Abstract
Primary studies have demonstrated that despite being useful, most of the drug-drug interaction (DDI) alerts generated by clinical decision support systems are overridden by prescribers. To provide more information about this issue, we conducted a systematic review and meta-analysis on the prevalence of DDI alerts generated by CDSS and alert overrides by physicians. The search strategy was implemented by applying the terms and MeSH headings and conducted in the MEDLINE/PubMed, EMBASE, Web of Science, Scopus, LILACS, and Google Scholar databases. Blinded reviewers screened 1873 records and 86 full studies, and 16 articles were included for analysis. The overall prevalence of alert generated by CDSS was 13% (CI95% 5–24%,
Keywords
Introduction
Medication prescription errors are quite prevalent worldwide and an important threat to patient safety. Although the most common results are only mild adverse effects, some cases significantly increase the risk of death. 1 In this context, harmful drug-drug interactions (DDIs), which can occur when the effects of one drug are influenced by the effects of another, are the leading cause of this risk. Research has already proven this and indicated that over half of adverse drug effects are directly related to prescription medication errors.2–5
To reduce these risks, healthcare systems around the world are developing and implementing electronic health records (EHR) with clinical decision support systems (CDSS) that warn prescribers of potential DDIs (pDDIs), thus protecting patients from adverse drug events (ADE). 6 Potential DDIs can be predicted from knowledge about the pharmacological properties of the drugs prescribed. There are different databases where healthcare professionals can query about potential drug interactions. In the Drugbank database alone, which is one of the most used drug databases, there are at least 365,984 interactions recorded. 4
Despite the concern with the information presented by CDSS to medical teams, it is also necessary to consider how these teams use the medical record and report their difficulties. The concept of user experience (UX) deals with how users interact with a product based on their expectations and needs in relation to the use of the product in question since bad experiences may decrease the use of the tool or even the complete discontinuation. 7 This phenomenon can also occur with CDSS, and in the case of detecting pDDIs, a negative UX can cause low adherence of healthcare professionals to the system’s guidelines.8–10
Given that it is important to verify whether this issue could impact patients’ clinical safety and further develop this tool by focusing on the UX, which is crucial to improving prescriber adherence. In order to better understand the issues related to these CDSS, we sought to conduct a systematic search to learn from the experiences of other CDSS implemented in other countries’ healthcare systems. Other systematic reviews have focused on prescribing errors that can be avoided by CDSSs,11–14 although none have focused on pDDI alerts generated by these systems.
Given the above, this study aimed to assess the frequency of CDSS-generated pDDI alerts in EHRs and evaluate adherence to these alerts through the alert override rate of physicians utilizing this tool to prescribe drugs.
Methods (study design)
The methodological procedure was defined by following the Preferred Reporting of Systematic Reviews and Meta-Analysis (PRISMA) guidelines.
15
The review question was formulated according to the PICOS approach (problem, intervention, control, outcomes, and study design): “
Literature search
A systematic literature search was conducted on April 12, 2023, in the electronic databases MEDLINE/PubMed, EMBASE, Web of Science, Scopus, and LILACS, searching for articles published between 2011 and 2023. Only manuscripts published in the English, Spanish, and Portuguese languages were included. We also searched for studies in gray literature using the Google Scholar database. A pilot search was conducted to define the Medical Subject Headings (MeSH) search terms and strategies, which was validated by three experts (CLC, JFH, and RSW). A list of reference articles (indicated by experts) was used to define the search strategy. A MEDLINE/PubMed search strategy was developed using a combination of the following keywords, which were divided into 3 research blocks: ((“Decision Support Systems, Clinical”[Mesh] OR “Decision Support Systems” OR “Information Systems”[Mesh] OR “Clinical Pharmacy Information Systems”[Mesh]) AND (“Drug Interactions”[Mesh] OR “Drug interactions”) NOT (“Herb-Drug Interactions”[Mesh] OR “Food-Drug Interactions”[Mesh])). The search strategy used in MEDLINE/Pubmed was adapted for other databases and is presented in Appendix B. The references retrieved from the searches were organized in EndNote Reference Manager web (2021) and Rayyan QCRI (Qatar Computing Research Institute - Data Analytics, Doha, Qatar) online software.16,17
Study selection
Independent pairs of reviewers (MF, RAS, DS, and EMD) selected the articles based on the inclusion and exclusion criteria. The inclusion criteria were: physicians’ prescriptions with pDDIs made on EHRs at a hospital and/or primary health care unit that uses CDSS. The reviewers began by reading the titles and abstracts independently while applying the eligibility criteria, followed by full-text reading, also applying the eligibility criteria. A third reviewer (CLC, JFH, and RSW) cross-checked all the retrieved information in both phases. The final selection was always based on the full text of the publication and according to the PICOS approach (Appendix A). We excluded studies that did not satisfy the inclusion criteria and those that met the exclusion criteria (Appendix C), and the remaining full-text articles were used for the study. We also searched for articles in the reference lists of eligible studies.
Data extraction
The articles were randomly distributed to four authors to extract the data independently using the Research Randomizer® software. 18 Data were extracted from the text, figures, or tables and added to a standardized table. In addition, three email attempts were sent to the authors whose studies were included in this review to acquire missing data and provide further clarification on the subject. The extracted data were validated by a pair of reviewers (MF, RAS, DS, and EMD). Any discrepancies were resolved through discussion or consulting a third reviewer. Afterward, the expert group validated the data extracted in the standardized form (Supplementary Material, Table S1). An expert group (CLC, JFH, and RSW) checked the extracted data, and disagreements were solved through discussion. The data were exported to the R software 19 for meta-analysis and quality assessment analysis.
Data synthesis
A random effects model for meta-analysis in the Rstudio software
19
was used for statistical pooling of data. The dichotomous outcomes were reported as prevalence ratios (events/total) according to 95% confidence intervals. Meta-analysis was performed using a minimum of three studies, and the pooled prevalence was presented with 95% confidence intervals. Heterogeneity was assessed by calculating I2 and Q value tests (I2 > 50% or
Quality assessment
Three review authors (MF, RAS, and DS) independently and blindly assessed the methodological quality using the Joanna Briggs Institute (JBI) critical appraisal tools. 20 Two reviewers independently and blindly checked a list of questions for each study type and answered “yes,” “no,” or “unclear.” The discrepancies were resolved by review experts (EMD and IGD). The final score for each article was calculated by the number of yes in the total number of quality criteria. The score indicated the quality as low (<50%), average (50–75%), and high (>75%).
Results
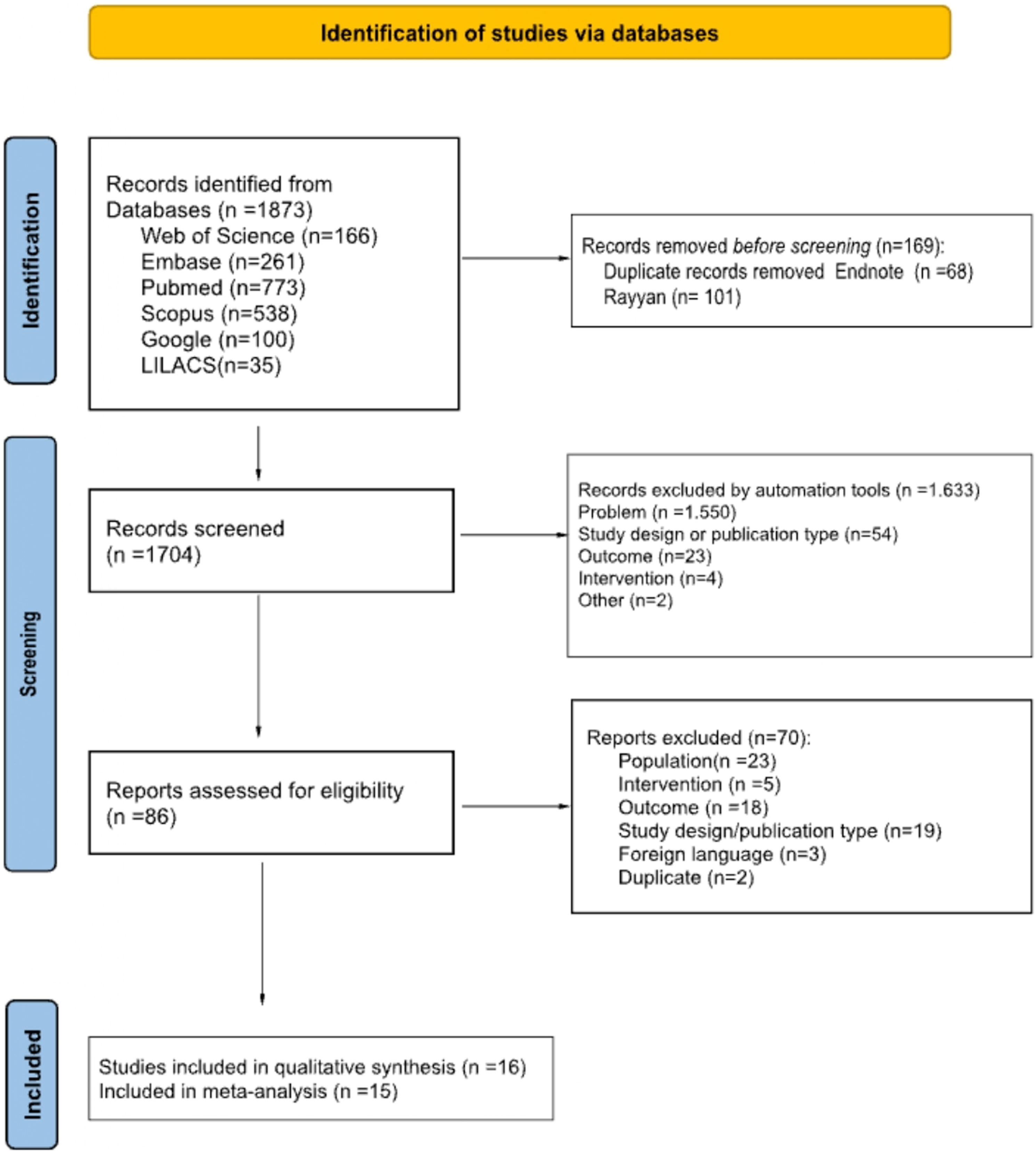
The systematic search of the electronic databases resulted in 1873 articles; after removing duplicated articles, 1704 manuscripts remained for title and abstract screening. Afterward, 86 articles were considered eligible for full-text reading. The reviewers excluded 70 studies as they met the exclusion criteria of wrong population ( Flowchart summarizing the identification and selection process of the studies for inclusion. Adapted from Page et al., 2020.
15
Characteristics of the studies
Summary of the characteristics of the included studies (
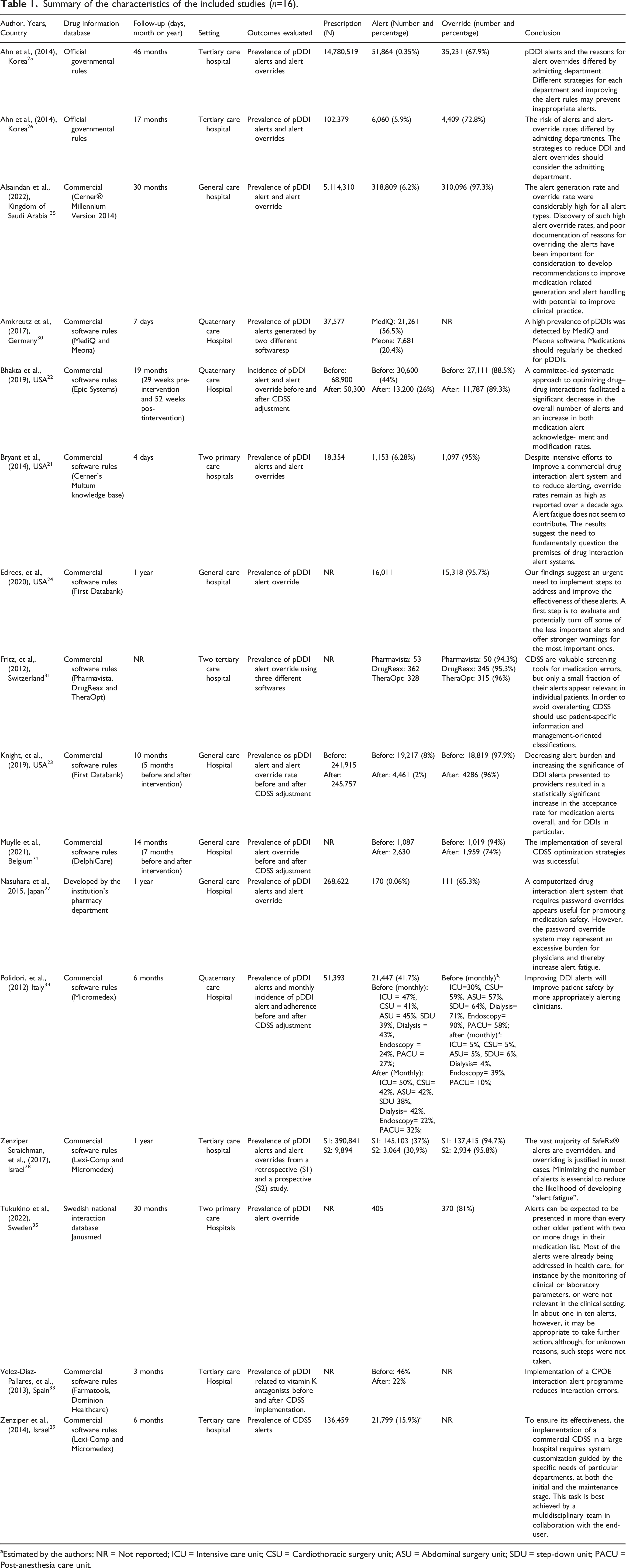
aEstimated by the authors; NR = Not reported; ICU = Intensive care unit; CSU = Cardiothoracic surgery unit; ASU = Abdominal surgery unit; SDU = step-down unit; PACU = Post-anesthesia care unit.
Regarding the studies’ research settings, two were conducted in three primary care hospitals,21,35 six studies in a tertiary care hospital,25,26,28,29,31,33 three studies in a quaternary care hospital,22,30,34 and five studies did not report the healthcare settings,23,24,27,32,36 so we counted them as general hospitals.
Among these healthcare settings, most employed commercial CDSS and drug information databases (
Main outcomes
Among the studies included in this systematic review, there were different types of reports on using CDSS to detect pDDIs in EHRs. Most of them evaluated the number of pDDI alerts generated by the CDSSs and the volume of alert overrides.21,23,25–28,36 Even in studies in which the period of the prevalence of alerts generated by the CDSS was below 10%, over 60% of these alerts were ignored by prescribers21,23,25–27,36 (Table 1).
Three other studies only evaluated the number of alerts for pDDIs and reported a high prevalence.29,30,33 Amkreutz et al. (2017) 30 compared two software (MediQ and Meona) and found that both showed a high prevalence of DDI alerts (56,5% and 20,4%). Four other studies only evaluated the prevalence of alert overrides,24,31,32,35 and one compared three software (Pharmavista, DrugReax, and TheraOpt). 31 These studies also reported a high prevalence of alert overrides ranging from 74% to 96%. Notably, two studies evaluated the prevalence of alerts and alert overrides before and after adjusting the rules for alerts generated by the CDSS. However, despite reducing the number of generated alerts, these adjustments did not significantly reduce the number of overrides, in one of the studies it decreased from 97% to 96%, 23 and in the other it decreased from 94% to 74%. 32 In addition, two studies reported the incidence of alerts generated and the volume of acceptance before and after adjustments in the drug interaction rules of these CDSS. In these studies, the number of alerts generated after CDSS adjustments was similar or lower than before, and the acceptance of these alerts increased after the intervention22,34 (Table 1).
The authors of the studies differ about the reason for the high prevalence of alerts and alert override, most of them reporting that system adjustments should improve acceptance of alerts and decrease the chances of adverse drug events.22–26,28,29,31,32,34,36 Others emphasized that these adjustments must be constant, made by a multidisciplinary team assembled for this purpose, and take into account the characteristics of the hospital sector25,26,29 and patient 31 (Table 1).
Moreover, some researchers reported that only generating alerts for more important interactions can increase acceptance.22,24,23,28,35 Other studies only evaluated the generation of alerts and concluded that implementing a CDSS is essential to avoid DDIs.30,33 One of the studies evaluated the use of a password for alert override and, due to the high rate of aversion, concluded that including an authentication step can increase the workload and generate alert fatigue. 27 In fact, one study concluded that the reason for the high levels of alert overrides is not related to professional fatigue but to the high number of alerts and that the real reason should be further investigated 21 (Table 1).
Meta-analysis
Of the 16 studies in the systematic review, 15 were included for quantitative analysis. Meta-analyses were performed separately for the subgroups, which were divided into studies that only dealt with generated pDDI alerts and used the number of alert substitutions. Ten studies were included in the meta-analysis of the prevalence of alerts generated by the CDSSs, considering that 21,435,597 prescriptions were analyzed. The overall prevalence obtained was 13.7% (CI95% 5.6–24.7%, Prevalence of alerts generated by the CDSS: 21,435,597 prescriptions were analyzed, and the overall prevalence obtained was 13.7% (CI95% 5.6–24.7%, p-value <0.0001, I^2 = 100%). Among them, a study compared two different software,
30
a study presented the prevalence of alerts generated before and after CDSS adjustments,
23
and another study consisted of two parts, one retrospective and one prospective.
28
Therefore, the respective prevalences appear in the meta-analysis.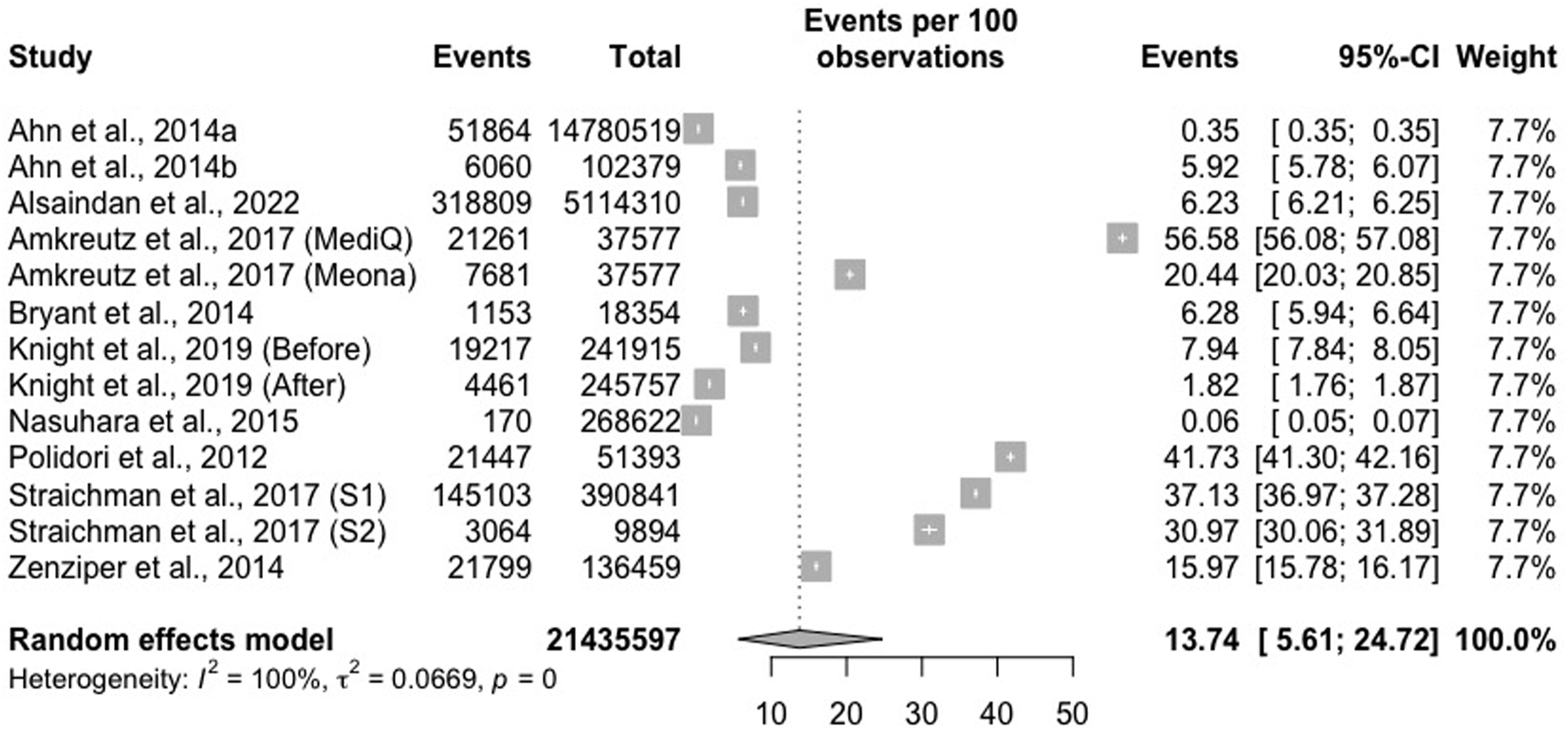
Regarding physicians’ adherence to receiving a pDDI alert in their prescription, eleven studies assessed the prevalence of alert overrides; 570,776 prescriptions were analyzed, and the overall prevalence obtained was 90% (CI95% 85.6–95.0%, Prevalence of alert overrides by physicians: 570,776 prescriptions were analyzed, and the overall prevalence obtained was 90% (CI95% 85.6–95.0%, p-value studies assessed the prevalence of disregard for alerts before and after adjustments to alert definitions in the CDSS,23,32 one study compared three different software,
31
and another study was divided into two stages, one retrospective and one prospective.
28
Therefore, the respective prevalences appear in the meta-analysis.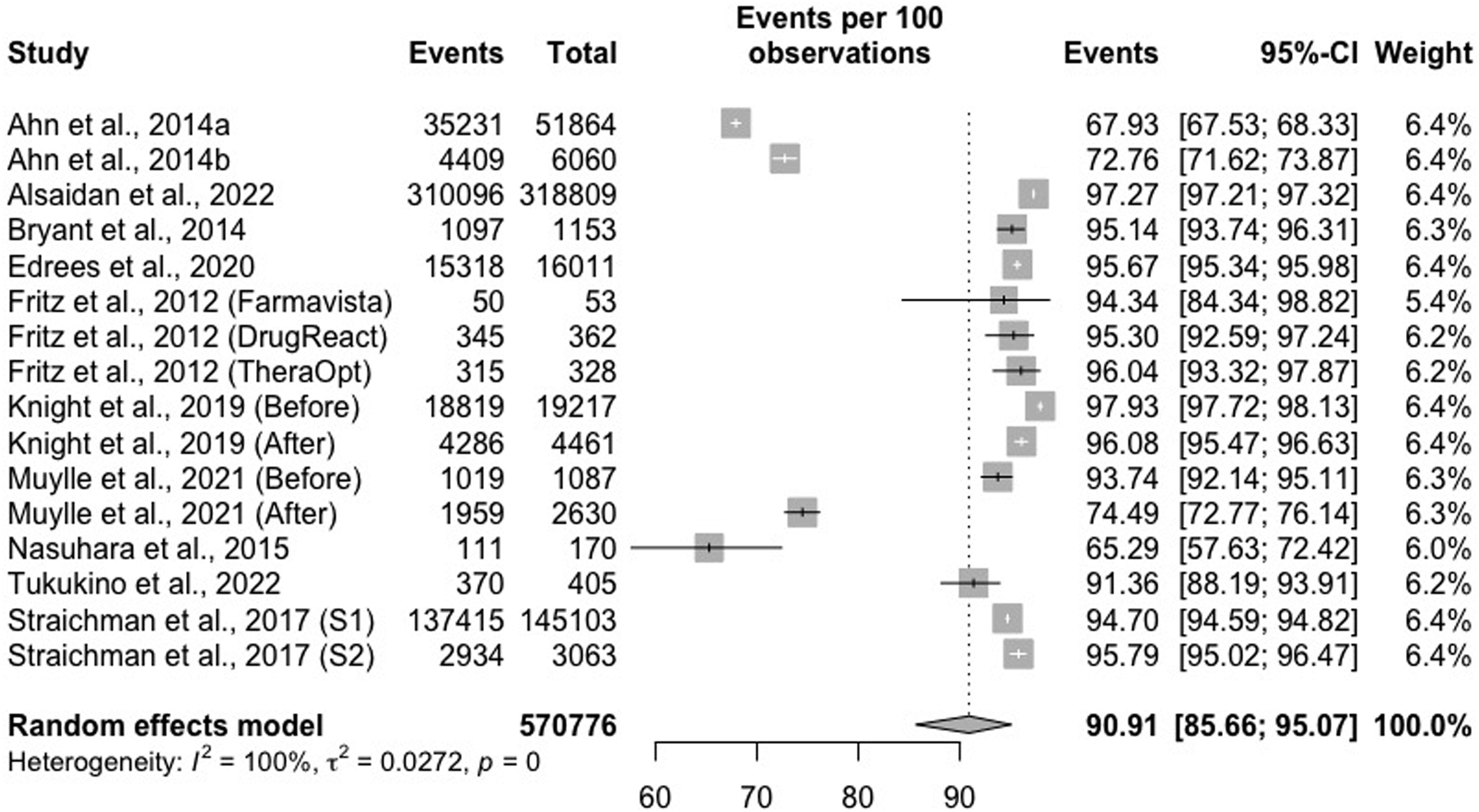
Quality assessment
Most studies demonstrated high methodological quality,21,23–27,29,32,33,35,36 one moderate,
22
and one low methodological quality
31
(Supplementary File S2). The questions that most indicated low methodological quality were questions 3 and 8: “(3)
Discussion
Studies have shown that most of the alerts generated at the time of prescription, indicating pDDIs, are ignored by physicians. Although there is no consensus on the ideal number of alerts that should be generated, it is already known that many inappropriate alerts can reduce users’ confidence in the system. 37 In this context, Wickens and Dixon (2007) 38 reviewed the literature in search of indirect evidence on this topic to establish a diagnostic reliability value below which automation becomes useless or even worse than the performance before its implementation. This analysis revealed that reliability of 70% was the “cutoff point” below which automation was worse than no automation. 38
In this review, we evaluated the frequency of alerts generated by these CDSS when physicians prescribe medication for their patients and found a prevalence of 13.7%. We could consider a low value, given the number of drugs that interact with each other and the difficulty of the prescriber remembering all of them. However, even though this tool was designed to help the prescriber, we observed that these physicians ignored the pDDI alerts generated by these CDSS, where the prevalence of alert override was 90%. Thus, it is evident that these systems require adjustments so that the adherence to their pDDI indications increases and the number of alert overrides decreases (Figure 4). The methodological quality of the studies was qualified according to the percentage of “Yes” responses, where: Low < 50%; moderate= 50<75%; High = 75-100%. Y= yes, N = no, U = unclear and NA = not applicable.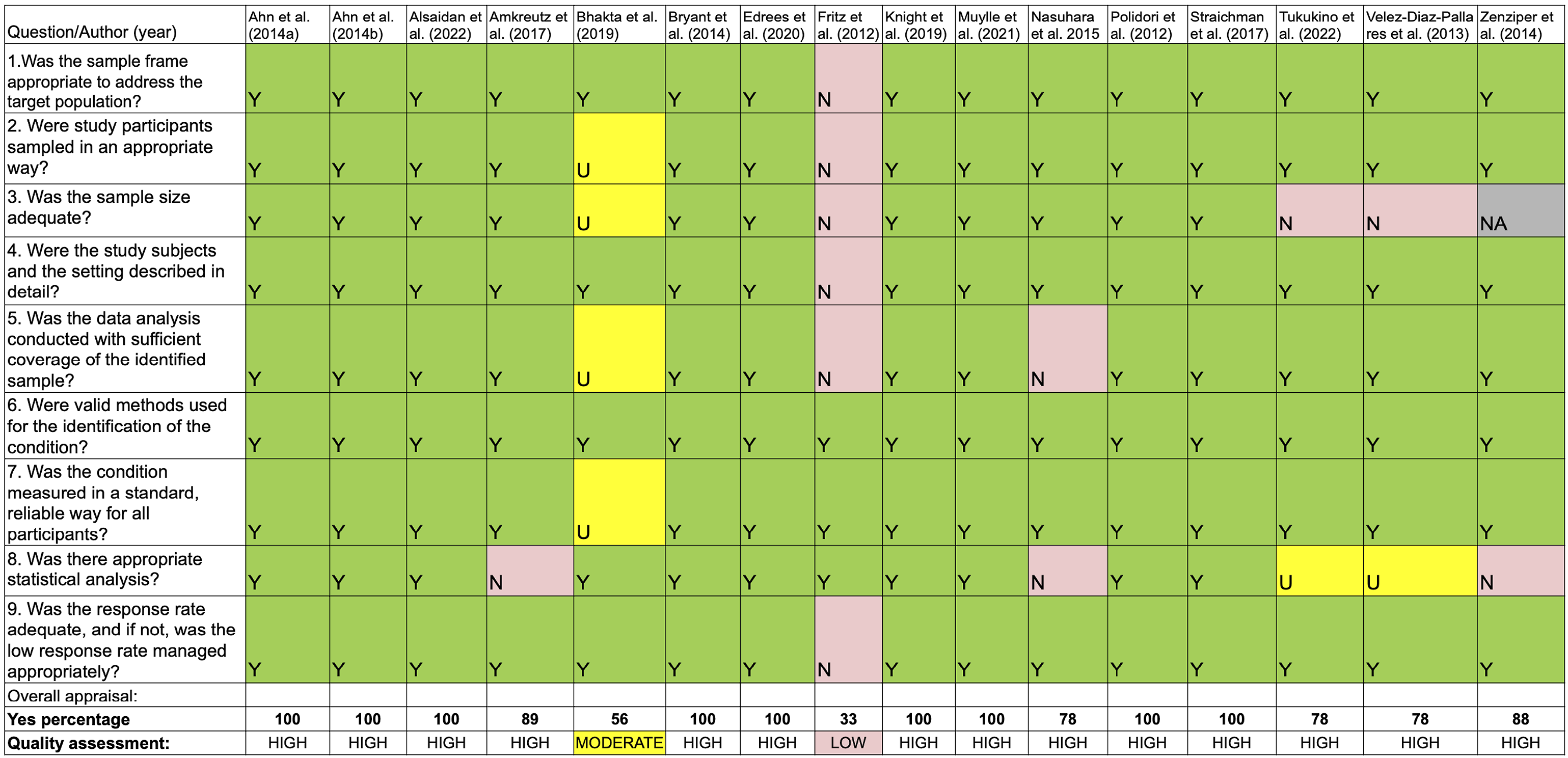
This is an important finding, which shows that despite being informed about pDDI, prescribers choose to ignore this information and subject the patient to possible ADE. However, some studies included in this review evaluated the consequences of alert override, where after the dispensing and administration of prescribed medications with pDDI, the authors checked whether ADE had occurred. Interestingly, these studies reported a low prevalence, ranging from 4% 31 to 7,8%, 24 or no occurrence of ADE 27 in the location where these studies were performed. However, it is important to remember that as these are studies carried out in hospitals, patients are monitored full-time, so physicians may have chosen to generate a greater benefit than the risk offered, as these patients would have all the necessary support in case of ADE. Thus, we could assume that pDDI alerts generated by CDSS would not represent a real risk to patients’ health, but when we extrapolate this data and apply this prevalence to a high number of patients, who often do not have the support of a hospital to manage this risk, we can consider CDSS as important tools to avoid ADE with serious consequences for patients.
Some studies have reported reasons why physicians override alerts. Among these most common reasons we can mention that prescribing doctors often consider that the benefit obtained from the administration of prescribed medications will be greater than the risk of developing an ADE.25,28,31 Furthermore, prescribers often considered the alert to have low clinical relevance and therefore overrode the alerts.25,28 In the latter case, it is important to highlight that many CDSS did not consider the patients’ laboratory tests, nor the route of administration, which directly influences the clinical relevance of the pDDI alert. But the most cited reason in one of the studies was “Other”, where prescribing doctors did not even indicate the reason for cancellation, which may indicate negligence on the part of the doctor. 27
Another issue to consider is the quantity and quality of these alerts; alerts of lesser clinical importance, when generated in excess, may tire these prescribers, also known as alert fatigue. This phenomenon has already been reported previously, showing that this problem can cause physicians to start ignoring these alerts after an excess of generated alerts, increasing the risk of alerts with greater clinical importance going unnoticed. 39 Some studies included in this review evaluated the effect of adjustments in the rules of the clinical decision support system to reduce the number of alerts generated. After these adjustments, only alerts of greater clinical importance were generated; with this, the number of alerts decreased significantly, although the acceptance of these alerts did not increase significantly, and physicians continued to override most of these alerts.21–23,32,34 These findings contribute to demonstrating the complexity of improving the acceptance of pDDI alerts. Among the studies that demonstrated the lowest alert override rates, we can highlight the studies by Ahn et al., (2014),25,26 which were conducted in South Korea, where the Korean government does not usually reimburse the value of prescribed medications that had pDDI alerts, so prescribers tend to pay more attention to the alerts. Another study that found a lower than average prevalence of alert override is the study conducted by Nasuhara et al., (2015), 27 where the authors report that because physicians had to consult directly with pharmacists to obtain override passwords, physicians could only decide to continue with their orders after receiving detailed pharmaceutical reasons for the contraindications. 27
After providing evidence-based information and removing minor alerts, a CDSS requires rigorous evaluation to determine the optimal sensitivity and specificity ratio to reduce patient harm. No system can achieve 100% sensitivity and specificity in a real-world setting. 40 Filtering lists of drug interactions in CDSS databases, keeping only clinically significant pairs, may improve the “alert fatigue” effect but also create liability concerns for clinicians, who could perceive these systems to be at risk of making mistakes. However, using a list of DDI based on consensus between professional societies or relevant regulatory bodies could increase confidence in these systems. 41
To reduce the number of alerts and increase their clinical relevance, the CDSS should not be used as an independent system but to work with the EHR and cross-check important patient information, such as laboratory test results, information on comorbidities, and other clinical parameters. Therefore, about 30% of alerts could be avoided if only five laboratory test results were integrated into the system, including potassium, white blood cell count, international normalized ratio, therapeutic drug monitoring, and glomerular filtration rate values. 36 Another way to reduce the number of alerts is to direct these alerts based on the specialty of the prescribing physician, for instance, not generating an excessive number of renal risk alerts for a kidney specialist with many years of experience. 40 With this, confidence in these systems tends to increase since physicians ignore alerts because of their lack of specificity; thus, alerts generated for the general population could be changed if the characteristics of patients and physicians were considered. 36
In this context, two literature reviews sought to analyze the reasons why doctors accept or do not accept the implementation of a CDSS.41,42 These reviews were conducted in different decades, however, their findings show similar results, where the authors report the barriers and facilitators in the acceptance of this technology, which represents a change in physicians’ routine. These reviews did not specifically focus on CDSS that generated drug interaction alerts and included other types of alerts related to medication prescriptions. However, we can use the findings of these studies as a basis to better understand the reason for the high prevalence of alert override shown in this study. As barriers to the acceptance of CDSS, the authors cite poor interface design, inadequate implementation (e.g. without prior training) and inadequate data maintenance. Furthermore, the authors of these studies report that for physicians to accept CDSS, they need to be easy to use, efficient, present clear and objective alerts and contain relevant information. It is worth noting that the definition of relevant information is subjective, as the same information may be relevant to one physician and not to another. Therefore, the authors suggested that physicians be involved during the development of these systems and that user-centered design may be a suitable method. 42
The strength of the present systematic review is consolidated in the following points: (a) analysis by paired blinded reviewers, (b) exhaustion of the literature search, (c) data validation, (d) search for expertise, and (e) quality assessment. Nonetheless, this study also had some limitations, including unclear outcomes in some of the analyzed articles; hence, they were estimated by the present article’s reviews. In addition, the variation in alert prevalence and alert override prevalence can be attributed to several factors. First, the rules of alerts issued by the systems varied considerably, and most studies did not provide information about this. Secondly, the data collection period was limited in most studies, where more than half of the included studies were conducted for less than a year. Third, institutions with different levels of health care complexity may value certain DDI as more important or relevant to the populations they serve, thus prioritizing a specific group of DDI alerts over others. Another limitation of this review was that the search strategy was limited to a time range between 2011 and 2023. This may have resulted in the low number of studies included, given that the subject is important and that many other studies on the topic may have been executed.
Conclusion
Our results show that prescribers ignore most of the alerts these clinical decision support systems generate since 90% of physicians override them. Even after adjustments were made to these systems to reduce the number of alerts and avoid professional fatigue, the number of alert overrides did not decrease satisfactorily. Therefore, these systems should be developed taking into account the particularities of each institution where this system will be implemented, as well as the experience and clinical specialty of the physician who will use this system. This could increase the users’ confidence and satisfaction with the CDSS and possibly decrease alert overrides, improving the clinical safety of the treatments offered to patients. Furthermore, integrated systems, where prescriptions take into account laboratory findings as well as individual characteristics of each patient, could also improve the acceptance of alerts by prescribers. In this systematic review with meta-analysis, we showed that despite the CDSS being recognized as an important tool to prevent adverse events related to drugs prescribed by physicians in health units, this instrument has been underused, wasting users’ time and money. Therefore, in some cases, to reduce the alert override rates, it may be necessary to apply more drastic solutions, such as not reimbursing the institution where the drugs with pDDI were prescribed.
Supplemental Material
Supplemental Material - Override rate of drug-drug interaction alerts in clinical decision support systems: A brief systematic review and meta-analysis
Supplemental Material for Override rate of drug-drug interaction alerts in clinical decision support systems: A brief systematic review and meta-analysis by Mariano Felisberto, Geovana dos Santos Lima, Ianka Cristina Celuppi, Miliane dos Santos Fantonelli, Wagner Luiz Zanotto, Júlia Meller Dias de Oliveira, Eduarda Talita Bramorski Mohr, Ranieri Alves dos Santos, Daniel Henrique Scandolara, Célio Luiz Cunha, Jades Fernando Hammes, Júlia Salvan da Rosa, Izabel Galhardo Demarchi, Raul Sidnei Wazlawick, and Eduardo Monguilhott Dalmarco in Health Informatics Journal.
Supplemental Material
Supplemental Material - Override rate of drug-drug interaction alerts in clinical decision support systems: A brief systematic review and meta-analysis
Supplemental Material for Override rate of drug-drug interaction alerts in clinical decision support systems: A brief systematic review and meta-analysis by Mariano Felisberto, Geovana dos Santos Lima, Ianka Cristina Celuppi, Miliane dos Santos Fantonelli, Wagner Luiz Zanotto, Júlia Meller Dias de Oliveira, Eduarda Talita Bramorski Mohr, Ranieri Alves dos Santos, Daniel Henrique Scandolara, Célio Luiz Cunha, Jades Fernando Hammes, Júlia Salvan da Rosa, Izabel Galhardo Demarchi, Raul Sidnei Wazlawick, and Eduardo Monguilhott Dalmarco in Health Informatics Journal.
Supplemental Material
Supplemental Material - Override rate of drug-drug interaction alerts in clinical decision support systems: A brief systematic review and meta-analysis
Supplemental Material for Override rate of drug-drug interaction alerts in clinical decision support systems: A brief systematic review and meta-analysis by Mariano Felisberto, Geovana dos Santos Lima, Ianka Cristina Celuppi, Miliane dos Santos Fantonelli, Wagner Luiz Zanotto, Júlia Meller Dias de Oliveira, Eduarda Talita Bramorski Mohr, Ranieri Alves dos Santos, Daniel Henrique Scandolara, Célio Luiz Cunha, Jades Fernando Hammes, Júlia Salvan da Rosa, Izabel Galhardo Demarchi, Raul Sidnei Wazlawick, and Eduardo Monguilhott Dalmarco in Health Informatics Journal.
Footnotes
Authors’ contributions
MF, GSL, ICC, JMDO, ETBM, JSR, CLC, and JFH performed the bibliographic search, and IGD and EMD validated it. MF, RAS, DHS, and ICC participated in elaborating the data extraction table. EMD, IGD, and RSW analyzed and critically reviewed the data. MF and JMDO performed the meta-analysis. MF, EMD, and IGD wrote the general text, and each stage was submitted for analysis and review by the co-authors, with their due criticism. MF, RAS, DHS, JMDO, ETBM, and IGD evaluated the quality assessment. IGD and EMD critically reviewed the drafts and subsequent steps. All authors approved the final version of the manuscript for submission. All authors had full access to all data in the study and assumed responsibility for the data’s integrity and data analysis accuracy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior [Coordination for the Improvement of Higher Education Personnel] – Brazil (CAPES) – Finance Code 001, and by Brazilian Ministry of Health (e-SUS PHC Project Stage 4). The RSW and EMD are productivity fellow in technology development and innovative extension of CNPq.
Ethical statement
Registration and protocol
This systematic review protocol was based on PRISMA-P and registered in the PROSPERO International Prospective Register of Systematic Reviews under registration number CRD42021261967.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
