Abstract
Screening for asymptomatic peripheral artery disease (aPAD) with the ankle–brachial index (ABI) test is hypothesized to reduce disease progression and cardiovascular (CV) events by identifying individuals who may benefit from early initiation of medical therapy. Using a Markov model, we evaluated the cost effectiveness of initiating medical therapy (e.g. statin and ACE-inhibitor) after a positive ankle–brachial index (ABI) screen in 65-year-old patients. We modeled progression to symptomatic PAD (sPAD) and CV events with and without ABI screening, evaluating differences in costs and quality-adjusted life years (QALYs). The cost of the ABI test, physician visit, new medication, CV events, and interventions for sPAD were incorporated in the model. We performed sensitivity analysis on model variables with uncertainty. Our model found an incremental cost of US $338 and an incremental QALY of 0.00380 with one-time ABI screening, resulting in an incremental cost-effectiveness ratio (ICER) of $88,758/QALY over a 35-year period. The variables with the largest effects in the ICER were aPAD disease prevalence, cost of monthly medication after a positive screen and 2-year medication adherence rates. Screening high-risk populations, such as tobacco users, where the prevalence of PAD may be 2.5 times higher, decreases the ICER to $24,092/QALY. Our analysis indicates the cost effectiveness of one-time screening for aPAD depends on prevalence, medication costs, and adherence to therapies for CV disease risk reduction. Screening in higher-risk populations under favorable assumptions about medication adherence results in the most favorable cost effectiveness, but limitations in the primary data preclude definitive assessment of cost effectiveness.
Introduction
Approximately half of the patients with peripheral artery disease (PAD) do not report symptoms.1,2 Population screening to identify asymptomatic PAD (aPAD) using the ankle–brachial index (ABI) test has been hypothesized to reduce cardiovascular (CV) related events and delay the progression of aPAD to symptomatic PAD (sPAD) with the initiation of medical therapy and lifestyle changes.3–6 The ABI tests can diagnose PAD (index <0.9) with high sensitivity (75%) and specificity (90%) in patients with symptomatic disease.7,8 The ABI test is relatively inexpensive and poses minimal risk to the patient. 9
Professional societies have varying recommendations regarding appropriate use of PAD screening with the ABI test (Table 1) as evidence supporting population based screening for aPAD using the ABI test is limited.10–15 A previous cost-effective analysis advocated screening for aPAD as it may be cost effective by initiating anti-platelet therapy as a result of a positive screen 16 ; however, no randomized controlled trials have been conducted to show that aPAD screening alone will reduce CV related events and mortality. 17 In 2006, the United States Preventive Services Task Force (USPSTF) recommended against ABI screening as the potential for unnecessary angiograms would outweigh the benefits. 18 Screening for aPAD was re-evaluated in 2013 by the USPSTF, considering risk factor modification with a positive ABI test, and the overall recommendation changed from ‘against’ to indeterminate. 10 This study aims to evaluate the cost effectiveness of implementing ABI screening in asymptomatic adults who may benefit from initiation of medical therapy and lifestyle changes.
ABI screening recommendations from societal guidelines.
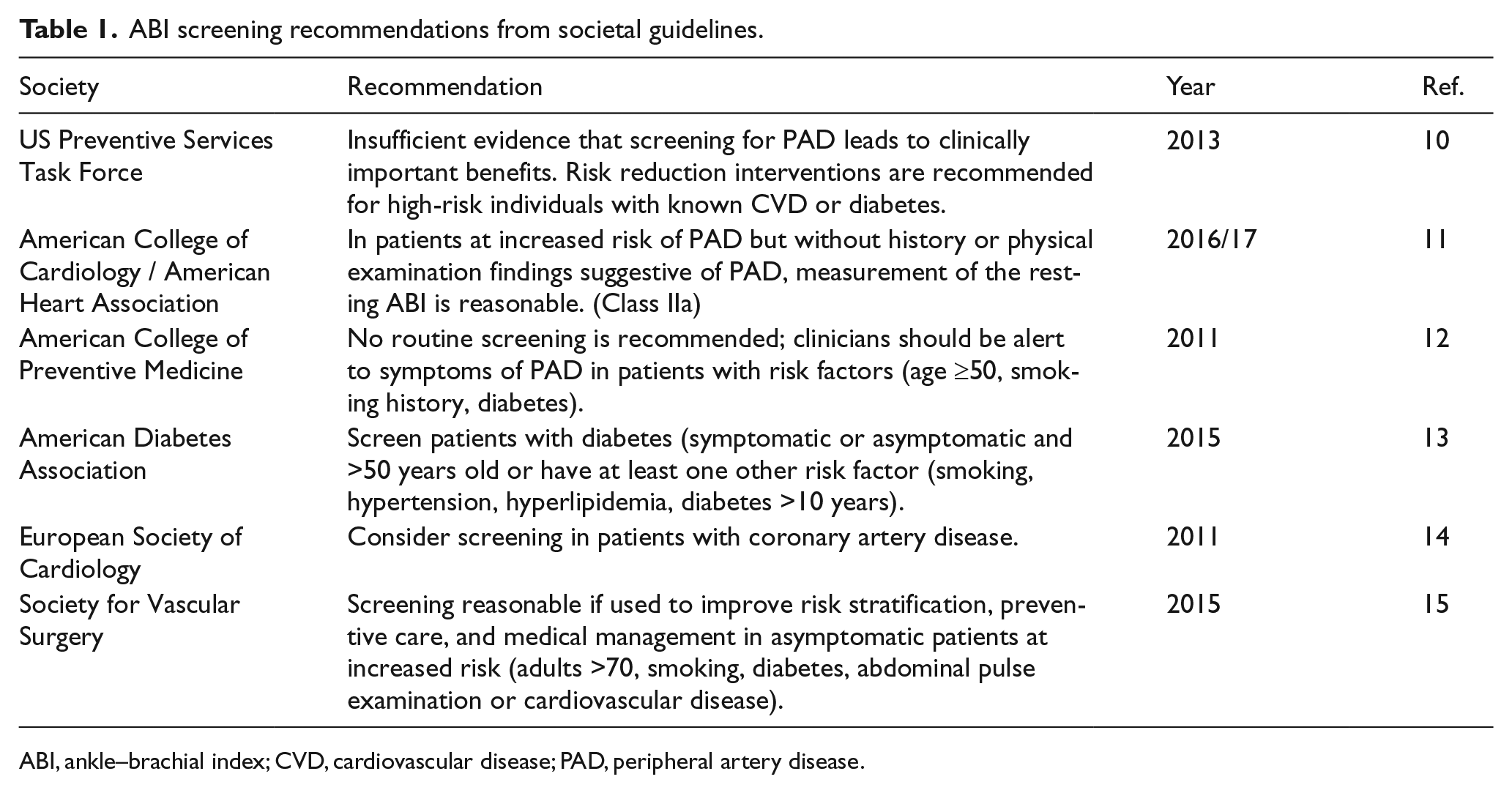
ABI, ankle–brachial index; CVD, cardiovascular disease; PAD, peripheral artery disease.
Methods
This cost-effectiveness analysis uses a decision tree and Markov model to predict the costs and health outcomes of aPAD screening with the ABI test. The model tracks costs in 2017 US dollars from the healthcare sector perspective, and evaluates health outcomes in quality-adjusted life years (QALYs). 19 All costs are discounted by 3% per year. Modeling was performed with TreeAge Pro Healthcare 2017 software (Williamstown, MA, USA). Published literature, including meta-analyses when available, were used for input values into our model. If specific values were not available for aPAD alone, values based on sPAD were used from the literature. Variables with uncertainty underwent sensitivity analysis to include the range of uncertainty.
Markov model
The overall model compares patients screened with the ABI test to patients without screening to determine differential costs and QALYs. The Markov model incorporates the natural history of PAD from the healthy state to aPAD followed by sPAD, surgical intervention, amputation, and death (Figure 1). Patients with aPAD, sPAD, and amputation can progress to CV related events modeled as acute myocardial infarction (MI) and acute stroke. All patients progress to death according to age adjusted 20 and disease related rates (Table 2). The benefits of aPAD screening are accounted for by a decrease in progression to sPAD from aPAD and lower rates of acute stroke and MI due to medication initiation and lifestyle changes after a positive ABI test.
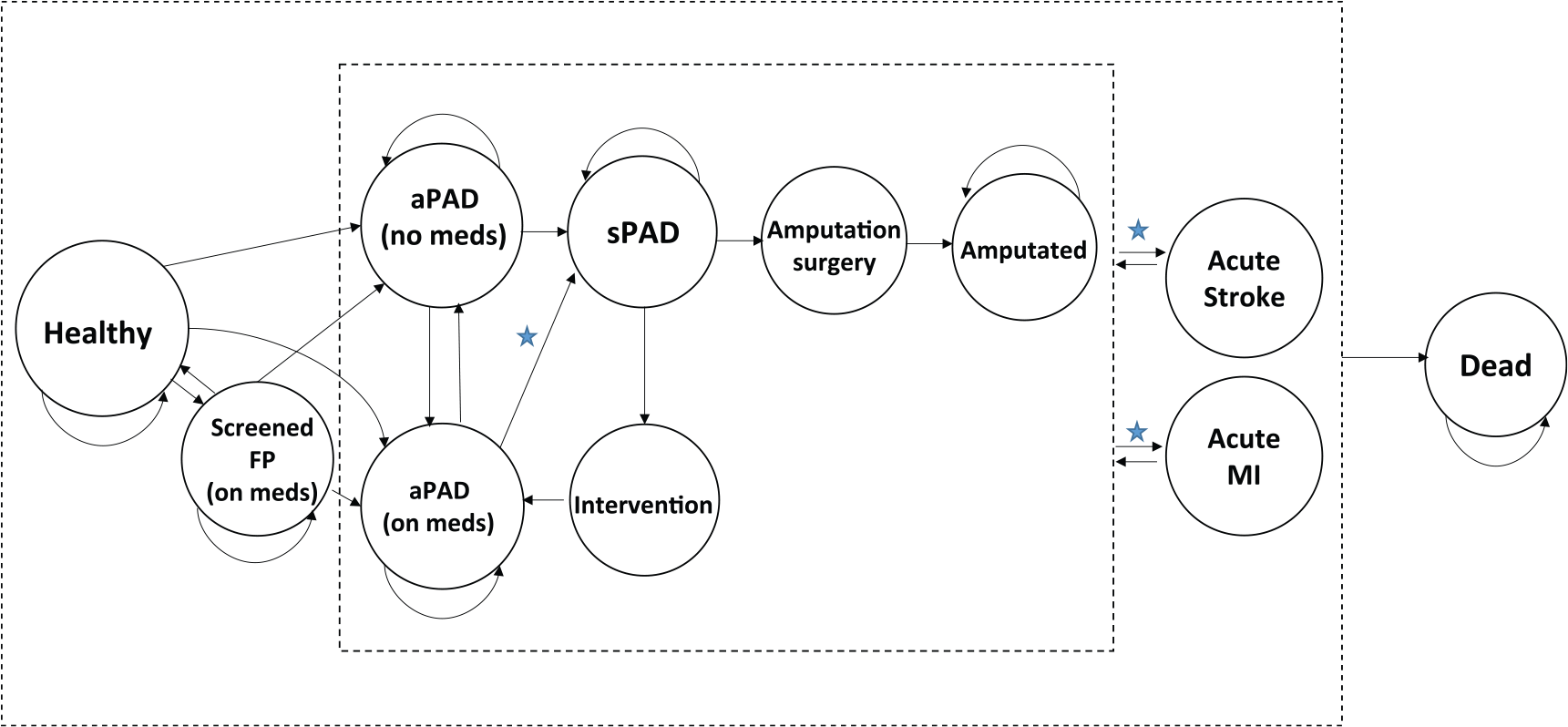
Markov model states and transitions. The cost effectiveness of the ankle–brachial index (ABI) test is performed by comparing ABI screening to no ABI screening. This captures the differences in cost and quality-adjusted life years in patients with aPAD not on medication (aPAD (no meds)) to patients with aPAD and assigned medication (aPAD (on meds)) after a positive ABI screening. The benefits of ABI screening are noted by stars, where there is a slower progression to sPAD and a lower incident rate of acute stroke and MI due to medication initiation and lifestyle changes after a positive ABI test. See the ‘Methods’ section for further details. (The stars indicate transitions where screening may be of benefit with initiation of medical therapy; aPAD, asymptomatic PAD; sPAD, symptomatic PAD; FP, false positive; MI, myocardial infarction.)
Markov model – baseline values.
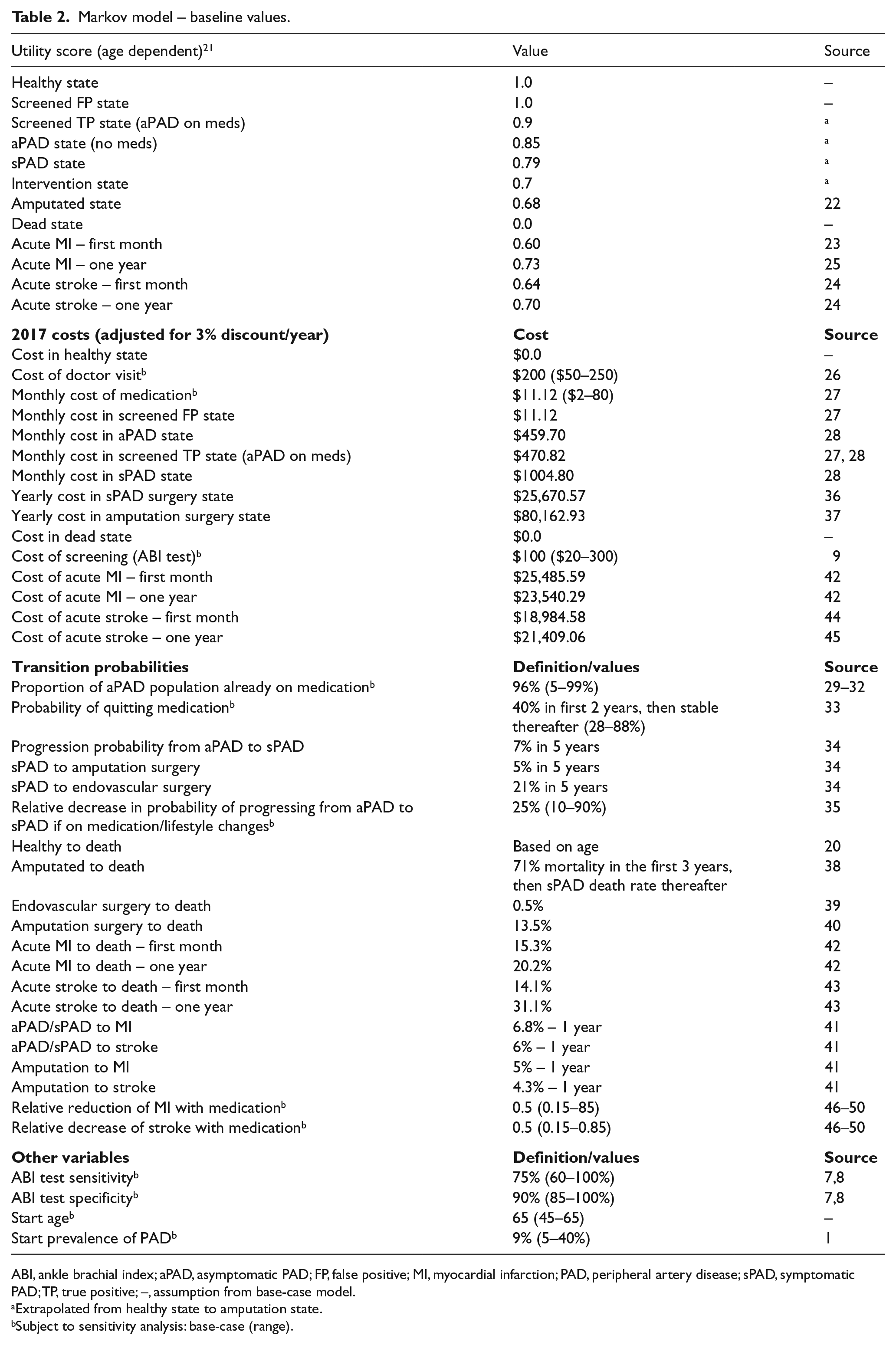
ABI, ankle brachial index; aPAD, asymptomatic PAD; FP, false positive; MI, myocardial infarction; PAD, peripheral artery disease; sPAD, symptomatic PAD; TP, true positive; –, assumption from base-case model.
Extrapolated from healthy state to amputation state.
Subject to sensitivity analysis: base-case (range).
The healthy and death states have a QALY of 1.0 and 0.0, respectively, and are adjusted according to the patient’s age adjusted QALY: at age 35, 45, 55, 65, and 75 the QALY is 0.8, 0.8, 0.78, 0.78, and 0.76, respectively. 21 The QALY assigned to aPAD, sPAD, or amputated state is extrapolated from the amputated state of 0.68. 22 Patients who experience an acute MI or stroke also have reduced QALYs at 1 month: 0.60 and 0.64, respectively.23,24 After 1 year the QALY for acute MI improves to 0.73 and 0.70 for stroke.24,25 Each cycle in the model is 1 month and the effects of aPAD screening are analyzed up to 35 years.
Patient population
We set our base-case model to screen patients in the US population at 65 years of age. The model considers age ranges above and below 65 years old incorporating the age related mortality and QALYs.20,21 Prevalence of PAD at specific ages are included in the model: at age 45, 55, 65, 75, and 85 the prevalence is 2%, 4%, 9%, 18%, and 30%, respectively. 1 High-risk populations (e.g. tobacco users) are considered in the model by increasing the initial prevalence rate of the population screened.
PAD screening
The model assumes ABI screening in a primary care setting, where a positive test triggers medicine and lifestyle intervention. We set the base-case cost of the ABI test in the outpatient setting at US $100 and include a sensitivity analysis for this cost. 9 The costs of accompanying physician visits are also included in the model set at a base-case of $200. 26 We set the base-case sensitivity and specificity of the ABI test in detecting aPAD at 75% and 90%, respectively, and perform sensitivity analyses of these values as the sensitivity and specificity of the ABI test is not well established for aPAD.7,8
Treatment strategies after a positive screening result
A positive ABI test assigns statin and angiotensin-converting-enzyme (ACE)-inhibitor medication as well as lifestyle modification counseling with the goal of improving both CV and PAD related outcomes. Patients with a positive ABI test are noted as ‘aPAD (on meds)’ in Figure 1. The monthly costs of starting an ACE-inhibitor and statin are $11.12, based on Veterans Affairs pharmaceutical prices and dispensing fees. 27 The cost of medications undergo sensitivity analysis given the variation in drug costs. The monthly cost of aPAD care without medications is $459.70, and $470.82 after medication costs are added with a positive ABI test. 28 Patients in the aPAD group may already be on a statin therapy and/or ACE-inhibitor therapy due to other comorbidities and we set the baseline of patients already on medication at 96%.29–32 This proportion of the population already eligible for similar medical therapy is considered in a one-way sensitivity analysis given that eligible patients may not be taking medication due to patient non-compliance or varying prescribing patterns. Long-term medication non-compliance after a positive ABI test is also incorporated into the model based on a 2-year non-compliance rate of 40%, which also undergoes sensitivity analysis. 33 Patients who are non-compliant with medical therapy transition back to the ‘aPAD (no meds)’ state (Figure 1). Patients can also be in a false positive state, ‘Screened FP (on meds)’, with a misdiagnosis from the initial ABI test.
Transitions
The starting healthy state progresses to aPAD according to age based prevalence. Patients with aPAD progress to sPAD over time (7% over 5 years) based on the natural history of disease. 34 This progression rate is reduced with medication initiation and lifestyle modification, set at a 25% reduction over 5 years. 35 Patients with sPAD can undergo surgical intervention to return to the aPAD state on medications, with an associated yearly cost of $25,670.57. 36 The sPAD state can also progress to the amputated state, which carries higher mortality and costs ($80,162.93/year).37,38 Peri-operative mortality is considered when patients undergo a surgical intervention for sPAD and amputation.39,40
The transition from aPAD, sPAD, and amputated states to CV events (i.e. acute MI and stroke) are based on yearly incident rates. 41 Patients who progress to acute MI and stroke have associated 30-day and 1-year mortality.42,43 If patients do not progress to death after an MI or stroke, they then return to their original state (e.g. aPAD, sPAD, or amputation), and can have a recurrence of an acute MI or stroke according to yearly incident rates. The associated first month and yearly cost of MI and stroke are noted in Table 2.42,44,45 The reduction of CV events and subsequent mortality is reduced by 50% with initiation of medication and lifestyle changes, and is subject to sensitivity analysis.46–50 All healthy and diseased states in the model can progress to death. For further details regarding transitions see Table 2 and Supplemental Table 1.
Sensitivity analysis
Model variables with uncertainty underwent sensitivity analysis as noted in Table 2. To determine variables that influence the incremental cost-effectiveness ratio (ICER) the most, we conducted one-way sensitivity analyses for each variable of interest given uncertainties in the literature.
Results
The base-case model for screening the general population costs $338 more compared to no screening. The increased cost is due to the ABI test, physician visit, and cost of medication for a positive ABI test. There is a mild offset in the overall costs due to slower progression of disease for screened patients, who require less surgical intervention and amputation. The screened patients also have an increase of QALYs of 0.00380, or 1.39 days, compared to no screening. The overall ICER is $88,758/QALY, demonstrating that aPAD screening increases the overall QALY with an associated increase in costs (Figure 2). Removing the benefits of medication effects on MI and stroke increases the ICER to $309,065 (248% increase) and removing the benefits of medication on PAD progression increases the ICER to $93,309 (5.1% increase).
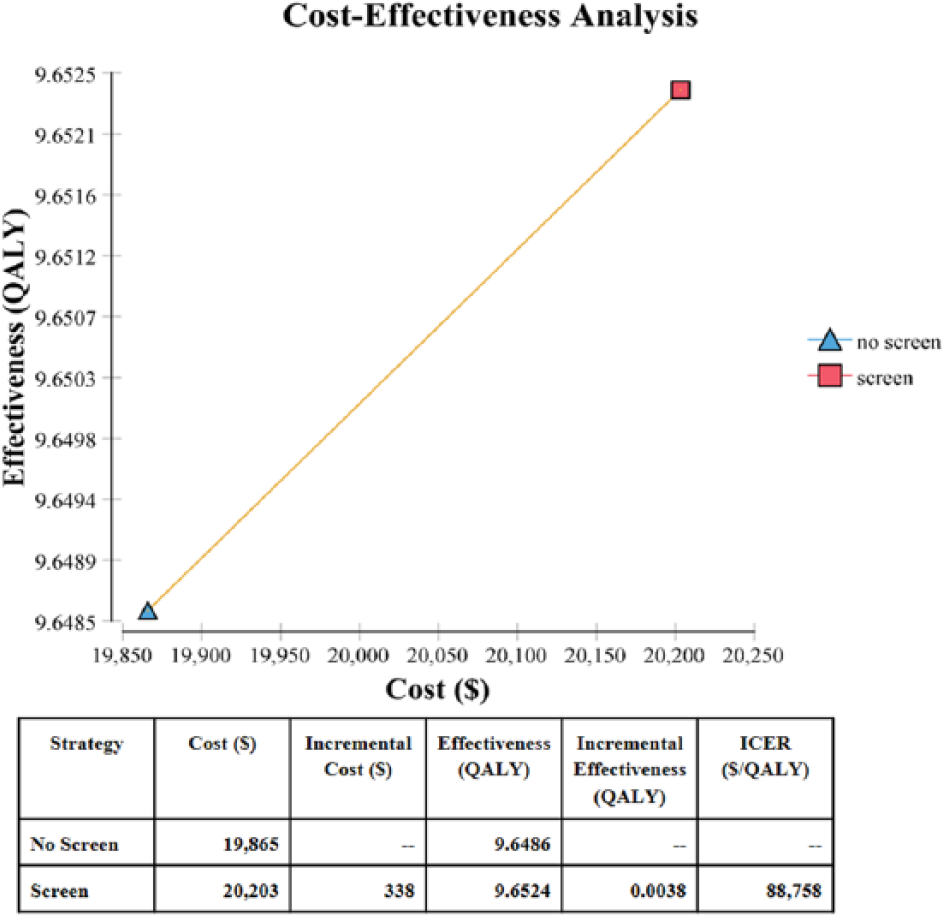
Cost-effectiveness frontier comparing screening and no screening with the ankle–brachial index (ABI) test by costs and quality-adjusted life years (QALYs). (ICER, incremental cost-effectiveness ratio.)
Our sensitivity analyses of variables with uncertainty are shown in Figure 3. The top three variables affecting the ICER are prevalence of aPAD in the population being screened, monthly cost of medication after a positive ABI screen, and medication adherence rates. One-way sensitivities for these variables are shown in Figure 4. The variables that decrease the ICER below $50,000/QALY are the starting prevalence, medication adherence, and percentage of patients already eligible for medical therapy. Screening high-risk populations, such as tobacco users, where the prevalence of PAD is predicted to be 23%, decreases the ICER to $24,092/QALY, a 73% decrease from the base-case (Figure 4a), with favorable assumptions about adherence to risk-reducing medications.
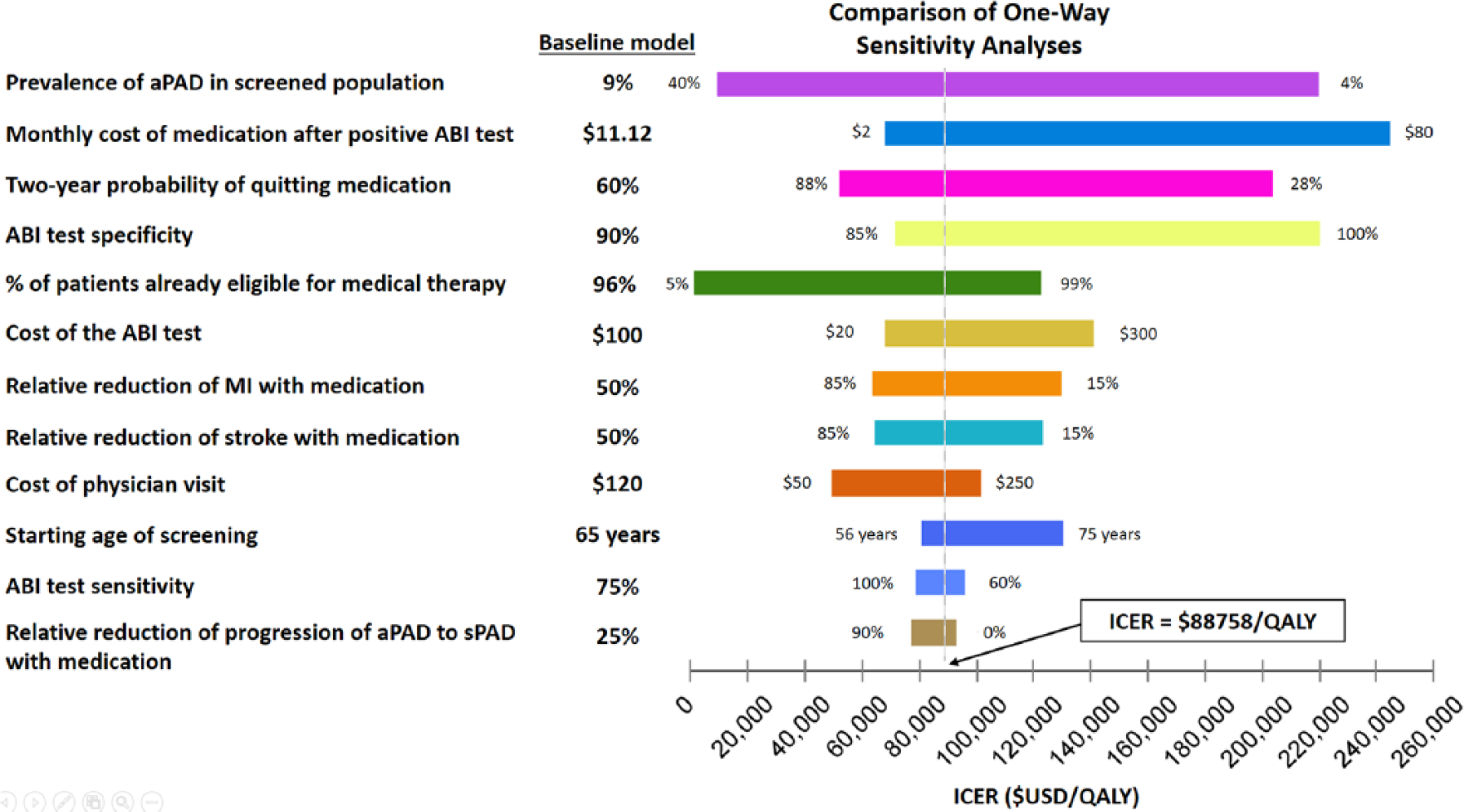
One-way sensitivity analysis for input variables with uncertainties. The variables are arranged from top to bottom, starting with the largest range of ICER, depending on the uncertainty range. The baseline value for each variable is noted by the vertical line indicating the ICER of $88,758/QALY. (aPAD, asymptomatic PAD; ICER, incremental cost-effectiveness ratio; MI, myocardial infarction; QALY, quality-adjusted life year; sPAD, symptomatic PAD.)
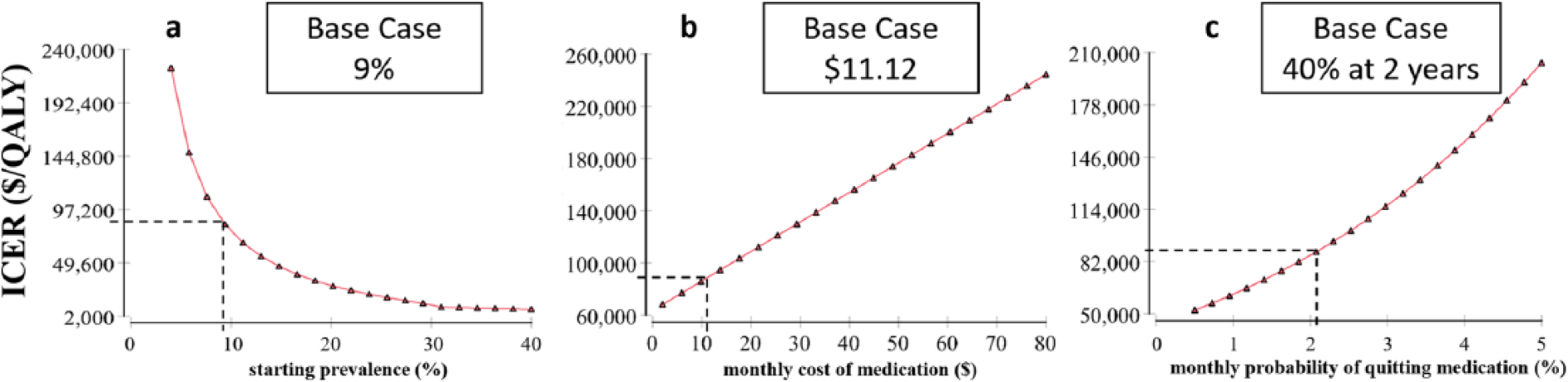
Sensitivity analysis for the top three variables affecting the ICER of aPAD screening with the ABI test: (a) starting prevalence, (b) monthly cost of medication, and (c) monthly probability of quitting medication. (ABI, ankle–brachial index; aPAD, asymptomatic PAD; ICER, incremental cost-effectiveness ratio; QALY, quality-adjusted life year.)
When the screened population already on medical therapy is reduced to 70% from the base model of 96%, the ICER decreases to $20,687/QALY (77% decrease; Supplemental Figure 1b). The optimal age for initial aPAD screening was found to be between 55 and 65 years old (Supplemental Figure 1f). The initial decrease in ICER from 45 to 55 years of age is attributed to an increasing prevalence of disease, while the increasing ICER after 65 years of age is due to the increasing all-cause mortality with age. Other variables affecting the ICER are shown with accompanying one-way sensitivity analyses in Supplemental Figure 1.
Discussion
Our base-case analysis found benefit from a one-time ABI screening to detect aPAD and initiate early medical therapy, reducing CV events and mitigating progression of aPAD to sPAD. With favorable assumptions about the effect of screening on the use of CV risk reducing medications, the ICER was $88,758 per QALY with ABI screening compared to no screening over a 35-year period, which is between the previously reported societal willingness to pay a threshold of $50,000 and $100,000. 51 Our results are similar to previous studies showing benefit from initiation of medication therapy in patients with aPAD.16,48 However, our model found a slightly higher ICER compared to these studies due to a variety of factors discussed below. As we also discuss below, empirical evidence regarding the effect of ABI screening on risk-reducing medication remains sparse, suggesting caution in interpreting our findings.
Effects of prevalence, high-risk populations, and age
The cost effectiveness for aPAD screening improves when there is a higher prevalence of disease in the screened population (Figure 4a). Screening high-risk populations, such as tobacco users, where the prevalence of PAD may be 2.5 times higher than the general population, decreases the ICER to $24,092/QALY (Table 1). Currently, the ABI test is not reimbursed in patients without symptoms. This favorable ICER shows ABI testing will be most cost effective in high-risk populations. Regarding age, our model found the highest return for screening when the patient is 55–66 years old (Supplemental Figure 1f). Although an older population has a higher prevalence of PAD, higher all-cause mortality diminishes the benefits of population screening at older ages.
Medication effects after a positive ABI test
We note that similar to abdominal aortic aneurysm screening, which may lead to a reduction in both disease and all-cause mortality,52,53 the focus of aPAD screening should lead to risk modifications to decrease CV related events and slow progression to sPAD. The medication cost after a positive ABI screen is relatively inexpensive and is noted at $11.12 per month in our base-case scenario, as statin and ACE-inhibitor prices have decreased with generic production. 27 At this low cost, we observe a benefit in ABI screening; however, small increases in medication costs can make ABI screening less cost effective (Figure 4b). We also note that prescribers may vary in choosing the cheapest medication, thus reducing the cost effectiveness of aPAD screening. 54
Medication non-adherence for chronic disease can cost the health care system $100 billion annually and it is unknown how a patient would increase adherence to medication and lifestyle recommendations with a positive ABI. 55 Previous reports have shown that statin adherence is only 40% at 2 years, even with financial incentives to patients and providers to reach LDL goals. 56 We speculate that medication adherence rates improve with screening as a diagnosis of PAD provides evidence of atherosclerosis disease. Patients with coronary artery disease (CAD) have higher compliance rates for statin medication when there is symptomatic disease compared to asymptomatic disease. 33 We also note that diagnosing PAD may help with risk stratification in CAD patients and support the use of more aggressive lipid goals similar to proposed adjunct testing with carotid ultrasound and coronary calcium scoring.57,58
A critique of PAD screening is that many patients who are eligible for screening may already be on statins, ACE-inhibitors, and anti-platelets for general management of their CV risk factors per current primary care guidelines and thus not benefit from screening. We considered 96% of patients already eligible for therapy in our model as this reflected previous population studies.29–32 Although 4–5% of patients eligible for new medication may seem small this can account for 4.9 million US adults with PAD without established CV disease. 32 Furthermore, we note that medication adherence is imperfect, as seen in real world data regarding patients who are eligible for cardio-protective medicine. 59 Therefore, the ICER may be lower if the screened population’s eligibility for therapy is below our baseline model of 96% (Supplemental Figure 1b).
The long-term effects of medication on aPAD patients encompass one of the larger uncertainties in the predicted ICER, including the hazard ratio for CV related events in patients with aPAD. Randomized controlled trials have not shown a benefit from an anti-platelet regimen in the primary prevention of CV related events, 60 but statins and ACE-inhibitors provide improvements to CV related mortality in aPAD patients.49,50 Our baseline model incorporates a 50% reduction in overall CV related events as this includes synergistic medication effects and possible lifestyle changes with a positive screen. Previous studies have also demonstrated favorable outcomes with lifestyle changes such as diet modification and exercise after CAD diagnosis. 61
Limitations
Limitations of the ABI test
The ABI test carries a wide range of uncertainty and limitations. The cost effectiveness of screening is limited by the specificity and sensitivity of the ABI test (Supplemental Figures 1a and 1h). More expensive diagnostic tests, such as duplex ultrasound, computed tomography (CT) angiography, and MR angiography, provide higher diagnostic capabilities assessing vascular flow and morphology but are associated with higher costs, risk and time. Although we do not consider these diagnostic imaging modalities in our model, as these tests are usually reserved for patients with symptoms or non-healing wounds, we note that increasing the cost of the ABI test from $100 to $300 in our model increases the ICER to $141,309 (Supplemental Figure 1c). Therefore, costly follow-up tests beyond the ABI test (e.g. a CT scan, which may cost thousands of dollars) need to be avoided in routine practice to make aPAD screening cost effective.
Although the ABI test has high accuracy with a receiver operating characteristic (ROC) area of 0.87 in symptomatic patients, there is far less data in asymptomatic patients and there are patients for whom screening may more commonly lead to inaccurate results, such as diabetic patients and those with pretibial calcification. 7 A previous study found that toe–brachial indices and skin perfusion may be more cost effective in diagnosing PAD in diabetic patients with foot wounds. 62 Currently, new and easy-to-use technology is being evaluated to asses flow to the lower extremities that has the potential to more accurately detect PAD with great ease of use. 63 We emphasize that the introduction of new technology must be evaluated for its sensitivity, specificity, and cost to determine cost effectiveness in future analyses. We also note that a lower specificity leads to a lower ICER for aPAD screening (Supplemental Figure 1h). This is explained by an advantage in false positive patients benefiting from early statin therapy,64,65 as well as disease prevalence increasing with age.
Limitations and barriers to implementing ABI screening
It is unclear what the direct and indirect costs would be from implementing aPAD screening in a primary care clinic. Given the increasing logistical demands placed on primary care physicians, it is of utmost importance that further evaluation of PAD screening evaluates the feasibility and cost of implementing and interpreting the ABI test. For example, examining how patient counseling and medication adherence will be affected with a positive ABI screen. 66 Furthermore, other studies are needed to determine when it is appropriate for primary care providers to refer to a specialist for a positive ABI test in the asymptomatic patient to balance ‘unnecessary’ revascularization procedures or supervised exercise therapy instead.18,67
We made simplifying assumptions in our analysis. First, we used sPAD literature values when aPAD values were unavailable, such as the true benefits of medication initiation in patients with aPAD and prevalence of asymptomatic disease. To address this limitation we performed sensitivity analyses, acknowledging that the aPAD population is difficult to identify without prior screening and when observational studies of aPAD are limited. Second, we did not differentiate between an open or endovascular approach to treat sPAD. As the level of disease (e.g. aorto-iliac vs femoral and tibial disease) and patient comorbidities vary, we considered the average cost of intervention over the course of 1 year after an intervention is pursued. We also interpolated QALYs given the lack of evidence and variability of disease states and patient demographics. 68 Furthermore, it is possible that aPAD patients may have worse functional status than patients with sPAD, highlighting the variation in patient complexity and the severity of asymptomatic disease. 69 Combining the paucity of studies and uncertainty in the variables outlined above, further investigation is required prior to implementing large-scale aPAD screening.
Conclusions
Our analysis demonstrates that the cost effectiveness of screening for aPAD with the ABI test depends on the prevalence of disease in the initial population being screened, monthly medication costs, and adherence to medical therapies for CV risk reduction. The lack of high-quality data leads to an indeterminate conclusion on whether aPAD screening with the ABI test is truly below the generally accepted thresholds for cost effectiveness. Further studies are warranted to not only understand the cost effectiveness of aPAD screening, but also to improve overall PAD and CV related health outcomes.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Douglas K Owens is a member of the United States Preventive Service Task Force (USPSTF). This article does not necessarily represent the views and policies of the USPSTF.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institutes of Health, National Center for Advancing Translational Science, Clinical and Translational Science Award (TL1 TR000084) and NIH / NHLBI (5T3HL098049). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
