Abstract
The Institute of Medicine estimates that 7,000 lives are lost yearly as a result of medication errors. Computerized physician and/or provider order entry was one of the proposed solutions to overcome this tragic issue. Despite some promising data about its effectiveness, it has been found that computerized provider order entry may facilitate medication errors.
The purpose of this review is to summarize current evidence of computerized provider order entry -related medication errors and address the sociotechnical factors impacting the safe use of computerized provider order entry. By using PubMed and Google Scholar databases, a systematic search was conducted for articles published in English between 2007 and 2019 regarding the unintended consequences of computerized provider order entry and its related medication errors. A total of 288 articles were screened and categorized based on their use within the review. One hundred six articles met our pre-defined inclusion criteria and were read in full, in addition to another 27 articles obtained from references. All included articles were classified into the following categories: rates and statistics on computerized provider order entry -related medication errors, types of computerized provider order entry -related unintended consequences, factors contributing to computerized provider order entry failure, and recommendations based on addressing sociotechnical factors. Identifying major types of computerized provider order entry -related unintended consequences and addressing their causes can help in developing appropriate strategies for safe and effective computerized provider order entry. The interplay between social and technical factors can largely affect its safe implementation and use. This review discusses several factors associated with the unintended consequences of this technology in healthcare settings and presents recommendations for enhancing its effectiveness and safety within the context of sociotechnical factors.
Keywords
Introduction
The cornerstone report released by the Institute of Medicine (IOM) in 1999 approximates that between 44,000 and 98,000 persons die per year as a result of preventable adverse events in hospitals. 1 Shockingly, medication errors (MEs) were estimated to account for about 7000 deaths per year; they account for 1 of 854 inpatient deaths and 1 of 131 outpatient deaths. 1 Another study considering ME-related deaths in US hospitals between 1983 and 1993 has shown that the mortality rate increased due to MEs from 2876 to 7391 deaths. 2 Accordingly, healthcare leaders have called for actions to increase patient safety within healthcare institutions. One of the important measures recommended by the IOM was to apply the capabilities of health information systems. Despite efforts to minimize adverse events, they continue to occur.3–5 Unfortunately, several reports have linked the implementation of computerized provider order entry (CPOE) to the occurrence of some of these adverse events.6–8
CPOE is a complex function of health information technology (HIT), commonly understood as a set of capabilities within the order entry application that permits clinicians to place medication and other therapy orders electronically with the assistance of pre-determined rules, alerts, and knowledge databases.9,10 This capability plays a key role in the patient management process, where it can impact a variety of systems, such as pharmacy, laboratory, radiology, and resource management. CPOE also involves different users and key players, such as physicians, pharmacists, nurses, and system developers. Implementing CPOE was supported with promises for improving the quality of patient care, decreasing costs, and reducing the risk of medical errors. 11
Numerous earlier studies have demonstrated the effectiveness of CPOE in reducing the unintended consequences (UCs) of healthcare therapies and treatments. Nuckols et al. 12 concluded that implementing CPOE is associated with an almost 50-percent decrease in preventable adverse drug events. Radley et al. 13 also found that using CPOE to prescribe drugs decreases the likelihood of error by 48 percent. Although the use of CPOE is linked to notable improvements in reducing and/or limiting MEs, others argue that the use of CPOE might facilitate MEs, or CPOE itself could become a source of errors.14–17 As an example, a recent study 18 found that nearly 6 percent (63,040) of the MEs reported to MEDMARX—an anonymous, confidential, Internet-accessible, ME-reporting program that is designed and developed by the US Pharmacopeia for hospitals and health systems to systematically collect, analyze, track, and report MEs 19 —between 2003 and 2010 were related to CPOE. Moreover, Amato et al. 20 conducted a study to evaluate different CPOE systems at six sites to review potential CPOE-related MEs occurring in the medication ordering phase. They reviewed 2522 ME reports and found that more than 50 percent of the MEs were related to CPOE. These findings suggest the need for a deeper understanding of the UCs to CPOE implementation is needed.
Following the release of several reports about CPOE-related MEs, numerous researchers have aimed to systematically investigate the faults behind such errors. It is crucial to explore the possible factors that hamper the safety and effectiveness of CPOE to achieve its fundamental purpose (i.e. avoiding medical errors and improving patient safety). Experts postulate that CPOE is not just about implementation of information technology; rather, it is a major organizational process change that requires a fundamental shift in the everyday practice of virtually all clinical providers. CPOE implementation is about redesigning complex clinical processes and changing administrative and clinical policies.21,22 Implementing a new intervention such as CPOE can significantly alter workflow, create more (or different) work, and change communication patterns. 23 Thus, CPOE developers and implementers must address several factors, including organizational readiness for change (i.e. articulating policies, building proper infrastructure) and individual willingness and preparedness to adopt the new intervention (i.e. change resistance, time constraints, and training requirements), before implementing such a system. 24
The literature contains a variety of recommendations for developing a CPOE system and maintaining it to be as error free as possible. However, each set of recommendations focuses on a specific element, such as addressing technical flaws by designing/redesigning and developing CPOE system, conducting usability tests to evaluate the ease of use of the system, and addressing human–computer interactions. The aim of this work is to review the recent evidence of CPOE-related MEs among hospitalized patients and to address sociotechnical factors associated with CPOE implementation and use.
Methodology
Study design
Pre-searching strategy
The determination of the type of literature review is largely dependent on many factors related to the primary/original data to be reviewed. The inconsistency of the outputs of the primary data requires the use of a specific approach for successfully analyzing them.
Initially, the term “CPOE-related MEs” has been defined as an error that resulted from or caused by the direct implementation and use of CPOE, and would not happen, or unlikely to occur, if the medication had been prescribed with the traditional way (i.e. handwritten orders). Then, content analysis was applied to deal with the heterogeneity of the available data on the CPOE-related MEs and sociotechnical systems–related factors. The content analysis was carried out in the following steps: (1) identifying the key concepts or theory (i.e. CPOE-related MEs that resulted from sociotechnical systems–related factors); (2) defining pre-determined coding categories that can help in managing and organizing the data (i.e. year of publication, language, healthcare settings, evidence on CPOE-related MEs, sociotechnical-related factors, etc.); (3) designing the sampling plan (i.e. defining inclusion/exclusion criteria); (4) collecting materials/literature to be reviewed and units of analysis (i.e. title, author and year, citation, purpose of the article, etc.); (5) determining operational definitions for each category (i.e. keywords within texts that indicate either the rate of CPOE-related MEs or specific recommendations related to a successful implemented CPOE); (6) reviewing all transcripts comprehensively and re-reading them to identify and highlight texts that describe the research question and its related answers; (7) coding the highlighted texts with the pre-defined coding categories or providing new codes if needed (i.e. when the text cannot be coded under the pre-defined codes); (8) examining the data to determine the necessity of constructing subcategories to provide a wider systematic approach for collecting and analyzing the context; (9) comparing the extent to which the data support the proposed theory; and (10) drawing summaries/conclusions from the reviewed article.
Search strategy
An integrative literature review was conducted using PubMed and Google Scholar databases for articles published in the English language between 2007 and 2019 and included the following keywords: computerized physician order entry, computerized provider order entry, medication errors, unintended consequences, health information technology, sociotechnical factors, and other recommendations for their corresponding MeSH (medical subject heading) term synonyms. The process of searching and data extraction was conducted by a single author and reviewed by another.
Inclusion and exclusion criteria
Articles that report CPOE-related MEs were critically appraised and summarized in greater detail within the text. However, studies that involved outpatients, studies that evaluated issues other than clinical or technical issues (e.g. economic burden), secondary studies and opinion reports, and studies that lacked a clear objective or methods were excluded. On the other hand, articles that discussed recommendations for CPOE system improvement, including secondary reports, were categorized based on the sociotechnical systems–related factors that expected to impact the CPOE implementation.
Data extraction
All the studies extracted using the pre-defined search strategy were scanned by title and/or abstract. Studies that matched the inclusion/exclusion criteria were read in full, critically appraised, and summarized within the context of the scope of this work (i.e. CPOE-related MEs).
Results
A total of 2228 papers were identified using PubMed and Google Scholar databases. Initially, they were screened by title and/or abstract, and after removing duplicates and irrelevant studies, this number was reduced to 288 relevant papers. We then classified them based on their use within the review into the following categories: (1) studies reporting rates and statistics about CPOE-related MEs, (2) studies reporting types of CPOE-related UCs, (3) studies reporting factors that contribute to CPOE failure, and (4) studies reporting recommendations based on addressing sociotechnical factors. Following screening and categorization, a full-text assessment was performed on the remaining articles for eligibility based on our inclusion criteria; this resulted in 106 papers that were read in full and included in this review together with 27 additional papers obtained from references (see Figure 1).
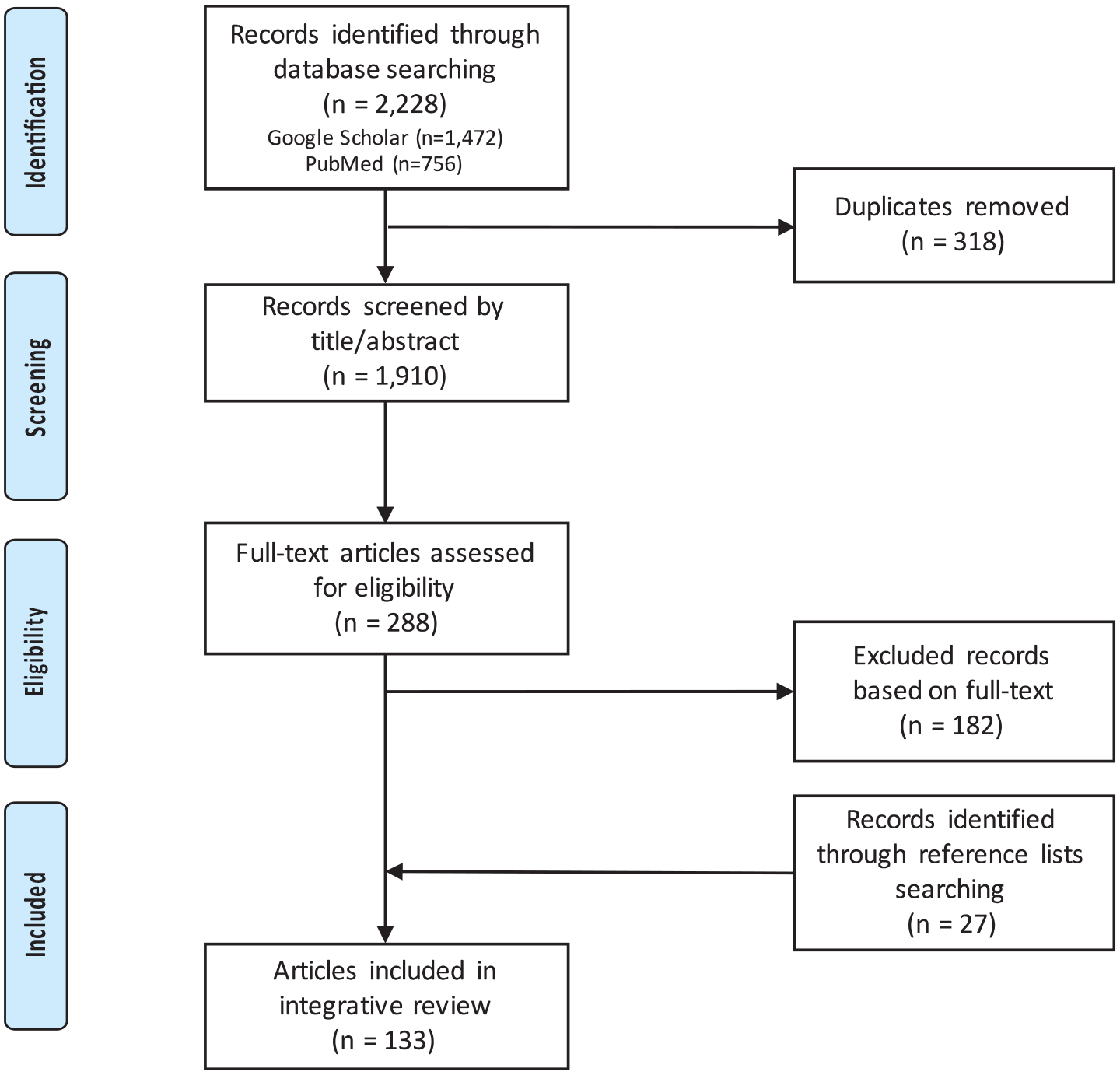
PRISMA flowchart of the search plan and exclusion criteria.
CPOE-related MEs: rates and statistics
Several studies suggest that CPOE is related to an increase in the rate of MEs (see Table 1). For instance, a group of researchers 36 prospectively investigated the incident of CPOE-generated MEs in a surgical intensive care unit of a large tertiary hospital, considering any MEs during prescribing, administration, and documentation phases. Over 4 months, 53.6 percent (286) of the total prescriptions contained at least one error related to CPOE; the majority (82.7%) of errors occurred within the documentation phase. Another retrospective study 3 compared the rates of MEs pre- and post-implementation of the CPOE system in a large tertiary pediatric hospital. MEs were identified and categorized based on their occurrence in the medication use process and their severity (see Table 2). 33 CPOE implementation was associated with a significant increase in the total MEs (2226 vs. 1741, p < 0.01), and after conducting further analysis of the error severity, there was a significant increase in the category (A and E). 3 Villamañán et al. 4 conducted a relatively short observational study to evaluate the rate of CPOE-related MEs in a large tertiary care hospital. In a 1-month period, 714 MEs were detected, where 77.7 percent were related to CPOE use, and 22.3 percent were unrelated to system use (i.e. occurred while using paper-based prescriptions). The main type of CPOE-related MEs was selection error (20.9%). Improper data placement (20.3%) was another source of errors (11.5% were due to data entry into a wrong location and 8.8% were due to inappropriate drug allergy registration).
Summary of recent studies of CPOE-related medication errors.

CPOE: computerized physician/provider order entry; PICU: pediatric intensive care unit; NCC-MERP: National Coordinating Council for Medication Error Reporting and Prevention; EMAS: electronic medication alert system; E-Rx: electronic prescription; MEs: medication errors; ICU: intensive care unit; ADEs: adverse drug events; HIT: health information technology; eMMS: electronic medication management systems; FDA: Food and Drug Administration.
NCC-MERP index for categorizing medication errors.
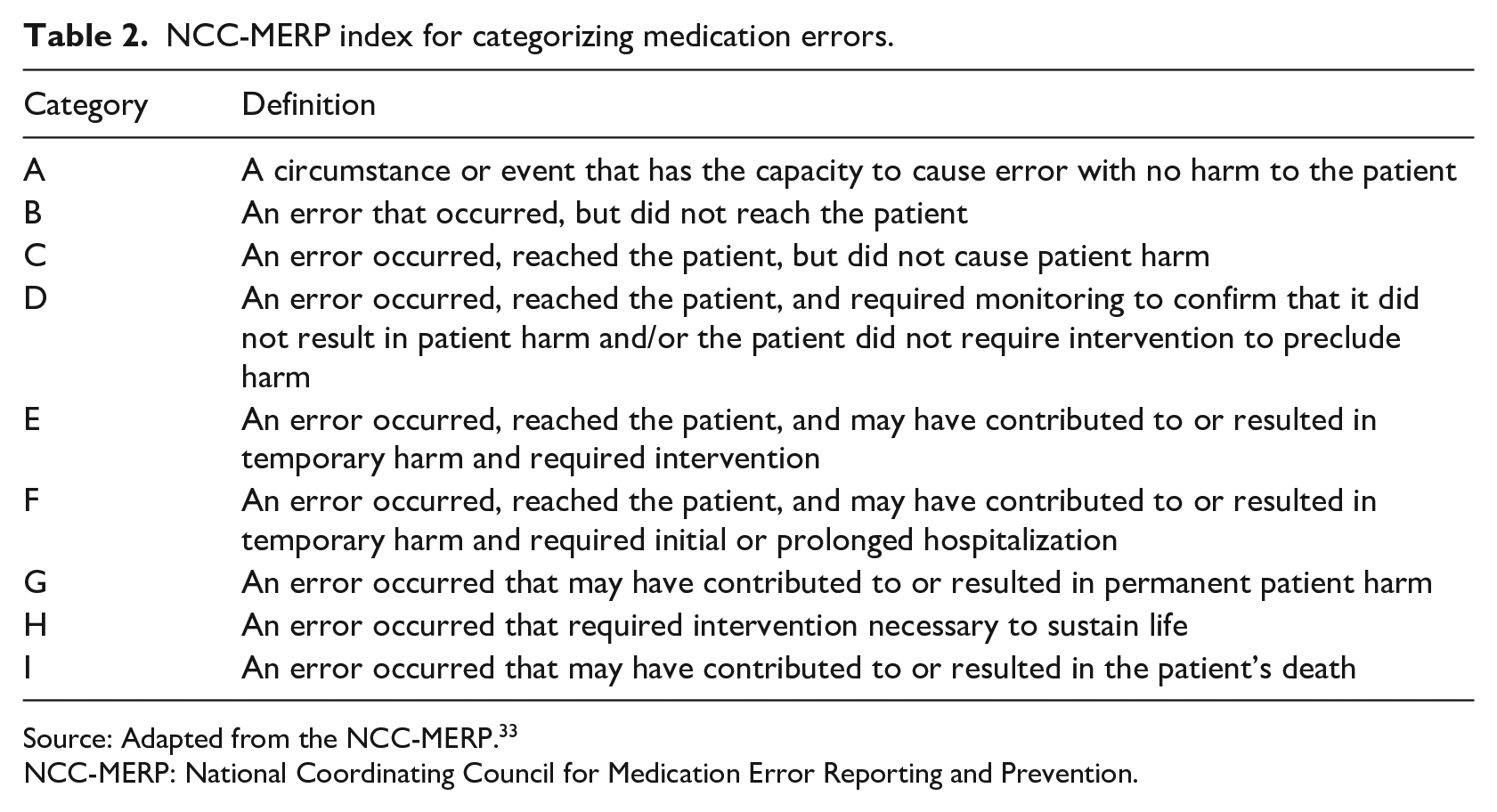
Source: Adapted from the NCC-MERP. 33
NCC-MERP: National Coordinating Council for Medication Error Reporting and Prevention.
Also, inappropriate use of the free-text field (caused by duplication or discrepancies between the structured template and the free-text comments) was another common type of CPOE-related errors and was accounted for 14.4 percent. In a study by Kadmon et al., 31 the authors estimated that 42–58 percent of all MEs were facilitated by CPOE implementation (1.8% and 0.4% of all prescriptions in 2015 and 2016, respectively). In another study that was conducted to quantify medication incidents related to electronic medication management systems (eMMS), a total of 93 of 5826 medication-related incidents were reported over a 3-month period (May–July 2014). Factors that facilitated eMMS-generated errors were suggested to be related to human factors (e.g. unfamiliarity or inadequate training), the application of cross-encounter or hybrid system, hardware malfunction, system build, and site build factors. 30
Types of UCs related to CPOE
Improving the quality of healthcare requires a full understanding of the UCs of CPOE, most particularly that they are unpredictable and they might jeopardize the safety of patient care when they occur, resulting in increased healthcare costs and greater patient and/or family suffering. For instance, CPOE might influence clinical workflow, change clinician behavior, and might generate new classes of errors.9,23,26 Identifying major types of errors resulting from CPOE implementation and addressing their causes and prevalence can help in developing implementation and precaution strategies for safe and effective CPOE.
Ash et al. 15 categorized and defined types of CPOE-related UCs for a better understanding of their nature to monitor and manage them. Accordingly, consequences related to CPOE were categorized into intended and unintended, desirable and undesirable, direct and indirect, and “two-sided” consequences (either desirable or undesirable, depending on how they are perceived). Campbell et al. 42 identify several types of unintended adverse consequences related to CPOE and classify them into nine major categories: (1) more/new work for clinicians, (2) unfavorable workflow issues, (3) never-ending system demands, (4) problems related to paper persistence, (5) untoward changes in communication patterns and practices, (6) negative emotions, (7) generation of new kinds of errors, (8) unexpected changes in the power structure, and (9) overdependence on the technology (see Table 3). Undesirable consequences related to CPOE could be direct consequences (e.g. error and security concerns) or indirect consequences (e.g. issues related to alerts, workflow, and ergonomics). Once the UCs are highlighted and fully understood, strategies and recommendations can be proposed and applied to prevent their occurrences.23,43,44
The nine categories for unintended consequences.

CPOE: computerized physician/provider order entry; HIT: health information technology.
This table is designed based on Campbell et al. 42
Factors contributing to CPOE-related MEs
Researchers 45 have postulated that the “generating new kinds of errors” category is the most serious type of CPOE-related UCs. These errors have been identified as e-iatrogenesis—patient harm caused by the use of HIT. Some examples of errors that can lead to patient harm include entering orders on a wrong patient’s file, ineffective communication among nurses and physicians (i.e. not knowing if an order has been generated), loss of information during care transitions, and overlapping medication orders. 23 Different risk factors, such as flaws in system design, unrecognized discontinued orders, unintended loss of orders, drop-down menu selection errors, and issues related to alerts (lack of drug dosing alerts, generation of inappropriate dosing alerts, and inappropriate duplication alerts), can be associated with the use of CPOE.46–48 Lichtner et al. 27 classified eMMS-related incidents into four categories: (1) technical issues (e.g. bugs), (2) issues related to user experience, (3) unanticipated workflow issues (e.g. duplicated or multiple orders, system incompatibilities with existing workflows), and (4) missing safety features (e.g. absence of specific decision support). Table 4 summarizes several risk factors associated with CPOE use.
Risk factors associated with CPOE use.
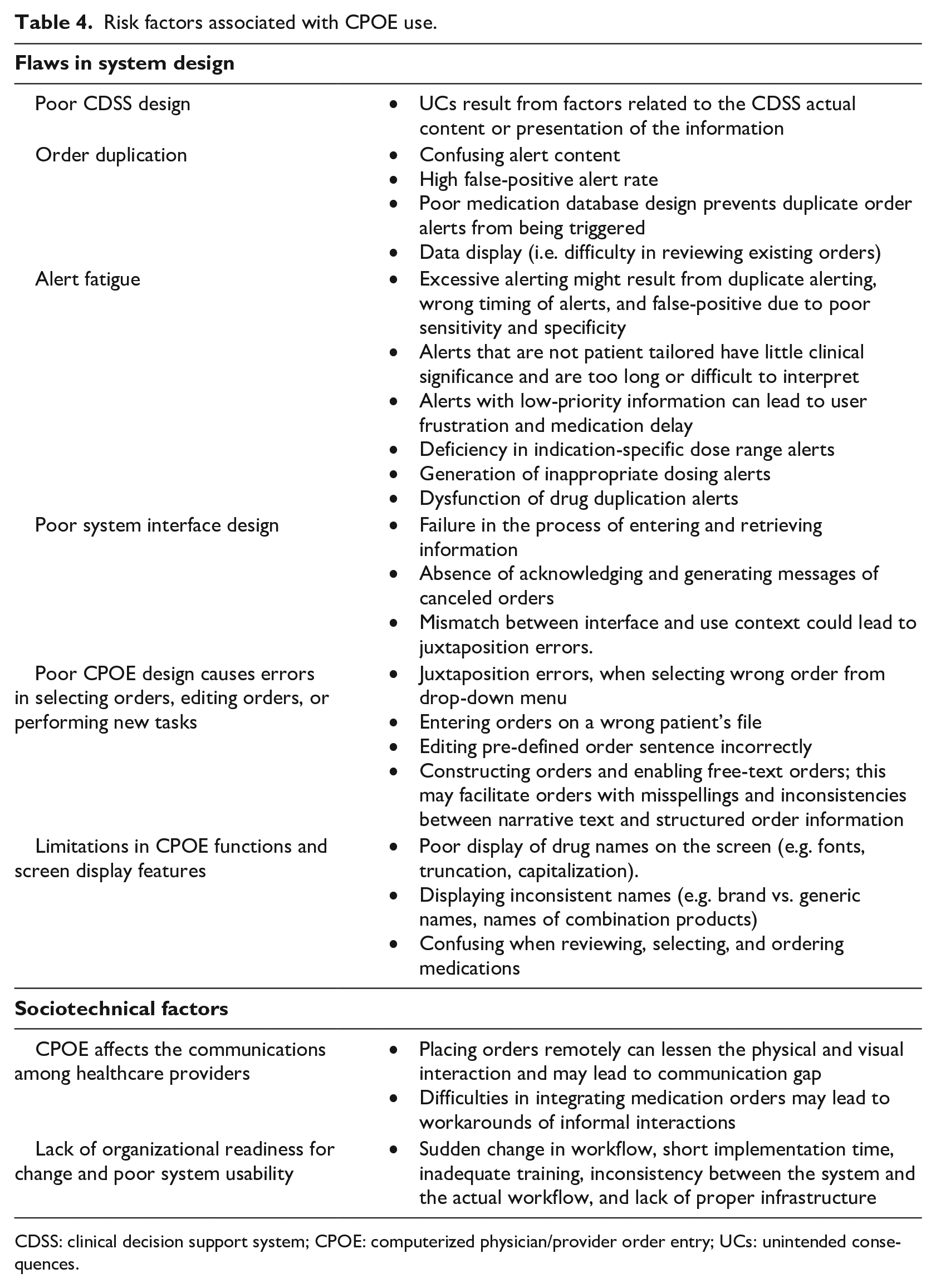
CDSS: clinical decision support system; CPOE: computerized physician/provider order entry; UCs: unintended consequences.
One of the primary factors contributing to CPOE UCs is system functionality and design.46,49,50 For instance, after analyzing the different types of CPOE-related UCs, researchers 51 found that many of them were related to the design of clinical decision support system (CDSS). CDSS is defined as “a computer-based system that offers passive and active referential information as well as reminders, alerts, and guidelines” (p. 524). 52 CPOE shows a high level of effectiveness when combined with CDSS. 53 The main goal of integrating the two systems is preventing adverse drug events and optimizing medication safety. 54 When both systems are designed and implemented carefully, they can improve the quality of clinical decisions by providing reminders or recommendations at the point of care.10,55,56 However, poor system design can lead to many errors, where the UCs related to CDSS can be generated by the actual content of the decision support module (e.g. elimination or shifting of human roles, ongoing necessity for updating the algorithm-based rules, wrong or misleading CDSS content) or by the presentation of the information (i.e. the way alerts and other modes of decision support are presented to the users). 51
Order duplication, defined as “two or more active orders for the identical medication regardless of dose” (p. 379), 57 is one of the CPOE-related UCs that might be generated by poorly designed systems. Wetterneck et al. 38 evaluated the duplicate medication orders before and after CPOE implementation; they found that duplication in medication orders increased significantly from 48 to 167 errors (p < 0.0001) after CPOE implementation. They identified several contributing factors, including ordering practice and computer availability (i.e. orders can be placed from different locations through computers by different providers on rounds within minutes); entering duplicate orders during care transitions and changing shifts (i.e. poor communication and hand-offs); poor system and medication database design (i.e. confusing alert content, high false-positive alert rate, and CDSS algorithms missing true duplicates); data display (i.e. difficulty in reviewing existing orders); and issues related to local CDSS design (i.e. medications in order sets defaulted as ordered). 38 Moreover, Magid et al. 57 studied duplicate orders by analyzing different aspects of the process such as specific drugs, role of prescribers, the source of the duplicate orders, and the workflow. They found that duplicate orders were generated mostly when the same medications were ordered by different prescribers than by the same prescribers, and from order sets than from single orders.
Another CPOE problem resulting from poor system design is alert fatigue—a phenomenon by which users tend to ignore many of these alerts, including the ones that notify them of potentially serious errors. 10 This mainly occurs when CPOE is integrated with a CDSS that generates an excessive number of warning alerts, which can lead to a high alert override rate and decision support overload.6,47,48,58–61 Excessive alerting can result from duplicate alerts, wrong timing of alerts, and false-positive alerts (non-relevant alerts due to poor sensitivity and specificity).62,63 Van der Sijs et al. 64 found that medication safety alerts were overridden by clinicians in 49–96 percent of cases (e.g. overdose, contraindications, allergies, and life-threatening drug–drug interactions). Another study evaluating the CDSS by monitoring CPOE order override rates shows that 87 and 81 percent of high-severity drug-drug interaction and drug allergy alerts, respectively, were overridden. 65 A study by Strom et al. 66 highlights the hard-stop alert feature designed to reduce undesired prescribing orders; this is defined as “an alert appeared as a pop-up window that notified the clinician that the order could not be processed because of a significant potential drug-drug interaction” (p. 1579). 66 Their study revealed that despite the effectiveness of hard-stop alerts in reducing prescribing errors, they caused a delay in medication orders, which led to an early termination of the study for ethical reasons (i.e. potential for harm in the intervention). In fact, alerts that are not patient tailored have little clinical significance and are too long or difficult to interpret; also, alerts with low-priority information cause user frustration and slow down the medication ordering task. 62 Moreover, several studies highlight other issues related to alert design, such as a lack of a dosing alert (deficiency in indication-specific dose range alerts), generation of inappropriate dosing alerts, and dysfunction of drug duplication alerts (order duplications do not trigger alerts due to the design of the medication database and the CDSS algorithms that miss true duplicates).38,67–71
Another CPOE problem resulting from system design flaws is poor system interface. The design of the CPOE system interface is required to be consistent with the physicians’ task behavior and decision-making processes; otherwise, it may obscure the appropriate order entry strategy and may lead to inefficient workflow and user frustration. 62 Moreover, researchers 72 found that some UCs were associated with the absence of adequate system interface functionality, such as the absence of acknowledging and generating messages of canceled orders. Poor CPOE interface design can result in usability problems, workflow interruptions, and subsequent MEs.62,73
Another risk resulting from CPOE use is entering orders on a wrong patient’s file 74 or selecting wrong orders. Such a problem is known as “juxtaposition errors” and can be considered an additional flaw in CPOE system design. 42 Errors resulting from drop-down selection menu are considerably common and may contribute to serious MEs.75–77 Researchers found that such system-related errors occur when prescribers need to select items from drop-down menus, construct orders, edit information within the system, and/or perform new tasks not previously required. 78 Selection errors were the most frequent type of error, accounting for 43 percent of total system-related errors, while editing orders accounted for 21 percent. In addition, failure to complete new tasks found to be accounted for 32 percent of all system-related errors. 78 It is important to note that such a failure is generally related to the limited functionality of the system, which leads to developing workaround processes and adopting hybrid systems (i.e. using computer-based orders along with some paper-based format). 79 Constructing or editing orders may also create risks and jeopardize patient care and safety. Enabling prescribers to use free-text instructions to enter orders may lead to inconsistencies between narrative text and structured order information.78,80,81 For instance, Zhou et al. 82 found that around 17 percent of free-text medication order entries contained misspellings and led to additional errors.
An additional CPOE-related factor that might contribute to MEs is the way drug names are displayed. 83 A number of MEs were found to be related to screen displays and workflow issues. 84 Displaying inconsistent names, especially the display of brand versus generic drug names, is a source of confusion when reviewing, selecting, and ordering medications. 83
Ineffective communications among nurses and physicians is another problem associated with CPOE use. The ability to enter orders through the system remotely can lessen the physical and visual interactions and may contribute to communication gaps. 85 A mixed method qualitative study was conducted by Pirnejad et al. 86 to evaluate the impact of CPOE on nurse–physician communication; the results showed that the communication was impacted due to difficulties in integrating medication orders among nurses and physicians, which eventually led to developing workarounds of informal interactions that represent risks for MEs.
The aforementioned factors may lead to MEs resulting from a lack of organizational readiness for change and poor system usability. 50 For instance, unanticipated and sudden changes in workflow, short implementation time, insufficient users training, inconsistency between the new system and the actual clinical workflow, and lack of proper infrastructure for incubating new interventions (e.g. physical space layout, policies and procedures, software and hardware capabilities) may contribute to generating new errors and affecting the workflow; this can lead to cognitive overload, user resistance, and eventual patient safety problems.15,62,87,88 Thus, addressing the way users interact with the system, how they perceive it, and to what level they accept it and understand it are critical elements for developing and implementing CPOE effectively and safely.
Published recommendations based on addressing sociotechnical factors
Adopting new technologies in a complex and dynamic setting such as healthcare is challenging. Implementing CPOE as a major intervention for reducing the rate of MEs was found to be a source of errors in many studies. Some may consider looking at the failure of CPOE as an opportunity for improvement by addressing potential factors that might lead to ineffective CPOE design and subsequent poor implementation. Like other health information systems, successful implementation of CPOE requires careful planning, consistent monitoring, and ongoing commitment. Identifying process flaws enables developers and implementers to successfully build a system capable of achieving its intended fundamental purpose, that is, reducing medication and order errors and maximizing patient safety.
The literature contains extensive valuable recommendations that may help in developing and implementing a successful and safe CPOE environment, such as system improvement, applying human factors, and considering sociotechnical factors. The majority of the recommended remedies were proposed by researchers based on different models of analyses of HIT interventions, such as the diffusion of innovations theory,89,90 human factors engineering and usability testing, 91 human factors and ergonomics models, 92 the acceptance and use of technology theory, 93 and interactive sociotechnical analysis (ISTA). 94 It is worth noting that there are several frameworks that address errors induced by HIT. Borycki et al. 95 have elegantly summarized a number of frameworks, models, and theories that would significantly impact safe HIT development and implementation. They organized the frameworks and models into three main groups based on works emerged on other literatures: (1) human factors models and frameworks (e.g. the theoretical and conceptual cognitive taxonomy of medical errors), 96 (2) organizational behavior and sociotechnical models and frameworks (e.g. the thematic hierarchical network model), 15 and (3) software engineering models and frameworks (e.g. continuum of methods for diagnosing technology-induced errors). 97 Parts of their work are summarized in Table 5.
Models and frameworks for considering technology-induced errors.

HIT: health information technology.
This table is designed based on Borycki et al. 95
System improvement
Improving the system design by assessing the existing ordering process and applying modifications and/or customizations of different functionalities based on the organizational needs is one of the recommended solutions. 105 CPOE effectiveness can be improved by enhancing the integrated CDSS. 55 For instance, selection errors can be minimized by shortening the drop-down menu and/or enhancing some of the system functionalities, such as adding new mouse trigger designs, safeguards, colors, bolded fonts, and tall-man lettering.46,106,107 In another study, a group of researchers 108 postulated that patient selection errors could be reduced by manipulating CPOE interface design, such as highlighting the selected item after the selection is made, and providing visual feedback of related information. In addition, building a more sophisticated CDSS that is capable of checking free text and providing dosing support features and indication-based alerts can help minimize CPOE-related UCs.46,109,110 Filtering the sensitivity and improving specificity of CDSS to provide prioritized alerts according to clinician preferences is another function that can be added to minimize alert fatigue.59,61,69,111–115
Considering human factors
Applying human factors engineering—a field of study that focuses on equipment/system/process design and analysis while addressing human characteristics, capabilities, limitations, and interactions with system’s components, processes, and work environment—in which addressing cognitive complexity, enhancing system usability (i.e. user-friendliness and usable user interface), and enhancing user interface design could help in facilitating the safe and effective implementation of any new HIT.46,99,116–120
Sociotechnical factors
Considering sociotechnical aspects is highly recommended in literature for addressing medical errors related to HIT implementation (i.e. understanding the relationship between human and computer/processes by addressing technical features, such as hardware, software, and techniques, as well as social factors, such as culture, acceptance, and environment).121–123 For instance, a study by Salahuddin et al. 124 highlights five major sociotechnical factors influencing the safe use of HIT, such as knowledge, system quality, task stressor, organization resources, and teamwork. One of the most comprehensive conceptual models that has been proposed is the eight-dimensional sociotechnical model: (1) hardware and software computing infrastructure; (2) clinical content; (3) human–computer interface; (4) people; (5) workflow and communication; (6) internal organizational policies, procedures, and culture; (7) external rules, regulations, and pressures; and (8) system measurement and monitoring (see Table 6). 102 It addresses a wide range of factors that should be carefully covered throughout all the CPOE adoption phases (planning, designing, developing, implementing, using, and monitoring). This important framework helps in identifying HIT-related contributing factors. It also assists developers and implementers in proactively planning and monitoring the CPOE implementation process by addressing sociotechnical factors that present challenges in the adoption of these systems.28,102 For instance, the first dimension can guide organizations to assess their technical infrastructure (i.e. the availability and capabilities of software and hardware) when they intend to adopt new interventions, such as CPOE. The second dimension concerns the availability and quality of the clinical knowledge and content and how it should be collected, managed, and employed effectively and safely; it guides organizations to configure specific software requirements, such as controlled vocabulary and order sets. The third dimension focuses on the human–computer interface, where it guides organizations to address system usability and how clinicians interact with system components; this helps organizations with ongoing assessments of system acceptance and usability (e.g. system navigation feasibility, applications response time, and screen display and alert notification simplicity). The fourth dimension concerns people who are involved in CPOE adoption phases; this is critical for addressing individuals’ needs, their capabilities and limitations, their level of acceptance, and how they perceive new interventions. The fifth dimension focuses on addressing communication and workflow processes, particularly where these guide organizations to ensure the new system does not negatively affect patient care delivery, and how the system must fit the clinical workflow (i.e. modifying the system to match the clinical workflow, and vice versa). The sixth dimension addresses the internal organizational aspects (e.g. policies, procedures, resource allocation), and it guides organizations to assess their readiness to adopt the new intervention by addressing challenges in the organizational culture and developing policies and procedures that are consistent with the clinical workflow in the presence of CPOE. The seventh dimension concerns the enablers and identifies constraints that are enforced by external bodies and regulators regarding the design, development, implementation, use, and monitoring of CPOE in the healthcare setting. These may include privacy and confidentiality legislation and fee-for-service policies. The final dimension focuses on ensuring ongoing measurement and monitoring to help organizations identify issues, monitor system usability, and measure the system impact on the overall workflow and outcomes, including the measurement of the rates and types of MEs following CPOE implementation.102,125
The eight dimensions of the sociotechnical model.
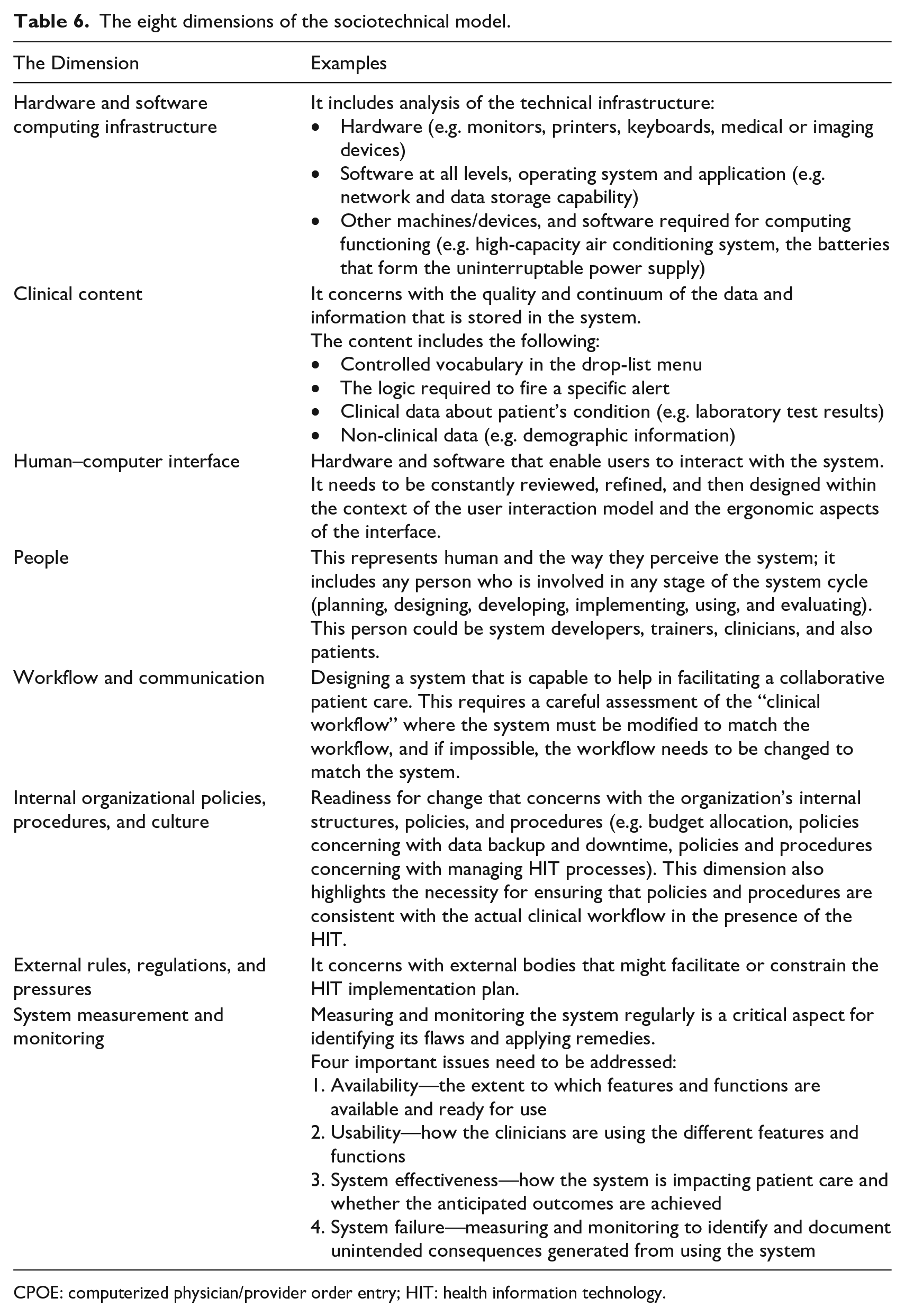
CPOE: computerized physician/provider order entry; HIT: health information technology.
Hierarchical decomposition and focusing on individual units/elements in the system is not effective in assessing HIT. 126 Thus, the eight-dimensional model described above should be used as an interactive standard that focuses on how each dimension is depending on and interacting with each other to comprehensively evaluate the effectiveness, usability, and safety of the system.102,125 Developing an effective implementation plan requires a comprehensive model to identify weaknesses. Identifying process flaws can work as lessons learned and permit developers and implementers to re-think about how to prevent them from recurring. This model has been found to be a successful tool for evaluating the system design, development, implementation, use, and ongoing monitoring. 102 Addressing the sociotechnical dimensions at each stage of the CPOE implementation plan may help software developers, healthcare organizations (implementers), and end users to proactively identify process flaws and accordingly reduce the risk of patient harm.123,127,128
Discussion
This integrative review has been written to highlight the burden of UCs of CPOE within the context of sociotechnical factors. It reviewed a wide range of research that focuses on illustrating the extent of CPOE-related MEs. The article summarized several studies that identified and classified CPOE-related UCs; recapped a number of research studies that investigated different factors and flaws associated with the CPOE design, implementation, and use; highlighted different factors associated with CPOE implementation; and underscored the importance of evaluating HIT by employing the eight dimensions of a sociotechnical model. Despite the large body of research that supports the effectiveness of CPOE in reducing MEs, some researchers have found that CPOE can facilitate the occurrence of MEs or become a source of them. For instance, a study by Cho et al. 36 found that nearly half of the medication orders entered through CPOE contained at least one error. Another group of researchers 3 found a significant increase in the rate of MEs after implementing CPOE. The persistence of high rate of MEs, despite the use of CPOE, has triggered an alarm about the safety and effectiveness of a system that was proposed to effectively reduce the rate of errors and substantially contribute toward improving patient safety. The CPOE-related MEs drove many researchers and experts in the field to study this matter more comprehensively assisted by the classification into nine major categories of CPOE-related UCs developed by Campbell and associates. 42 Also, different factors and flaws associated with the CPOE use and implementation have been studied to identify a successful CPOE design and implementation.
Identifying the problem sets up only part of the eventual solution. Categorizing the UCs of CPOE and understanding its flaws have guided many researchers to study and develop strategies for minimizing the occurrences of such consequences, including those contributing to MEs. As an example, remedies for technical and social-related issues have been proposed to assist in addressing this matter. One of the recommended models documented for analyzing sociotechnical related factors intended to evaluate HIT is the eight-dimensional framework previously described. 102 The domains in this model are intended to assist healthcare organizations and system developers address challenges related to sociotechnical factors at each stage of system adoption (i.e. planning, designing, developing, implementing, using, and monitoring).
Evaluating CPOE systems needs to be started at the planning stage, as soon as CPOE has been recommended for implementation. This is initiated by conducting a comprehensive assessment of the organizational readiness, which starts by assessing the appropriate infrastructure (e.g. hardware, software, the physical layout, policies, and procedures) and addressing the perspectives and involvement of healthcare professionals and other stakeholders (e.g. patients, policymakers, and system suppliers).24,129 Adopting a new technology such as CPOE requires a strong culture in which people understand and accept the change; this may require strong top management support for allocating sufficient resources for the implementation process (e.g. staffing, procurement, maintenance, and training). In addition, having organizational champion leaders who can drive implementation, change culture, and bridge knowledge with developers would be an important step to consider. 130 Also, it is important that the system’s design serves its intended purposes, especially in the clinical arena. For instance, clinical content stored in the system including a controlled vocabulary, specific order sets, and rules/alerts needs to be carefully designed—more specific and sensitive—to minimize possible related errors. To carefully conduct this step, the organization should clearly define knowledgeable users to be involved in each stage of the CPOE implementation plan, including clinicians, trainers, and technical experts. Moreover, providing periodic training sessions, understanding users’ needs, and addressing their perceptions before implementing the system are important for detecting and correcting problems, minimizing change resistance, and increasing user acceptance and satisfaction. These actions need to be regularly monitored and assessed for meeting user expectations and maintaining system feasibility.131,132 Addressing user–computer interactions is another factor that must be carefully addressed. For instance, an organization should conduct usability tests, simulation scenarios, and pilot trials to observe how users perceive the system and how they interact with it. These actions may help identify possible system gaps and may help in evaluating the system’s usability. 133 Any new system must be carefully monitored and measured on an ongoing basis, 134 which may give implementing organizations the opportunity to identify and address system flaws and hidden safety risks. 127 Besides, implementers should not underrate the importance of monitoring MEs post-CPOE implementation. For instance, it is crucial to constantly review and measure the rates and types of MEs prevented, not prevented, and/or MEs facilitated by the use of CPOE during the following years after CPOE implementation to identify CPOE problems and find solutions. By measuring and evaluating system success or failure, implementers can consider a sufficient timeline for planning and implementing the CPOE system to allow for adequate system adaptation. 135 One more aspect organizations must address is the external laws and regulations, where forces outside the organization’s control might suppress or negatively impact the CPOE implementation process. Addressing them early may provide implementers the opportunity of not rushing the adoption of new systems without a full assessment for both the system and the organization’s preparedness to address external impacts. To drive the effective and safe use of CPOE functions, organizations should automate with caution. Full assessment of the current culture and infrastructure together with a comprehensive understanding of users’ needs are important keys for a successful and safe CPOE system.
For future research and new HIT projects, organizational leadership must recognize that focusing on technical capabilities alone to implement safe HIT is not sufficient; other factors, including organizational culture, work environment, and human–computer interactions, must be considered. Evaluating sociotechnical factors by utilizing the eight-dimensional model might contribute to safe and effective CPOE implementation. This can help implementers proactively address their preparedness to implement CPOE by assessing the internal culture and developing policies and procedures consistent with the clinical workflow in the presence of the change under consideration. Also, addressing the technical infrastructure, users’ needs, and how they interact with CPOE is crucial to success. By adopting and employing the eight-dimensional model, organizations can understand how each dimension can influence one another, and they can work to build a comprehensive and careful implementation plan based on their assessments. To this end, organizations must recognize that implementing CPOE is multifaceted and requires careful consideration of internal and external factors that might impede its effectiveness.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
