Abstract
Digitalized healthcare services offer remote and cost-effective treatment of diabetes patients. Thus, the present online study analyzed the readiness to use telemonitoring among Austrian diabetes patients. We developed and validated a German version of the patient telehealth readiness assessment tool and performed quantitative context analysis of free-text comments on perceived barriers and benefits of telemonitoring. Participants (n = 41, 42.6% females) achieved a medium average readiness level for telemonitoring. The three top benefits were intensified care, shorter travel and waiting times, and better therapy adjustment. The top three barriers were data privacy issues, loss of personal communication and focus on blood sugar, and teledoctor competence. Diabetes patients represent a suitable target group for remote treatment opportunities. However, a shift from traditional face-to-face medical care to exclusive telemonitoring treatment from diagnosis to consultation and treatment requires fundamental new legal framework conditions.
Introduction
Diabetes mellitus is a common chronic metabolic disorder characterized by elevated blood glucose levels as a result of imbalanced insulin secretion. 1 Type 1 and type 2 diabetes are the two most common types of diabetes, both of which are associated with loss of insulin-producing beta cells in the pancreas. To reduce the risk for concomitant complications such as diabetic retinopathy, nephropathy, and vasculopathy, diabetes patients need lifelong drug therapy accompanied by close-meshed medical counseling.
Approximately 450 million adults are affected by diabetes worldwide in 2017. 2 In Austria, an estimated number of 573,000–645,000 people live with diabetes. 3 With about Euro (EUR) 4000 spent per person with diabetes, Austria spends 12 percent of the nation health expenditures for diabetes treatment, which corresponds to the respective average global estimates. Diabetes patients face nearly twofold higher health expenses compared to people without diabetes, mainly due to inpatient treatment. 4 Duftschmid et al. 5 showed that about three quarters of diabetes patients visit two or more different care providers during an observation period of 2 years.
Recently, telemonitoring systems that continuously or intermittently capture relevant clinical parameters are increasingly implemented for the follow-up of diabetes. 6 Diabetes patients’ self-reliant use of an automated support tool to collect, transmit, analyze, and respond to medical data has the potential to sustainably reduce care costs and increase health and well-being. Relevant research has shown beneficial effects on objective health parameters including blood pressure, blood glucose levels, and body weight as well as reduced hospital admission rates.6–9 However, the majority of telemonitoring pilot projects—although proven to be effective—fail to be implemented in standard medical care, mostly due to a lack of user acceptance, public funding, and incentives for health professionals.6,10–12 User awareness and perceptions of innovative health technologies aimed at influencing compliance and self-management of chronic conditions are essential for their successful adoption in everyday life. As respective data are scarce for the Austrian situation, we conducted a cross-sectional study that aimed at (1) evaluating telemonitoring readiness as well as perceived benefits and barriers in the context of diabetes care among Austrian diabetes patients and (2) creating and validating a German version of the patient telehealth readiness assessment tool (PatRAT) on the basis of the original English tool.11,13,14
Methods
Study population
The study protocol, which was adapted from previous research, was approved by the ethical committee of the Medical University of Vienna, Austria (no. 1197/2017) and conducted in accordance with the principles of the Declaration of Helsinki and established guidelines.15,16 We reported the study findings following established guidelines. Study subjects gave their consent to participate explicitly by filling in the online questionnaire. Participation was voluntary, anonymous, and could be interrupted without reason at any time. IP check prevented multiple entries from the same individual. We did not perform randomization of questionnaire items. Respondents had the possibility to review and change their answers through a back button. We checked data for completeness as incomplete questionnaires were not eligible for analysis. Survey data were stored securely and were protected from unauthorized access.
The web survey was accessible from 10 April to 30 June 2017 via SoSci Survey, 17 addressing German-speaking adult diabetes patients across the nine federal states of Austria. We posted the call for survey participation and the according link on various online platforms and official Facebook pages of relevant organizations including Diabetes Austria, 18 the Austrian Diabetes Association, 19 and Medtronic Diabetes Austria. 20 In addition, the survey link was posted to the Austrian Facebook groups for type 1 and type 2 diabetes.
Survey tool
We developed a German 36-item study questionnaire including an adapted version of the English PatRAT. 21 The German study questionnaire can be obtained upon request from the authors. The original PatRAT allows for adaption to specific research questions, countries, and study populations, which in our case was telemonitoring readiness among Austrian diabetes patients. 21 The questionnaire was forward–backward translated and validated following respective guidelines.22–24 Five laypersons and five researchers pre-tested the final German questionnaire for appearance and content. Subsequent adaptions included omitting the option “other,” as its meaning was unclear to testers, and replacing the original 5-point Likert-type scale with a 4-point scale to avoid neutral answers with limited relevance.
Three readiness domains for telemonitoring are covered. Core readiness deals with aspects of discontent with the current situation, desire for change, and need for telemonitoring services. Engagement readiness deals with awareness and assessment of telemonitoring-related benefits and obstacles. Structural readiness deals with technical pre-requirements and soft skills for telemonitoring. Summed means of all three readiness categories and also a global score specified the level of readiness for telemonitoring in the context of diabetes care, with higher scores indicating higher readiness. 25 Internal consistency was acceptable for core readiness (5 items, Cronbach’s α = 0.796, range 5–20 points) and engagement readiness (7 items, α = 0.737, range 7–28 points), moderate for structural readiness (5 items, α = 0.696, range 5–20 points), and poor for the global score (17 items, α = 0.543, range 17–68 points). The global score is interpreted in three levels of readiness, that is, high level: above 48, indicating that patients were in a good position to use telemonitoring; moderate level: between 34 and 48, indicating that patients faced certain items that adversely affect telemonitoring use; and low level: below 34, indicating that patients faced barriers toward the successful use of telemonitoring.
We also collected socio-demographic information such as age (in years), gender (male, female), education level (primary, secondary, and tertiary), and place of residence (East, South, and West of Austria according to the Nomenclature of Territorial Units for Statistics (NUTS), a hierarchical classification of administrative areas used across the European Union for statistical purposes). 26 We also asked participants for diabetes-specific details regarding diabetes type, methods of diabetes self-control, and partaking in the Austrian diabetes management program “Therapy active—Diabetes Under Control” predominantly for type 2 diabetes patients. 8 Furthermore, we collected opinions on telemonitoring on a 5-point Likert-type scale ranging from negative to positive by asking “How do you feel about telemonitoring?” We asked study subjects to rate their self-perceived level of innovativeness using the item “How would you rate your degree of innovativeness?” As possible options, we offered the adopter groups based on Rogers’ diffusion of innovation model, namely, innovator, early adopter, early majority, late majority, and laggard. 27 We asked participant to indicate their willingness to use telemonitoring services offered by their physician and to pay a deductible for their use on a 4-point Likert-type scale ranging from strongly disagree to strongly agree. Using a free-text comment box, we also investigated the participants’ actual annual diabetes treatment costs and the financial burden they would be willing to bear for as a deductible. For qualitative content analysis, we coded and sorted responses to the free-text questions in the categories benefits and barriers for telemonitoring. 28 Steps for preparing, organizing, and reporting findings from the free-text material were initial text work, developing main thematic categories, first coding, compiling coded passages, determining subcategories, second coding, category-based evaluation, and presentation of results.28,29 The codes were reviewed for accuracy by the research team members independently.
The link to the survey was clicked 73 times, whereas the questionnaire was fully completed 48 times. The final data set consisted of 47 questionnaires, as we did not include one record of a survey participant who was no diabetes patient (completion rate: 64.4%).
Statistical analysis
We conducted all statistical data analyses using SPSS version 25.0 (SPSS Inc., Chicago, IL, USA). We descriptively presented categorical data as absolute frequencies and percentages, and continuous data as mean, standard deviation (SD), and median, where appropriate. We reported 95 percent confidence intervals (95% CIs) of observed percentages based on 1000 bootstrap samples to provide population estimates for readiness levels. We calculated differences between variables using chi-square tests and correlations using Spearman’s rho correlation coefficient (rs). We used Cronbach’s alpha (<0.6 poor, 0.6–0.7 moderate, and >0.7 acceptable) to determine internal consistency of the scales. 30 Furthermore, we investigated the factor structure of the 17 items of the German telemonitoring assessment tool using principal component analysis (PCA) with Varimax rotation as used in similar studies.31,32 Our data were suitable of factor analysis with a Kaiser-Meyer-Olkin measure of sampling adequacy score of over 0.5, that is, 0.620, indicating an acceptable sample size and a significant Bartlett’s test of sphericity, that is, p < 0.001, indicating that there are adequate inter-correlations between the items. 33
Results
Average age of participants (n = 47) was 44.2 years (SD 15.6, range 18–77 years), 42.6 percent were females. Participants lived in all nine federal states of Austria, with the majority living in the Eastern part including Vienna, the capital of Austria. The majority of participants were type 1 diabetes patients (63.8%) and 34.1 percent were type 2 diabetes patients, with 21.3 percent had insulin-dependent and 12.8 percent had non-insulin-dependent type 2 diabetes. One participant did not further specify his or her diabetes type. Average duration of diabetes was about 16 years (mean 15.7, SD 11.0, range 1–45 years). The minority of the total sample participated in the Austrian therapy active program (8.5%). Only 10.6 percent of participants indicated to visit private doctors for diabetes treatment, as 80.9 percent have only statutory health insurance. For diabetes self-control, use of electronic documentation tools was more common (70.2%) than paper-based diaries (19.1%). In total, 21 (44.7%) participants reported on their predominantly used app for diabetes management in free-text box comments. Of those, MySugr (n = 10, 47.6%) and FreeStyle Libre (n = 8, 38.1%) were the two most commonly mentioned apps.34,35 Furthermore, 61.7 percent participants expressed a rather positive or positive attitude toward telemonitoring. The majority perceived their openness to innovation as high, with 85.1 percent counting to innovator, early adopter, or early majority.
The actual annual privately covered costs for diabetes treatment were in average EUR 914.5 (SD 893.7, range 0–4000) based on the reports of 32 participants without differences between diabetes types (type 1 diabetes: n = 19, mean EUR 840.3, SD 771.0; type 2 diabetes: n = 13, mean EUR 1225.0, SD 1368.3).
The majority of participants was not inclined to pay a deductible for telemonitoring treatment (mean 3.3, SD 0.8), with 70.2 percent strongly disagreed or disagreed to do so. Twenty participants filled out the free-text box to indicate the potential sum of a deductible in EUR (mean EUR 30.5, SD 27.2, range 0–100) without differences between the diabetes type (type 1 diabetes: n = 15, mean EUR 30.3, SD 22.6; type 2 diabetes: n = 5, mean EUR 30.0, SD 38.8). Most participants indicated their willingness to participate in telemonitoring services (mean 1.9, SD 0.9), with 83.0 percent agreed or strongly agreed to do so.
Table 1 shows scores of patient telemonitoring readiness stratified by the three categories: core readiness, engagement readiness, and structural readiness among Austrian diabetes patients obtained by the German translation of the original English telehealth readiness tool. Ratings for core readiness indicated a quite low perceived level of dissatisfaction with the prevailing healthcare provision (mean range 1.6–2.0), but also for desire for change and active involvement in health decisions (mean 2.7, SD 1.2). We found high approval for most of the items in the engagement readiness category, with highest ratings for “I have a sense of ownership regarding my well-being and that of my community.” (mean 3.8, SD 0.5). In the structural readiness category, the item “I have access to training on telehealth equipment and the ability to use the equipment.” yielded outstanding ratings (mean 3.7, SD 0.6) compared to the other items.
Ratings of patient telemonitoring readiness.
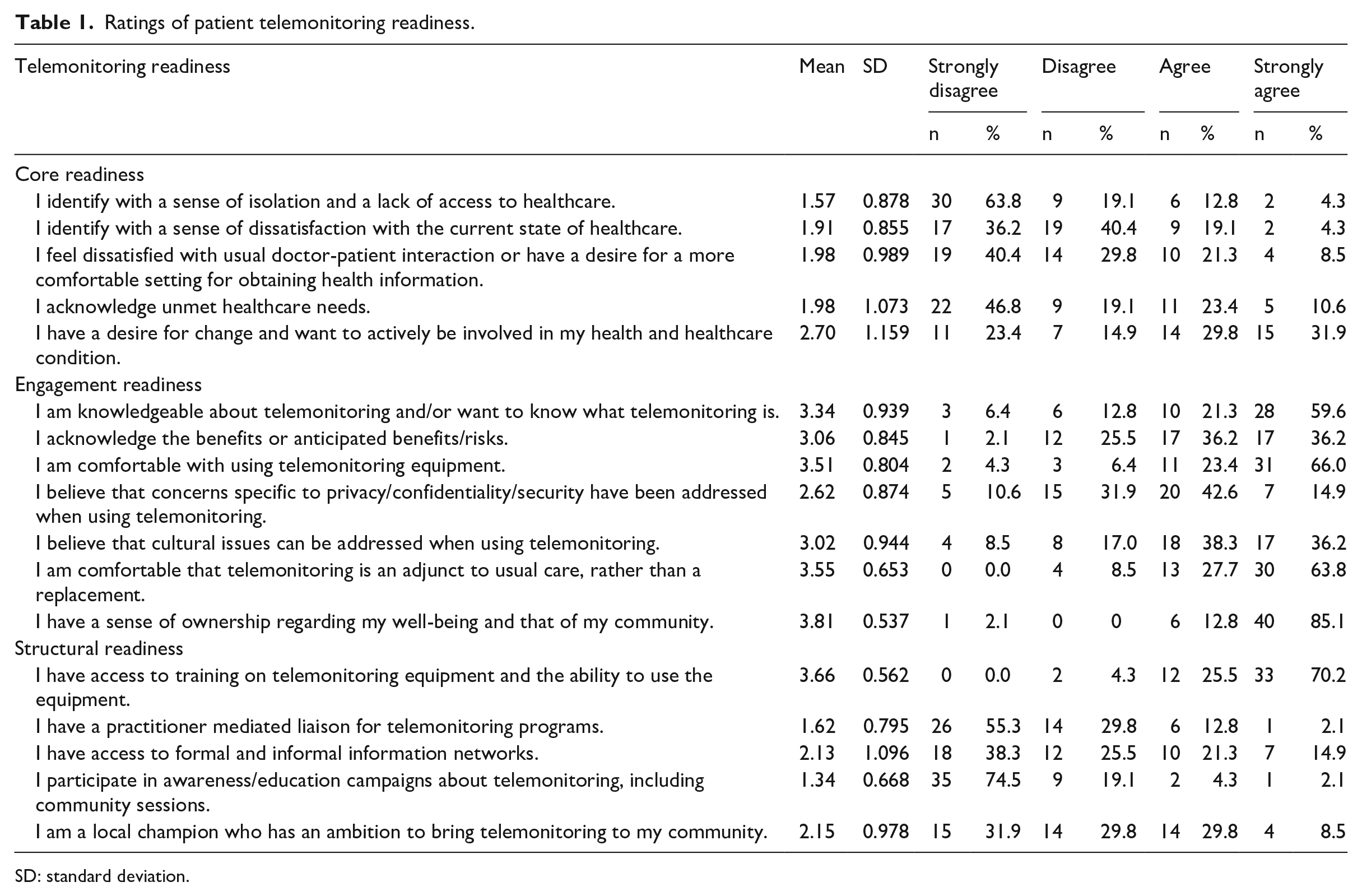
SD: standard deviation.
The average score for core readiness was 10.1 (SD 3.7), that is, 50.5 percent of maximum points were comparable to those for structural readiness (mean 10.9, SD 2.8, 54.5% of maximum points), both categories consisting of five items, while with 81.8 percent of maximum points, engagement readiness yielded much higher ratings (Table 2). The total scores ranged from 17 to 68 points. Average total readiness was in the category between 34 and 48 points (mean 44.0, SD 5.2), according to which several points have an unfavorable influence on telemonitoring use by patients. While 23.4 percent (95% CI 12.8%–36.2%) were in a good position, 4.3 percent (0%–10.6%) perceived barriers, and for the majority (72.3%, 59.6%–83.0%), certain items might adversely affect the use of telemonitoring to telemonitoring use. The level of readiness was positively correlated with participation in telemonitoring (rs = 0.33, p = 0.022) and attitude toward telemonitoring (rs = 0.47, p = 0.001).
Total and average subscores of patient telemonitoring readiness.
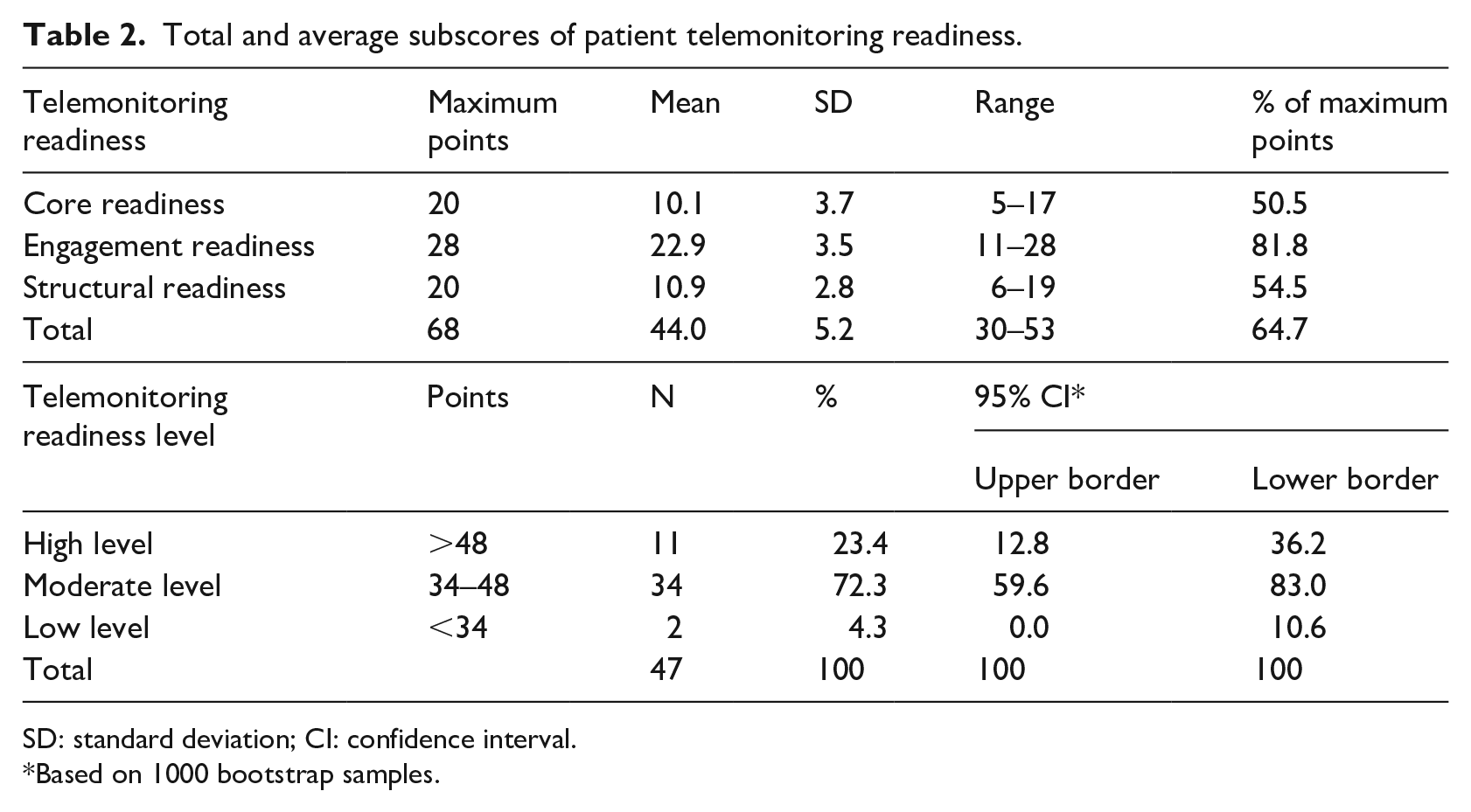
SD: standard deviation; CI: confidence interval.
Based on 1000 bootstrap samples.
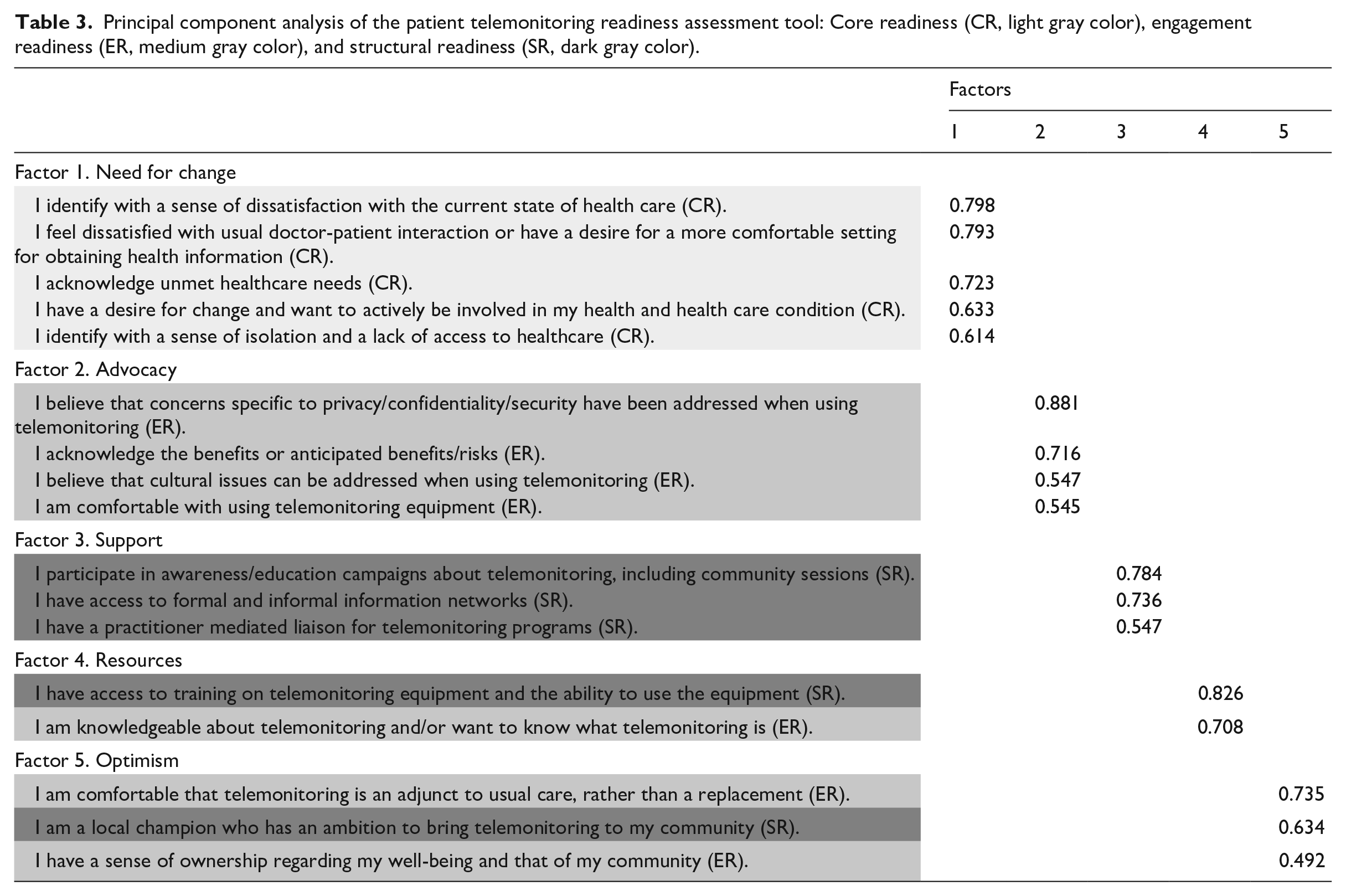
PCA identified a five-factor structure with all factors loading higher than 0.4, explaining 66.3 percent of the variance (Table 3). These factors were renamed according to the descriptive character of the items loading on them. All five items of the core readiness category loaded on the first factor (which corresponded to “need for change,” α = 0.796). Four items from engagement readiness loaded on the second factor (which corresponded to “advocacy,” α = 0.713), three items of structural readiness loaded on the third factor (which corresponded to “support,” α = 0.653), one further item from each structural readiness and engagement readiness loaded on the fourth factor (which corresponded to “resources,” α = 0.587), whereas the remaining items from structural readiness (one item) and engagement readiness (two items) loaded on the fifth factor (which corresponded to “optimism,” α = 0.537). The latter three factors included less than four items. However, a reduced two-factor PCA would only explain 41.4 percent of the variance.
Principal component analysis of the patient telemonitoring readiness assessment tool: Core readiness (CR, light gray color), engagement readiness (ER, medium gray color), and structural readiness (SR, dark gray color).
We collected in total 29 comments on expected benefits (61.7% of participants). The three most often mentioned benefits related to the possibility of intensified care by telemonitoring use (n = 18, 62.1% of total comments), potential shortening of travel and waiting times (n = 8, 27.6%), and better therapy adjustment (n = 7, 24.1%).
Examples for single quotes are “My sugar levels change very much. If I visit my doctor once every three months, he only checks on the last two weeks. I would like to have the whole time period examined,” “Avoid waiting times and respond quickly to spontaneous problems,” and “Closely-meshed controls for identifying treatment failures.”
We collected in total 31 comments on expected barriers (66.0% of participants). The three main barriers related to data security, privacy, and confidentiality concerns (n = 13, 41.9% of total comments), loss of personal communication and focus on blood sugar as well as concern regarding the competence of the teledoctor (both: n = 6, 19.4%). Also, 12.9 percent (n = 4) of study subjects explicitly stated that they would see no disadvantages of telemonitoring.
An example for a single quote is
Discussion
The present online study investigated prevailing opinions on telemonitoring among Austrian diabetes patients and whether they are ready to use these services. Assuming that Austria is a country with adequate financial and professional capacities to implement these services, we were especially interested in diabetes, a chronic health condition that is very suitable for digital remote self-monitoring.7,36 The study contributes to the existing literature by providing a so far unpublished German version of the well-known PatRAT, which we adapted and validated to study telemonitoring in an Austrian context. 21
Participants expressed an overall moderate readiness level and positive feelings about telemonitoring. We found high approval for most of the items in the engagement readiness category, which could be interpreted as a high empowerment level in the online Austrian diabetes community. Thus, the potential for telemonitoring implementation is very likely not fully exploited yet.
A healthy lifestyle adapting physical activity, dietary, smoking, and alcohol habits reduces the risk for late-onset diabetes. 37 So, in addition to medication-based treatment, telemonitoring applications should also strive for a behavior-related risk factor reduction by, for example, motivating for increasing sport activities following the principles of gamification.38,39 A lack of viable business models hampers successful integration of these innovative diabetes telemonitoring services in Austrian standard care.10,11,15 Despite experiencing positive effects on health parameters in the course of a longer term pilot telemonitoring project, diabetes patients were not willing to pay for these services for themselves. 6 Likewise, in our sample, the minority would be willing to contribute financially to advanced telemonitoring-based services, even though they expected to benefit from advanced treatment options, especially due to intensified care, shortening of travel and waiting times, and better therapy adjustment. However, it is questionable whether a deductible would increase the sense of responsibility and motivation of the diabetes patients.
Although some participants explicitly perceived no obstacles for telemonitoring, concerns in regard to data security and confidentiality, loss of face-to-face-communication, and the teledoctor proficiency were raised among others. These findings are in line with other publications reporting on similar perceived obstacles for electronic disease management tools, with concerns on data security and privacy being most frequent.40,41 Noteworthy, Demiris et al. 42 did not find privacy concerns regarding telemonitoring among elderly home residences. In contrast, Ignatowicz et al. 43 found concerns regarding confidentiality as well as a shift of the boundaries of the patient–doctor relationship in the context of electronic health communication among a UK study population of young people with chronic disease.
According to Emani et al., 40 innovativeness increases willingness to engage in electronic health communication among patients. In our study, the majority of participants showed a quite high degree of self-rated innovativeness. Choice of telemonitoring services may influence compliance rates among users with mobile applications showing higher acceptance compared to web browser–based user interfaces. 44 High use of digital devices in consultation settings by healthcare professionals has been shown to decrease patient satisfaction and lead to communication problems. 45 Noteworthily, electronic health communication between doctors and patients could paradoxically lead to dependency rather than establishing autonomy on the patient side. 43
A closer evaluation of collected quotes identified several observations on how telemonitoring adoption among diabetes patients might be increased. IT-related health services might not have to be delivered permanently but could be most efficiently employed in specific phases in a diabetes career such as diabetes diagnosis, therapy adaptation, stressful events, unexplainable values, or vacation and when offered to a well-defined target population. 46 Also, some individuals might feel motivated by regular monitoring to adhere to therapy and healthy lifestyle. For these patients, permanent or longer term telemonitoring would be more effective in diabetes treatment. 6
Awareness of one’s own illness is the most important prerequisite for compliance. 6 Reasons for non-compliance can range from strict denial to a situation where telemonitoring is too much effort for the perceived current problem and will, therefore, not be used. 39 Cognitive understanding, especially language skills, and technology affinity are required personality traits that often, but not exclusively, correlate with age and, notably, also relatives can support. 47 Our survey could not find correlations between readiness for telemonitoring and age, sex, or education. Notably, we did not analyze whether relevant diabetes-related complications or other diseases of participants affected their telemonitoring readiness. However, particularly suitable types of patients for telemonitoring could include newly diagnosed, insulin-dependent, gestational, or multimorbid diabetes patients, patients who live in rural areas, and very health conscious or anxious patients.6–9
We designed and validated a German version of a well-established survey tool for the first time, thus providing a valuable instrument for further research in this area. We conducted PCA to present the items from the assessment tool as a set of new orthogonal variables and to display patterns of similarity of the observations, as our data met the assumptions for this procedure in accordance with comparable studies.11,15,31,32 Further validation of the three readiness scales is required to establish the construct validity in a larger representative sample, potentially confirming our suggested five-factor structure. This cross-sectional study aimed at testing design feasibility for consecutive research projects. Thus, the study questionnaire could serve as a useful research instrument for assessing trends and new developments in a larger scale survey among representative study samples and evaluate progress of national telemonitoring implementation.
The findings of this study are subject to several limitations. We expected a low response rate in a cross-sectional online study among a purposive sample of diabetes patients and abstaining from sample size justification due to the feasibility nature also limiting representativeness and generalizability of the study results. However, statistical measures showed that the power of the study was adequate for PCA. We also collected self-reported information, thus introducing response bias. Nonetheless, participants were internet-savvy diabetes patients who very likely resembled the target population for telemonitoring-based diabetes care. The single item assessing innovativeness might have limited power to measure the underlying construct. However, the item is derived from the well-established and reliable diffusion of an innovation model. We did not provide incentives for participation, which might have increased participation rates. 48 We developed a study questionnaire in German on the basis of its original English version to use it as an online survey instrument to collect data nationwide in a short period of time. Main challenges regarding translating the questionnaire were adapting the specific terminology to concepts used in German as well as gender-neutral language. We also modified the answer options of the questionnaire to respond to the results of the pre-testing, which might decrease comparability of the instrument the original PatRAT. Nevertheless, we aimed at generating a novel survey tool suitable for capturing the opinions of the German-speaking online diabetes community in Austria.
Conclusion
The high expectations toward digitalization of healthcare services are in contrast to the low diffusion of telemonitoring services. To provide so far lacking evidence for prevailing perceptions and expectations in the context of the Austrian digital health economy, our study analyzed the readiness to use telemonitoring services for diabetes care. Our data revealed that Austrian patients, who are important stakeholders for successfully implementing a nation-wide telemonitoring strategy, showed a moderate readiness for telemonitoring. Study participants perceived themselves as open to innovations but did rather not want to pay for telemonitoring themselves.
This study provides so far unknown empirical knowledge on how Austrian diabetes patients perceive telemonitoring and which factors might affect telemonitoring adoption in diabetes care, as a relevant example for chronic disease requiring a high level on lifelong self-care and compliance. To overcome potential barriers, we suggest initiating public information campaigns to improve knowledge among all healthcare stakeholders and increase their mutual understanding and cooperation. Financial aid for evidence-based and secure telemonitoring tools might probably be the most effective way to increase quality of life and empowerment of diabetes patients.
Footnotes
Acknowledgements
The authors are grateful to the participants of the pre-test for their helpful and valuable detailed comments. The authors also express their sincere appreciation to all survey participants.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
