Abstract
Background:
Mitochondrial abnormalities exist in lower-extremity peripheral artery disease (PAD), yet the association of the ankle–brachial index (ABI) with mitochondrial respiration in gastrocnemius muscle is unknown. The association of gastrocnemius mitochondrial respiration with 6-minute walk distance in PAD is unknown. This objective of this study was to describe associations of the ABI with mitochondrial respiratory function in gastrocnemius muscle biopsies and associations of gastrocnemius mitochondrial respirometry with 6-minute walk distance in people with and without PAD.
Methods:
People with (ABI ⩽ 0.90) and without (ABI 1.00–1.40) PAD were enrolled. ABI and 6-minute walk distance were measured. Mitochondrial function of permeabilized myofibers from gastrocnemius biopsies was measured with high-resolution respirometry.
Results:
A total of 30 people with PAD (71.7 years, mean ABI: 0.64) and 68 without PAD (71.8 years, ABI: 1.17) participated. In non-PAD participants, higher ABI values were associated significantly with better mitochondrial respiration (Pearson correlation for maximal oxidative phosphorylation PCI+II: +0.29, p = 0.016). In PAD, the ABI correlated negatively and not significantly with mitochondrial respiration (Pearson correlation for PCI+II: –0.17, p = 0.38). In people without PAD, better mitochondrial respiration was associated with better 6-minute walk distance (Pearson correlation: +0.51, p < 0.001), but this association was not present in PAD (Pearson correlation: +0.10, p = 0.59).
Conclusions:
Major differences exist between people with and without PAD in the association of gastrocnemius mitochondrial respiration with ABI and 6-minute walk distance. Among people without PAD, ABI and walking performance were positively associated with mitochondrial respiratory function. These associations were not observed in PAD.
Keywords
Background
Mitochondria produce energy that is essential for cellular function. Oxygen is necessary for normal mitochondrial activity, but in people with lower-extremity peripheral artery disease (PAD), reduced oxygen delivery to lower-extremity skeletal muscle impairs mitochondrial activity during walking activity.1 –5 In addition to inadequate oxygen delivery to skeletal muscle mitochondria, it is unclear whether the mitochondria are directly damaged by lower-extremity ischemia in people with PAD. If lower-extremity ischemia directly damages lower-extremity muscle mitochondria, then people with more severe PAD may have greater mitochondrial damage and poorer mitochondrial activity, even in settings where oxygen supply is adequate. Measuring mitochondrial activity with respirometry in muscle biopsy specimens allows assessment of mitochondria function when oxygen supply is adequate.
To better understand the association of lower-extremity ischemia with mitochondrial activity in PAD, this study described associations of ischemia severity, measured by the ankle–brachial index (ABI), with mitochondrial respiratory function in gastrocnemius muscle biopsies from people with PAD. In people without PAD, prior work has documented significant associations of lower ABI values with significantly greater abundance of mitochondrial DNA but also with poorer mitochondrial energy production.6,7 Therefore, this study also evaluated the association of the ABI with mitochondrial activity, measured by gastrocnemius muscle biopsy, in people without PAD.
More severe lower-extremity ischemia, measured by the ABI, is associated with greater walking impairment in people with and without PAD.8 –11 However, the extent to which mitochondrial activity impairment is associated with walking impairment in PAD remains unclear. Hou et al. reported that in 15 people with PAD, mitochondrial activity in gastrocnemius muscle, measured by respirometry using three energy substrates, was not consistently associated with pain-free or maximal treadmill walking performance. 12 In 11 people without PAD, Hou et al. reported no significant associations of gastrocnemius mitochondrial activity, measured by respirometry, with treadmill walking performance. 12 In contrast, McDermott et al. reported that a greater abundance of mitochondrial DNA in gastrocnemius muscle biopsies was associated with a better 6-minute walk distance in people without PAD, but this association was not observed in people with PAD. 6 Similarly, Picca et al. reported that a greater abundance of mitochondrial complexes in gastrocnemius muscle biopsies was associated with a better 6-minute walk distance in people without PAD, but this association was not observed in people with PAD. 13 For these reasons, this study described associations of gastrocnemius mitochondrial activity, measured by respirometry, with 6-minute walk distance in people with and without PAD, respectively. Study hypotheses were that in people both with and without PAD, a better gastrocnemius mitochondrial function would be associated with a better 6-minute walk distance.
Mitochondrial dysfunction in people with PAD may be due to poor mitochondria quality or inadequate mitochondria abundance. Prior work documented that lower ABI values were associated with a significantly greater abundance of gastrocnemius mitochondrial DNA in people both with and without PAD. 6 Therefore, in exploratory analyses, associations of mitochondrial respirometry function with mitochondrial protein abundance were evaluated in people with and without PAD. Associations of the abundance of these proteins with the ABI, 6-minute walk distance, and physical activity were evaluated in people with and without PAD. This study described associations of gastrocnemius mitochondrial activity, measured by respirometry, with 6-minute walk distance in people with and without PAD, respectively. Study hypotheses were that in people both with and without PAD, a better gastrocnemius mitochondrial function would be associated with a better 6-minute walk distance.
Methods
Participants with and without PAD were enrolled from the Chicago, Illinois area. Northwestern University’s Institutional Review Board approved the protocol. All participants provided written informed consent.
Participant identification
Participants with and without PAD were identified using lists of patients from Northwestern Medicine’s Chicago medical center, with advertisements placed on Chicago Transit Authority buses and trains, and by mailed recruitment postcards using a purchased mailing list. Some participants were identified from individuals who completed studies in the past with a study investigator (MMM). Enrollment occurred between July 23, 2018 and August 26, 2021.
Inclusion criteria
PAD was defined as an ABI ⩽ 0.90 in either leg. 14 Absence of PAD was defined as an ABI of 1.00–1.40. 9 Potential participants with an ABI of 0.90–0.99 were excluded because they may have mild lower-extremity atherosclerosis.7,8 To attain similar ages between people with versus without PAD, only people aged 60 and older were included among those without PAD.
Exclusion criteria
Individuals taking part in a randomized clinical trial or who had completed a randomized clinical trial in the past 3 months were not eligible. Among potential participants with PAD, those with chronic limb-threatening ischemia (CLTI) and those whose walking was limited by a condition other than PAD were excluded. For all potential participants, those with recent or planned major surgery, those with a Mini-Mental Status Examination score < 23, 15 those with major medical illness (such as recent cancer treatment), and those unwilling to have a gastrocnemius biopsy were excluded. Potential participants with diabetes were initially excluded; however, owing to challenges with recruitment, diabetes mellitus was not an exclusion criterion from September 20, 2019.
ABI measurement
A hand-held Doppler probe (Pocket Dop II; Nicolet Biomedical Inc., Golden, CO, USA) was used to measure systolic pressures twice in the right and left brachial, dorsalis pedis, and posterior tibial arteries while the participant was supine. The ABI was measured by staff, following the study protocol. Staff were certified by co-investigator MMM prior to beginning data collection and approximately every 6 months. The ABI was calculated by dividing the mean of the dorsalis pedis and posterior tibial pressures in each leg by the mean of the four brachial pressures. 16
Six-minute walk
The 6-minute walk test was used to assess functional performance because it is a well-validated measure of walking endurance that reflects walking ability in daily life better than treadmill walking performance. 17 For example, the 6-minute walk test was found to be more closely correlated with physical activity in daily life than treadmill walking performance. 18 Participants walked up and down a 100-foot (30.48 m) hallway after receiving scripted instructions from a trained and certified research coordinator. Participants were advised that the goal of the test was to cover as much distance as possible in 6 minutes.17,18 The distance completed after 6 minutes was recorded. Distance to first report of leg symptoms during the walk and whether the participant reported leg symptoms at the end of the walk were recorded. The intra-class correlation coefficient for the test-retest reliability of the 6-minute walk test among 155 participants who performed the 6-minute walk test twice, 1–2 weeks apart, was 0.90 (p < 0.001). 18
Physical activity
Physical activity was measured over 7 days with the ActiGraph (Pensecola, FL, USA) accelerometer. Low, medium, and high-intensity activity were combined into a single ‘activity count’ outcome as previously described.19,20
Gastrocnemius biopsy
An open muscle biopsy was performed in the medial head of the gastrocnemius muscle of the leg with the lower ABI.6,13,21 If the left and right legs had identical ABI values, the leg for biopsy was selected randomly. All muscle biopsies were performed by two co-investigators (RS and KJH). Anesthesia was achieved with subcutaneous lidocaine. Subcutaneous tissue was dissected until muscle was identified. After muscle tissue was removed, 10–20 mg was transported in cold biopsy media for immediate respirometry testing. Remaining muscle was immediately prepared for freezing at −80°C.
Mitochondrial respiratory function
High-resolution respirometry was performed using an Oroboros O2k (Oroboros Instruments, Innsbruck, Austria) per established protocols.22,23 Respirometry was performed on muscle immediately after the biopsy was obtained. Muscle fiber bundles were separated in ice-cold preservation media (BIOPS) under a dissecting microscope to obtain four replicates per participant (approximately 2–3 mg each), which were permeabilized with saponin for 20 minutes followed by a 10-minute wash in mitochondrial respiration media (MiR05). All data were collected at 37°C in hyperoxygenated (200–450 µM O2) conditions in MiR05 to avoid limitations to oxygen diffusion. Two substrate-uncoupler-inhibitor titration (SUIT) respiration protocols (carbohydrate and fatty acid) assessed maximal phosphorylation (OXPHOS) and electron transport chain (ETC) capacity of complex-I and complex-II-mediated respiration. Malate, pyruvate (carbohydrate) or octanoylcarnitine (fatty acid), adenosine diphosphate (ADP), glutamate, cytochrome c, and succinate were sequentially added, followed by titrations of carbonyl cyanide m-chlorophenyl hydrazine (CCCP), rotenone, and antimycin A. The state of respiration in the presence of malate, pyruvate, glutamate, and ADP was defined as carbohydrate complex-I-mediated maximal OXPHOS capacity (PCI). Respiration after subsequent succinate addition was defined as carbohydrate complex-I+II-mediated maximal OXPHOS capacity (PCI+II). The state of respiration after adding CCCP (uncoupler) was considered maximal ETC capacity (ECI+II). Fatty acid-supported respiration in the presence of malate, octanoylcarnitine, and ADP was measured using complex-I and electron-transferring flavoprotein complex-mediated maximal OXPHOS (PCI/FA). Results from replicates that increased by more than 15% after adding cytochrome c were considered over-permeabilized and not included in analyses.
Mitochondrial protein measures
Protein expression of mitochondrial ETC complexes I, II, III, IV, and V (adenosine triphosphate [ATP] synthase) subunits; voltage-dependent anion channels (VDAC); and peroxisome-proliferator-activated receptor-γ coactivator (PGC-1α) were measured by Western blot as previously described (Supplemental Figure),24,25 using the following primary antibodies: Total OXPHOS Human WB Antibody Cocktail (1:2000; #ab110411, Abcam, Waltham, MA, USA) for the mitochondrial electron transport chain complex subunits; anti-VDAC (#4661s, Cell Signaling, Danvers, MA, USA); and anti-PGC-1α (1:1000; #516557, Millipore/Sigma, Burlington, MA, USA).
Additional measures
Height and weight were measured and body mass index (BMI) was calculated as weight (kg)/height (m2). Comorbidities were ascertained according to participant report, based on whether a physician had previously diagnosed them with each comorbidity. 25 Race was identified with self-report. Participants were asked for a list of their current medications, and medications were adjudicated by a study investigator (MMM) to categories including statins, antiplatelet drugs, angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor blocker (ARB), cilostazol, or pentoxifylline medications. Participants were asked to report the number of blocks walked and stair flights climbed in the past 7 days, the number of days they went walking for exercise in the past 2 weeks, and the number of walking exercise minutes per exercise session.
Statistical analyses
Characteristics of participants with and without PAD were summarized as means and SD for continuous variables and as frequencies and percentages for categorical variables. Two sample t-tests were used to compare continuous characteristics. Chi-squared or Fisher’s exact tests were used to compare the categorical characteristics of participants with versus without PAD. Pearson correlation coefficients were used to evaluate associations of the ABI, 6-minute walk distance, and physical activity, respectively, with mitochondrial respiratory function and with abundance of the mitochondrial proteins. Pearson correlation coefficients were used to evaluate associations of physical activity with ABI, 6-minute walk distance, and mitochondrial function in people with and without PAD. Analyses were performed separately among people with and without PAD. We studied whether these associations were significantly different between people with and without PAD by testing the interaction between PAD status and ABI in a linear regression model with ABI as the independent variable of interest and mitochondrial respirometry as the dependent variable of interest. We also tested the interaction between PAD status with mitochondrial respirometry as the independent variable of interest and 6-minute walk distance and physical activity as separate dependent variables of interest. Among people with and without PAD, mitochondrial function was compared between participants who reported walking for exercise at least 3 days/week for at least 30 minutes/day and those who did not walk for exercise at least 3 days/week for at least 30 minutes/day using two-sample t-tests. Among people without PAD, statistical testing for interaction was performed to assess whether associations of mitochondrial function with 6-minute walk distance or physical activity differed significantly between people with versus without comorbidities. Statistical tests were two-sided an the criterion for statistical significance was p < 0.05. Statistical analyses were conducted using SAS 9.4 (SAS Institute, Inc., Cary, NC, USA).
Results
Of 111 potential participants without PAD who signed informed consent, 42 met at least one exclusion criterion and 69 were enrolled (Figure 1). Of 53 potential participants with PAD who signed informed consent, 22 met at least one exclusion criterion and 31 were enrolled (Figure 1). One participant with PAD and one without PAD had insufficient gastrocnemius muscle for respirometry, leaving 68 without PAD and 30 with PAD for analyses.
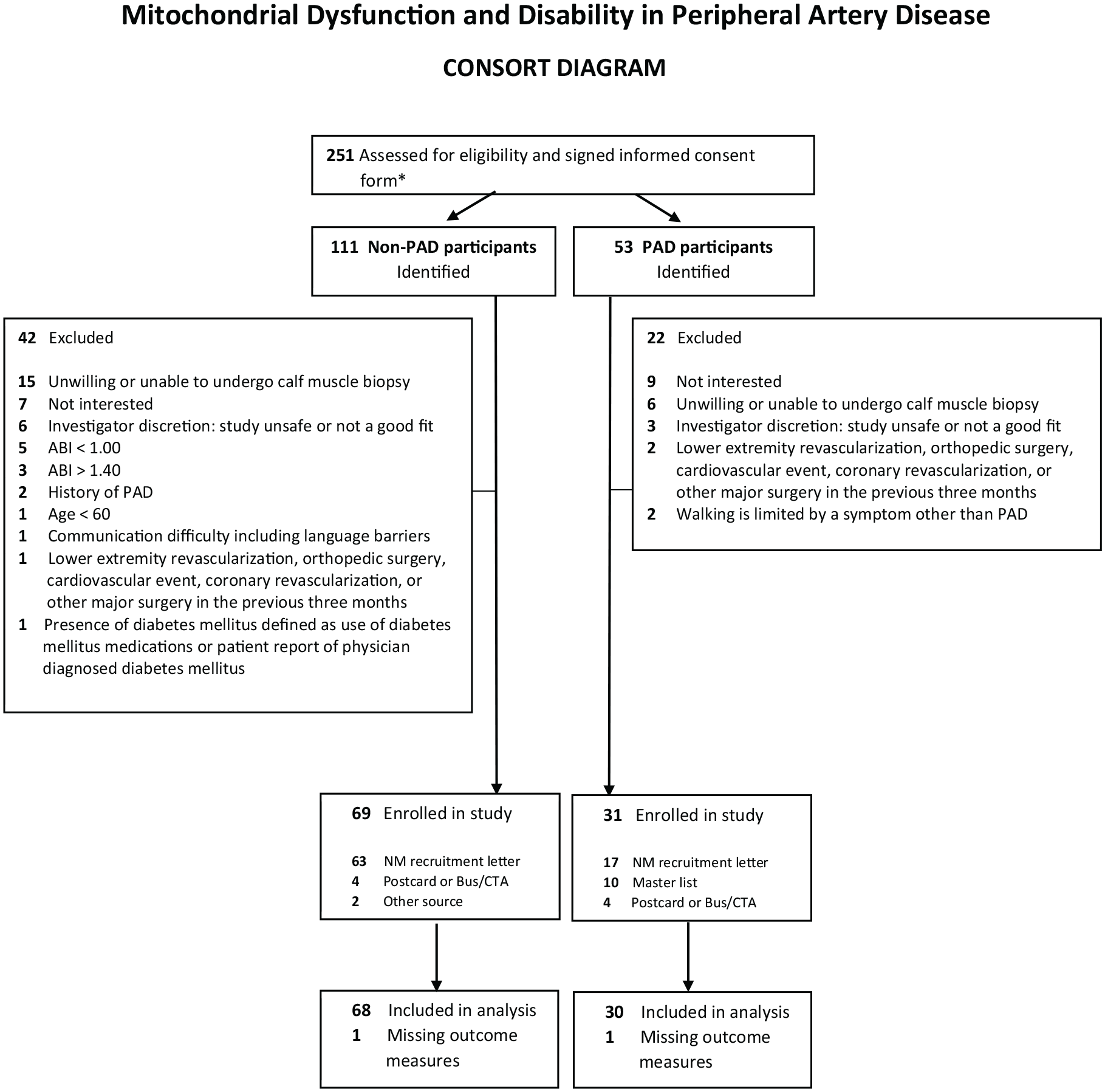
Number of excluded and included participants with and without lower-extremity peripheral artery disease (PAD).
The cohort included 38 (39%) women and 36 (37%) Black individuals. Mean ABI values were 0.64 (SD: 0.17) and 1.17 (SD: 0.09) in participants with and without PAD, respectively (Table 1). Participants with PAD included a higher proportion of men, people with history of angina, myocardial infarction, diabetes mellitus, and heart failure, and people who smoked cigarettes compared to those without PAD (Table 1). The mean 6-minute walk distance, physical activity measured by ActiGraph, and blocks walked in the past week were significantly lower in people with PAD, compared to those without PAD (Table 1). A significantly higher proportion of people with PAD were taking statin medications and ACE inhibitors or ARB medications, compared to those without PAD (Table 1). No participants were taking pentoxifylline. Correlations between ABI, 6-minute walk distance, and mitochondrial measures, respectively, with physical activity measured by ActiGraph are shown in Supplemental Table 1.
Characteristics of study participants.
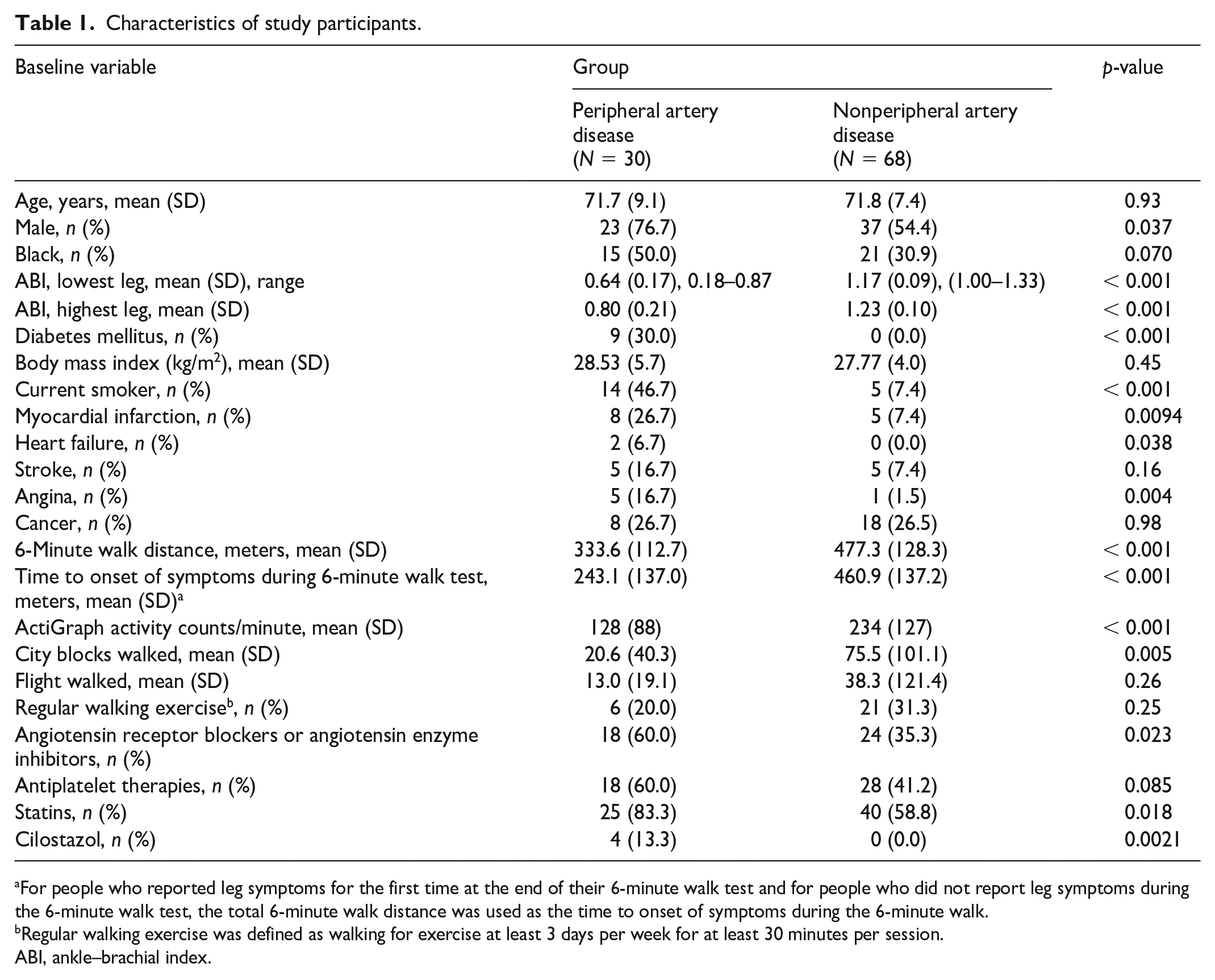
For people who reported leg symptoms for the first time at the end of their 6-minute walk test and for people who did not report leg symptoms during the 6-minute walk test, the total 6-minute walk distance was used as the time to onset of symptoms during the 6-minute walk.
Regular walking exercise was defined as walking for exercise at least 3 days per week for at least 30 minutes per session.
ABI, ankle–brachial index.
ABI and mitochondrial function
Among people without PAD, higher ABI values were associated significantly with better mitochondrial respiratory function for PCI+II (correlation +0.29, p = 0.016), ECI+II (correlation +0.29, p = 0.019), and PCI/FA (correlation +0.47, p < 0.001) (Figure 2). There were no statistically significant associations of the ABI with gastrocnemius biopsy respirometry function in people with PAD (Figure 2). A statistically significant interaction was present between the presence of PAD and the association of ABI with mitochondrial function, indicating that the correlation of ABI with mitochondrial function in people with PAD was significantly different from the correlation of ABI with mitochondrial function in people without PAD (Figure 2).
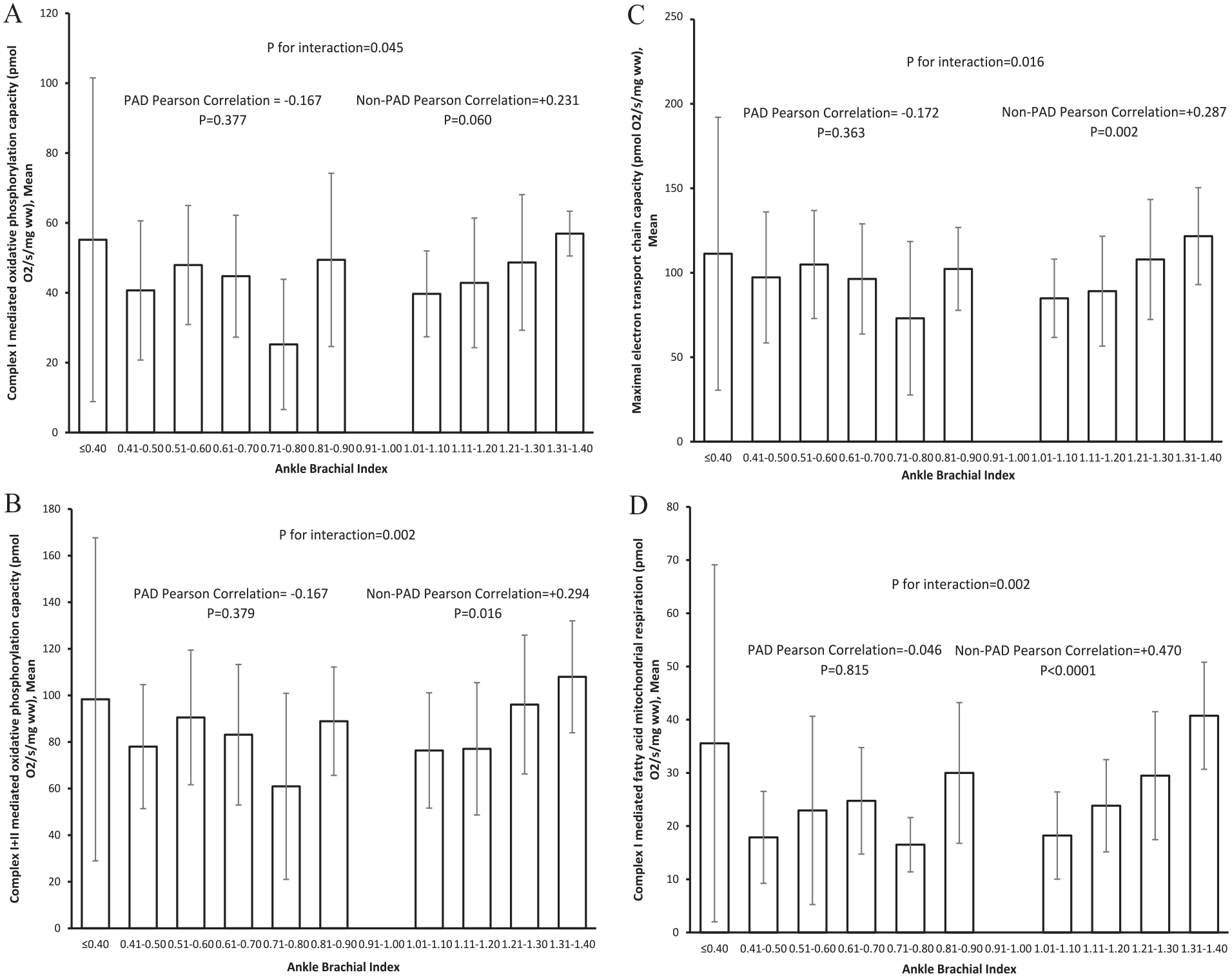
Associations of the ankle–brachial index with mitochondrial function, measured by respirometry, among people with and without peripheral artery disease for complex I-mediated oxidative phosphorylation
Mitochondrial function and 6-minute walk distance
Among people without PAD, better mitochondrial function was associated significantly with higher 6-minute walk distance, when carbohydrates were used as substrates (PCI: +0.38, p = 0.002; PCI+II: +0.51, p < 0.001; ECI+II carbohydrate substrate: +0.49, p < 0.001) (Figure 3). In contrast, among participants with PAD, there were no statistically significant associations of mitochondrial function with 6-minute walk distance (Figure 3).
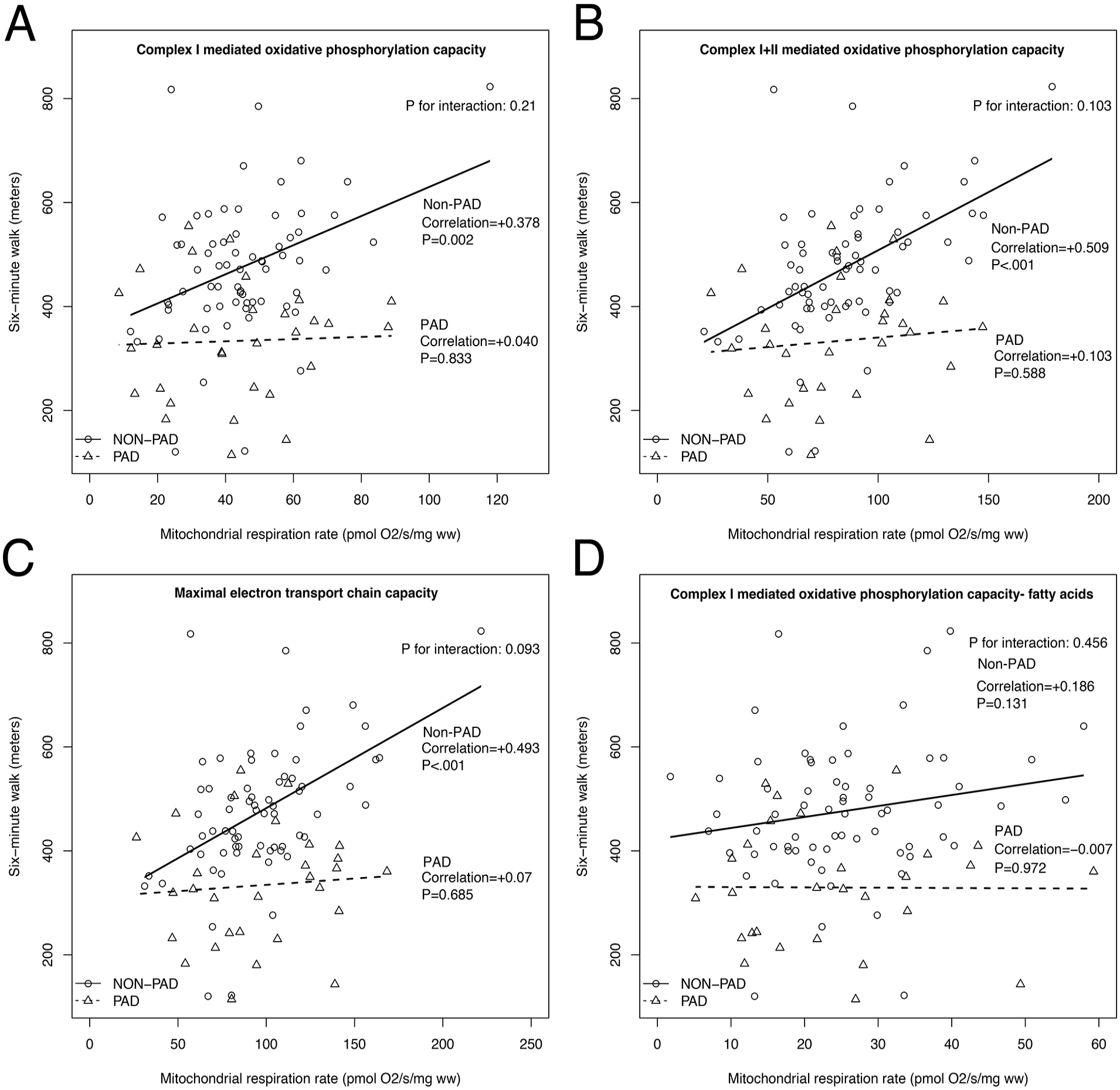
Associations of mitochondrial function with 6-minute walk distance among participants with and without peripheral artery disease for complex I-mediated oxidative phosphorylation
Mitochondrial function and accelerometer-measured physical activity
Among people without PAD, better mitochondrial respiratory function was associated with significantly higher physical activity, when carbohydrates were used as substrates (PCI+II: +0.37, p = 0.002; ECI+II carbohydrate substrate: +0.33, p = 0.007) (Figure 4). Among participants with PAD, there were no statistically significant associations of mitochondrial function with physical activity, perhaps due to inadequate statistical power (Figure 4).
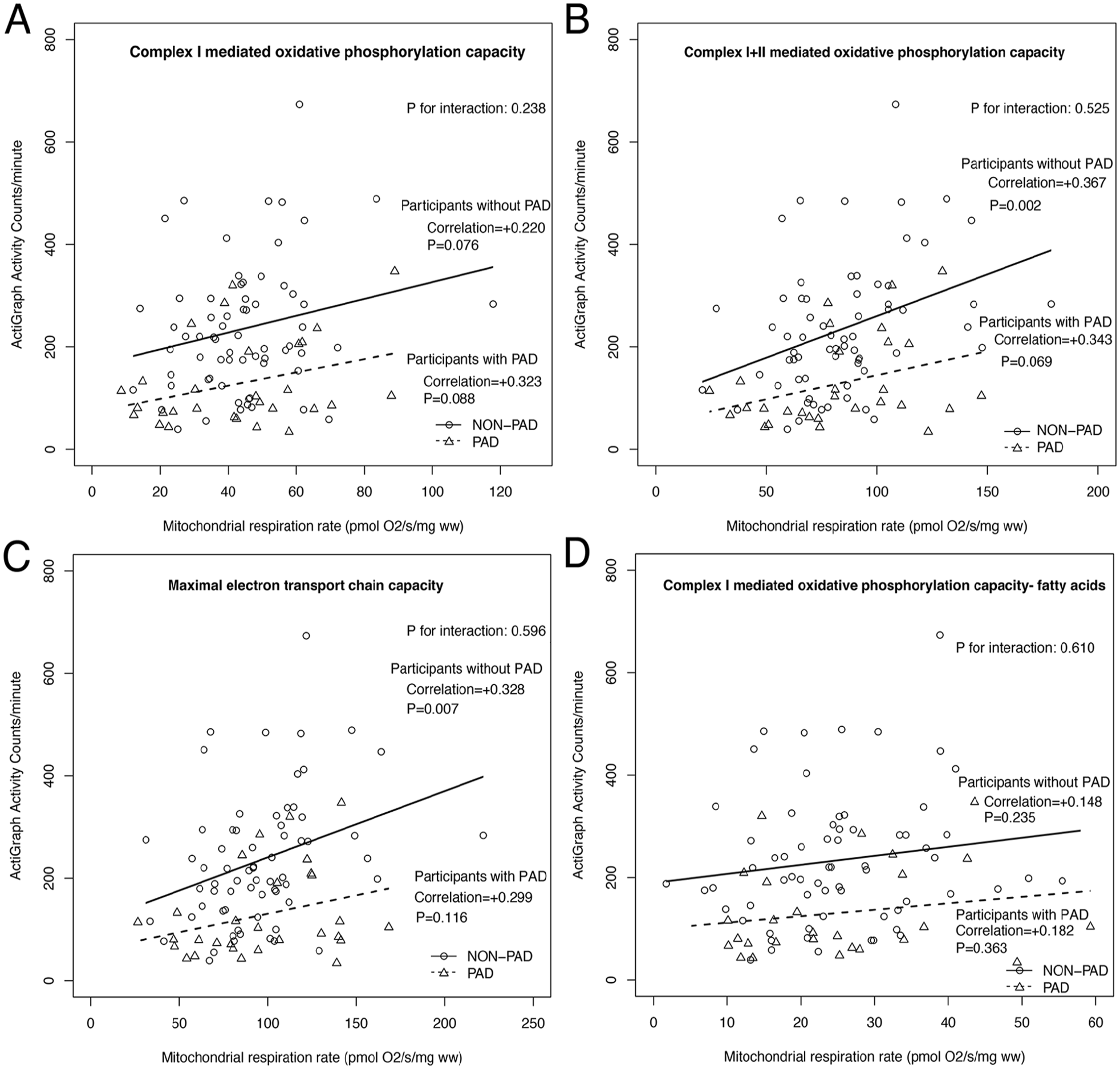
Associations of the ankle–brachial index with physical activity among people with and without peripheral artery disease for complex I-mediated oxidative phosphorylation
ABI, 6-minute walk, physical activity, and mitochondrial proteins
There were no statistically significant associations of the ABI with abundance of any mitochondrial complexes. Among people without PAD, greater abundance of each mitochondrial protein, except complex IV, was associated with significantly better 6-minute walk distance (Table 2). These statistically significant associations were not present in people with PAD (Table 2). Among people without PAD, greater abundance of each mitochondrial protein, except complex III, IV, and V, was associated with significantly greater physical activity level. Among people with PAD, greater abundance of complex I mitochondrial protein was associated with greater physical activity. Among people with PAD, other mitochondrial proteins were not associated with physical activity (Table 2).
Associations of gastrocnemius muscle abundance of mitochondrial proteins with ankle–brachial index, 6-minute walk distance, and physical activity in participants with and without peripheral artery disease (PAD).
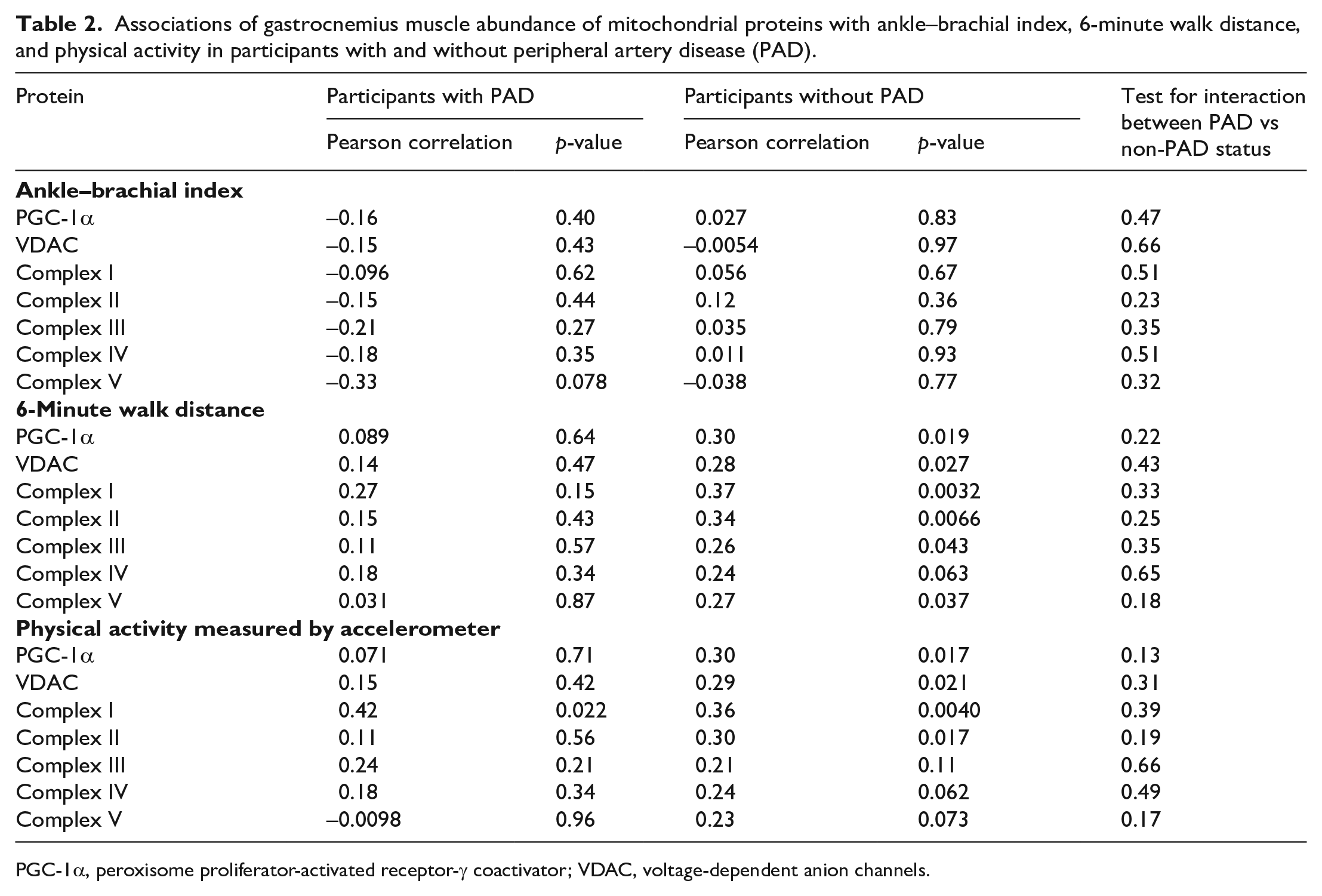
PGC-1α, peroxisome proliferator-activated receptor-γ coactivator; VDAC, voltage-dependent anion channels.
Mitochondrial proteins and mitochondrial function
Among participants without PAD, a greater abundance of PGC-1α, VDAC, and mitochondrial complexes I–IV were associated significantly with better PCI, PCI+II, and ECI+CII respiration. Complex V was associated significantly with PCI+II respiration, but not with other respirometry measures, perhaps due to lack of statistical power or due to chance (Table 3). Among participants with PAD, greater abundance of PGC-1α, VDAC, and each mitochondrial complex was associated significantly with better mitochondrial respiratory function (Table 3). There were statistically significant interactions for the presence of PAD and associations of VDAC, complex I, and complex IV with PCI/FA, indicating that the strength of the correlations for these associations was significantly stronger among participants with PAD, compared to those without PAD (Table 3).
Associations of mitochondrial proteins with mitochondrial function in gastrocnemius muscle biopsies.
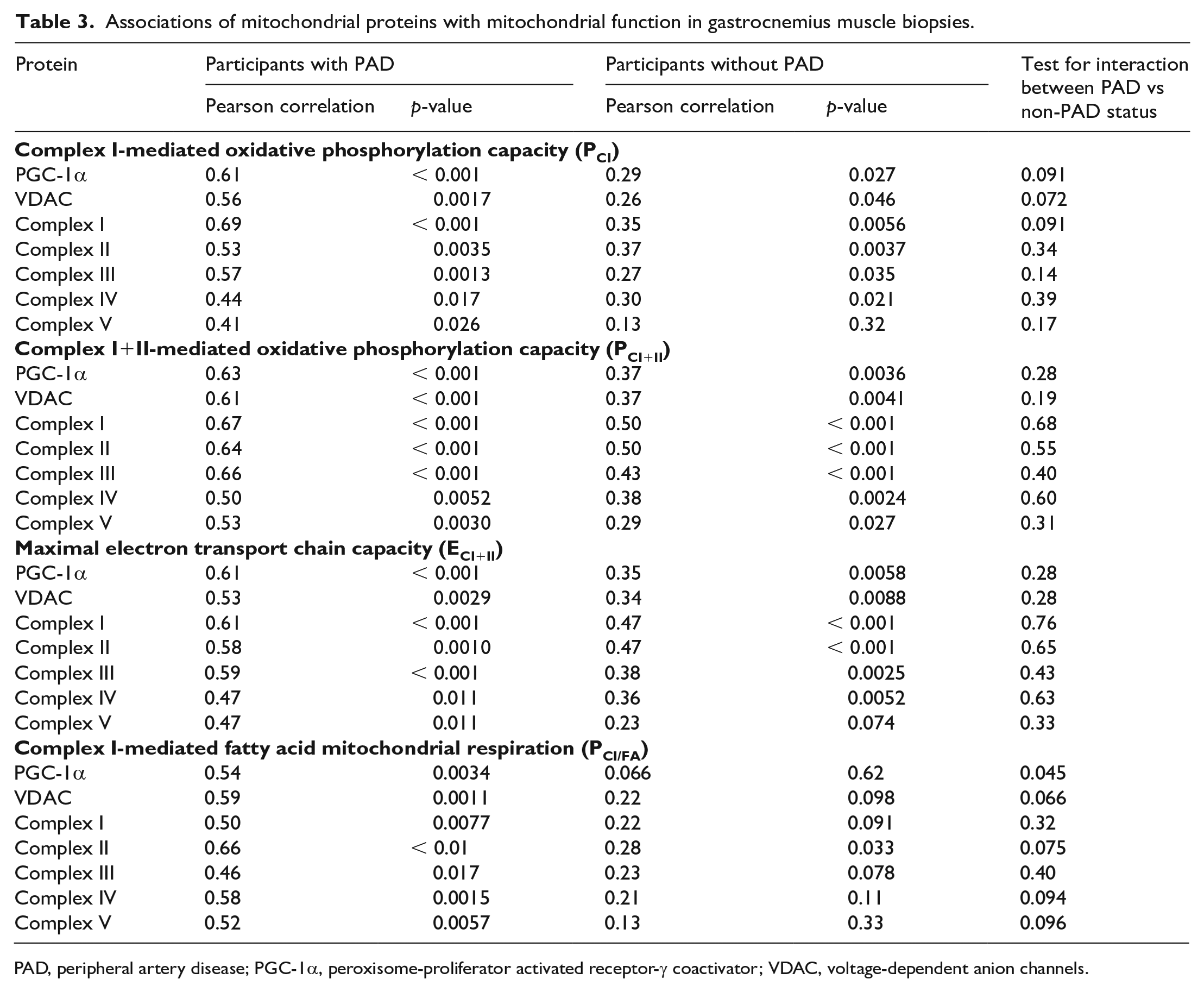
PAD, peripheral artery disease; PGC-1α, peroxisome-proliferator activated receptor-γ coactivator; VDAC, voltage-dependent anion channels.
ABI, functional performance, and physical activity
Among people with PAD, lower ABI values were associated significantly with poorer 6-minute walk distance, but this association was not observed in people without PAD (Figure 5A). Among people with PAD, lower ABI values were not associated significantly with lower physical activity levels, but among people without PAD, higher ABI values were associated with greater physical activity (Figure 5B).
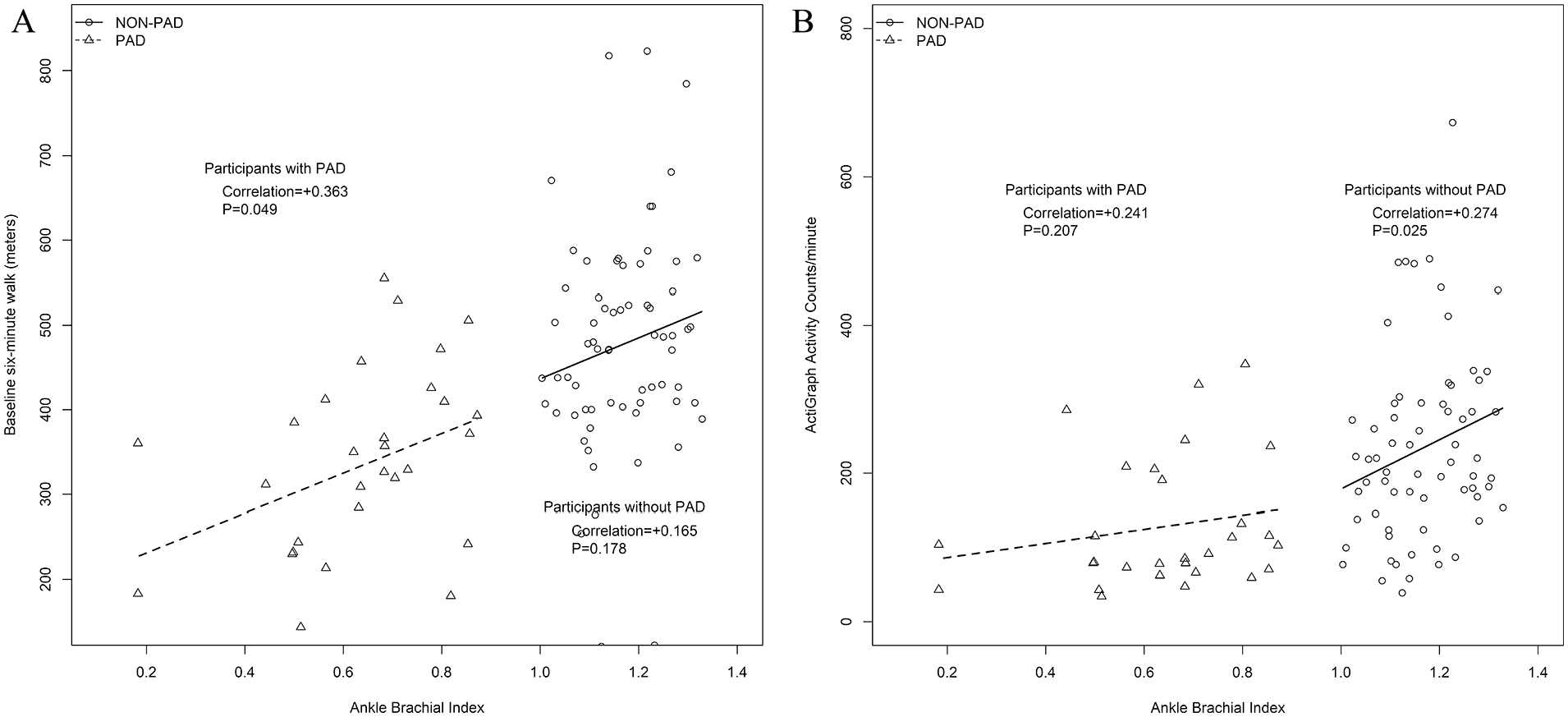
Association of the ankle–brachial index with 6-minute walk distance
Mitochondrial function according to self-reported exercise behavior
Mitochondrial function according to self-reported exercise behavior among people with and without PAD is shown in Supplementary Table 2.
Comorbidities and associations of mitochondrial function with 6- minute walk distance
Among people without PAD, there were no statistically significant interactions for the presence versus absence of comorbidities and associations of mitochondrial function with 6-minute walk distance or physical activity (Supplementary Tables 3 and 4).
Discussion
Mitochondrial structural abnormalities and increased oxidative stress have been documented in gastrocnemius muscle of people with PAD, compared to those without PAD.1 –4 However, to our knowledge, no prior studies have reported associations of a range of lower-extremity ischemia severity, measured by ABI among people with PAD who did not have CLTI, with gastrocnemius muscle mitochondrial respirometry of permeabilized muscle fibers after muscle biopsy. In this descriptive report of older people, lower ABI values were associated with significantly poorer gastrocnemius mitochondrial respiratory function in people without PAD, but not in people with PAD (Figure 2). The presence of a statistically significant interaction for PAD on the association of ABI with mitochondrial respiratory function demonstrated that the correlations of ABI with mitochondrial activity significantly differed between people with versus without PAD. These findings are consistent with prior studies demonstrating that even in people without PAD, higher ABI values were associated with better walking performance, greater gastrocnemius muscle area, less gastrocnemius fatty infiltration, lower rates of mobility loss, and better phosphocreatine recovery, measured by phosphorous magnetic resonance spectroscopy ( 31 P MRS).7 –11,26,27 Preliminary evidence suggests that lower-extremity perfusion declines with age even in people without PAD.28,29 The lack of association of ABI with mitochondrial activity among people with PAD may signify damaged mitochondria function in PAD. However, further study is needed to better understand these associations in people both with and without PAD.
Results reported here documented that better gastrocnemius muscle mitochondrial function, measured with respirometry on muscle biopsies, was associated with greater 6-minute walk distance in people without PAD, but not in people with PAD (Figure 3). Consistent with these findings, prior work documented that a greater abundance of mitochondrial DNA and a greater abundance of mitochondrial complex proteins in gastrocnemius muscle were significantly associated with a better 6-minute walk distance in people without PAD, but these associations were not observed in people with PAD.6,12 Better mitochondrial activity, measured by respirometry in muscle biopsies from the vastus lateralis muscle, was also associated with faster walking velocity in a 400-meter walk test in people without PAD.10,30 Results suggest that impaired perfusion of the lower extremities may be the over-riding factor limiting walking performance in PAD (Figure 5). It is possible that damage to gastrocnemius muscle mitochondria by repeated episodes of lower-extremity ischemia in people with PAD obliterates the association of mitochondrial activity with 6-minute walk distance that was observed in those without PAD, but further study is needed.
Prior studies using MRS reported that lower ABI values were associated with a longer time to phosphocreatine recovery, a measure of mitochondrial function, in people with PAD.31,32 However, 31 P MRS is performed in vivo, where impaired perfusion reduces oxygen delivery to the legs in people with PAD, thereby lengthening the phosphocreatine recovery time and impairing mitochondrial function. 22 In contrast to MRS, gastrocnemius muscle biopsy respirometry measures mitochondrial respiratory function under optimal conditions, when mitochondrial oxygen supply or substrate availability is not limited by reduced lower-extremity perfusion.
This study has several limitations. First, the study was cross-sectional. No causal inferences can be made. Second, these results do not apply to people with CLTI. Third, it is possible that associations of mitochondrial activity with treadmill walking performance may have differed from the associations of mitochondrial activity with 6-minute walk distance. Fourth, it is possible that some differences in findings for participants with versus without PAD may have been due to residual or unmeasured confounders. Fifth, muscle measures such as capillary density, measures of the microcirculation, and muscle phenotype were not available for this cohort. Sixth, the sample size was relatively small.
Conclusion
Major differences exist between people with and without PAD in the association of gastrocnemius mitochondrial respiratory function with ABI and walking performance. Among people without PAD, ABI and walking performance were positively associated with mitochondrial respiratory function. These associations were not observed in PAD.
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X241268893 – Supplemental material for The ankle–brachial index, gastrocnemius mitochondrial respirometry, and walking performance in people with and without peripheral artery disease
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X241268893 for The ankle–brachial index, gastrocnemius mitochondrial respirometry, and walking performance in people with and without peripheral artery disease by Mary M McDermott, Sudarshan Dayanidhi, Christiaan Leeuwenburgh, Stephanie E Wohlgemuth, Luigi Ferrucci, Charlotte A Peterson, Lu Tian, Robert Sufit, Lihui Zhao, Joshua Slysz, Tamar S Polonsky, Jack M Guralnik, Melina R Kibbe, Karen J Ho, Michael H Criqui, Dongxue Zhang, Shujun Xu and Philip Greenland in Vascular Medicine
Supplemental Material
sj-png-1-vmj-10.1177_1358863X241268893 – Supplemental material for The ankle–brachial index, gastrocnemius mitochondrial respirometry, and walking performance in people with and without peripheral artery disease
Supplemental material, sj-png-1-vmj-10.1177_1358863X241268893 for The ankle–brachial index, gastrocnemius mitochondrial respirometry, and walking performance in people with and without peripheral artery disease by Mary M McDermott, Sudarshan Dayanidhi, Christiaan Leeuwenburgh, Stephanie E Wohlgemuth, Luigi Ferrucci, Charlotte A Peterson, Lu Tian, Robert Sufit, Lihui Zhao, Joshua Slysz, Tamar S Polonsky, Jack M Guralnik, Melina R Kibbe, Karen J Ho, Michael H Criqui, Dongxue Zhang, Shujun Xu and Philip Greenland in Vascular Medicine
Footnotes
Acknowledgements
Kevin Wu of University of Florida performed some of the mitochondrial Western Blot measures.
Data availability statement
Data for analyses are available from the corresponding author upon reasonable request
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. McDermott has received research support from Helixmith, Mars, and ArtAssist. The other authors have no conflicts of interest.
Funding
This study was supported by the American Heart Association (18SFRN33900142, 18SFRN33900097, 18SFRN33900136, 18SFRN3390010). The funding agency played no role in the study design or the decision to submit the manuscript for publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
