Abstract
Background:
Underweight patients may be at an increased risk of bleeding while receiving venous thromboembolism (VTE) prophylaxis. Additional evidence is needed to identify patient-specific factors associated with bleeding. The objective of the study was to describe the incidence and identify risk factors associated with bleeding in low-weight (⩽ 60 kg) adult patients receiving subcutaneous unfractionated heparin (SQH) for VTE prophylaxis.
Methods:
This was a single-center, retrospective, nested case–control study of low-weight patients receiving SQH for VTE prophylaxis for ⩾ 48 hours. Cases, patients with clinically relevant bleeding while receiving SQH, and controls, patients without a bleeding event, were matched in a 1:3 ratio for age, sex, primary service (surgical or medical), and time at risk of bleeding on SQH to determine factors associated with bleeding.
Results:
A total of 3761 patients met the inclusion criteria, of which 38 cases of clinically relevant bleeding were identified. The bleeding incidence was 1% at hospital day 6 and 2.8% at hospital day 14. Most patients in this study (69%) received SQH 5000 units three times daily. ICU admission at SQH start was associated with bleeding, OR 2.97 (95% CI 1.21–7.29).
Conclusion:
Bleeding in low-weight patients on prophylactic SQH was uncommon. Patients admitted to the ICU at time of SQH start may be at a higher risk of bleeding. Further studies are needed to detect additional risk factors associated with bleeding and investigate the effects of reduced dosing in this population.
Background
Venous thromboembolism (VTE), consisting of deep venous thrombosis (DVT) and pulmonary embolism (PE), affects up to 900,000 Americans each year, leading to 100,000–300,000 potentially preventable deaths.1 –3 Thromboprophylaxis is proven to reduce VTE by up to 65%.4 –6 Current guidelines favor the use of chemoprophylaxis with either low-molecular-weight heparin (LMWH) or low-dose subcutaneous unfractionated heparin (SQH) over mechanical prophylaxis in medically ill patients who qualify for therapy,7,8 but the evidence supporting an optimal agent or dose of VTE prophylaxis is less clear.
The benefit of VTE prevention with widespread use of chemoprophylaxis must be weighed against the risk of bleeding associated with anticoagulation. Patients with reduced weight specifically may be at an increased risk of bleeding due to use of one-size-fits-all dosing strategies without empiric dose reductions.9,10 However, the incidence of bleeding in this population has been poorly characterized to date. Though low body weight appears to be a risk factor for bleeding associated with VTE prophylaxis, it is likely not the only factor that needs to be accounted for in determining individual risk in patients.
To help identify medical patients who may be at an increased risk of bleeding, the IMPROVE trial retrospectively reviewed over 10,000 medical patients and found major bleeding to occur in only 1.2% of patients within 14 days of admission, 11 an incidence similar to that described in prospective randomized studies assessing VTE prophylaxis in medical patients,12 –14 but lower than in critically ill patients. 15 Active gastroduodenal ulcer, prior bleeding, and low platelet count were found to be the strongest risk factors associated with bleeding whereas advanced age, hepatic or renal failure, and ICU stay were of lesser significance. 11 Of note, the median weight of the study population was 69 kg and less than 50% received chemoprophylaxis during hospitalization, making it difficult to assess whether these same risk factors apply to patients with lower weight receiving chemoprophylaxis.
The primary objective of the study was to describe the incidence of clinically relevant bleeding in low-weight patients receiving prophylactic dose SQH. Secondary objectives included identifying risk factors for bleeding events and describing the prescribing practice of SQH in low-weight patients.
Methods
Study design and patients
This was a single-center, retrospective, nested case–control study conducted in adult patients admitted May 8, 2018 to August 15, 2021 at Mayo Clinic Hospital Rochester, MN. The study was exempt from the Mayo Clinic institutional review board as it used preexisting de-identified data.
Patients were included if they were ⩾ 18 years of age, had a body weight ⩽ 60 kg, received VTE prophylaxis with SQH for ⩾ 48 hours, and were admitted to hospital for ⩾ 72 hours. Patients were excluded if they received therapeutic anticoagulation during admission or any dose of an alternative VTE prophylaxis agent, were pregnant, or declined authorization for use of their medical record for research purposes. Patients who experienced a clinically relevant bleeding event while receiving SQH for VTE prophylaxis (cases) were directly matched to patients without a bleeding event (controls) in a 1:3 ratio based on age (± 5 years), sex, and primary service (medical or surgical). Controls were also required to have at least as much time at risk of bleeding as their matched case. Baseline demographics collected included age at admission, sex, height, weight, body mass index (BMI), primary service (surgical or medical), intensive care unit admission at first SQH dose, and laboratory values (serum creatinine, hemoglobin, and platelet count).
Definitions
Patients with a body weight ⩽ 60 kg were defined as having low weight. Although historically a body weight ⩽ 50 kg has been arbitrarily recognized as a cutoff for underweight patients in previous studies, 10 our study utilized a higher weight in its inclusion criteria. The rational for this was to allow comparison between patients weighing 50–60 kg and < 50 kg to determine if bleeding risk differs between these groups and to increase sample size for risk factor analysis. Prophylactic dose SQH was defined as any SQH dose ⩽ 15,000 units/day. The daily dose was calculated as the total amount of SQH received over a 24-hour period. Standard dose prophylaxis was defined as 15,000 units/day and reduced dose prophylaxis as < 15,000 units/day.
Clinically relevant bleeding cases were defined by a fall in hemoglobin (Hgb) of ⩾ 2 g/dL and transfusion of ⩾ 1 units of packed red blood cells (PRBC) or whole blood within 24 hours or bleeding at a critical site as defined by International Statistical Classification of Diseases and Related Health Problems, 10th Revision (ICD-10) codes (i.e., intracranial, intraspinal, intraocular, pericardial, intra-articular, intramuscular with compartment syndrome, retroperitoneal, thoracic, airway bleeding; online Supplementary Table 1 16 ). Patients meeting the above criteria underwent validation via manual chart review to ensure the event was clinically relevant, which was defined as bleeding resulting in interruption or discontinuation of SQH for ⩾ 24 hours.
Statistical analysis
Baseline demographics were summarized with descriptive statistics including frequencies and percentages for categorical data and either means and SDs or medians and IQRs for continuous data. Risk factors were evaluated for an association with bleeding events on conditional logistic regression. Associations were summarized using odds ratios and 95% CIs. Univariate analyses were conducted to identify exposures with a significant association with bleeding and identify factors for further multivariable analysis. Prespecified variables of interest for univariate analysis included reduced dose SQH, low body weight < 50 kg, chronic kidney disease (ICD-10 codes; online Supplementary Table 2), cirrhosis (ICD-10 codes; online Supplementary Table 2), antiplatelet use, anemia (Hgb < 13 g/dL for men or < 12 g/dL for women at baseline), thrombocytopenia (platelets < 100,000 cells/microliter at baseline), and ICU admission at SQH start.
Results
Patient characteristics
A total of 106,045 patients received a dose of prophylactic SQH during the study period, of which 3761 met the inclusion and exclusion criteria (Table 1 includes demographic data for the 3736 patients that had data available). Among these patients, 38 clinically relevant bleeding cases were identified. Cases were matched directly in a 1:3 ratio resulting in 36 cases and 108 controls (Figure 1). In the case–control cohort, the median age was 67 years old (IQR 52, 77), 77.8% were women, and 47.2% received care from a surgical service (Table 1).
Baseline characteristics.
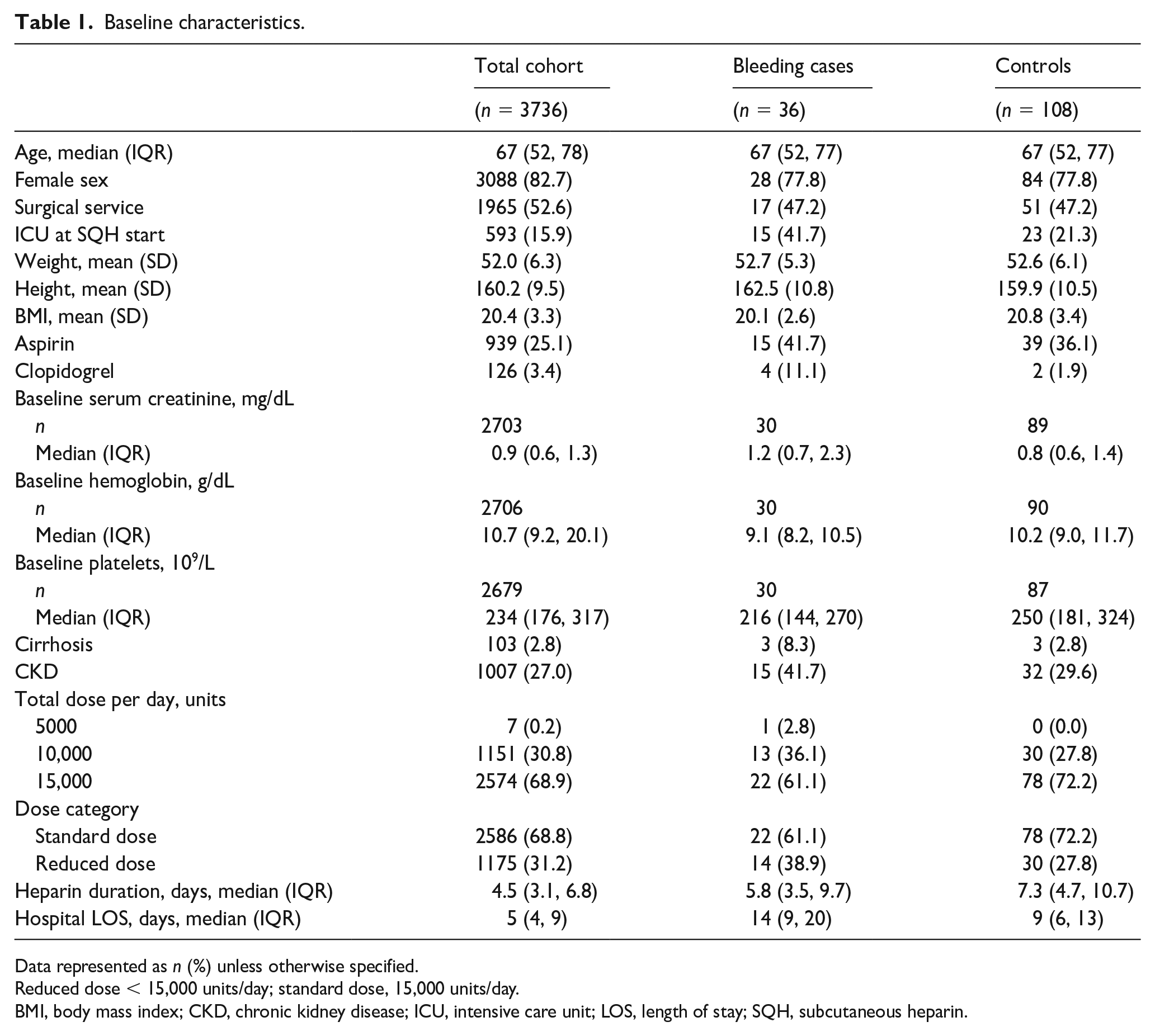
Data represented as n (%) unless otherwise specified.
Reduced dose < 15,000 units/day; standard dose, 15,000 units/day.
BMI, body mass index; CKD, chronic kidney disease; ICU, intensive care unit; LOS, length of stay; SQH, subcutaneous heparin.
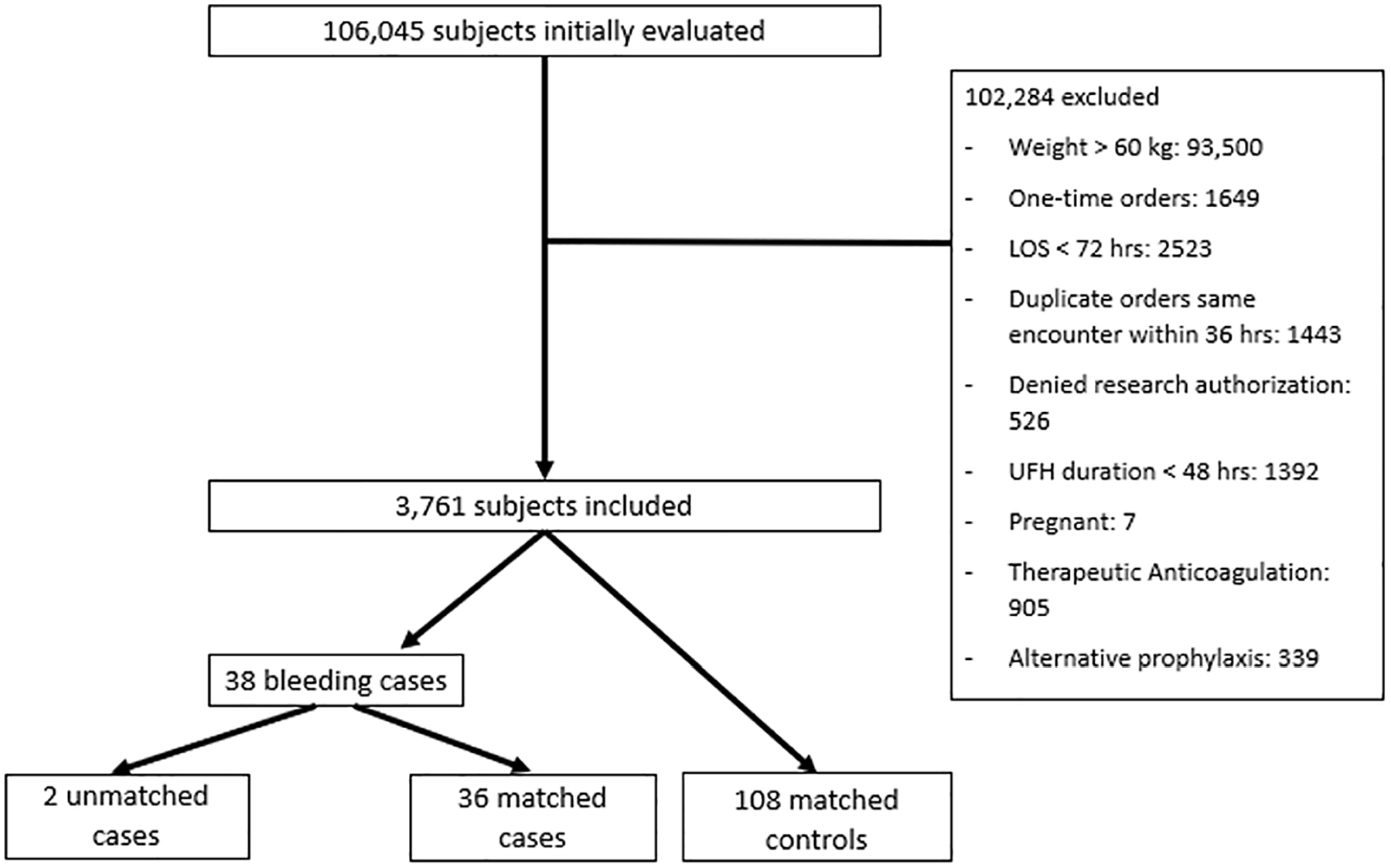
Flowchart of study participants.
Outcomes
The bleeding incidence was calculated from the 3761 subjects meeting inclusion criteria and occurred at a rate of 1% (95% CI 0.6–1.4%) at hospital day 6 when 36.3% of subjects were still followed and 2.8% (95% CI 1.4–4.2%) at hospital day 14 when 6.3% of subjects were still followed (Figure 2). Of the 38 bleeding events, a majority were identified based on a Hgb decrease of ⩾ 2 g/dL and requirement of ⩾ 1 units PRBC (n = 21, 55.3%) with the remainder meeting criteria based on ICD-10 codes (n = 17, 44.7%). The median change in Hgb was a decrease of 2.6 g/dL (IQR-2.3-3). The most common type of bleeding as identified by ICD-10 codes was melena (n = 6) (Table 2).
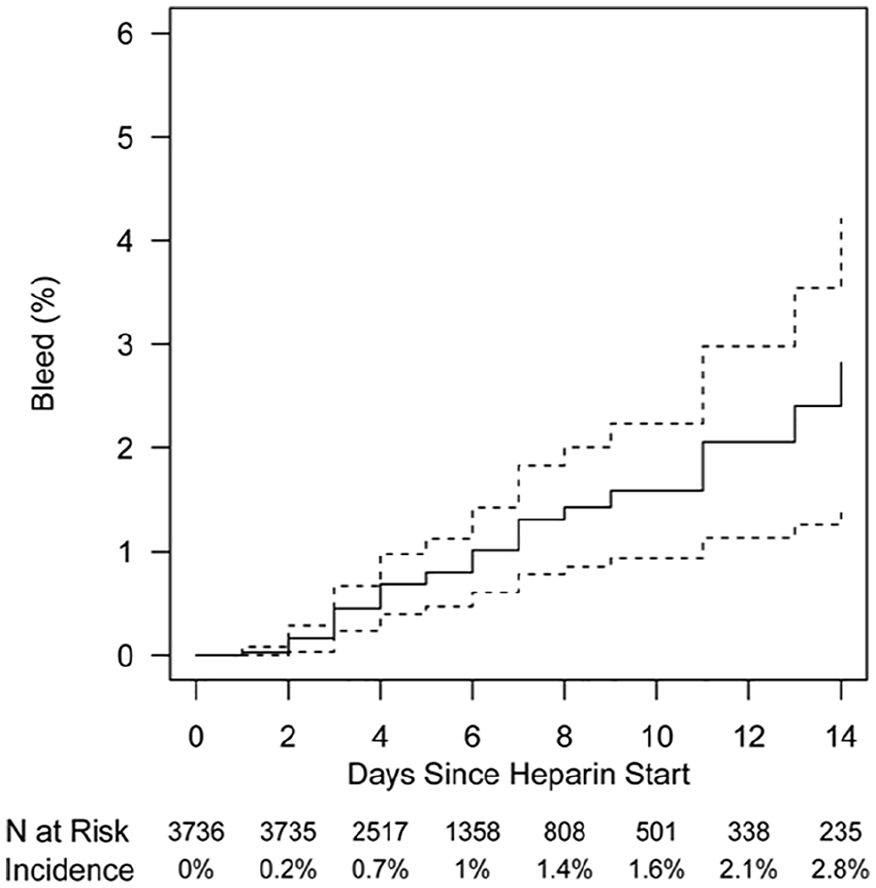
Kaplan–Meier plot showing cumulative bleeding incidence (solid line). Dashed lines represent the 95% CI.
Bleeding characteristics.
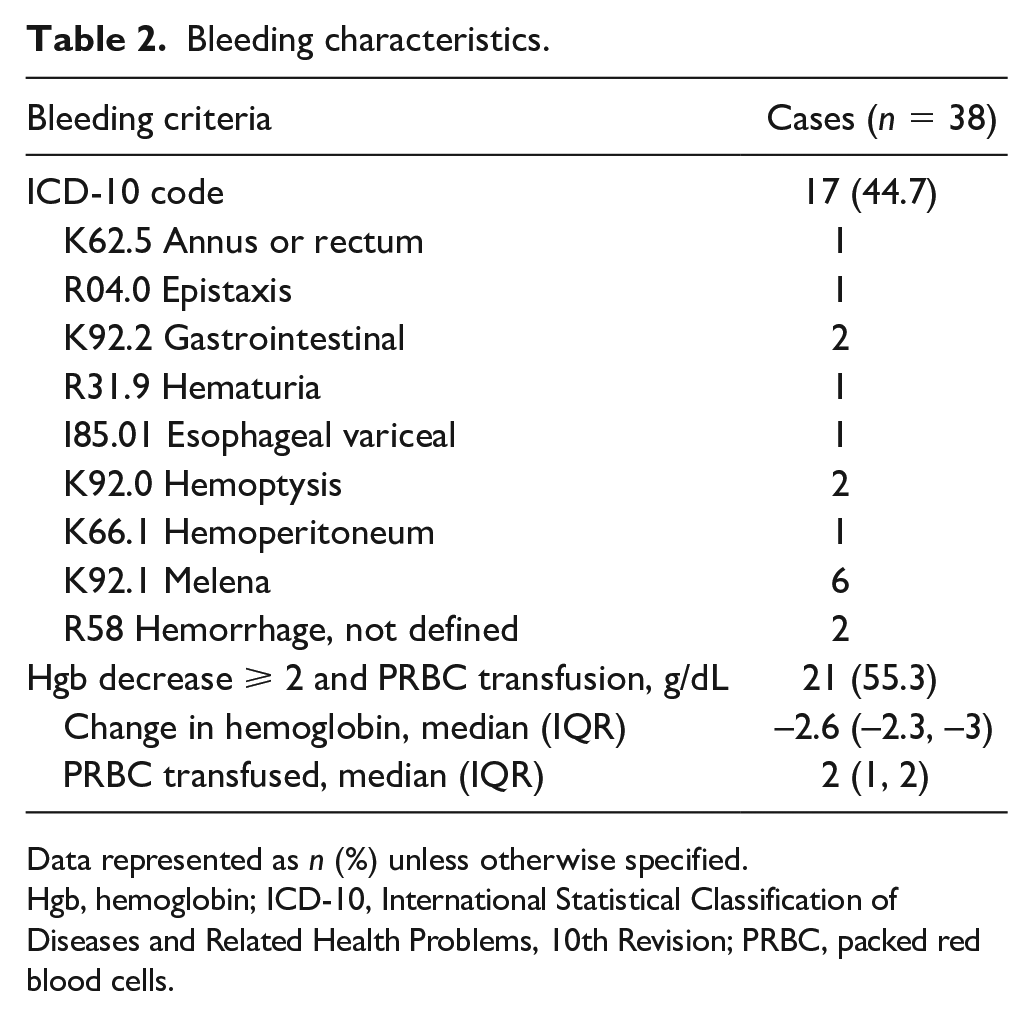
Data represented as n (%) unless otherwise specified.
Hgb, hemoglobin; ICD-10, International Statistical Classification of Diseases and Related Health Problems, 10th Revision; PRBC, packed red blood cells.
ICU admission at time of SQH start was associated with bleeding on univariate analysis, OR 2.97 (95% CI 1.21–7.29). There was a trend towards increased bleeding risk for patients with CKD, cirrhosis, antiplatelet usage, anemia, and thrombocytopenia, although none of these characteristics were found to be statistically significant (Table 3). Given the low bleeding event rate and limited number of statistically significant risk factors on univariate analysis, multivariable analysis was unable to be performed.
Univariate analysis.
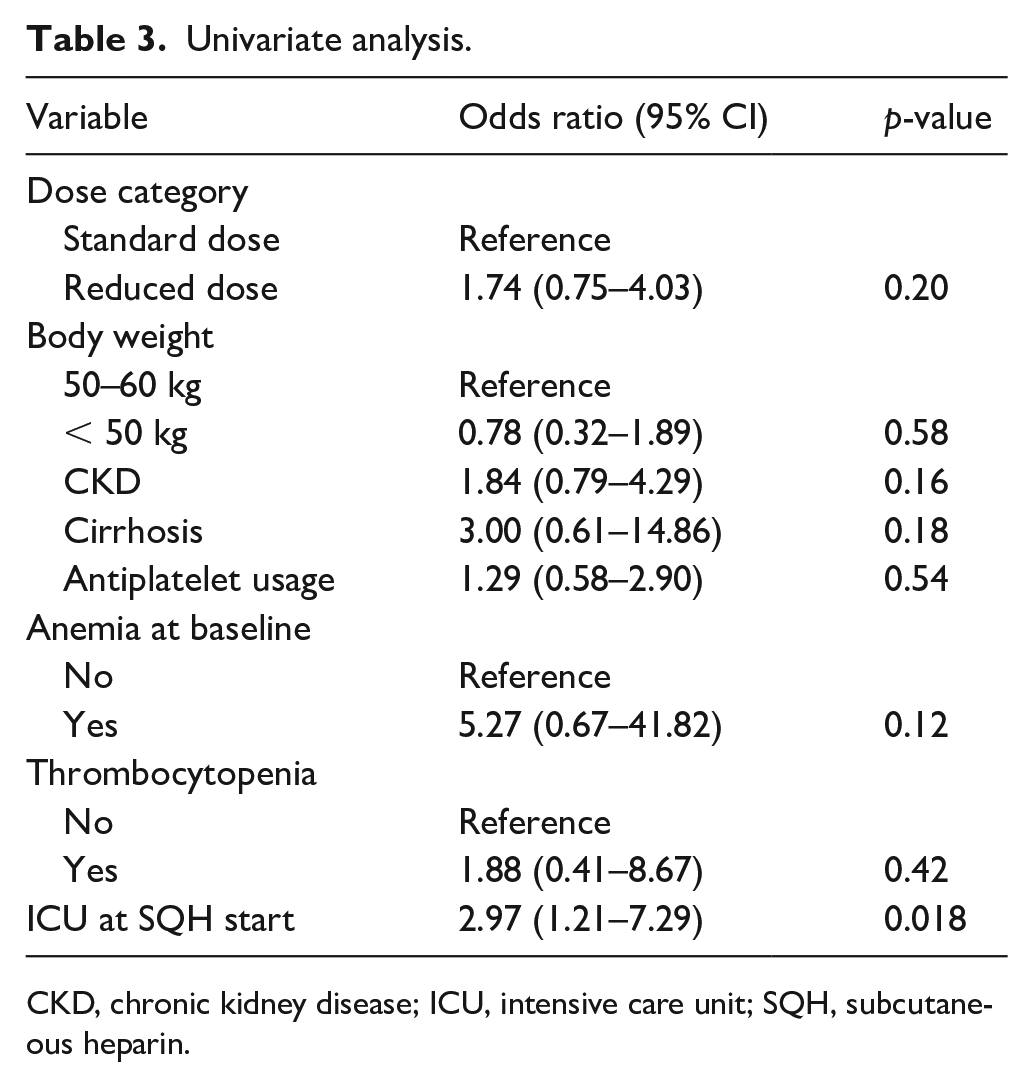
CKD, chronic kidney disease; ICU, intensive care unit; SQH, subcutaneous heparin.
The most prescribed regimen observed in the total cohort was standard dose SQH at 5000 units three times daily (68.8%) followed by reduced dose SQH at 5000 units twice daily (30.9%). Only 0.3% of subjects received a reduced dose of 2500 units twice or three times daily. The reduced dose SQH regimen was used more frequently in patients ⩽ 50 kg compared to > 50 kg at 76.2% versus 8.9% (p < 0.001) (Table 4).
Subcutaneous heparin prescribing practice.
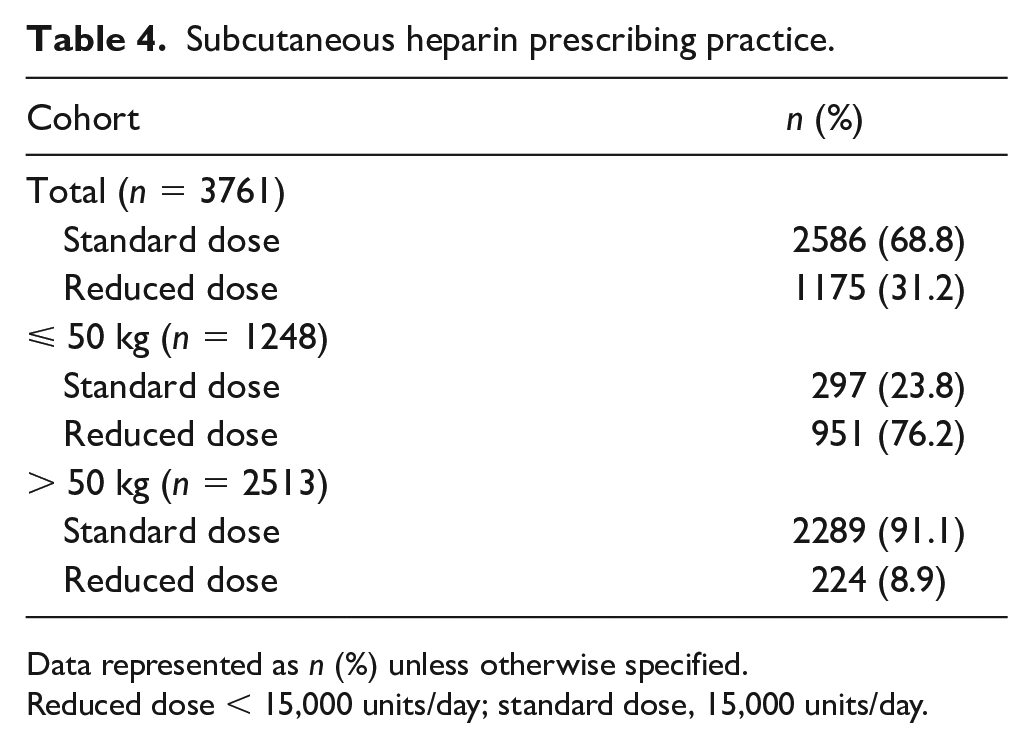
Data represented as n (%) unless otherwise specified.
Reduced dose < 15,000 units/day; standard dose, 15,000 units/day.
Discussion
This study is the first to report the incidence of bleeding in low-weight patients receiving prophylactic dose SQH. We found that at hospital day 6, 1% of patients experienced a clinically relevant bleed, which increased to 2.8% at hospital day 14. In contrast to these findings, two smaller retrospective reviews, the first enrolling medically ill underweight adults (⩽ 45 kg) and the second critically ill underweight adults (BMI ⩽ 18.5 kg/m2 or ⩽ 50 kg) receiving VTE prophylaxis, reported rates of major bleeding or clinically relevant bleeding of 5% and 10.5%, respectively.9,10 A higher major bleeding rate of 5.6% was also demonstrated in the PROTECT trial, which included 3746 critically ill patients that received SQH or dalteparin, although the median BMI in this study was notably higher at 28 kg/m2.13,17 The increased rate of bleeding seen in these studies may be explained by an overall higher severity of illness and greater risk of bleeding at baseline. In contrast, just 16% of our total cohort (593/3761) was admitted to the ICU at time of first SQH dose. Additionally, it is possible that the difference in bleeding definitions and adjudication of events led to a lower rate of events.
The bleeding incidence reported compares similarly to the IMPROVE trial where the incidence of major and nonmajor bleeding at hospital day 14 was 1.2% and 2%, respectively. 11 The IMPROVE trial included patients regardless of receipt of chemoprophylaxis (47.5% received chemoprophylaxis), yet in this study all patients received chemoprophylaxis with SQH. Furthermore, the median weight of patients in the IMPROVE trial was 69 kg compared to the average weight of 52 kg in our study. 11 Our study provides additional context for the safety of SQH in low-weight patients and suggests that the incidence of bleeding is relatively low.
The conditional logistic regression model showed that ICU admission at the start of SQH was statistically associated with bleeding on univariate analysis. This mirrors the increased rate of major bleeding seen in critically ill patients requiring chemoprophylaxis, as previously mentioned.10,15,17 In addition, ICU admission was an independent risk factor associated with bleeding upon multivariable analysis in medically ill patients in the IMPROVE trial. 11 The rationale for increased risk of bleeding in critically ill patients is likely multifactorial due to higher incidence of thrombocytopenia, hepatic and renal dysfunction, and disseminated intravascular coagulation compared to medically ill patients. 18 There was a trend towards increased bleeding risk for patients with CKD, cirrhosis, antiplatelet usage, anemia, and thrombocytopenia at baseline, but the wide confidence intervals and low number of bleeding cases limit the interpretation of this finding. Given that thrombocytopenia, liver failure, and kidney dysfunction were independent risk factors associated with bleeding in the IMPROVE trial, 11 it is possible with a larger sample size a statistically significant association with bleeding may be detected for these variables in low-weight patients. These findings further emphasize the importance of selecting a VTE prophylaxis agent and dose in the context of specific patient clinical factors, particularly for critically ill patients.
Reduced dose SQH (5000 units twice daily or < 15,000 units/day) showed a trend towards increased bleeding compared to standard dose SQH (5000 units three times daily or 15,000 units/day), which contradicts previous findings.9,10 Clinical bias likely led to selection of reduced dose SQH more often in patients at higher risk of bleeding at baseline that was unable to be controlled for on univariate analysis. Overall standard dose SQH was used in 68.8% of the total cohort, which is similar to findings from prior studies assessing VTE prophylaxis in low-weight patients.9,10 Future studies should further assess the dose–response relationship between SQH and bleeding risk to determine the optimal dose in lower weight patient populations.
Study limitations
Despite matching cases and controls at baseline on key characteristics, the retrospective nature of our study is susceptible to unknown confounding, limiting the establishment of a causal relationship between exposures and outcomes. Therefore, results should be interpreted as hypothesis generating. Additionally, this was a single-center experience including only patients receiving SQH. As such, results may not be applicable to other centers and cannot be extrapolated to patients receiving LMWH for VTE prophylaxis. Use of ICD-10 codes and chart review to determine clinically relevant bleeding events is subject to reporting bias, although we adjudicated and confirmed each bleeding event with the requirement that SQH was held for at least 24 hours. Given our low bleeding event rate and number of cases, our ability to detect the association between additional exposures associated with bleeding risk on univariate analysis was limited. This further precluded our ability to test variables on multivariable analysis. Finally, it was outside the scope of our study to assess the incidence and risk factors for VTE in low-weight patients receiving SQH.
Conclusion
This is the first study to report the incidence of clinically relevant bleeding specifically in low-weight patients receiving prophylactic SQH. Study results suggest that patients receiving prophylactic SQH at time of ICU admission may be at higher risk of bleeding. Bleeding incidence was overall low: 1% at hospital day 6 and 2.8% at day 14. Further investigation is needed to improve risk stratification, especially in the ICU population, and to assess the safety and efficacy of reduced dose SQH strategies for those identified to be at the highest risk of bleeding.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231189758 – Supplemental material for Assessment of bleeding risk in low-weight patients receiving prophylactic subcutaneous unfractionated heparin
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231189758 for Assessment of bleeding risk in low-weight patients receiving prophylactic subcutaneous unfractionated heparin by Andrew J Jatis, Scott D Nei, Joseph J Zieminski, Kristin Mara and Abby K Krauter in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
