodies, then images was taken by using Olympus-IX81-FV1000 laser scanning confocal microscopy with FITC (green) channel at the excitation wavelength of 488 nm, and further analyzed by computer.
Statistical methods
All the data were treated with SPSS 17.0 statistical software for statistical evaluation, and the calculated results were expressed with
The results
Morphological changes of podocytes after puromycin aminonucleoside (PAN) damage and FK506 intervention
The cells were observed and photographed under inverted microscope, and the relative area of podocytes was calculated by ImageJ and SPSS. The cell areas at eight hours, 24 hours and 48 hours of the Ang II stimulation group were significantly larger than those of the Ang II group, p < 0.05; the cell areas at eight hours, 24 hours and 48 hours for the FK506 intervention group were significantly larger than those of the Ang II stimulation group, p < 0.05 (Figure 1).
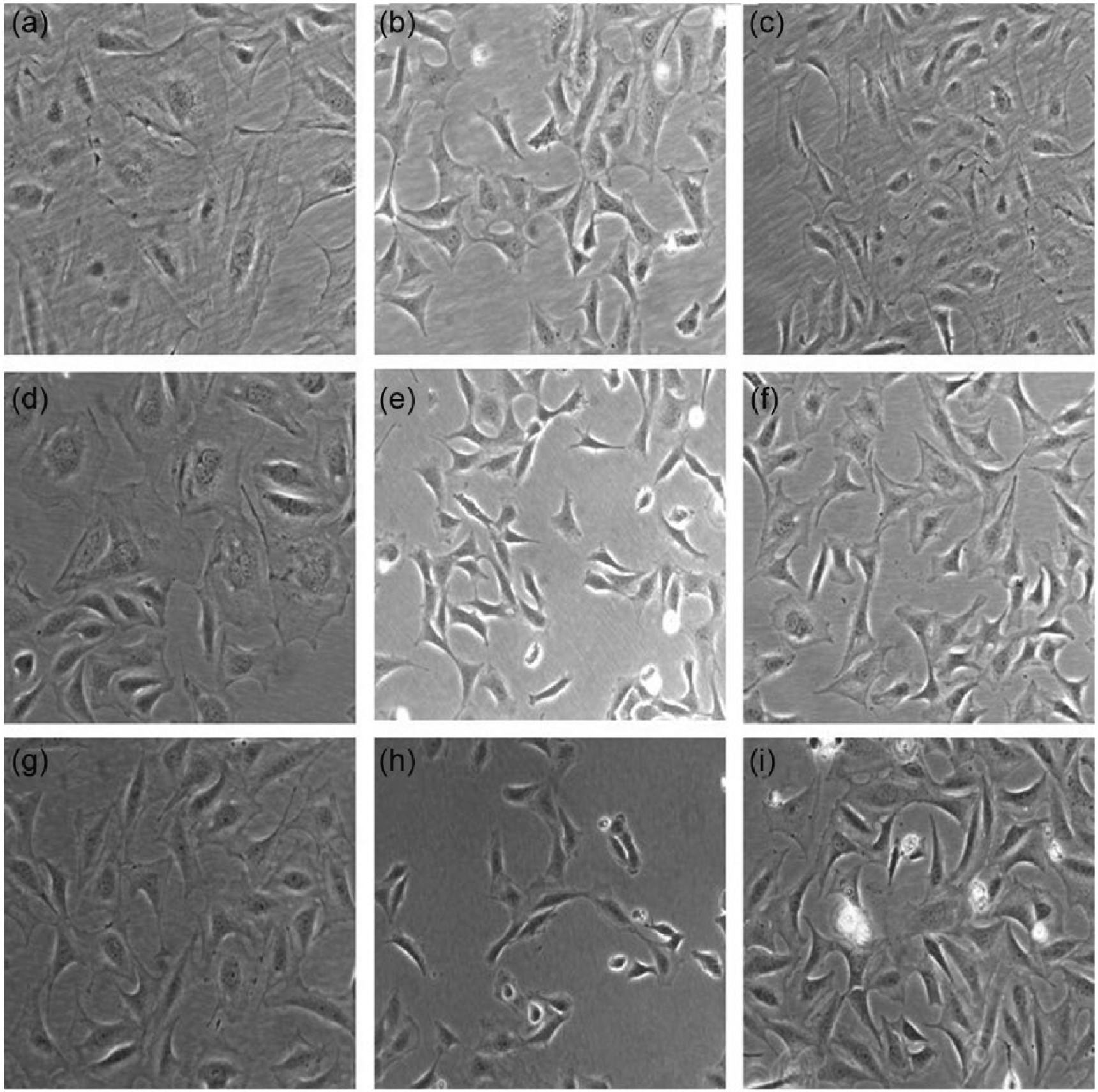
Changes of podocyte activity and apoptosis rate after Ang II damage and FK506 intervention.
The apoptosis rate of podocytes was detected by flow cytometry using a PI and Annexin V-FITC double staining apoptosis kit, and the obtained results indicated that the apoptosis rate of podocytes at eight hours for the Ang II stimulation group had no significant changes when compared with the normal control group (p > 0.05); the apoptosis rates of podocytes at 24 hours and 48 hours were obviously higher than those of the control group (p < 0.01); the apoptosis rates of podocytes at 24 hours and 48 hours of the FK506 group decreased significantly more than the Ang II stimulation group (p < 0.05) (Figure 2).

The apoptosis rate of podocytes detected by flow cytometry in the control group, Ang II stimulation group and FK506 intervention group at different time points.
TRPC6 messenger RNA (mRNA) expression changes
Under normal circumstances, podocytes have a certain amount of TRPC6 mRNA expression. Using GAPDH as an internal control, TRPC6 mRNA expression significantly increased after Ang II stimulation for 24 hours and 48 hours (p < 0.01); TRPC6 mRNA expression after FK506 intervention for 24 hours and 48 hours decreased significantly more than that in the Ang II stimulation group (p < 0.05).
TRPC6 protein expression changes
Western blot analysis showed that TRPC6 and GAPDH displayed the specific bands at the 106 kd and 36 Kd. Under normal circumstances, there is a certain amount of TRPC6 protein expression in podocytes. Compared with the control group, TRPC6 protein expression did not show significant change after Ang II stimulation for eight hours, and significantly increased after 24 hours and 48 hours (p < 0.01); compared with the Ang II stimulation group, TRPC6 protein expression in the FK506 intervention group did not change obviously after eight hours, decreased after 24 hours (p < 0.05) and significantly decreased after 48 hours (p < 0.01), then became normal (Figure 3).

Western blot band of TRPC6 of the control group, Ang II stimulation group and FK506 intervention group at different time points.
TRPC6 distribution changes in podocytes
Immunofluorescence staining showed that TRPC6 in the control group was linearly and uniformly distributed in the cell membrane, and only a little in the cytoplasm; after Ang II stimulation for 24 hours, TRPC6 was discontinuously distribution along the cell membrane and increased in the cytoplasm; after 48 hours’ stimulation, TRPC6 increased in the cell membrane, lost in part of the cell membrane, gathered into granular forms, and was widely distributed in the cytoplasm. TRPC6 at different time points after FK506 intervention was distributed more uniformly in the cell membrane, and the distribution remarkably improved in the whole cells more than before, and tended to be normal (Figure 4).

TRPC6 distribution and expression changes of the control group, Ang II stimulation group and FK506 intervention group at different time points (fluorescence microscopy, ×600).
Discussion
TRPC6 is a non-selective transient receptor potential cation channel positioned in the cell membrane. SD in podocytes constitutes the last barrier of glomerular filtration, and plays an important role in maintaining the hematuria barrier and preventing leakage of high molecular weight protein. 5 However, podocytes are highly differentiated cells, and once damaged they are hard to repair, so the pathological changes of many kidney diseases show as foot processes fusion and loss, podocytes injury and exfoliation. This study observed the effect of FK506 on the morphology, activity, and apoptosis rate of podocytes, and TRPC6 expression and distribution after podocyte damage, and investigated the protective effect of FK506 in podocytes and the correlation of TRPC6 with podocyte injury repair.
Ang II has been widely used in mechanism research of podocyte disease. It can induce foot process effacement of podocytes and lead to cytoskeletal damage and abnormal molecular expression and distribution of podocytes.6–12 In this study, we established podocyte injury models using Ang II in this part of the study by referring to the previous work of our group.13,14 The experimental results demonstrated that the Ang II stimulation group showed foot process retraction at eight hours, and the cell area is reduced to 38.9% of the control group; the foot process significantly was reduced at 24 hours’ stimulation, and the cell area declined to 5% of the control group; after stimulation for 48 hours, the cell area declined to 4.5% of the control group, and part of the podocytes showed foot process effacement and loss of intercellular junctions. After Ang II stimulation for 24 hours and 48 hours, the cell activities of podocytes significantly decreased and the apoptosis rate obviously increased; after FK506 intervention for 24 hours and 48 hours, the cell activities of podocytes increased and the apoptosis rate significantly decreased. Accordingly, Ang II stimulation can result in glomerular podocyte morphology changes, increased apoptosis, decreased podocyte numbers, and the damaged structural integrity of the SD, then the damage of the SD structure would inevitably affect the function of the filtration barrier and finally lead to the occurrence of proteinuria. FK506 improved the cell morphology, apoptosis, and podocyte number stability through inhibition of Ang II damage, and maintained the SD structure integrity of the podocytes, protected kidney tissue, and played an important role in anti-proteinuria.
Our study found that FK506 could inhibit Ang II damage on podocytes to improve cell morphology, reduce apoptosis and stabilize the number of podocytes, whereas its molecular mechanism is still not clear. In 2005, Winn’s research group 15 found that the TRPC6 mutation is the virulence gene, which caused familial focal segmental glomerulosclerosis (FSGS), and this mutation resulted in an SD dysfunction and proteinuria, eventually developed into glomerulosclerosis and renal failure. Reiser’s research group 16 found that in patients suffering from nonhereditary proteinuric renal disease, such as non-familial FSGS, membranous glomerulonephritis, minimal change nephritis, TRPC6 mRNA and protein expression were higher than those of the normal control group. Accordingly, TRPC6 mutation or its increased expression is closely related to genetic and acquired kidney diseases. Ichimura et al. 17 applied cytochalasin D as the cytoskeleton-cracking agent in in vitro differentiation and maturation of mouse podocyte MPC5, and found that TRPC6 protein expression after application of cytochalasin D significantly increased in comparison with the control group, which suggested that increased TRPC6 expression correlated with podocyte injury. Our study observed that, after Ang II stimulation for 24 hours and 48 hours, TRPC6 mRNA and protein expression were higher than those of the control group, which was consistent with the literature that Ang II can stimulate podocytes and induce abnormal intracellular TRPC6 transcription, translation and protein translocation. 18 At the same time, our experimental results also demonstrated that TRPC6 was highly expressed with mRNA and protein at the same time points, and the peak values are consistent with each other, which indicated that the effect of Ang II on TRPC6 expression in podocytes affects mRNA transcription and protein translation at the same level. Our research confirmed that TRPC6 overexpression can activate the RhoA pathway; RhoA promotes the formation of contractile actin-myosin and the stress fibers on the cell body, and their inappropriate activation may further cause structure disorder of foot process. 19 The in vivo study showed that TRPC6 mutations would eventually cause Ca2+ dysfunction and result in glomerular disease.20–22 Therefore, we proposed that FK506 can reduce the intracellular calcium overload by inhibiting TRPC6 expression in podocytes, thereby reducing the podocyte apoptosis and stabilizing the number of podocytes, and further protect the podocytes and decrease proteinuria excretion, whereas the specific molecular pathways still need to be verified by related experiments that will be the research direction in the future.
In conclusion, the results suggest that FK506 can reduce apoptosis and stabilize the number of podocytes by inhibiting TRPC6 overexpression and improving TRPC6 distribution in podocytes, as well as inhibit the damage effect of Ang II on podocytes, which may provide a theoretical basis for clinically using FK506 for kidney diseases.
Footnotes
Authors’ contributions
Yu Shengyou designed the study and performed the acquisition and analysis of data. Yu Li, Hao Zhihong and Ma Yuanyuan helped to draft and revise the paper.
Conflict of interest
None declared.
Funding
This work was supported by a grant from the National Natural Science Foundation of China (grant 81273205).
