Abstract
Disturbed wound healing (DWH) following elective foot and ankle surgery is associated with a number of known risk factors. The purpose of this study was to determine if peripheral artery disease (PAD) is a potential risk factor that contributes to an increase in postoperative DWH. In a case–control study, we analyzed all patients undergoing elective foot and ankle surgery between January 1, 2014 and December 31, 2017 at two institutions and identified 51 patients with postoperative DWH. After matching with 51 control patients without DWH, all 102 patients were evaluated for PAD. The prevalence of PAD was significantly higher in the DWH group compared to the control group (41.2% vs 19.6%, p < 0.01). This difference was even more distinctive for patients with any abnormal ankle–brachial index (ABI) (51.0% vs 19.6%, p < 0.001). After adjustment for diabetes, hypertension, hypercholesterolemia, and smoking, any abnormal ABI or a history of PAD remained an independent risk factor for DWH (odds ratio 3.28; 95% CI 1.24–8.71). In this dual-center study, postoperative DWH was associated with significantly higher rates of PAD. These findings suggest that preoperative evaluation for PAD could be a helpful tool to identify patients at high risk for postoperative wound complications undergoing foot and ankle surgery.
Introduction
Musculoskeletal disease of the foot and ankle requiring reconstructive surgery are common medical conditions in the western world and often result in a prolonged rehabilitation and considerable productivity losses. 1
Disturbed wound healing (DWH) is a relevant and feared postoperative complication encountered by patients undergoing orthopedic foot and ankle surgery, with a reported incidence of 2–15%. Reported risk factors of DWH in the literature include, among others, age, cigarette smoking, diabetes, and obesity.2–7
Peripheral artery disease (PAD) is a frequent manifestation of atherosclerosis in the western world, showing prevalence rates of 17–20%, rising with age. Men above 80 years of age are the subgroup most affected, with prevalence rates of up to 25%. 8 Signs and symptoms of PAD typically include intermittent claudication and critical limb ischemia, prone to resulting in trophic lesions comprising ulcerations and necrosis. However, epidemiological data show that the majority of patients with PAD are asymptomatic, emphasizing the need for a dedicated diagnostic workup in order to identify these patients. 8 PAD is a well-described risk factor for the development of chronic wounds, ulcerations, and prolonged wound healing of traumatic lesions. 9
Yet, despite the potential connection between the two conditions, there is a lack of systematic data on the impact of PAD on the development of DWH following orthopedic foot and ankle surgery. Although many established risk factors for PAD and DWH are overlapping, current guidelines concerning elective foot and ankle surgery fail to address PAD or preoperative screening for compromised circulation as a risk stratification regarding the development of DWH. 10 The aim of our clinical trial was to evaluate if PAD is a potential (as yet to be described) independent risk factor which may contribute to an increase in postoperative DWH following orthopedic foot and ankle surgery.
In perspective, preoperative screening and an adjusted treatment approach may help to reduce the incidence of postoperative DWH.
Methods
Patients
All patients above 45 years of age undergoing foot and ankle surgery in two large orthopedics departments (Klinik und Poliklinik für Orthopädie, Klinikum rechts der Isar, Munich and Schön Klinik München Harlaching, Munich) between January 1, 2014 and December 31, 2017 were assessed regarding the development of postoperative DWH defined as wound dehiscence or secretion (defined as persistent production of fluid from the wound) 21 days postoperatively or wound necrosis or deep wound infection. Patients meeting criteria for DWH following foot and ankle surgery were matched with patients experiencing normal postoperative wound healing. Matching was performed based on the age and sex of the individual patient. Written informed consent was obtained prior to the evaluation for PAD, and the local ethics committee approved the study. This trial is registered with drks.de, number DRKS00012580.
ABI measurement
All subjects were screened for PAD in the outpatient clinic of our angiology department (Klinikum rechts der Isar, Munich, Germany), unless the diagnosis was previously established and could be obtained from medical files. Ankle–brachial indices (ABI) were determined using Doppler ultrasound according to current guidelines. 11 PAD was defined as an ABI < 0.9 or > 1.4 in both ankle arteries of one leg (with the higher value being taken for diagnosis according to European Society of Cardiology (ESC) guidelines) or a history of PAD of the lower extremities.
Statistical analysis
The predefined primary endpoint was the prevalence of PAD in both case and control patients. Characteristics of the patients and PAD screening outcomes were summarized using descriptive statistics: mean ± SD for quantitative data and frequency (%) for qualitative data. The chi-squared test was used to compare distributions of categorical data, including the primary endpoint, between independent groups. The t-test for independent samples was used for group comparisons regarding continuous data. A logistic regression model was fitted to the data to adjust for differences in smoking habits, hypercholesterolemia, hypertension, and diabetes between the groups. Data were analyzed using SPSS version 20.0 (IBM Corp., Armonk, NY, USA) and SigmaPlot version 12.5 (Systat Software Inc., San Jose, CA, USA).
Results
Patient population
A total of 51 patients, 45 years of age and older, meeting criteria for DWH following foot or ankle surgery were identified and matched with 51 patients with normal postoperative wound healing. Baseline characteristics of all patients evaluated are shown in Table 1. The average age was 62 ± 10 years, and half of the patients were female. Hypertension was the most common cardiovascular risk factor and was present in about 60% of patients in the DWH group. A history of smoking or active smoking was significantly more frequently found in the DWH group as compared to the group with normal wound healing. Diabetes was equally distributed between the two groups. Coronary artery disease and a history of myocardial infarction or vascular intervention/surgery had a higher prevalence in the DWH group as compared to the control group. Consequently, permanent medical therapy, including anticoagulation (20%) and antiplatelet therapy (31%), was more common in the DWH group. Surgical characteristics were similarly distributed, exhibiting no significant differences between the two groups (online Supplemental Table 1). All patients underwent a clinical examination by the orthopedic surgeon prior to the foot and ankle surgery. No absent foot pulses and no signs of neuropathy were documented. There were no amputations or diabetic ulcers present in the studied population.
Characteristics of the patients at baseline.
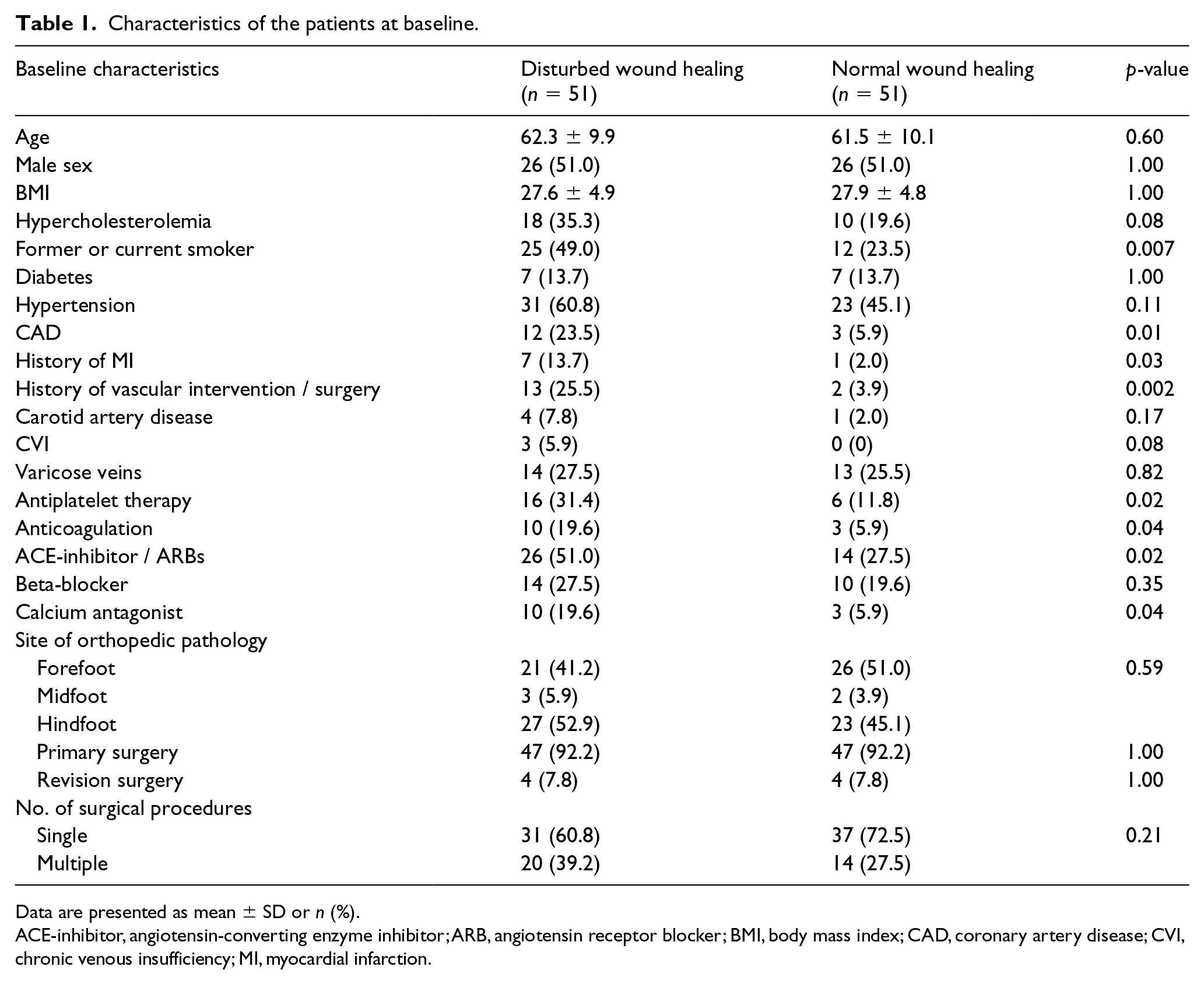
Data are presented as mean ± SD or n (%).
ACE-inhibitor, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; BMI, body mass index; CAD, coronary artery disease; CVI, chronic venous insufficiency; MI, myocardial infarction.
PAD distribution
The prevalence of PAD as defined by the ESC was significantly higher in the DWH group than in the control group (41.2% vs 19.6%, p < 0.01) (Figure 1A). This difference was even more distinctive for patients with an abnormal ankle–brachial index (ABI) in at least one ankle artery (51.0% vs 19.6%, p < 0.001) (Figure 1B). After adjustment for diabetes, hypertension, hypercholesterolemia and smoking, any abnormal ABI or a history of PAD remained an independent risk factor for the occurrence of DWH (odds ratio 3.28; 95% CI 1.24–8.71).
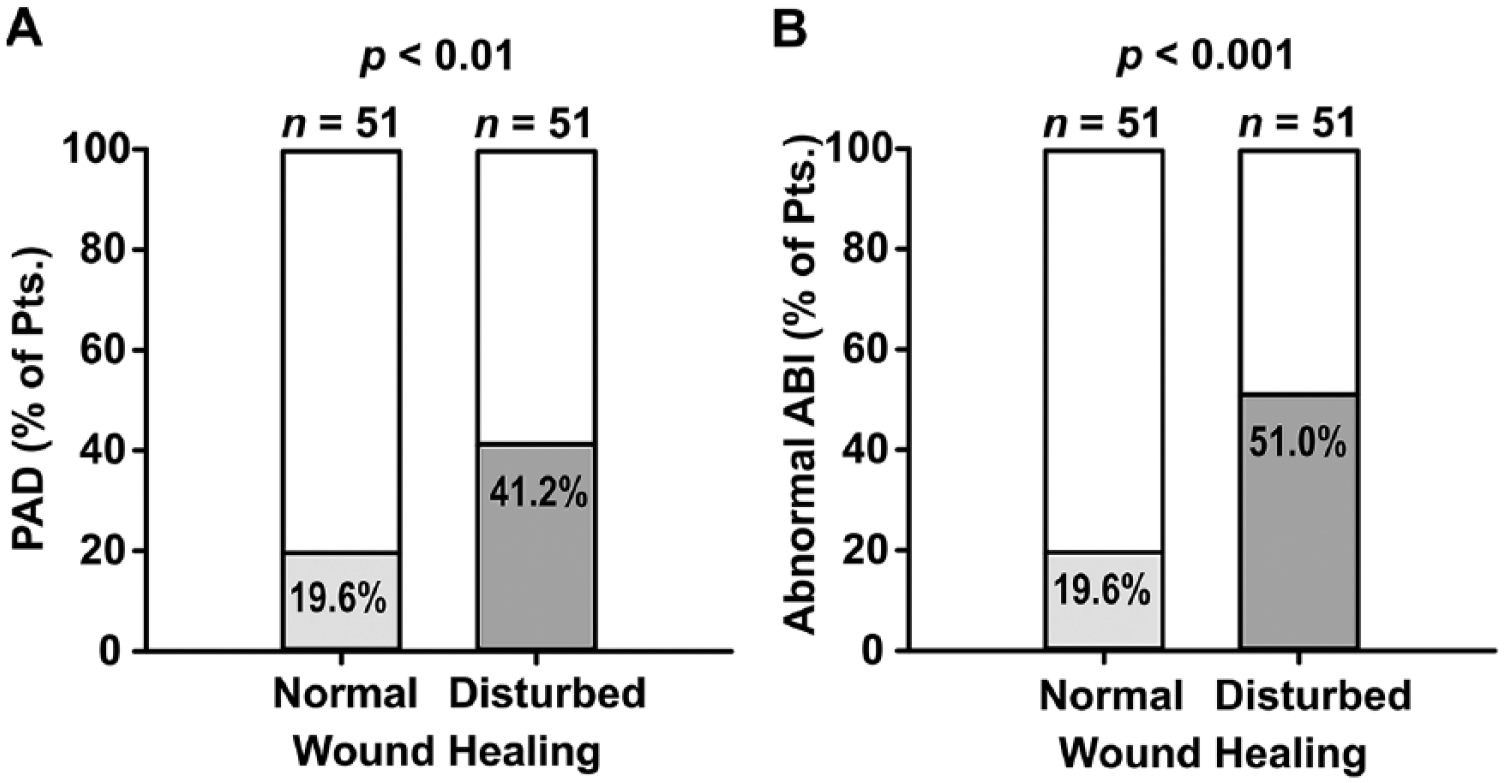
Prevalence of PAD as defined by ESC guidelines 11 (A) or an abnormal ABI in at least one ankle artery (B) in patients with and without DWH after foot and ankle surgery.
Most of the patients with PAD in both groups were asymptomatic before surgery. Roughly 10–20% of patients presented with symptomatic PAD (i.e. intermittent claudication) (Figure 2).
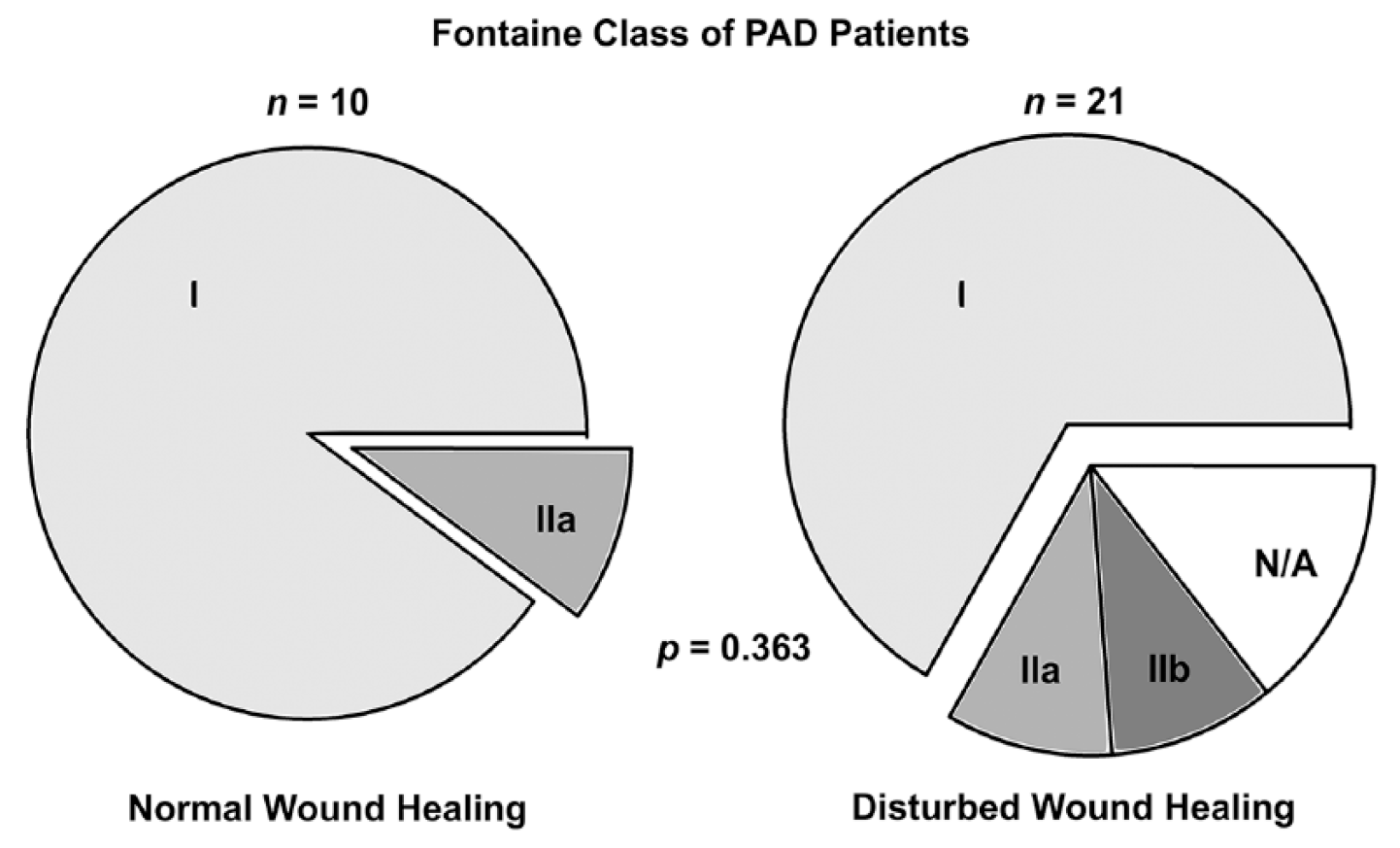
Symptoms of PAD patients prior to orthopedic surgery.
In patients with DWH, an abnormal ABI was numerically more common in the operated leg and more frequent than in patients without DWH (73.1% vs 60.0%, p = 0.322) (Figure 3).
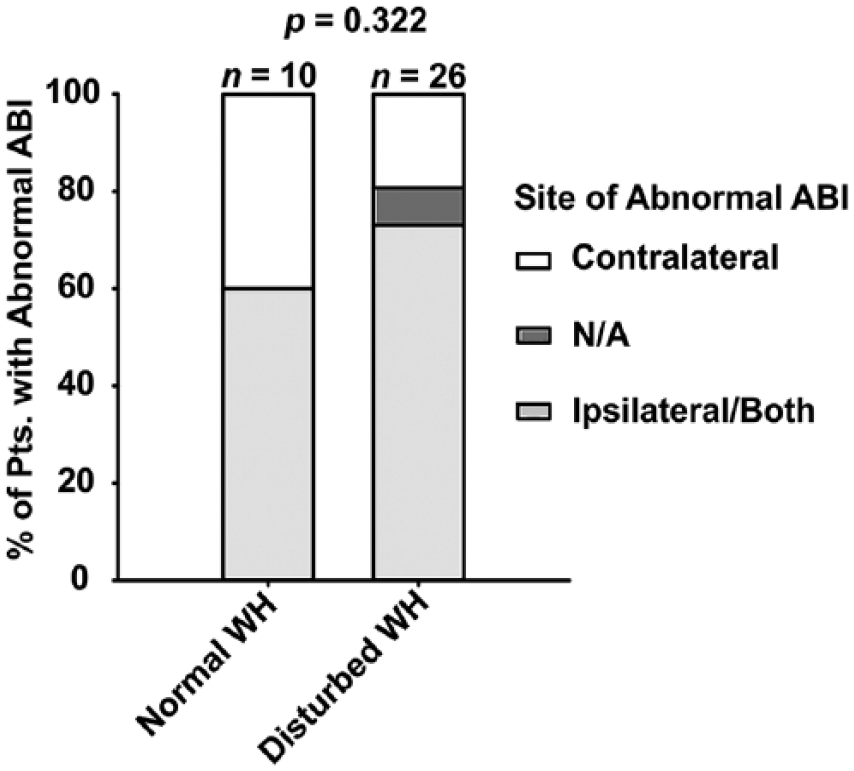
Location of abnormal ABI regarding operation site.
In the group with DWH, surgical wound revisions were slightly more often performed in patients with proven PAD compared to patients with normal peripheral blood supply (46.2% vs 36.0%, p = 0.461), suggesting an at least equal severity of wound healing disorders and suggesting that abnormal ABI is one important risk factor among others. In three patients with proven PAD, a secondary revascularization was performed (Figure 4).
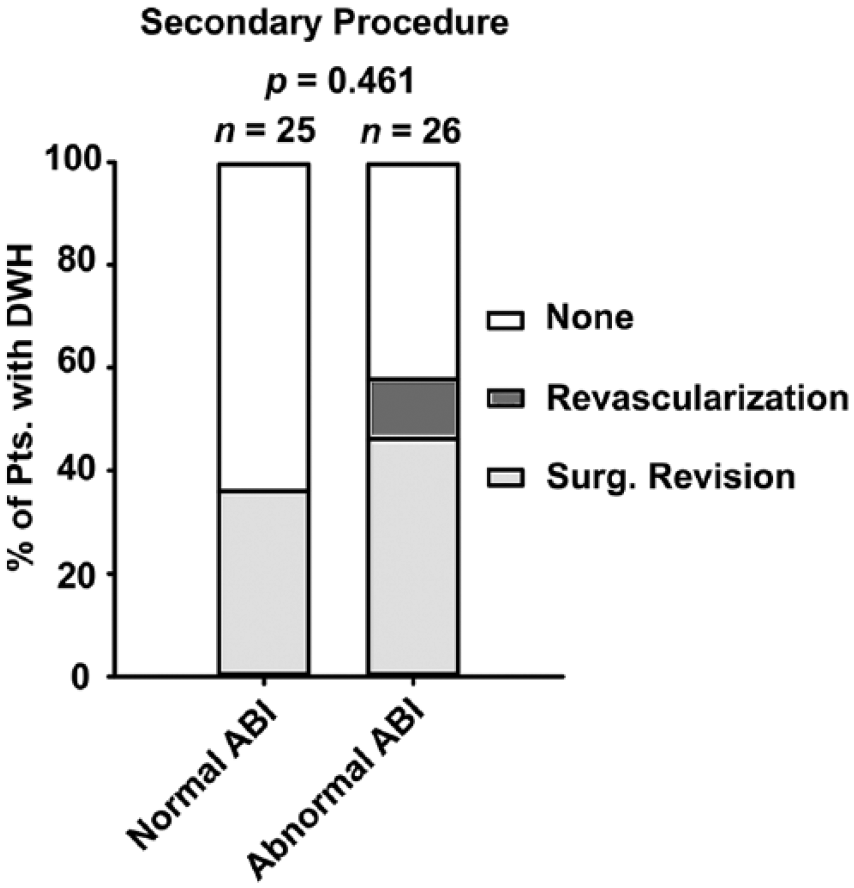
Necessity of secondary procedures in patients with DWH.
To evaluate the question of whether further clinical predictors (beyond the study’s inclusion age > 45 years) can help to preselect patients with a high likelihood of abnormal ABI, we compared the count of classical cardiovascular risk factors (hypertension, cholesterol, diabetes, smoking, family history) as well as a PAD risk score based on the REACH registry 12 of patients with normal and abnormal ABI for all study patients. Both risk factor count (normal ABI: median 1, range 0–4; abnormal ABI: median 2, range 0–5) and PAD risk score (normal ABI: median 29, range 17–43; abnormal ABI: median 34, range 20–47) did not allow a useful discrimination of patients at risk by a specific cut-off value.
Discussion
The key finding of the current study is that the PAD prevalence was significantly higher in patients with DWH following orthopedic foot and ankle surgery compared to patients with normal wound healing.
Although PAD is a well-established risk factor for chronic wounds, the connection appears to be commonly overlooked during the preoperative evaluation of patients, if no wounds are present. A situation that is also reflected by the current surgical guidelines. Our study clearly demonstrates that even asymptomatic patients with slightly abnormal blood supply to the extremities may be at high risk for DWH.
ABI in preoperative screening
The data arising from this trial suggest that systematic preoperative screening for PAD by ABI measurement may be helpful to identify patients at risk for the development of DWH. Furthermore, it appears reasonable to further evaluate affected patients for the need of revascularization, possibly prior to surgery – at least in elective cases. Based on our study results, it may be justified to screen all patients > 45 years, as early stages of vascular disease may not be predicted reliably by risk profile and risk scores.
Therapeutic consequences
Interestingly, the majority of patients with abnormal perfusion of the extremities were not symptomatic for PAD at the time of orthopedic surgery. In the absence of wounds, these patients would not be candidates for revascularization therapy according to current recommendations. Finding a sensible therapeutic approach for these individuals will be demanding. Revascularization being the preferred strategy in patients with abnormal ABI, apparent arterial stenosis in duplex sonography, and matching symptoms, no such clear pathway exists for asymptomatic patients with an abnormal ABI and/or duplex sonography finding. In this subgroup of patients, several approaches, ranging from patient education or an intensified medical therapy (such as vasoactive drugs, statins or platelet inhibitors) to revascularization, can be discussed.11,13 Furthermore, the angiosome (i.e. the anatomic unit of tissue fed by an artery) with altered perfusion and the site of the surgical procedure should be taken into account in preoperative planning. 14
Alteration of the surgical approach may be a just as important option in patients with pathological vascular findings. Where applicable, using a less invasive and therefore less traumatic operation technique may lead to less tissue damage and might be a sensible way to reduce the incidence of DWH in individuals with altered peripheral perfusion. In the same way, these high-risk patients may benefit from the absence of a tourniquet or minimal tourniquet time leading to minimal perioperative ischemia. 6 In any case, further research will be needed to define the optimal approach to asymptomatic vascular patients undergoing orthopedic surgery.
Evaluation of foot pain
Finally, since the distinction between ischemic and musculoskeletal pain in clinical practice can be challenging, standard preoperative ABI measurement could improve patient selection for both orthopedic procedures and vascular interventions, as sometimes intermittent claudication may be misjudged as musculoskeletal pain and vice versa. An interdisciplinary approach encompassing orthopedists and angiologists could prove to be helpful in finding an optimal strategy. Further data derived from prospective studies is needed to underline this concept and to achieve integration into guidelines for foot and ankle procedures.
Limitations
The current study is a dual-center case–control study. Owing to the small sample size, it cannot adjust for all unidentified risk factors that could be important for the occurrence of postoperative DWH. In three-quarters of patients with abnormal ABI and DWH, the foot affected by DWH also had an abnormal ABI, supporting the idea of a pathophysiological connection between PAD and DWH in our patients. However, the study design does not allow conclusions about causality in the relationship between PAD and DWH. Furthermore, owing to the retrospective identification of patients, wound evaluation was based on medical files. To minimize bias from this fact, clear definitions for postoperative DWH were utilized. Our study aimed to provide a simple approach feasible in primary care and orthopedics settings. Therefore, ABI measurement was chosen as the method to screen for PAD. However, ABI measurement may miss obliterating arteriosclerosis below the ankle and the values are less meaningful in uncompressible arteries of Mönckeberg’s sclerosis. Measurement of toe–brachial index (TBI) – usually performed by vascular specialists – can overcome these limitations and should be included in a dedicated diagnostic workup for patients with DWH and suspected underlying vascular disease. Lastly, new cases of PAD may have occurred in the time window between surgery and the evaluation for this study. Therefore, no patients operated before January 1, 2014 were included in the study.
Conclusions
The current study demonstrates that patients with DWH following foot and ankle surgery had a higher prevalence of PAD compared to patients experiencing normal postoperative wound healing. Systematic preoperative assessment of ABI may help to identify patients at risk for postoperative DWH.
Supplemental Material
10.1177_1358863X19883945_Supplementary_table – Supplemental material for Significant prevalence of peripheral artery disease in patients with disturbed wound healing following elective foot and ankle surgery: Results from the ABI-PRIORY (ABI as a PRedictor of Impaired wound healing after ORthopedic surgerY) trial
Supplemental material, 10.1177_1358863X19883945_Supplementary_table for Significant prevalence of peripheral artery disease in patients with disturbed wound healing following elective foot and ankle surgery: Results from the ABI-PRIORY (ABI as a PRedictor of Impaired wound healing after ORthopedic surgerY) trial by Arne M Müller, Andreas Toepfer, Norbert Harrasser, Bernhard Haller, Markus Walther, Rüdiger von Eisenhart-Rothe, Karla Gemperlein, Katharina Bergmann, Christian Bradaric, Karl-Ludwig Laugwitz, Tareq Ibrahim and Ralf J Dirschinger in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
