Abstract
Health Information Technology is now widely promoted as a means for improving patient safety. The technology could also, under certain conditions, pose hazards to patient safety. However, current definitions of hazards are generic and hard to interpret, particularly for large Health Information Technology in complex socio-technical settings, that is, involving interacting clinical, organisational and technological factors. In this article, we develop a new conceptualisation for the notion of hazards and implement this conceptualisation in a tool-supported methodology called the Safety Modelling, Assurance and Reporting Toolset (SMART). The toolset aims to support clinicians and engineers in performing hazard identification and risk analysis and producing a safety case for Health Information Technology. Through a pilot study, we used and examined the toolset for developing a safety case for electronic prescribing in three acute hospitals. Our results demonstrate the ability of the approach to ensure that the safety evidence is generated based on explicit traceability between the clinical models and Health Information Technology functionality. They also highlight challenges concerning identifying hazards in a consistent way, with clear impact on patient safety in order to facilitate clinically meaningful risk analysis.
Introduction
The introduction of Health Information Technology (HIT) can have positive and negative impact on patient safety.1–3 In this article, we focus on the potential hazards and associated risks rather than on the expected benefits. In this regard, different international and national standards and initiatives have emphasised the importance of adopting safety risk management principles for the design and deployment of HIT.4–6 These typically are centred on an explicit description of the technology and its context, the identification of the hazards and the assessment and management of the risks associated with these hazards during the design, use and maintenance of the technology.7,8
The systematic and proactive implementation of these principles is well understood and established in traditional engineering domains such as aviation and nuclear power. 9 In such domains, the technology, procedures and organisations are well defined and controlled, as are the system boundaries and interfaces. This enables focused hazard identification and risk analysis. 10
However, the above cannot be assumed in healthcare. 11 This is mainly due to the inherent complexity, flexibility and scale of healthcare services, some of which are irreducible, for example, varying the care in order to suit patients with different clinical conditions, together with individual and social needs and constraints. 12 This, in turn, complicates the safety analysis processes for HIT. Such processes require a clear and structured description of the clinical environment within which the technology is deployed. 13 These safety processes focus on two notions: hazards and risks.
A hazard is defined as ‘
Despite its central role in safety processes, the concept of hazard is loosely defined in the safety literature. The above definition by National Health Service (NHS) Digital in England is consistent with the definition offered by the International Organization for Standardization (ISO) of a hazard as a ‘
Our recent review of HIT safety practices in England
21
supports this view, highlighting that the
It is important to note that, like hazards, there is much debate about risk and the way in which it has been approached from different perspectives.22,23
In this article, we present a tool-supported methodology for supporting proactive and systematic hazard identification and risk analysis for HIT. In particular, our article considers three questions:
In order to answer
The rest of the article is organised as follows. We first present our conceptualisation of hazards for HIT. We then introduce SMART as a tool-supported methodology for implementing this conceptualisation. Next, we present a pilot study on the use of SMART for analysing the safety of electronic prescribing in three acute hospitals. Finally, we discuss our results and explore the strengths and weaknesses of our approach, followed by our conclusions.
Conceptualising hazards for HIT
The list of hazard definitions discussed in ‘Introduction’ shows a lack of clarity, focus and consistency in how the fundamental concept of hazard is described. Building on the results and recommendations in the recent review of HIT safety practices by Habli et al., 21 we define the following criteria for the concept of hazards as a basis for improving hazard identification practices for HIT:
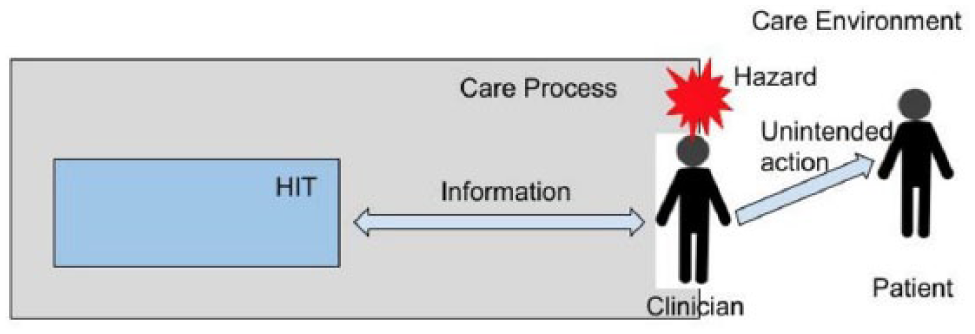
Hazard boundary illustration.
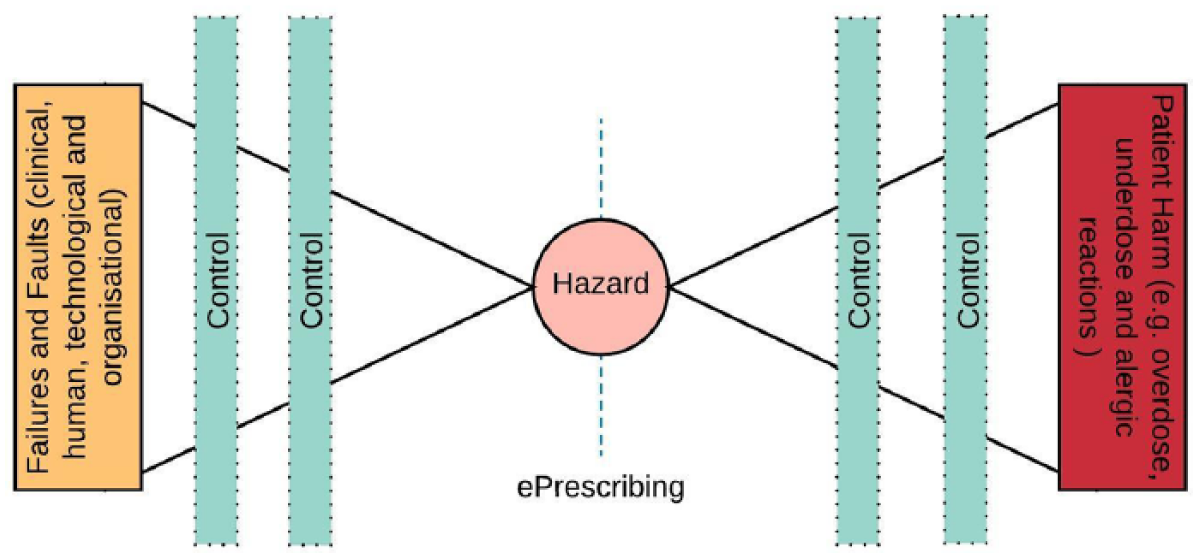
An example bowtie model.
SMART: tool-supported methodology for hazard identification and risk analysis
A fundamental aspect of the above conceptualisation is the linkage between the clinical processes and settings on the one hand and the HIT functionality on the other hand. A hazard associated with the technology cannot be identified, and its risk analysed, in isolation from the healthcare setting.
To support the above and to operationalise the hazard conceptualisation in ‘Conceptualising hazards for HIT’, we developed the SMART. SMART provides a self-contained platform for developing explicit clinical safety cases for HIT. 5 A safety case documents an argument, based on the evidence, that is, mainly based on hazard identification and risk analysis, for why the system is considered to be safe for a given application in a given environment. 14 In SMART, the safety analysis is clinically driven through a structured model of the clinical processes and an explicit description of the care settings.
The development of SMART has been model driven, based on the Eclipse Modeling Framework (EMF), 26 which provided the basic structure of the Java code. The core aspects of the data model are described in ‘Hazard identification’. The development of the toolset has been agile and iterative, allowing continuous feedback from the clinical users, representing NHS Digital and different healthcare providers and technology firms. The primary users of the tool are the Clinical Safety Officers (CSOs) 14 who, in their capacity as experienced clinicians, are expected to lead the HIT risk management activities. CSOs are typically supported by a team of clinicians, engineers and analysts.
From a methodological perspective, SMART implements the hazard conceptualisation by supporting three central HIT risk management activities: 14
Modelling the clinical context and the HIT system, ensuring that (1) the clinical setting, including the decision making process and the flow of clinical activities, (2) the HIT functions and (3) the mapping between the clinical setting and HIT functions are explicitly defined;
Identifying hazards in the modelled clinical setting, ensuring that the hazards associated with the HIT functions are explicitly traced to the contextual factors;
Analysing the risks associated with the identified hazards, showing how these risks, and their control measures, could vary across different clinical settings and HIT functions.
These activities are described in the next three sections, with emphasis on the explicit interlinks between the clinical and technological factors, including the underlying SMART data model and the key user-interface aspects of the tool design.
Clinical context and HIT modelling
Central to the design of SMART is a graphical flow-charting interface through which a clinical process of activities and decisions can be modelled. This is in essence similar to process mapping with which clinicians are already familiar. The diagrams produced using the toolset represent the sequence of steps that a patient may encounter as they progress through the care system (Figure 3). We refer to these diagrams as ‘Care Processes’, and it is through them that the core information for Hazard Identification and Risk Analysis is collected (i.e. safety in the clinical context). This simple representation provides an expressive means for describing pathways of care while being simple and intuitive enough to use with little training. Within a care process, an activity represents some action carried out in a health or social care setting. In order to ensure specificity in the identification of HIT-related hazards, the care process editor in SMART allows activities and decisions to be associated with particular HIT functions. The HIT systems and their associated functions are defined by the user in a separate section of SMART – the ‘System Editor’. SMART also allows the user to define ‘Care Settings’, which describe and correspond to services and locations, for example, Maternity Unit or Pharmacy, that a patient may visit or use during the process of receiving care. A setting may be associated with an activity/decision in the Care Process, thus providing additional context for the activity/decision, for example, the level of staffing or noise in the defined setting.
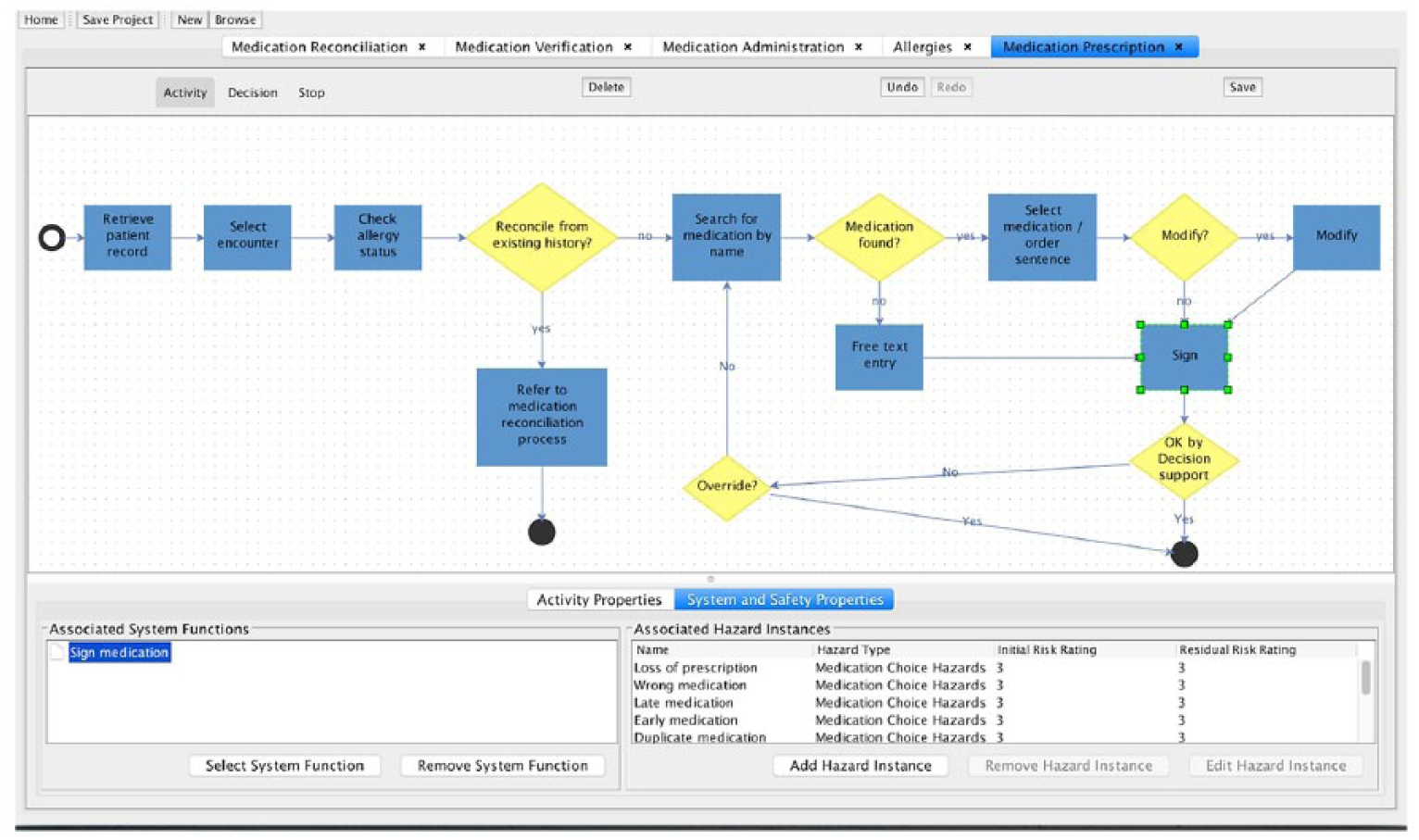
Example Care Process linked to a HIT function and list of Hazard Instances.
Hazard identification
Within SMART, the concept of a hazard is decomposed into two related aspects. First, the user may create ‘Hazard Types’ with no reference to the context within which they may occur (e.g. late prescription or wrong dose). Second, the user can associate a ‘Hazard Instance’ to any Hazard Type. Instances are specific occurrences of the hazard defined by the associated Hazard Type. To enforce contextualisation of Hazard Instances, they must be associated with both an Activity or Decision in a Care Process and a System Function. In effect, this ensures that Hazard Instances are only discussed in a specific context (e.g. ‘wrong dose prescribed by an Obstetrician in a Maternity ward’), thereby helping to provide a shared contextual understanding about the relative risk of a hazard. The degree of specificity in describing the relevant contextual factors will inevitably depend on the clinical setting and the stage at which the analysis is performed (i.e. during design or deployment). For instance, a high-level description of a hazard as ‘wrong dose prescribed in secondary care’ might be sufficient for a technology firm to allow the engineers to design holistic controls such as automated rules for dose range checking. However, once a HIT system is selected for deployment in a specific clinical setting, the clinicians and engineer will have to be more detailed in the description of the contextual factors, for example, ‘wrong dose of
In essence, in SMART, a Hazard Instance is a product of four components
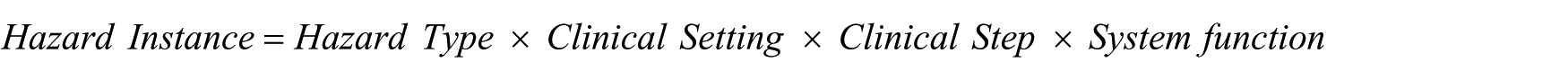
The above is shown in the SMART data model extract in Figure 4, and illustrated in grey. The model, defined in the Unified Modelling Language (UML), 27 specifies the relationships between the primary concepts in SMART. A Hazard Instance has mandatory links with a Hazard Type and a Clinical Step (which in turn has to be associated with a clinical setting). A Hazard Instance has a link with a System Function, which can be optional in order to allow the identification of non-HIT-related Hazard Instances. It is important to note that SMART, in this respect, aims to ensure that the necessarily types of links are established. However, it is neither possible nor desirable for the tool to dictate the content of the hazard description, especially as the analyses and the safety case are expected to evolve in an incremental manner as a collaboration between the engineers and clinicians.
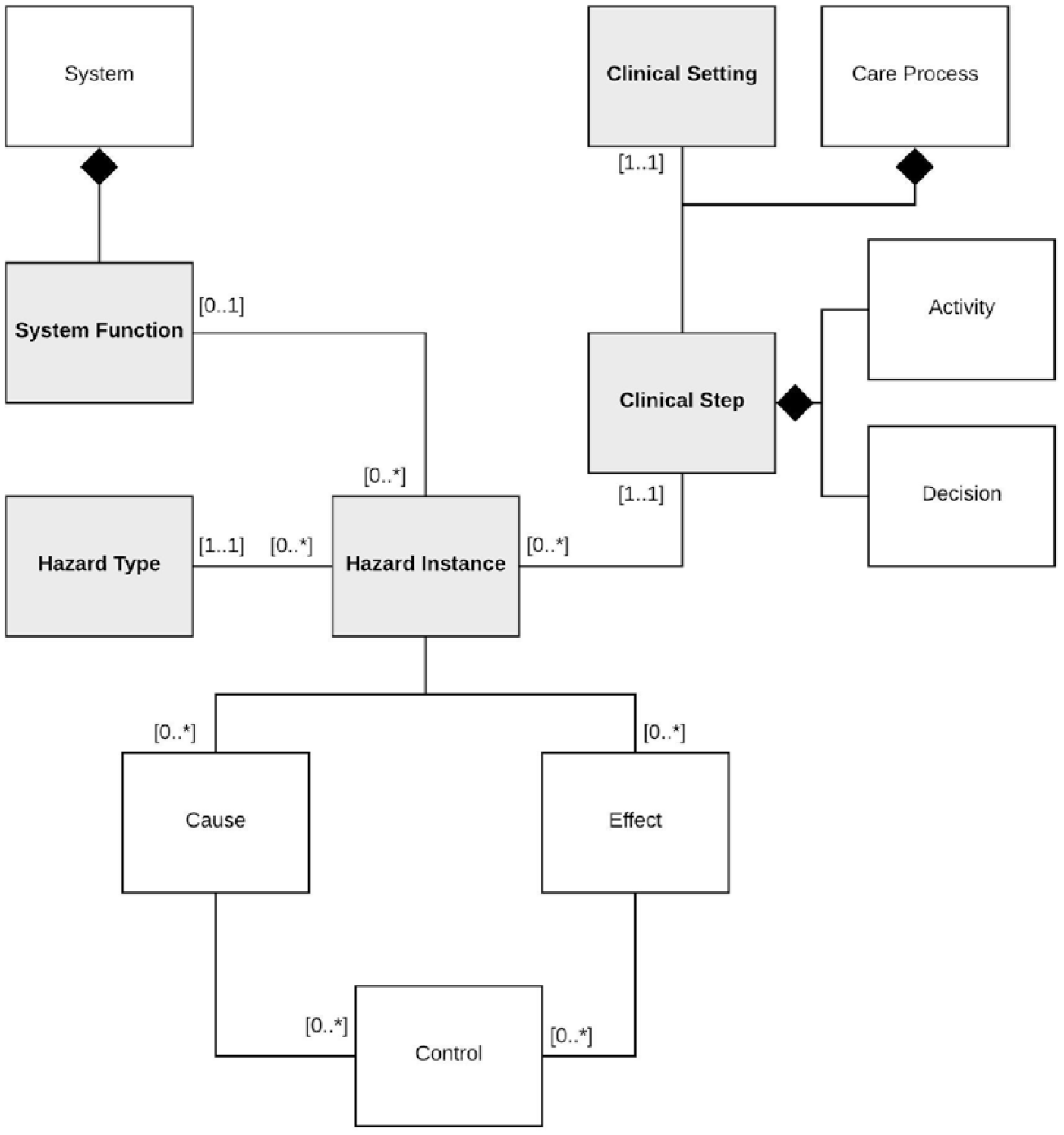
Hazard model in SMART.
Risk analysis
Hazard Instances are associated with several other pieces of information. Severity and likelihood ratings must be specified for all Hazard Instances, hence determining the level of risk, typically based on a Risk Matrix, that is, determining the level of risk based on the likelihood of the harm against the potential severity of the harm. It is important to identify the difference between initial and residual risk ratings. Initial risk ratings are those which describe the hazard instance before the implementation of controls. Residual risk ratings define the same three elements (severity, likelihood and risk) after the implementation of controls. Much of these data corresponds to the entries in the ‘Hazard Log’ section of the clinical safety case report template developed by NHS Digital. 14 Thus, with the addition of some extra information provided in another integrated editor, SMART automatically generates and exports conformant reports using the data from modelling and analysis in the tool.
Traceability
The traditional approach of presenting HIT safety evidence as written clinical safety case reports or spreadsheets makes it difficult to understand the connections between various components of the analysis. SMART’s data model was structured to ensure that the logical relationships between the model components as depicted in Figure 4 are automatically established and managed. All of the data is user defined as they require expertise and judgement of both clinical and engineering staff. However, by enforcing these relationships, the inherent structure of dependencies between the constituent parts of the hazard and risk analysis is clarified.
Although SMART insists on specifying, as a minimum, a care setting, a care process, a HIT function and a hazard type as a prerequisite for declaring a hazard instance (i.e. in order to ensure that the hazard instances are clinically meaningful and traceable), the approach is flexible in the level of detail that a user enters in describing care settings and processes, leaving it to the user to select an appropriate socio-technical model28,29 and the granularity of the clinical activities and decisions, considering the complexity and criticality of the HIT deployment.
Pilot study
As we are interested in the impact on practice, a pilot study, through an actual in-depth experience, is best suited for exposing the strengths and weaknesses of the hazard conceptualisation and its implementation in SMART. Essentially, we are interested in identifying and examining insights from the use of SMART, that is, hurdles, mitigations and workarounds, as well as sharing the outcomes of the tool, that is, list of hazards and associated risks.
Study overview
The initial evaluation of SMART included the use of the approach to develop safety cases for two HIT systems in four large acute hospitals, one national clinical service, one app and one telehealth platform. In this article, we describe a HIT deployment in acute care, covering three hospitals, in which SMART was used in the Hazard Identification and Risk Analysis for an electronic Prescribing and Allergies Management (ePAM) system.
Medication management was selected as it is one of the most safety-critical processes in healthcare. 30 Increasingly, the process is supported by HIT, covering the different phases, mainly prescribing, preparation and verification, administration and monitoring, including supporting activities for reconciliation and allergies management. Both the potential safety benefits and risks associated with HIT for medication management are highlighted in the literature.31,32 Here, we limit the scope to analysing and managing the safety risks of ePAM. More specifically, we examine how proactive and systematic Hazard Identification and Risk Analysis, supported by SMART, can generate the evidence in order to support an explicit safety case for a complex socio-technical ePAM. The system is selected not on its own unique merits but because it represents a typical use of HIT, supplied by an external technology firm, in order to support prescribing and allergies management as part of a wider HIT deployment, for example, including electronic health records.
Eight multidisciplinary workshops were organised, including a meeting dedicated for planning. The purpose of these workshops was to identify the hazards and analyse the risks associated with the deployment of ePAM. SMART was used as the primary approach for driving the analysis and recording the outcomes, that is, producing a Hazard Log for ePAM. The multidisciplinary team included three clinical consultants, two nurses, two pharmacists, two safety engineers, two researchers and two systems engineers (representing the HIT supplier). All the clinicians have clinical lead roles in the overall HIT deployment project. The workshops were used to satisfy three fundamental requirements of the safety standard SCCI 0160, which is mandated by NHS England: (1) defining the scope of the system, including the clinical settings, (2) identifying hazards to patients and (3) estimating and evaluating risks before and after the deployment of risk control measures. The analysis is based on a critical reflection about our experiences with the use of SMART for proactive HIT hazard identification and risk analysis. Critical reflection is a frequently used technique to learn from experience and to improve practice.33,34
Using SMART for the safety analysis of ePAM
The results are grouped based on the application of SMART to define the clinical scope and system and perform hazard identification and risk analysis.
Scope definition
The scope of the analysis is Electronic Prescribing, including allergies management, in its clinical context. As such, the first challenge was to describe the relationship between prescribing, as a clinical activity, and Electronic Prescribing, as a digital information system. This was seen as essential in order to highlight that hazards caused by clinical factors were outside the scope of the analysis, for example, hazards due to an incorrect clinical decision. That is, electronic prescribing is one of many interrelated human, social and technological systems that are used to support prescribing. Figure 5 shows the main modules covered by medication management, including prescribing and allergies management. These modules define the clinical scope within which the HIT functionality is used.
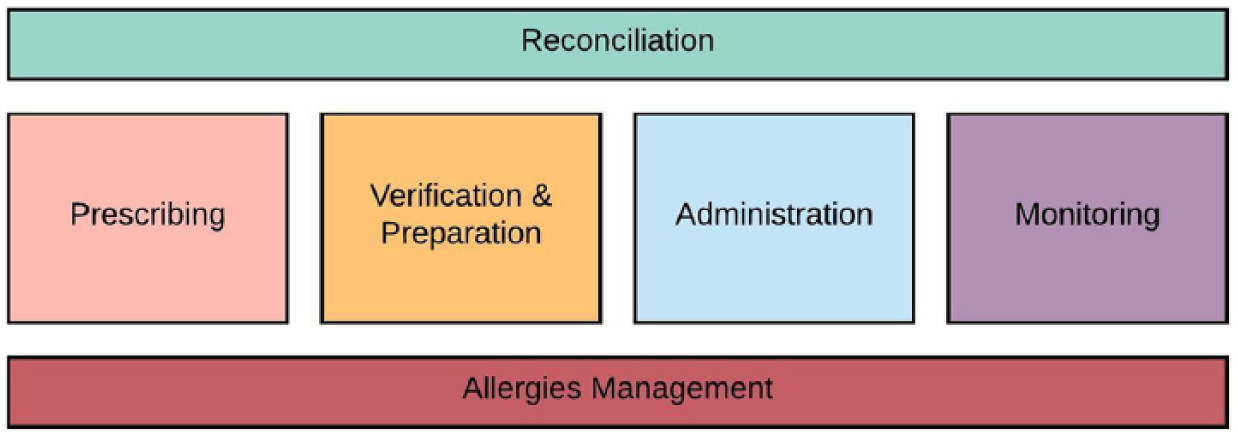
Medication management scope.
Prior to this study, models of the clinical processes existed, created through an exercise of mapping clinical practices and HIT functionality. However, for the purpose of Hazard Identification, this was seen as too detailed and IT-centric, for example, ‘click to approve’ or ‘right click for more options’. Further, many of these process models were generated based on predefined templates provided by the supplier. As a result, in order to improve the validity of the processes and emphasise the clinical focus of the Hazard Identification, the multidisciplinary team re-created the clinical process models to describe the flow of activities and decisions as perceived and performed by the users, that is, healthcare staff. Figure 6 shows the transition from (Figure 6(a)) a detailed IT-centric process model to (Figure 6(b)) a refined model created manually by the clinical team and (Figure 6(c)) a further refinement of the model by the multidisciplinary team in SMART that formed the basis for the Hazard Identification.
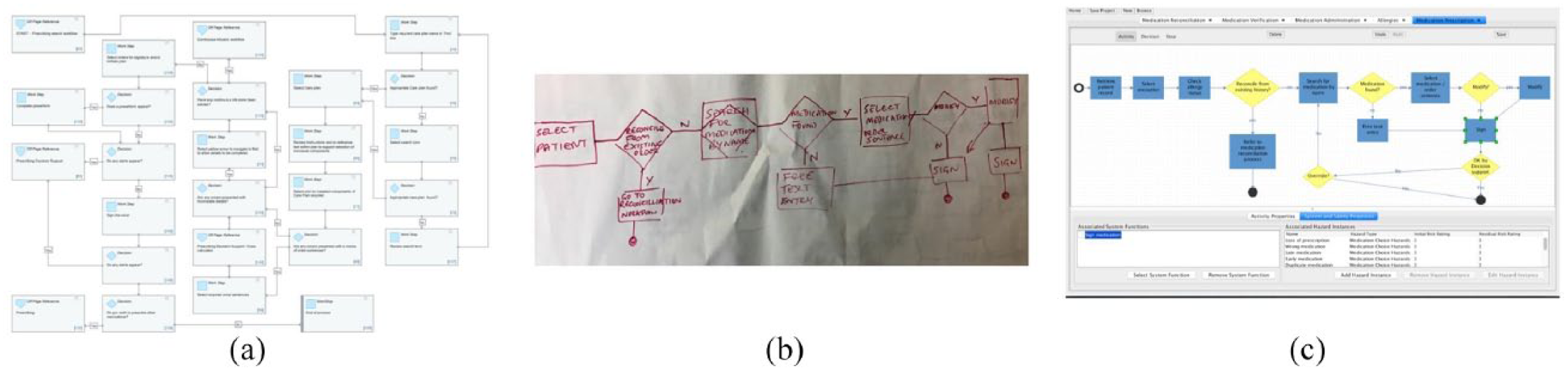
Clinical process models: (a) Initial process model, (b) Refined process model by clinical team and (c) Final model used for hazard identification.
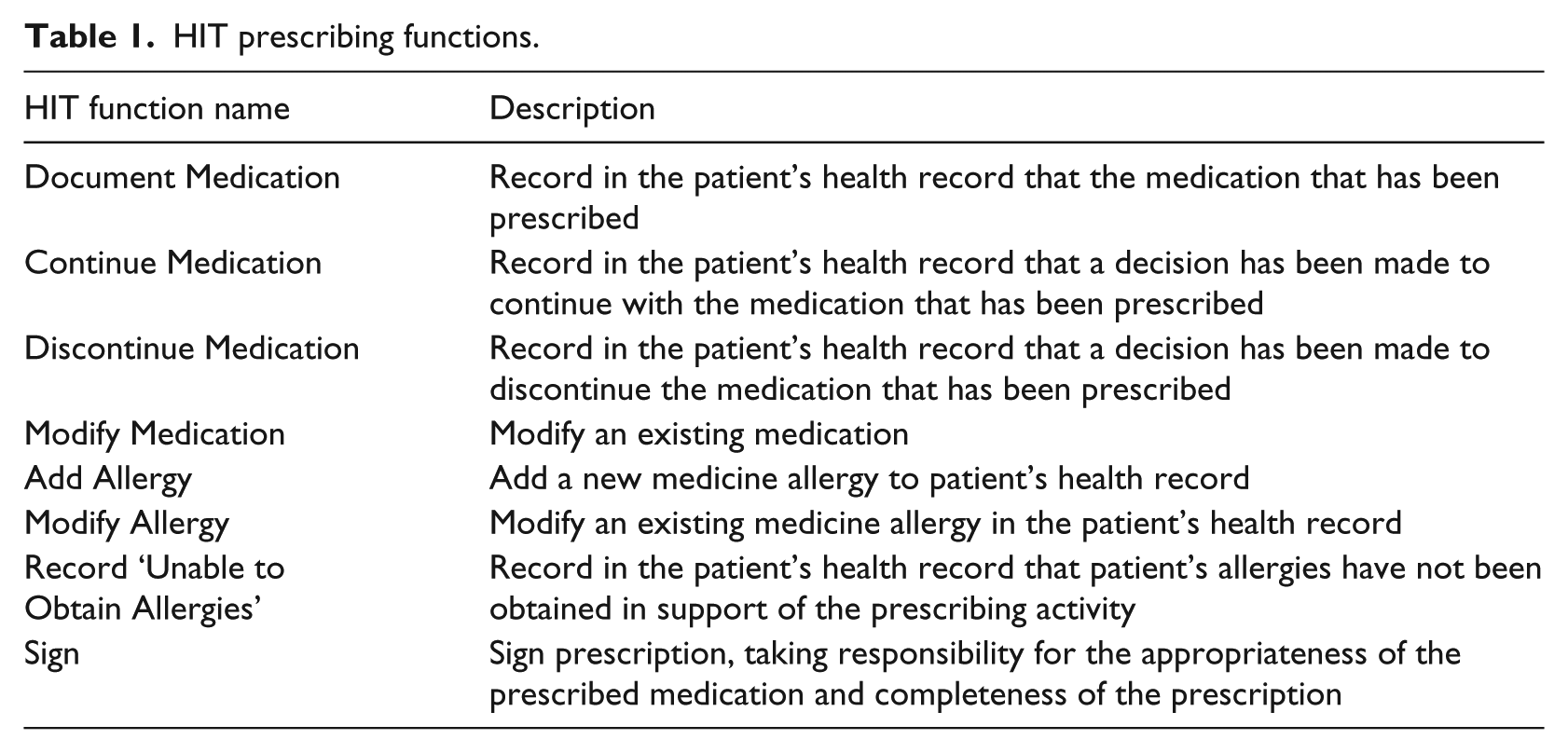
Concerning the care settings in which ePAM is used, the following were specified: (1) Inpatient, (2) Outpatient, (3) Pharmacy and (4) Community. Each of the process steps in the care models had to be associated with a specific care setting. Finally, 18 different HIT functions were specified for ePAM. Importantly, these functions were specified from the user perspective (i.e. making it clear how they serve a clinical purpose). A subset of these functions is listed in Table 1.
HIT prescribing functions.
At the end of this phase, each HIT function was explicitly traced to specific clinical activities and decisions and the settings within which it is used. This provided a basis for identifying the main
Hazard identification
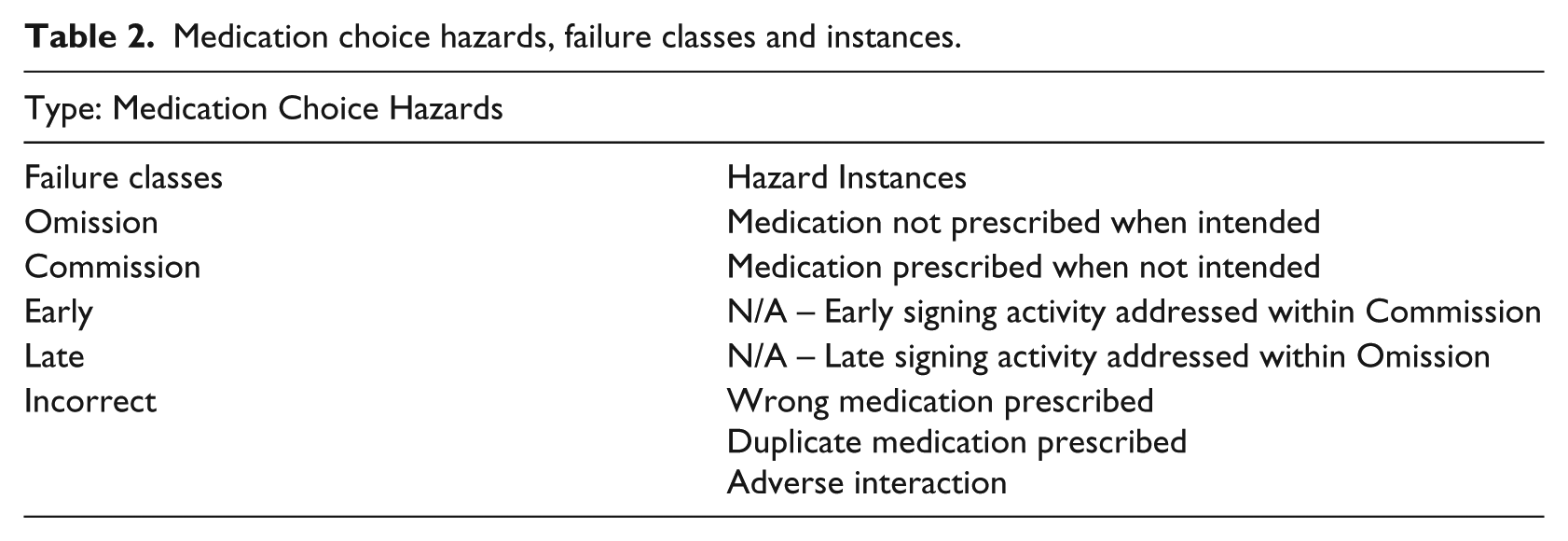
The starting point for Hazard Identification was to agree on an overall hazard classification that is clinically meaningful. We classified the hazards associated with prescribing through ePAM into five types:
Medication
Medication
Medication
Medication
Patient
These types are based on the five rights in medication safety. 35 Allergies management is a critical issue in the medication process. A hazard such as ‘prescribing a medication to which a patient is allergic’ can have severe consequences. 36 The team had two options, either to consider this hazard under the ‘Medication Choice Hazards’, that is, wrong medication, or create a new type, that is, ‘Allergies Hazards’. The team adopted the latter option in order to highlight the significance of this type of hazard, which is consistent with hospital policy, and the National Institute for Health and Care Excellence (NICE) and Medicines and Healthcare products Regulatory Agency (MHRA) guidance concerning allergies.37,38
Next, each type was refined further based on the failure classes defined in the Software Hazard Analysis and Resolution in Design (SHARD) method,39,40 which is a variant of the process industries’ Hazard and Operability Study (HAZOP) technique.
41
These failure classes are as follows:
Medication choice hazards, failure classes and instances.
In order to declare a Hazard Instance for ePAM, the context had to be clearly defined in terms of the clinical settings and steps. This was performed based on the clinical processes described in ‘Scope definition’. For example, in the prescribing process model (Figure 3), a number of Hazard Instances were declared against the activity of digitally signing a prescription. This clinical activity results in issuing an electronic prescription. An electronically signed prescription is on the boundary between multiple systems and authorities, that is, typically prescription by doctors, verification by pharmacists and preparation by nurses.
The Hazard Identification of ePAM is consistent with both the medication error classification based on the medication process (prescribing, transcribing, dispensing, administering and monitoring)
42
and that based on the type and modality, which are often referred to as ‘five rights’.
43
In this particular case, type and modality helped provide a detailed characterisation of the hazard, for example, adverse interaction. The classification based on the medication process helped describe the context of the hazard, for example,
Risk analysis
At this stage, the causes and effects of the Hazard Instances were identified, considering human, organisational and technological factors, but excluding clinical factors. Importantly, using SMART, this analysis was performed given the specific clinical context associated with the Hazard Instance, that is, the already defined activity/decision in a specific care process within a specific care setting and using a specific HIT function. This helped ensure that the causes and effects were relevant to the Hazard Instance. This also helped ensure that the risk estimation and evaluation, including any necessary controls, were performed given the relevant contextual factors.
The risk of each Hazard Instance was then estimated, based on
Each risk was then evaluated against predefined acceptability criteria, for example, as defined in the risk matrix provided by NHS Digital. Next, options were identified and analysed for controlling the risks that were deemed unacceptable, for example, through training and supervision. Figure 7 shows an example Hazard Instance form in SMART, covering Causes, Effects and Controls and including traceability to the Hazard Type, HIT function and Care Process Step.
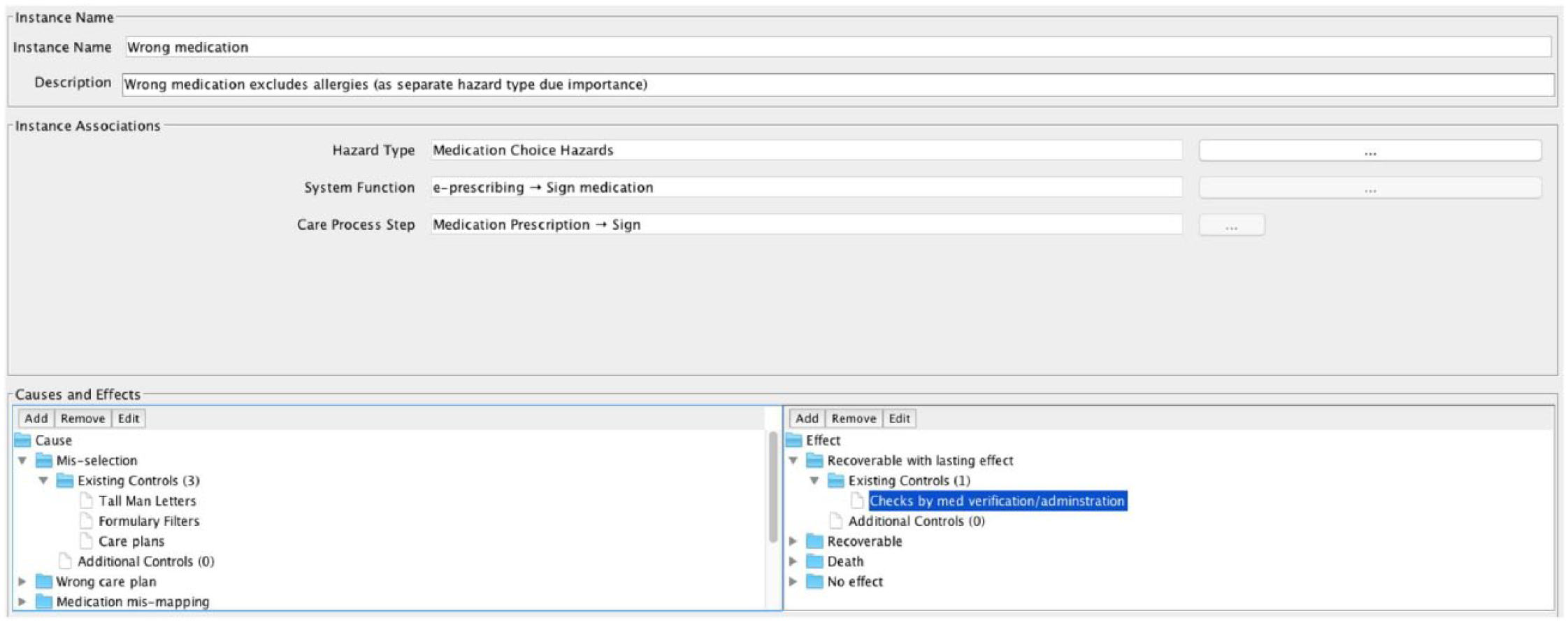
Example Hazard Instance Causes, Effects and Controls.
Discussion
In this research, we provided four criteria for refining the notion of hazards, combined with SMART as a modelling methodology and toolset. The four criteria helped ensure that the hazards identified are clinically meaningful, particularly in how they relate to patient care and clinical practice. SMART builds on these principles by creating a platform that brings together clinical and technology models as a prerequisite and a structured basis for Hazard Identification and Risk Analysis. In this section, we discuss the main findings of the pilot and the overall strengths and weaknesses of SMART.
Analysis of ePAM using SMART
We report on the key findings of the ePAM Hazard Identification and Risk Analysis that we performed using SMART. The findings are described against the four criteria for hazard conceptualisation that are discussed in ‘Conceptualising hazards for HIT’.
Clarity of the impact on patient care
Deciding on how and when the technology meets clinical practice was the most significant factor in identifying clinically meaningful hazards with potential impact on patient care. In our analysis, the explicit models of clinical processes and the inherent traceability with the ePAM functions helped ensure that all associated Hazard Instances were identified and analysed in their clinical context. SMART does not allow a Hazard Instance to be declared without specifying the clinical process, step and setting within which it might occur. To achieve this, the existing medication processes had to be simplified in order to abstract detailed technology-oriented functions and focus on the Clinical Steps and context. These functions are important for system implementation, but can often be a distraction when performing a clinically meaningful Hazard Identification.
Hazards on the boundaries of clinical systems
ePAM covered two clinical systems: prescribing and allergies management. Hazards were identified on the outputs of these systems, for example, ‘wrong medication dose’ or ‘omission of an allergy in patient records’. However, distinguishing between technological factors and clinical factors proved a challenge. For example, when estimating the risk of a ‘wrong medication’, the clinicians estimated the totality of the risk and not just the risk due to non-clinical factors, that is, which is the scope of the analysis. This issue highlights the difficulty of distinguishing between clinical and non-clinical factors in complex healthcare services, especially when HIT functions are intertwined with clinical processes. The ideal situation, though not realistic in this case, was to perform Hazard Identification and Risk Analysis on medication management as a clinical service and consider HIT as one of many factors. However, unfortunately, similar to the majority of HIT deployments, the sphere of control of the multidisciplinary team was the technology and its clinical context rather than the wider clinical practice. To some extent, the safety analysis had to be performed bottom up.
Sufficient space for detection and mitigation
The hazards were defined in such a way that controls were specified in order to reduce the risk to acceptable levels. Most of these controls were already in place, for example, training or supervision in clinical processes and tallman lettering for drug names. The results of the analysis did not generate any significant new controls, which might raise the questions as to whether the safety analysis was necessary. On reflection, the analysis was essential for two main reasons (excluding the need for compliance with SCCI 160). First, the analysis clarified how HIT can compromise patient safety and highlighted the importance of the existing controls, that is, the safety significance of certain practices and design features such as redundancy and cross-checking. Second, the analysis highlighted the need to monitor the effectiveness of the controls and the importance of revising the risk estimates based on real usage data. For example, making it hard for prescribers to use free text rather than use predefined drop-down lists is an existing control. It is now clear why it is important to regularly evaluate and monitor the extent to which prescribers are using the predefined list rather than free text.
Distinction between hazards and other major failures
Emphasising that hazards are specific events with specific characteristics was a challenge. Initially, the multidisciplinary team tended to label every significant event as a hazard. Their rationale was to ensure that the event was controlled. By calling an event a hazard, it was felt that the hazard label elevated the significance of the event. However, this could result in unnecessarily large Hazard Logs. In the ePAM Hazard Identification, this issue was partly resolved by agreeing on Hazard Types prior to declaring Hazard Instances. This helped show that many failures that would otherwise be labelled as hazards were still specified and controlled as causes or effects. As discussed in ‘Introduction’, the source of this confusion is the generic definition of hazards as sources of patient harm.
Finally, the above observations, particularly the importance of the explicit traceability among the clinical setting, HIT functionality and hazards, potentially address the weaknesses reported by Habli et al. 21 in their review of the Hazard Logs for 20 different HIT systems. Issues reported included confusing clinical hazards and HIT failures, with many hazards lacking a clear clinical impact and context. These issues made it difficult to assess the risk associated with the hazards and judge the suitability of the risk controls.
Overall strengths and weaknesses of SMART
Although the scope of the pilot study was limited to a specific setting and technology, it provided insights and highlighted practical challenges that will inform the future development and evaluation of the SMART.
Despite the additional clarity and automated support provided through SMART, hazard identification remains a complex task. This stems from the scale of healthcare, which is inherently a complex and adaptive socio-technical system, and the uncertainty about actual practice, that is, work-as-imagined versus work-as-done. 48 For example, estimating the likelihood and severity of the potential harms concerning a late prescription hazard in an Intensive Care Unit is extremely hard to perform. This is due to the sheer number of variables that have to be estimated, prior to the system deployment, concerning issues such as the medication type, the profile of the patients, the complexity of the clinical conditions and the state of the clinical setting. As such, proactive Hazard Identification and Risk Analysis is useful as long as it accompanied with a through-life safety management process that continuously updates and revises the clinical and technology models, and the associated Hazard Log, based on real-time usage data. That is, workarounds are common in healthcare. They often stem from the realities of complex clinical practice, which are hard to completely model prior to deployment. The continuous evaluation and updating of the clinical process models and their associated safety evidence are essential for maintaining the validity of the overall safety cases. One approach to achieving this, and reducing the gap between work-as-imagined and work-as-done, is via the notion of dynamic safety cases 49 that supplement proactive safety analysis with reactive analysis of data collected from actual practices (e.g. via Bayesian Network). 50
Further, the use of standard risk matrices for HIT, for example, five likelihood ratings versus five severity ratings, seems to be too coarse to cater for the inherent risks that are due to the clinical conditions or the complexity of clinical decision making. That is, a difficulty was the fact that the likelihood and severity ratings seem to be ‘overall’ as opposed to risk profiles that cater for combinations that cover high likelihoods of non-severe events and low likelihoods of severe events. As such, the extent of the harms caused by the technology compared to those resulting from the clinical condition or disease is hard to determine. This highlights the need to explore alternative means for risk estimation for HIT, building on measures such as Quality-Adjusted Life-Years (QALYs). 51 This also emphasises the importance of monitoring and collecting usage data in order to continuously revise the risk estimates and the underlying models of the clinical processes and settings.
A related important matter is risk proportionality. SMART provides clinicians and engineers with traceability data needed for analysing how the level of rigour and detail in the safety evidence is commensurate with the criticality of the clinical setting and HIT functionality. What is proportionate and therefore acceptable is a debatable and an ethically sensitive matter on which standards and legal systems have differed, that is, similar to the discussion regarding the ‘As Low As Reasonably Practicable’ (ALARP) principle. 52 Most standards provide templates for risk matrices that distinguish between acceptable, tolerable and intolerable levels of risks.14,16,17 Under exceptional circumstances, these standards allow engineers and clinicians to appeal to risk-benefit analysis to show that the clinical benefits outweigh the technological risks.
Our study provides further evidence concerning the importance of treating HIT safety assurance as a socio-technical process, involving both clinical and engineering stakeholders.13,53 This was exemplified in the difficulty of modelling the HIT functions given the variable nature of clinical settings; a significant issue that has been highlighted in the patient safety literature.11,54 The current literature on Resilience Engineering 55 and Safety 2.0 56 emphasise the need to redefine the notion of variability. This is in order to help distinguish between, on the one hand, unsafe violations and, on the other hand, desirable performance adjustments that are necessary to ensure the ability of the system to maintain safety, given changing demands and disturbances. The Systems Engineering Initiative for Patient Safety (SEIPS 2.0) 54 also now more explicitly considers variability through the concepts of configuration and adaptation.
Finally, there are cultural challenges with risk analysis for HIT. On the one hand, many organisations still adopt a strategy of ‘
Conclusion
Current definitions of hazards are high level and generic. As such, they are hard to interpret. This is particularly the case for large HIT systems used in complex socio-technical settings. Although hazard-directed safety processes are prominent in other safety-critical industries, the notion of hazards has to be refined and evaluated further in order to fit the complexity and scale of healthcare services. Publicly available exemplar Hazard Logs and safety cases are needed in order to inform the debate concerning appropriate safety analysis approaches to HIT.
Two areas of further work are important: (1) further development of SMART in order to incorporate established socio-technical models such as SEIPS 2.0 54 and (2) incorporating means for updating the clinical models and safety evidence dynamically based on real-time data. The combination of these two areas would help ensure that the safety analysis reflects the complex and adaptive socio-technical realities of clinical practice and reduces the gap between work-as-imagined and work-as-done.
Footnotes
Acknowledgements
We are grateful to colleagues who supported SMART and this study: Alistair Morris, Kay Pagan, Paul Southern, Beve Smith, Jackie Murphy, Hannah Mccann, Wale Lawal, Chris McLernon and Damon Horn.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported, in part, through a grant by the UK Royal Academy of Engineering (ISS1516\8\8) and a PhD Fellowship by the Yorkshire and Humber Patient Safety Translational Research Centre.
