Abstract
During the past decade, direct oral anticoagulants (DOACs) have advanced and simplified the prevention and treatment of venous thromboembolism (VTE). However, there remains a high incidence of bleeds, which calls for agents that have a reduced risk of bleeding. Factor XI (FXI) deficiency is associated with lower rates of venous thrombosis and stroke compared to the general population with a lower risk of bleeding. In conjunction with this, phase 2 studies have demonstrated safety and the potential for reduced thrombotic events with FXI inhibitors as compared to currently available medications. The aim of this review is to summarize key data on the clinical pharmacology of FXI, the latest developments in clinical trials of FXI inhibitors, and to describe the efficacy and safety profiles of FXI inhibitors for the prevention of venous and arterial thromboembolism.
Keywords
Introduction
Since the advent of oral factor Xa and direct thrombin inhibitors more than a decade ago, these direct oral anticoagulants (DOACs) have advanced significantly in terms of ease of administration and use in venous thromboembolism (VTE) and atrial fibrillation (AF). DOACs have a lower risk of bleeding, especially intracranial bleeding, when compared to vitamin K antagonists (VKAs) in patients with nonvalvular atrial fibrillation (NVAF) for cardioembolic stroke prevention.1,2 Despite this, there are still risks for bleeding in DOACs and an unmet need for safer anticoagulants that are effective with improved safety profiles in terms of major bleeding events. This review briefly discusses the rationale, and current and future roles of factor XI/XIa inhibitors for the prevention of VTE, prevention of and treatment for arterial thromboembolism (ATE), stroke prevention in AF, and other potential therapeutic indications in the complex subgroup of medically ill patients. This review reports the existing data on factor XI (FXI) inhibitors for various clinical conditions and its safety.
The contact pathway as a target for FXI inhibitors
Hemostasis is a process involving platelets, clotting factors, and endothelium at the site of vascular injury to form a blood clot to prevent or decrease the extent of bleeding. 3 The coagulation cascade is stratified into the contact activation (intrinsic) and the tissue factor (extrinsic) pathways which subsequently converge to form the common pathway. 4 The tissue factor pathway is activated by tissue factor expressed in subendothelial tissue which binds with factor VIIa and calcium to convert factor X to factor Xa.3,4 In the contact activation pathway, negatively charged molecules such as dextran sulphate, silica, extracellular nucleic acids, neutrophil extracellular traps, long-chain polyphosphates from infectious pathogens, platelet-derived polyphosphates, and negatively charged artificial surfaces from indwelling medical devices can activate factor XII, which then activates FXI followed by factor IX.5,6 The activated factor VIII, activated factor IX, and calcium ions in turn activate factor X, which leads to the common pathway. 6 The common pathway consists of factor Xa forming a complex with factor Va that cleaves prothrombin (factor II) to thrombin (factor IIa). 6
The contact system, also known as the plasma kallikrein-kinin system, consists of coagulation factors XIIa, XIa, plasma pre-kallikrein along with nonenzymatic cofactor high molecular weight kininogen. They are often called the contact system as they require contact with artificial, negatively charged surfaces for activation. FXI is activated by factor XIIa, which is involved in thrombus initiation. 7 Compared to the mechanism of factor Xa and thrombin, FXI is more specific in its functions where it is involved in clot formation but does not affect hemostasis. In patients with hereditary FXI deficiency, there is a decreased incidence of VTE and cardiovascular events without the complication of spontaneous bleeding.8,9 Moreover, these patients confer a relatively lower risk for bleeding, which mainly occurs in different mucosal surfaces, characterized by high fibrinolytic activity. These findings suggest that FXI plays only a modest role within the coagulation cascade, since FXI do not contribute to initiation but rather thrombi stabilization and expansion.8,9 This led to the development of medications that specifically inhibit FXI and/or XIa, hoping to harness similar thromboembolic benefits without the risk of bleeding. A schematic representation of the mechanism of action of DOACs and different classes of FXI inhibitors is displayed in Figure 1. 10
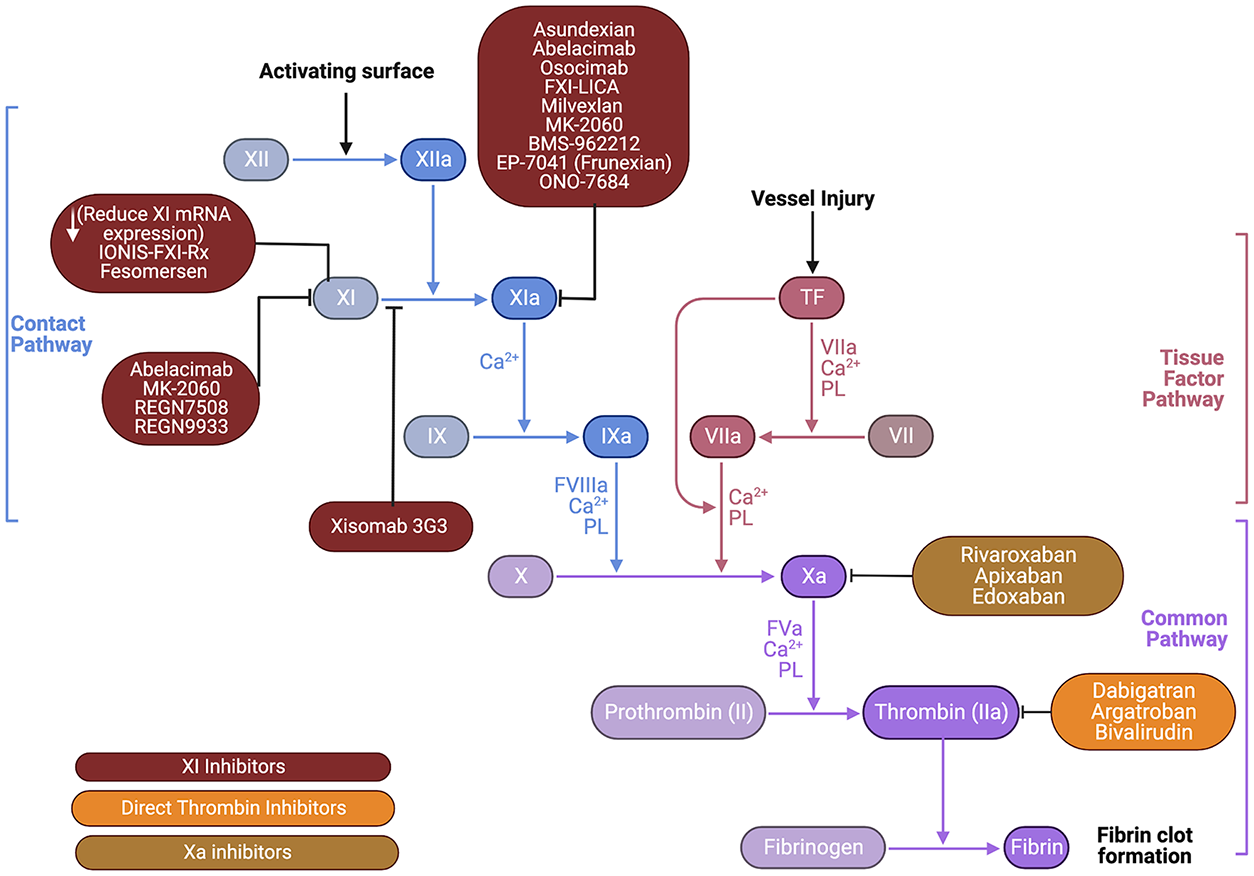
Mechanism of action of common anticoagulants versus factor XI inhibitors.
Development of novel agents targeting FXI
There are several novel agents that are directed against FXI, which include monoclonal antibodies, small molecules, natural inhibitors, antisense oligonucleotides (ASOs), and aptamers.10,11 Natural inhibitors and aptamers have not been discussed in this article as they have not been tested in humans. Pharmacologic properties of different classes of FXI inhibitors are displayed in Table 1. Table S1 in supplementary material summarizes ongoing and completed clinical trials studying Factor XI inhibitors.
Pharmacological properties of novel anticoagulants targeting factor XI and factor XIa.
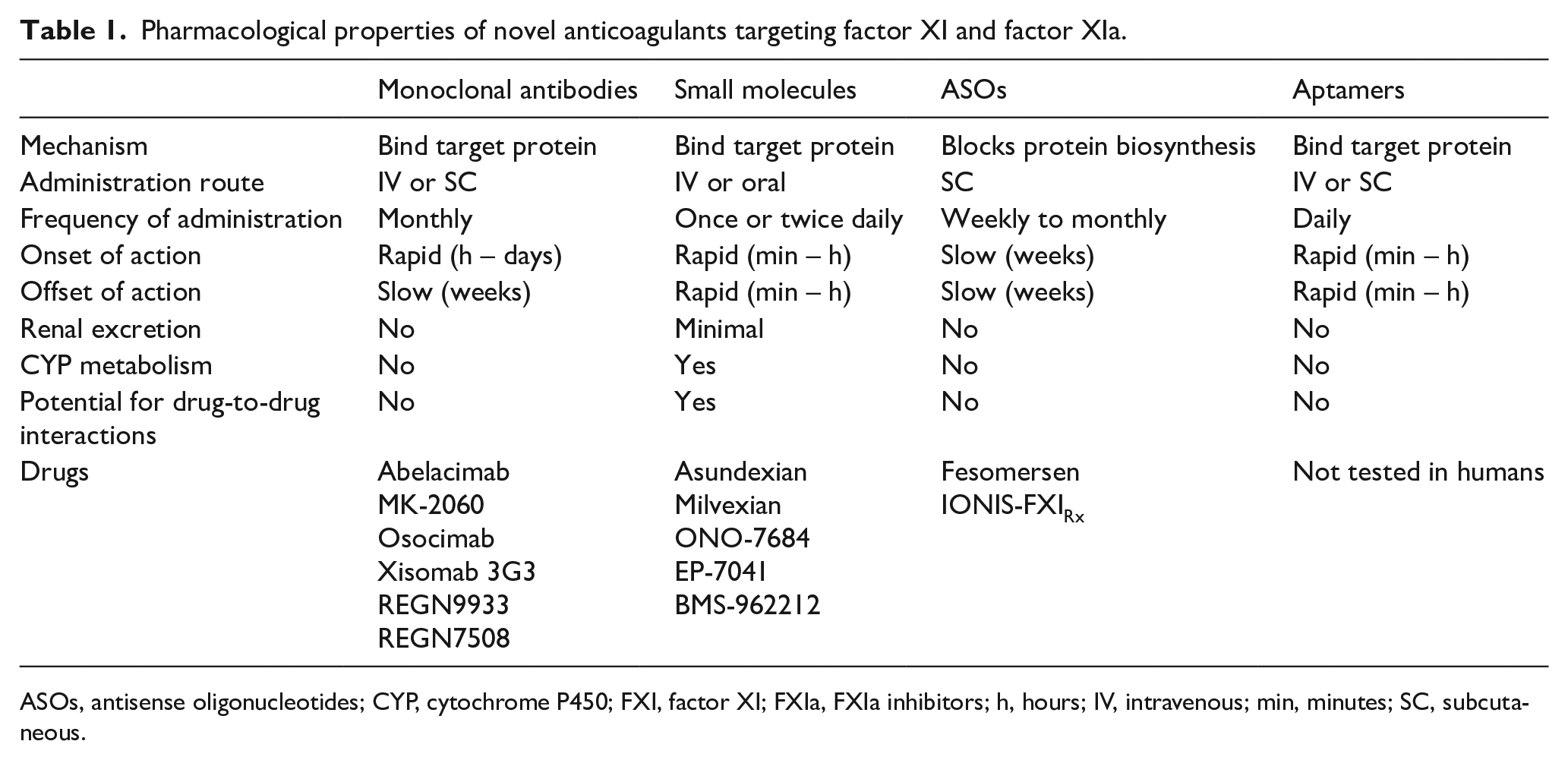
ASOs, antisense oligonucleotides; CYP, cytochrome P450; FXI, factor XI; FXIa, FXIa inhibitors; h, hours; IV, intravenous; min, minutes; SC, subcutaneous.
Methods
We performed a search strategy for clinical trials from www.clinicaltrials.gov from inception to February 1, 2023 using the following key terms: “factor XI inhibitors, thrombosis”. We searched for other clinical trials, observational studies, retrospective studies, and prospective studies and reviews using MEDLINE/PubMed, EMBASE, Cochrane Library, and Web of Science database using the following MeSH terms: “factor XI inhibitors AND thrombosis AND thromboembolism”. The data obtained from this search were further screened based on clinical relevance pertaining to this article, which included medically ill patients (e.g., VTE, AF). The titles and abstracts of the studies were analyzed to evaluate relevance for this review. There were no strict eligibility criteria other than English written articles relevant to the topic and clinical trials evaluating FXI inhibitors.
Factor XI/XIa inhibitors
Antisense oligonucleotides (ASOs)
Fesomersen
Fesomersen, also known as BAY2976217 or FXI-LICA, is an antisense oligonucleotide inhibitor of FXI which facilitates receptor-mediated hepatic uptake and decreased circulating FXI. 12 One of the major advantages of fesomersen is its property of having triantennary N-acetyl galactosamine (GalNAc) conjugation, which allows for maximum drug delivery to hepatocytes where FXI synthesis occurs. This conjugation also allows for once-a-month dosing when compared to other ASOs having once-a-week dosing. 12 Given that this medication has no renal clearance, the RE-THINc ESRD (ClinicalTrials.gov identifier: NCT04534114) trial studied the safety and tolerability of fesomersen in end-stage renal disease (ESRD) patients requiring hemodialysis (HD). The goal of the study was to learn more about the safety of BAY2976217, how it is tolerated and the way the body absorbs, distributes, and eliminates the study drug given as multiple doses in participants with renal impairment who require HD. The primary outcome for this study was incidence of major bleeding and clinically relevant nonmajor bleeding (CRNMB) events during the main treatment period and within the on-treatment time window (up to 24 weeks) assessed by the blinded Central Independent Adjudication Committee (CIAC) where fesomersen is being compared against matching placebo.
IONIS-FXIRx
IONIS-FXIRx (BAY2306001 or ISIS 416858) is an antisense oligonucleotide inhibitor of the synthesis of FXI in the liver, which shows a reduction in FXI activity. 12 IONIS-FXIRx is an unconjugated ASO which results in weekly dosing when compared to fesomersen, as discussed earlier. 12 In a phase 2 study in ESRD patients on HD, a significant, dose-dependent, sustained reduction in FXI antigen and activity were noted. There were no CRNMB events in the study and no major bleeding events occurred in the group that received the 200 mg dose but there was only one major bleeding event in the group that received the 300 mg dose. 13 Another trial studying the effects of ISIS 416858 on the incidence of thromboembolism in patients undergoing total knee arthroplasty where two doses were given preoperatively and one dose postoperatively showed noninferior results in the 200 mg group (27% rate) and superior results in the 300 mg group (4%) when compared to enoxaparin (30% rate). Bleeding events were numerically higher in the 300 mg group (8%) and equivalent in the 200 mg group and enoxaparin group (3% for both). 14 Currently, the Study of ISIS 416858 Administered Subcutaneously to Participants with End-Stage Renal Disease (ESRD) on Hemodialysis (EMERALD) (NCT03358030) trial is studying the pharmacokinetics and pharmacodynamics of ISIS 416858 in patients with ESRD on HD. The primary outcome is number of participants with major bleeding and CRNMB.
Monoclonal antibodies
Abelacimab/MAA868
Abelacimab is a monoclonal antibody that binds to the catalytic domain of FXI and prevents its activation by factor XIIa and thrombin. 15 In the ANT-003 study, a single intravenous dose of 30, 50, and 150 mg abelacimab in healthy subjects was safe and well tolerated without any major bleeding or CRNMB events. 16 In the ANT-004 study, patients with AF were administered monthly subcutaneous (SC) injections of abelacimab, which led to a sustained reduction in free FXI concentrations when compared to placebo. No major bleeding or CRNMB events were noted in the ANT-004 study. The trial was not designed to assess clinical endpoints (i.e., the incidence of stroke and systemic embolism events in patients with NVAF). 16 The ANT-005 trial revealed that for the efficacy outcome of preventing VTE, the single 30 mg dose of abelacimab is noninferior to enoxaparin in patients undergoing total knee arthroplasty, whereas the single 75 mg and 150 mg doses of abelacimab were shown to be superior to enoxaparin. 17 The rates of major bleeding and CRNMB events were low in all arms of the study. 17 In the ongoing clinical trial AZALEA-TIMI 71 (NCT04755283), major and CRNMB events will be compared to rivaroxaban in patients with NVAF. The clinical trial ASTER (NCT05171049) compares abelacimab to apixaban in patients with cancer-associated VTE, and the clinical trial MAGNOLIA (NCT05171075) compares abelacimab against dalteparin for the prevention of VTE recurrence in gastrointestinal or genitourinary cancers associated with VTE. The ongoing LILAC-TIMI 76 (NCT05712200) trial studies the time to ischemic strokes, systemic embolisms, VTE, myocardial infarction (MI), and acute limb ischemia in patients with diagnosed AF or atrial flutter.
MK-2060
MK-2060 is a monoclonal antibody against FXI currently being studied in ESRD patients on HD to assess for arteriovenous graft (AVG) thrombosis prevention and bleeding events (NCT05027074). The primary endpoint is time to first AVG thrombosis event.
Osocimab
Osocimab, also known as BAY1213790, is a monoclonal antibody involved in the allosteric inhibition of FXIa. 18 The FOXTROT trial studied pre- and postoperative thromboprophylaxis using osocimab in patients undergoing knee arthroplasty. In that trial, osocimab was noninferior to enoxaparin in the postoperative phase and superiority in the preoperative phase with regards to prevention of VTE events. 19 A recent meta-analysis showed a significant reduction of VTE events and bleeding events in patients undergoing orthopedic surgery. 20 The ongoing CONVERT (NCT04523220) trial will assess the safety and tolerability of low and high-dose osocimab in patients with ESRD undergoing HD. Primary outcome measures are the composite of major and clinically relevant nonmajor bleeding events as assessed by blinded CIAC (time frame: from the first dose at month 1 and up to 6 months) and the composite of moderate and severe adverse events (AEs) and serious adverse events (SAEs) (time frame: from the first dose at month 1 and up to 6 months).
Xisomab 3G3
Xisomab 3G3, also known as AB023 or Gruticibart, is a monoclonal antibody that binds to FXI and prevents its activation by factor XIIa. 21 A single dose of AB023 reduced intradialyzer clotting during heparin-free HD in ESRD. 22 There is another ongoing clinical trial (NCT04465760) regarding the efficacy of xisomab at preventing catheter-associated thrombosis in individuals with a central venous catheter.
REGN9933
REGN9933 is a monoclonal antibody against FXI which is currently undergoing phase 1 clinical trials to assess the pharmacokinetics and pharmacodynamics in healthy subjects (NCT05102136). 23
REGN7508
REGN7508 is another monoclonal antibody against FXI which is undergoing phase 1 clinical trials to assess the safety, tolerability, pharmacokinetics, and pharmacodynamics in healthy subjects (NCT05603195).
Small molecules
Asundexian/BAY 2433334
Asundexian is a small molecule inhibitor of FXIa activity which produces a direct, reversible inhibition of FXIa activity. 24 Asundexian is primarily eliminated via liver metabolism but does not involve the cytochrome p450 system. 24 The PACIFIC-AF (NCT04218266) trial showed that asundexian at 20 mg and 50 mg once-daily doses led to similar suppression of FXIa with lower rates of bleeding as compared to apixaban. 25 The PACIFIC-STROKE (NCT04304508) trial concluded that there was no increase in major or CRNMB events when asundexian was compared to placebo. Recognizing that the PACIFIC-STROKE trial was not powered for thrombotic outcome comparison, there was no difference in the recurrence of stroke or covert brain infarctions seen in the asundexian and placebo arms. 26 A secondary analysis of the PACIFIC-STROKE trial revealed a reduction of ischemic stroke/TIA in the asundexian 50 mg arm when compared to placebo. 27 A third clinical trial, known as the PACIFIC-AMI (NCT04304534), studied the secondary prevention of MI, finding that when asundexian was added to aspirin and a P2Y12 inhibitor, there was a dose-dependent inhibition of FXIa activity without a significant increase in bleeding rates and a decreased rate of ischemic events. 28 The ongoing OCEANIC-AF trial (NCT05643573) compares the efficacy of asundexian when compared to apixaban for stroke prevention in AF. The ongoing OCEANIC-STROKE trial (NCT05686070) assesses the time to occurrence of a stroke in patients who recently had an ischemic stroke or high-risk transient ischemic attack (TIA).
Milvexian
Milvexian, also known as BMS-986177, is a small molecule reversible inhibitor with high affinity for activated FXI.29,30 Milvexian is a cytochrome p450 inhibitor with hepatic metabolism which may interfere with other drugs that interact with cytochrome p450.7,29 In the recently completed AXIOMATIC-SSP phase 2 trial, participants were randomized to milvexian or placebo along with aspirin and clopidogrel following an acute ischemic stroke or high-risk TIA. 31 The primary efficacy outcome, symptomatic ischemic stroke, and the outcome of covert brain infarction at 90 days were not different between milvexian and placebo. Of note, no increase in bleeding was noted with milvexian. 32 The primary efficacy endpoint was numerically lower at the 50 mg and 100 mg twice-daily doses; there was no apparent dose–response (placebo, 16.6%; 25 mg once daily, 16.2%; 25 mg twice daily, 18.5%; 50 mg twice daily, 14.1%; 100 mg twice daily, 14.7%; 200 mg twice daily, 16.4%).
In the AXIOMATIC-TKR trial of patients undergoing knee arthroplasty surgery, the 25 mg (25%) and 50 mg (24%) oral milvexian groups were noted to have similar incidence rates of VTE when compared to the enoxaparin group (21%). However, the 200 mg oral milvexian group was noted to have decreased VTE incidence rates (7%). The bleeding rates of any severity are 4% in both milvexian and enoxaparin groups. 33
The ongoing LIBREXIA-STROKE (NCT05702034) trial primarily studies stroke prevention after an acute ischemic stroke or high-risk TIA. The ongoing LIBREXIA-ACS (NCT05754957) studies major adverse cardiovascular events in patients who experienced acute coronary syndrome in the past 7 days, with the trial comparing milvexian versus placebo in addition to standard of care antiplatelet therapy (either single or dual, per investigator discretion).
ONO-7684
ONO-7684 is an oral small molecule FXIa inhibitor with competitive and reversible inhibition. 34 In the first human, double-blind, clinical trial in healthy volunteers, ONO-7684 was well tolerated at all doses and in repeated doses without any evidence of bleeding risk. 34
EP-7041
EP-7041 is an intravenous small molecule FXIa inhibitor with a short half-life of 45 minutes, which may not necessitate the need for a reversal agent. 35 EP-7041, also known as frunexian, was found to be safe and well tolerated at all doses with minimal bleeding risk (infusion site bleeding) and mild headaches (NCT02914353). 36 A prospective study of frunexian in the thromboprophylaxis of COVID-19 patients is currently being conducted, which included two different doses of EP-7041 (NCT05040776).
BMS-962212
BMS-962212 is an intravenous, selective, reversible, active-site inhibitor of FXIa which produced significant antithrombotic activity with minimal effects on bleeding risk. 37 The first in-human study revealed a quick onset of action, short half-life, and was found to be well tolerated by all subjects with no signs of bleeding (NCT03197779). 38
Clinical applications of FXI inhibitors
Atrial fibrillation
The PACIFIC-AF trial was a randomized dose-finding trial for asundexian in patients with AF where it was found that asundexian dosed once a day at 20 mg and 50 mg led to 81% and 92% reduction percentages of FXIa at trough, respectively. 25 Overall, bleeding rates were lower in the asundexian group compared to the apixaban group. 25 The rate of adverse effects was similar in all treatment arms, whereas thrombotic endpoints such as cardiovascular death, MI, systemic embolism, and ischemic stroke were the least in the asundexian 20 mg arm. 25 It has less than 15% renal elimination, making it relatively safe in chronic kidney disease (CKD), although more trials regarding its safety in CKD patients are required. Abelacimab is also currently being studied for its use in AF, but results are currently not available.
VTE prevention and treatment
Abelacimab is currently undergoing several clinical trials in VTE prevention/treatment for which results have not been published yet. However, the ANT-005 trial studied the incidence of VTE postoperatively in patients undergoing total knee arthroplasty with a single intravenous dose of abelacimab (30 mg, 75 mg, or 150 mg) versus enoxaparin 40 mg SC daily. The primary efficacy outcome being VTE incidence was 13%, 5%, 4%, and 22% in the groups for abelacimab 30 mg, 75 mg, 150 mg, and enoxaparin 40 mg, respectively. Bleeding occurred in 2%, 2%, none, and none of the patients in abelacimab 30 mg, 75 mg, and 150 mg groups, and enoxaparin 40 mg, respectively. 17 Abelacimab also has zero renal clearance and would be a safe option in patients with CKD. 16
IONIS-FXIRx (also known as FXI-ASO) was studied in patients undergoing total knee arthroplasty where they received either 200 mg or 300 mg FXI-ASO or enoxaparin 40 mg SC daily. It was found that the primary efficacy outcome which was VTE incidence was found to be 27%, 4%, and 30% in the FXI-ASO 200 mg, 300 mg, and enoxaparin 40 mg groups, respectively. The 300 mg FXI-ASO group was also found to be superior, with p < 0.001, but the 200 mg group was noninferior. Bleeding occurred in 3%, 3%, and 8% in the FXI-ASO 200 mg, 300 mg, and enoxaparin 40 mg groups, respectively. 14 Patients with creatinine clearance < 60 mL/min should be avoided as evidenced in a clinical trial, 14 but it appears to be relatively safe in patients undergoing HD without any accumulation. 13
Milvexian was studied in patients undergoing elective unilateral total knee arthroplasty where they received one of the following postoperative regimens of milvexian (25 mg, 50 mg, 100 mg, or 200 mg twice daily or 25 mg, 50 mg, or 200 mg once daily) or enoxaparin (40 mg once daily). Patients with creatinine clearance < 30 mL/min, history of severe hepatic impairment, prior history of VTE, and use of long-term anticoagulation except for aspirin were excluded. There was a VTE incidence of 25% in milvexian 25 mg twice daily, 11% in 50 mg twice daily, 9% in 100 mg twice daily, and 8% in 200 mg twice daily, whereas it was 25% in 25 mg once daily, 24% in 50 mg once daily, and 7% in 200 mg once daily compared to 21% in enoxaparin 40 mg once daily. There was a similar bleeding risk of 4% in milvexian and enoxaparin. 33
The FOXTROT trial for osocimab studied patients undergoing knee arthroplasty where single intravenous osocimab postoperative doses of 0.3 mg/kg, 0.6 mg/kg, 1.2 mg/kg, or 1.8 mg/kg; preoperative doses of 0.3 mg/kg or 1.8 mg/kg; or 40 mg of SC enoxaparin once daily or 2.5 mg of oral apixaban twice daily were administered. There was a VTE incidence of 23.7% in groups who received 0.3 mg/kg, 15.7% in 0.6 mg/kg, 16.5% in 1.2 mg/kg, and 17.9% in 1.8 mg/kg postoperative doses of osocimab. There was a VTE incidence of 29.9% in 0.3 mg/kg and 11.3% in 1.8 mg/kg preoperative doses of osocimab when compared to 26.3% VTE incidence in the enoxaparin groups and 14.5% in the apixaban groups. 19 Major or clinically relevant nonmajor bleeding was seen in up to 4.7% in the osocimab group, 5.9% in the enoxaparin group, and 2% in the apixaban group. 19
Stroke
The PACIFIC-STROKE trial studied asundexian in patients with acute (< 48 h) noncardioembolic ischemic stroke where the primary efficacy outcome was the composite of incident magnetic resonance imaging (MRI)-detected covert brain infarcts and recurrent symptomatic ischemic stroke. The primary efficacy outcome was seen in 19% in the placebo group, 19% in the asundexian 10 mg group, 22% in the asundexian 20 mg group, and 20% in the asundexian 50 mg group. Major bleeding or CRNMB was noted in 2% of the placebo group, 4% of the asundexian 10 mg group, 3% of the asundexian 20 mg group, and 4% of the asundexian 50 mg group. 39 Several other trials, such as the OCEANIC-STROKE trial, AXIOMATIC-SSP trial, AZALEA-TIMI 71 trial, and LIBREXIA-STROKE trial are currently in progress.
Myocardial infarction
The PACIFIC-AMI trial studied oral asundexian versus placebo in patients with a recent MI on dual antiplatelet therapy (aspirin and P2Y12 inhibitor). The efficacy outcome was a composite of cardiovascular death, MI, stroke, or stent thrombosis. The efficacy outcome occurred in 6.8%, 6.0%, 5.5%, and 5.5% of patients in groups of asundexian 10 mg, 20 mg, or 50 mg, and placebo, respectively. According to the main safety outcome, which was Bleeding Academic Research Consortium types 2, 3, or 5, bleeding occurred in 7.6%, 8.1%, 10.5%, and 9.0% of patients receiving asundexian 10 mg, 20 mg, or 50 mg, and placebo, respectively. This shows the efficacy outcomes among the asundexian and placebo groups, whereas the bleeding outcomes were the least in asundexian 10 mg. 28 Online Supplemental Table S1 summarizes ongoing and completed clinical trials studying FXI inhibitors.
Future perspectives for FXI/XIa inhibitors
Despite promising early phase studies of FXI inhibitors on cardioembolic stroke prevention in NVAF, prevention of VTE, and other thromboembolic conditions, asundexian is the only molecule studied alongside dual antiplatelet therapies to prevent cardiovascular death, MI, ischemic stroke, and stent thrombosis. Additional studies on FXI inhibitors need to be conducted to assess their role in the prevention of stent thrombosis and cardiovascular outcomes in patients with coronary artery disease.
Though certain thrombotic conditions are common and have strong evidence supporting the anticoagulant therapies (e.g., AF, VTE), others are less common or have less evidence to support current antithrombotic therapies. These include acute limb ischemia without a cardioembolic source and ESRD. Finally, other clinical scenarios have a clear thrombotic link, but currently available therapies have not provided an optimal risk–benefit balance. The hope for factor XI and XIa inhibitor agents is that they can provide an improved risk–benefit balance that can be applied both in situations without currently available antithrombotic therapies (e.g., ESRD) and in those where prior therapies have led to excessive bleeding (e.g., acute MI, embolic stroke of unknown source).
Other potential areas of research and exploration may include problematic, challenging clinical scenarios in which DOACs have been proven ineffective. These include so called ‘artificial contact surfaces-associated thrombosis’ (ACSAT), such as mechanical valves, extracorporeal membrane oxygenation (ECMO), left ventricular assisting devices (LVADs), and central venous catheter-associated thrombosis.40–43 Dabigatran posed an increased risk for stroke and bleeding complications in patients with mechanical heart valves. 44 Current guidelines recommend against the use of DOACs (favoring VKAs), specifically in patients with mechanical valves. 45
The American Society of Hematology (ASH) and the International Society of Thrombosis and Haemostasis (ISTH) guidelines recommend against using DOACs in patients with antiphospholipid syndrome (APS), particularly patients with high-risk APS who carry a triple positivity for lupus anticoagulant, anticardiolipins, and anti-beta-2 glycoprotein-1 antibodies.46–48 A recent systematic review and meta-analysis of four randomized controlled trials involving 472 patients with APS that compared DOACs with VKAs, published by Khairani et al., demonstrated that the use of DOACs compared with VKAs was associated with increased odds of subsequent arterial thrombotic events (odds ratio [OR]: 5.43; 95% CI: 1.87–15.75; p < 0.001), particularly ischemic stroke, and the composite of ATE or VTE (OR: 4.46; 95% CI: 1.12–17.84; p = 0.03); investigators concluded that patients with thrombotic APS randomized to DOACs compared with VKAs appear to have increased risk for ATE. 49 In APS, one of the mechanisms involves the reduction of inactivation of FXI by antithrombin, which may pave the way for the use of FXI inhibitors in APS. 50
Several different agents targeting factor XI and XIa are in various stages of development. Two of the oral agents targeting factor XIa are currently in phase 3 clinical trials (milvexian and asundexian). Others that use different routes of administration (e.g., SC injection) and those that target FXI in addition to factor XIa are currently being developed (e.g., abelacimab).
Reversal of FXI inhibitor drugs remains another area where research may be required. Although FXI is a component of fresh frozen plasma (FFP), FFP also contains other clotting factors, along with physiologic anticoagulants, making it a diluted blood product to administer to individuals who are bleeding. A large quantity of FFP may also be required to reverse FXI inhibitors for this reason. FXI concentrates is another option to reverse FXI inhibitor bleeding complications. 51 Management of bleeding in FXI deficiency includes administration of FFP, FXI concentrate, recombinant factor VIIa (rFVIIa), tranexamic acid, or epsilon-aminocaproic acid. 52 Because FXIa inhibitors can potentially impair thrombin activatable fibrinolysis inhibitor (TAFI) activation, tranexamic acid represents a cornerstone reversal agent for the prevention and treatment to control bleeding, and it should be given in patients undergoing urgent surgery or surgical interventions with a high risk for major bleeding. 53 The drawback of administering these products includes the risk of thrombosis, except in FFP. 52 Despite having blood products to reverse FXI inhibitors, this may not be enough to reverse all bleeding complications induced by FXI inhibitors, especially in the agents with long half-lives, which necessitate the development and further research of specific reversal agents. 53 Any agent that inhibits the coagulation cascade has the potential to increase bleeding risk. Although FXI inhibitors are known to have fewer bleeding complications due to the mechanism of action, there are limited data to suggest the complete absence of bleeding complications. Furthermore, clinician behavior is such that the use of an anticoagulant at the time of bleeding may lead to a strong desire to eliminate the anticoagulant as a potential source/facilitator of bleeding.
Conclusions
FXI/XIa inhibitors have shown great promise in improving the thrombosis/bleeding balance. Given the tremendous impetus and enthusiasm among thrombosis clinicians/researchers focusing on this fascinating arena, further confirmation of their efficacy and safety is highly anticipated in ongoing, large, phase 3, randomized controlled clinical trials. It is certainly not unreasonable that multiple medical specialties including hematologists, cardiologists, and vascular medicine specialists will incorporate the frequent use of FXI/XIa inhibitors into their daily clinical practice. Though many hope that FXI/XIa inhibitors become the safer and ideal anticoagulant of choice in many clinical scenarios, we must wait for the high-quality, phase 3 randomized trial data to better understand if those hopes will be reflected.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231206778 – Supplemental material for Factor XI/XIa inhibitors for the prevention and treatment of venous and arterial thromboembolism: A narrative review
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231206778 for Factor XI/XIa inhibitors for the prevention and treatment of venous and arterial thromboembolism: A narrative review by Swathi Prakash, Adriana C Mares, Mateo Porres-Aguilar, Debabrata Mukherjee and Geoffrey D Barnes in Vascular Medicine
Footnotes
Declaration of conflicting interests
Dr. Barnes serves on the Board of Directors for the Anticoagulation Forum and as a consultant for Pfizer, Bristol-Myers Squibb, Janssen, Bayer, AstraZeneca, Sanofi, Anthos, Abbott Vascular, and Boston Scientific. The other authors have no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
