Abstract
Introduction:
Direct oral anticoagulants (DOACs) are associated with bleeding. Patients often stop taking DOACs due to nonmajor bleeding, which may lead to venous thromboembolism (VTE) recurrence. We aimed to determine the risk of nonmajor bleeding using different DOACs to prevent and treat VTE.
Methods:
PubMed, Embase, Web of Science, and Cochrane Library databases were searched from inception until January 6, 2021. The incidence of clinically relevant nonmajor bleeding and minor bleeding was investigated. In frequentist-based network meta-analysis, we analyzed the odds ratio (OR) with 95% CI and the surface under the cumulative ranking curves (SUCRA).
Results:
Twenty-seven randomized controlled trials (RCTs) (involving 64,493 patients) were included. For preventing VTE, the risk for clinically relevant nonmajor bleeding was lowest for apixaban, followed by that for low-molecular weight heparin (LMWH), dabigatran, edoxaban, and rivaroxaban. The risk for minor bleeding was lowest for apixaban, followed by that for rivaroxaban, LMWH, dabigatran, and edoxaban. For treating VTE, the risk for clinically relevant nonmajor bleeding was also lowest for apixaban, followed by that for edoxaban, vitamin K antagonists (VKAs), and rivaroxaban. The risk for minor bleeding was lowest for apixaban, followed by that for rivaroxaban and VKAs.
Conclusions:
Regardless of whether it was used for preventing or treating VTE, apixaban had the lowest risk of nonmajor bleeding. This suggests that apixaban may have a lower risk of nonmajor bleeding than other anticoagulants and may help provide some clinical reference for choosing a more appropriate drug for the patient.
Introduction
Direct oral anticoagulants (DOACs) have been developed for venous thromboembolism (VTE) in the past 20 years. 1 DOACs are not only convenient to take and have low complexity in treatment management and few drug interactions, but also have predictable pharmacokinetic characteristics; therefore, there is no need for routine anticoagulation monitoring. 2 Clinically relevant nonmajor bleeding and minor bleeding involve hematuria, nose bleeding, gum bleeding, and bruising. 3 Nonmajor bleeding also has a significant impact on the patient’s quality of life. Statistically, there is 11.81% nonmajor bleeding in DOACs for VTE prevention and treatment.4–10 At the same time, as anticoagulation is a long-term treatment, doctors and patients should be aware of potential bleeding adverse effects. Even nonmajor bleeding may become a reason patients do not adhere to treatment, and treatment nonadherence may lead to VTE recurrence. Therefore, there should be focus on the risk of nonmajor bleeding with different DOACs for VTE treatment and prevention.
We conducted a systematic review and a network meta-analysis (NMA) to separately conduct a comprehensive analysis of clinically relevant nonmajor bleeding and minor bleeding that occurred as a result of DOACs being used for preventing and treating VTE. We ranked the bleeding incidence of different DOACs and screened the DOACs with lower incidences of bleeding for clinical applications.
Methods
We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses for NMA (PRISMA-NMA) extension guideline and checklist for reporting systematic reviews and NMA. 11
Data sources and searches
A systematic search was conducted of the PubMed, Embase, Web of Science, and Cochrane Library databases for relevant articles published in English before January 6, 2021. The keywords and retrieval strategies are presented in the online Supplemental Table 1. For completeness, we checked the reference lists of relevant studies found in the database searches. Two researchers (JC and ML) searched and screened the literature independently.
Inclusion and exclusion criteria
The inclusion criteria were: (i) randomized controlled trial (RCT); (ii) comparison between DOACs commonly used in clinical practice (apixaban, edoxaban, dabigatran, rivaroxaban) and control, including low-molecular weight heparins (LMWHs) and vitamin K antagonists (VKAs); (iii) patients aged ⩾ 18 years undergoing VTE prevention or treatment; (iv) outcome indicators, including at least one clinically relevant nonmajor bleeding and minor bleeding case; and (v) for RCTs comparing various DOAC doses with a control dose, only the US Food and Drug Administration (FDA)-approved doses (see the online supplementary materials) were included in our study.
The exclusion criteria were: (i) cancer-associated thrombosis; (ii) combined use of other antithrombotic drugs; (iii) studies that compared DOACs with placebo; and (iv) duplicate studies, incomplete/unavailable original research data.
The definitions of clinically relevant nonmajor bleeding and minor bleeding were consistent across all trials. Online Supplementary Table 2 shows the definitions of bleeding.
Data extraction and risk of bias assessment
Two researchers (JC and ML) extracted data from the relevant RCTs independently. These data were: first author and publication year; number of patients; age; sex ratio; number of patients with bleeding (clinically related nonmajor bleeding and minor bleeding); follow-up duration. Disputes were discussed and resolved by a third researcher (SJ) to reach a consensus.
Included studies were assessed using the Cochrane Collaboration risk of bias tool, 12 which included random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome assessment, selective reporting, and other biases. Each independent field was assessed as having low, high, or unclear risk of bias. Two researchers (JC and ML) evaluated the risk of bias in each RCT independently. During the evaluation, disputes were resolved by having another researcher (SJ) conduct an evaluation.
Statistical analyses
We classified the studies as prevention or treatment according to the inclusion and exclusion criteria in the methods section of the original RCTs. We used NMA to compare the risk of nonmajor bleeding due to DOACs used for VTE and to analyze VTE prevention and treatment separately. The NMA combines direct evidence (direct comparison) and indirect evidence (comparison through other reference interventions). Combining direct and indirect evidence can improve the accuracy of estimation and provide estimates for all pairwise comparisons without direct evidence. We used Stata 14 (College Station, TX, USA) to perform a frequentist NMA. The node size and line thickness in the network evidence map represented the number of patients included in the corresponding intervention and number of directly compared interventions, respectively. We used the inconsistency test to verify whether the results between direct and indirect evidence were consistent. If the inconsistency factor was close to 0, or if p > 0.05, then inconsistency was absent. To examine similarity, the population, intervention, comparison, and outcome (PICO) methodology was used to assess each selected article. 13 We used a fixed effect model with odds ratio (OR) and 95% CI to assess whether there was a significant difference in the incidence of bleeding between the test and control groups. The surface under the cumulative ranking curves (SUCRA) was used for ranking of bleeding risk. The advantages and disadvantages of the intervention measures were evaluated using the SUCRA.13,14 A larger area under the curve indicated a lower risk of bleeding. Network league tables were produced to detail the results of the comparisons between interventions. We performed statistical analyses of the bleeding data of the four DOACs, VKAs, and LMWH, and compared the safety of each drug in clinically relevant nonmajor bleeding and minor bleeding.
Results
Our search strategy identified 9076 studies, of which 27 were included in our analyses (Figure 1).5–10,15–35
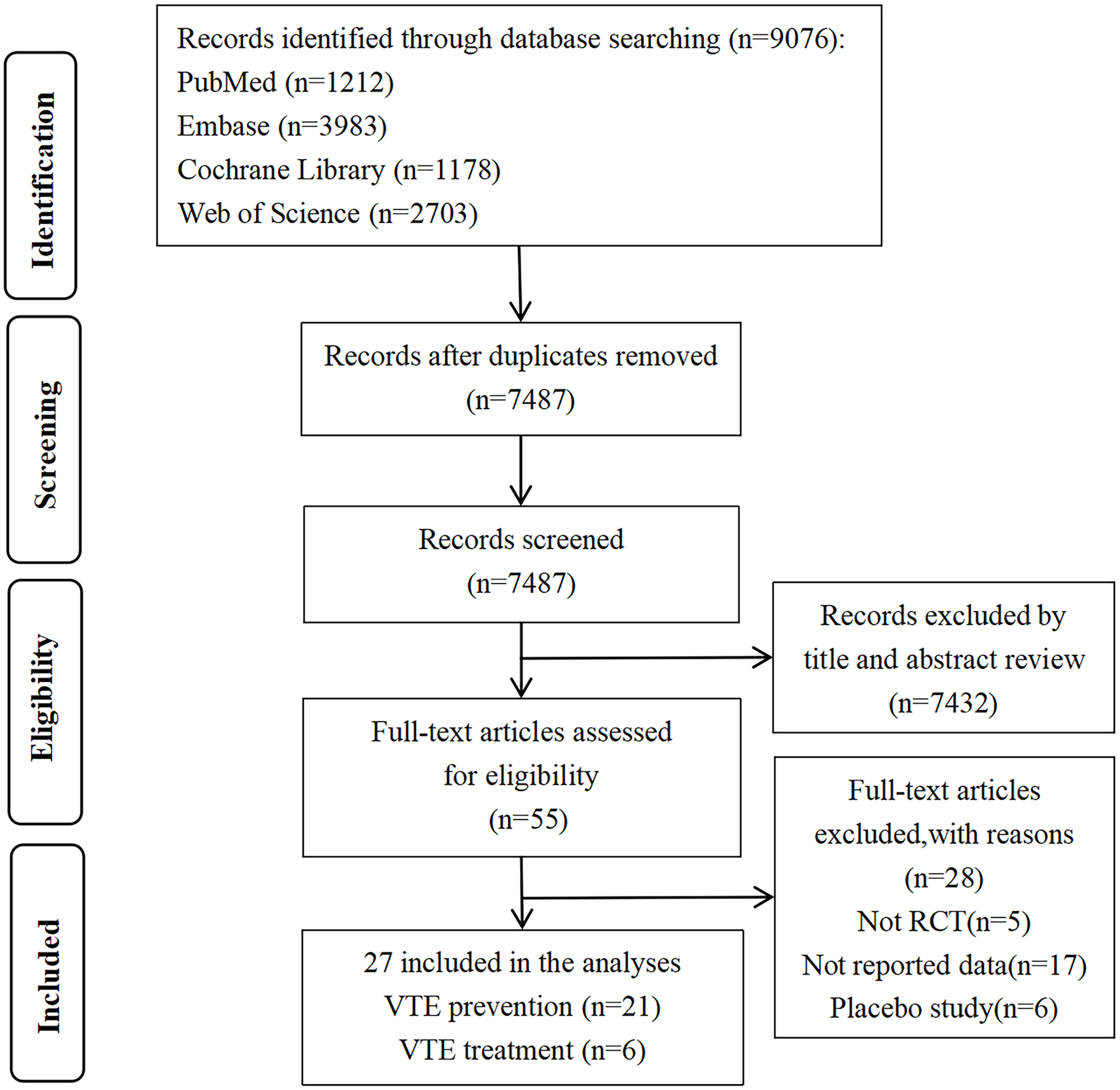
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flowchart showing study selection.
The 27 RCTs involved 64,493 patients. Six articles involved VTE treatment and 21 articles involved VTE prevention. Among the 64,493 patients, 8992 (13.94%) used VKAs, 23,093 (35.81%) took LMWH, 8938 (13.86%) used apixaban, 4082 (6.33%) took dabigatran, 5261 (8.16%) used edoxaban, and 14,127 (21.90%) took rivaroxaban. The characteristics of the 27 included RCTs are shown in online supplementary Table 3.
Presentation of network geometry and assessment of inconsistency
Online supplementary Figure 1 shows the network evidence map between interventions. The node size and line thickness in the network evidence map represent the number of patients included in the corresponding intervention and the number of directly compared interventions, respectively. Online supplementary Table 4 shows the comparisons between interventions. There is a lack of direct comparative evidence for different DOACs in the same RCT, although there are direct comparisons of DOACs with LMWH or VKAs. Inconsistency could not be evaluated because the network did not form a closed loop.
Risk of bias assessment
Figure 2 depicts the assessment of the risk of bias of the included studies (online supplementary Figure 2 shows the detailed evaluation of each study). Overall, the risk of bias of the included RCTs was reasonable. Three articles10,18,26 did not blind the participants and researchers to the study protocol and were assessed as high risk of bias.
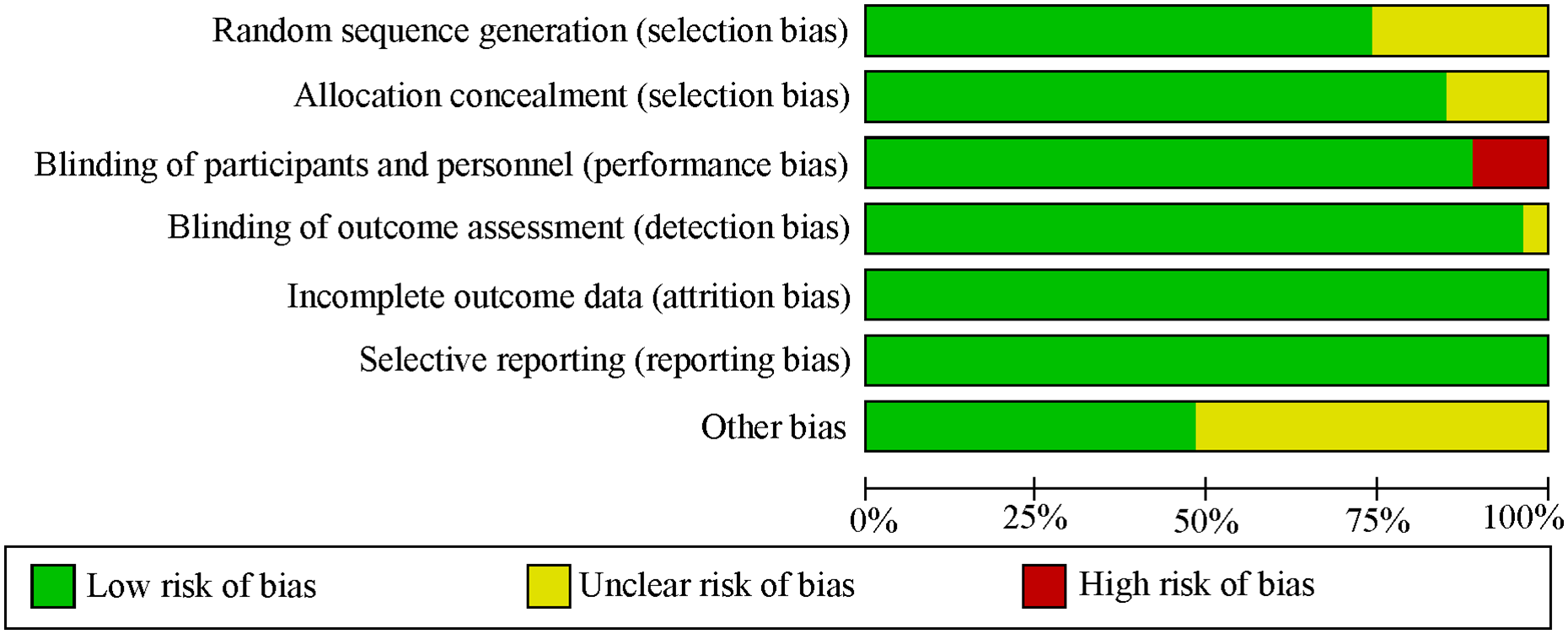
Evaluation of the overall risk of bias of the included studies.
Similarity assumption
In the similarity assumption, indicators such as sex ratio, age, dose, route of administration, outcome definition, follow-up time, and duration of treatment were assessed. The studies selected in the NMA differed with regard to length of follow up and duration of treatment, but the remaining PICO parameters were basically uniform, thus satisfying the similarity assumption.
Network meta-analysis (NMA) outcomes of VTE prevention
Clinically relevant nonmajor bleeding
Figure 3A and online supplementary Figure 3A (league table) show the NMA results for clinically relevant nonmajor bleeding. Rivaroxaban (OR, 1.78; 95% CI, 1.27–2.51) was associated with a significantly higher risk of bleeding compared to apixaban. LMWH (OR, 0.69; 95% CI, 0.55–0.86) was associated with a significantly lower risk of bleeding compared to rivaroxaban.
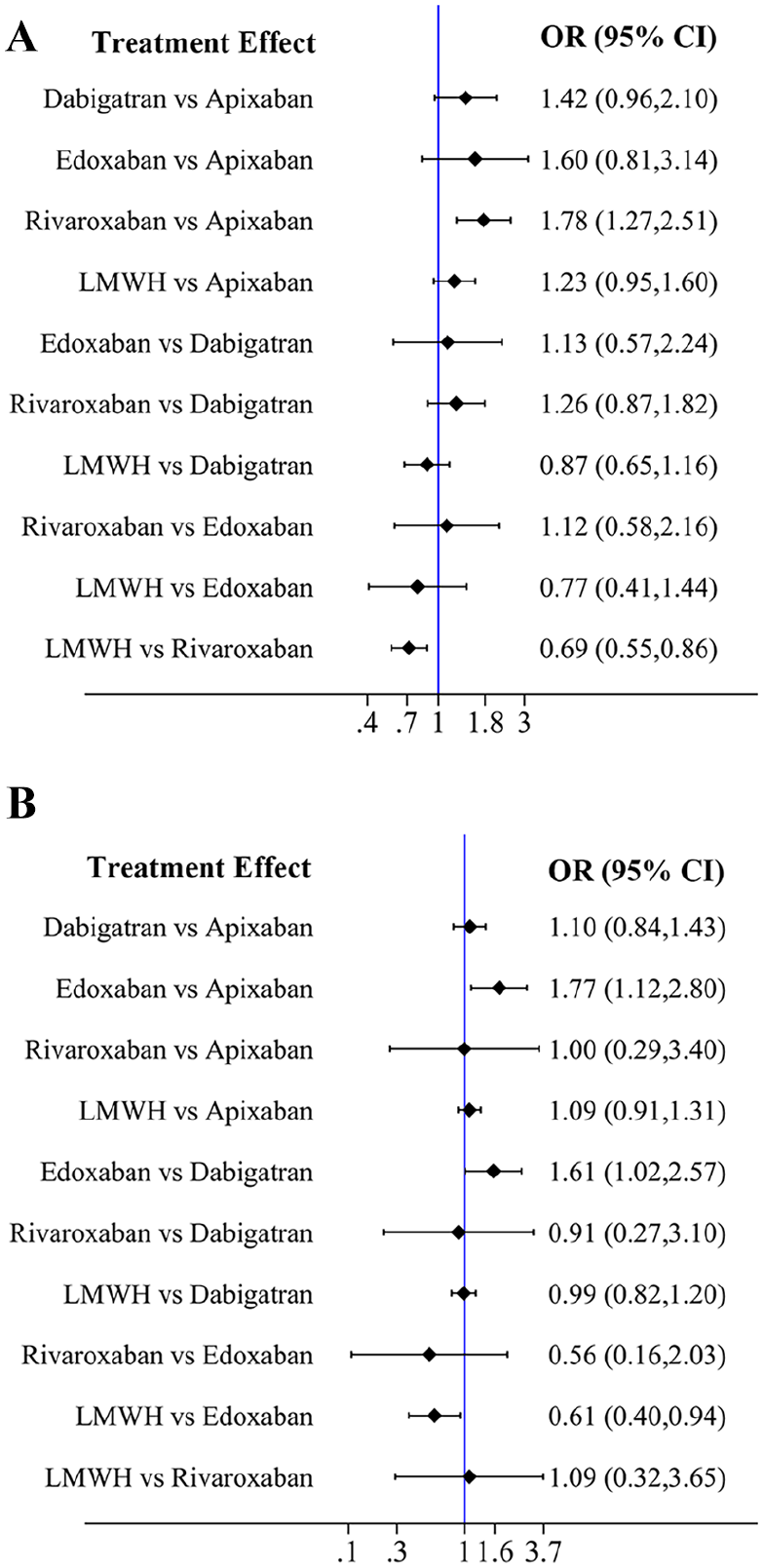
Forest plots of VTE prevention.
In the ranking of the cumulative risk of clinically relevant nonmajor bleeding (Table 1 and Figure 4A), the risk of clinically relevant nonmajor bleeding for apixaban (SUCRA, 95.1) was lowest, followed by that for LMWH (SUCRA, 67.1), dabigatran (SUCRA, 43.4), edoxaban (SUCRA, 32.2), and rivaroxaban (SUCRA, 12.2).
Bleeding risk ranking.
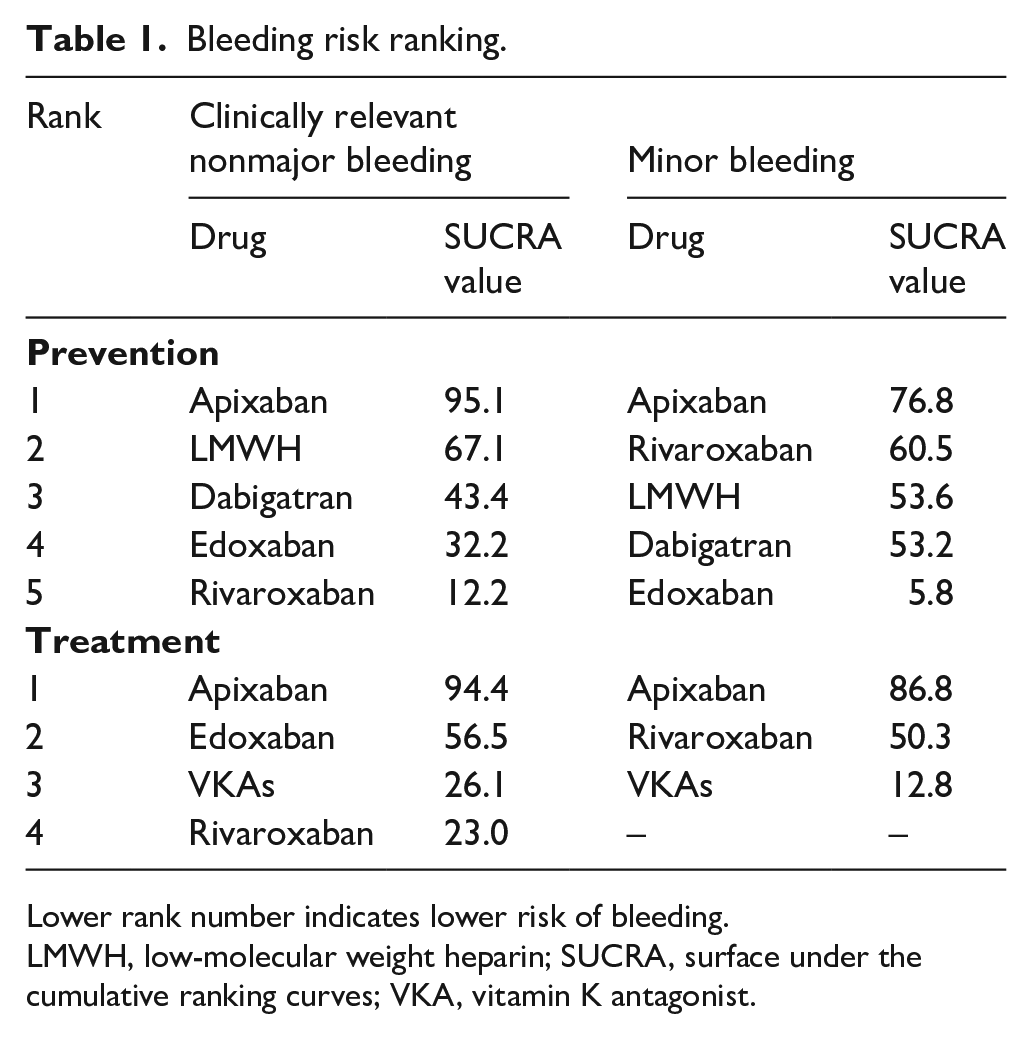
Lower rank number indicates lower risk of bleeding.
LMWH, low-molecular weight heparin; SUCRA, surface under the cumulative ranking curves; VKA, vitamin K antagonist.
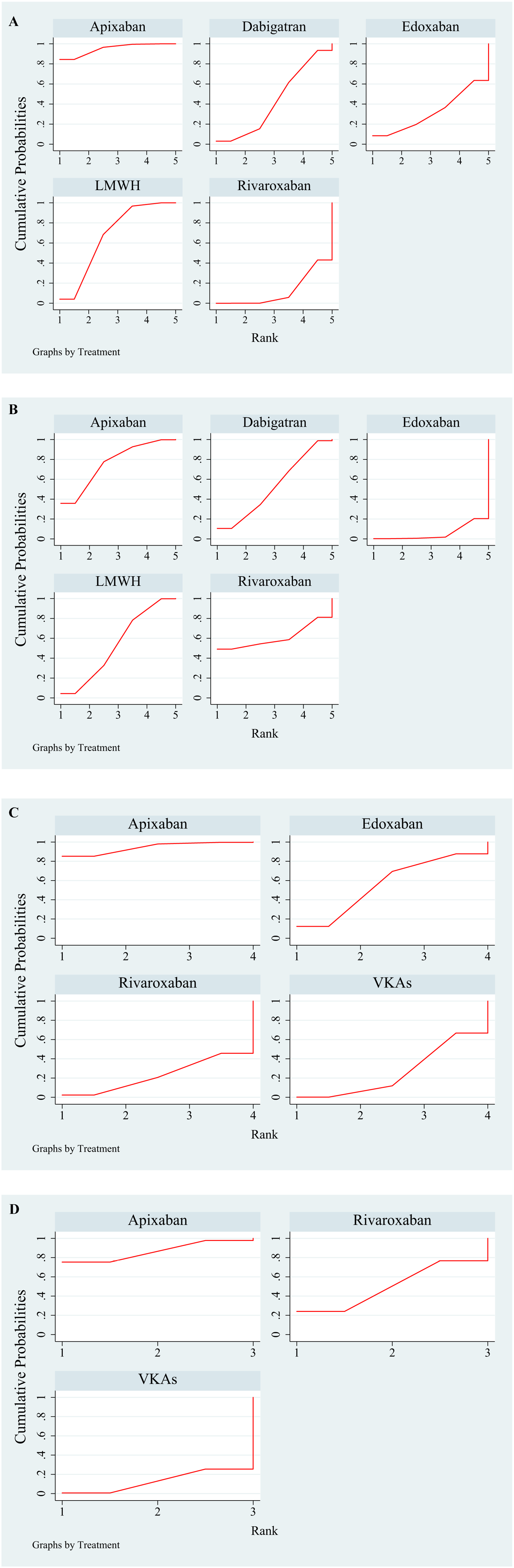
Chart showing the ranking for cumulative risk.
Minor bleeding
Figure 3B and online supplementary Figure 3B (league table) show the NMA results for minor bleeding. Edoxaban (OR, 1.77; 95% CI, 1.12–2.80) was associated with a significantly higher risk of bleeding compared to apixaban. Edoxaban (OR, 1.61; 95% CI, 1.02–2.57) was associated with a significantly higher risk of bleeding compared to dabigatran. LMWH (OR, 0.61; 95% CI, 0.40–0.94) was associated with a significantly lower risk of bleeding compared to edoxaban. Among the DOACs, the risk for minor bleeding was lowest for apixaban.
In the ranking of the cumulative risk of minor bleeding (Table 1 and Figure 4B), the risk for minor bleeding for apixaban (SUCRA, 76.8) was lowest, followed by that for rivaroxaban (SUCRA, 60.5), LMWH (SUCRA, 53.6), dabigatran (SUCRA, 53.2), and edoxaban (SUCRA, 5.8).
NMA outcomes of VTE treatment
Clinically relevant nonmajor bleeding
Figure 5A and online supplementary Figure 3C (league table) show the NMA results for clinically relevant nonmajor bleeding. VKAs (OR, 1.96; 95% CI, 1.16–3.34) was associated with a significantly higher risk of bleeding compared to apixaban.
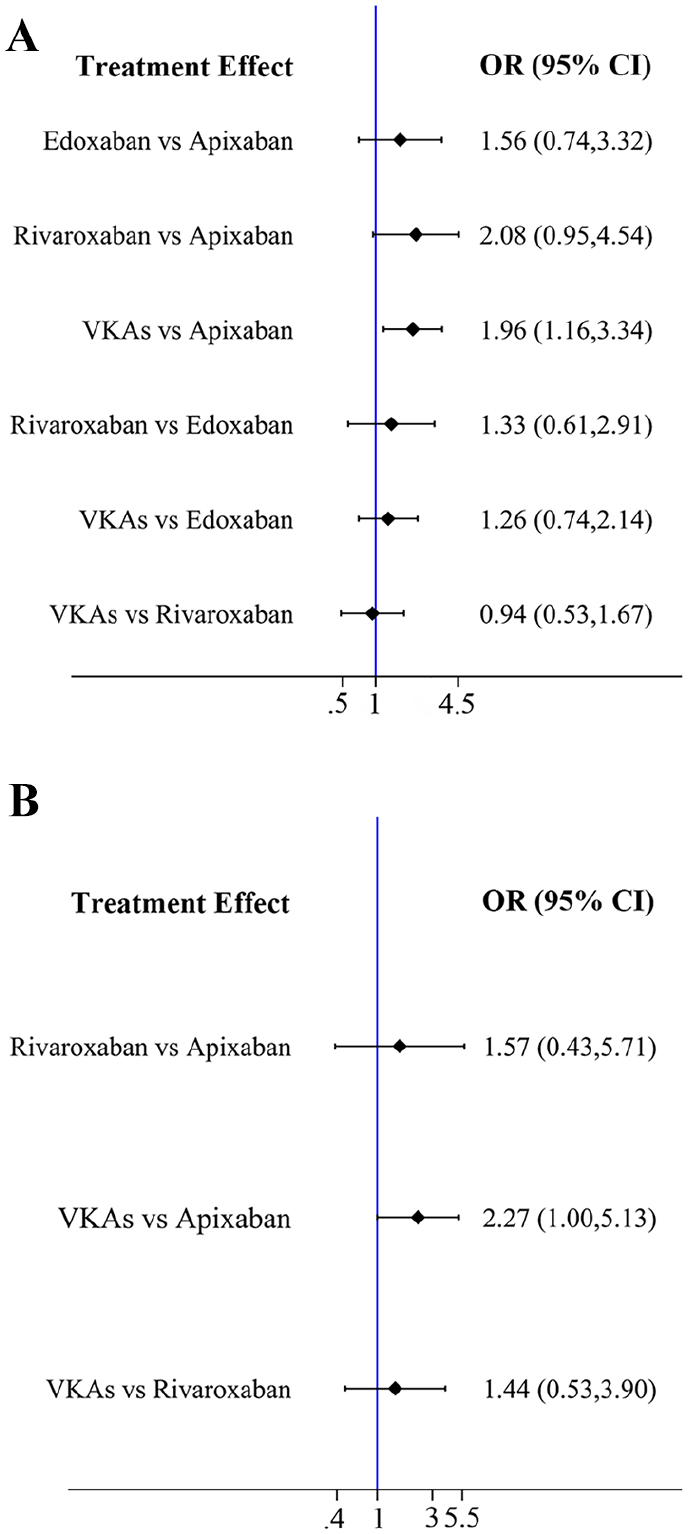
Forest plots of VTE treatment.
In the ranking of the cumulative risk of clinically relevant nonmajor bleeding (Table 1 and Figure 4C), the bleeding risk was lowest for apixaban (SUCRA, 94.4), followed by that for edoxaban (SUCRA, 56.5), VKAs (SUCRA, 26.1), and rivaroxaban (SUCRA, 23.0).
Minor bleeding
Figure 5B and online supplementary Figure 3D (league table) show the NMA results for minor bleeding. There was no statistical difference between all interventions.
In the ranking of the cumulative risk of minor bleeding (Table 1 and Figure 4D), the bleeding risk was lowest for apixaban (SUCRA, 86.8), followed by that for rivaroxaban (SUCRA, 50.3) and VKAs (SUCRA, 12.8).
Discussion
Although nonmajor bleeding does not pose a threat to the patient’s life, it will have an important impact on the patient’s quality of life and needs to be taken seriously. In this systematic review and NMA, we aggregated the data of 27 RCTs and compared the risk of clinically relevant nonmajor bleeding and minor bleeding in VTE prevention and treatment with six interventions. As far as we know, we are the first to use NMA to independently compare the risk of clinically relevant nonmajor bleeding and minor bleeding occurring as a result of DOACs being used for preventing and treating VTE. We analyzed VTE treatment and prevention trials separately, and discuss the risk of bleeding for the different drugs.
For preventing VTE, the drugs were ranked from lowest to highest risk for clinically relevant nonmajor bleeding as follows: apixaban, LMWH, dabigatran, edoxaban, and rivaroxaban. For treating VTE, the drugs were ranked from lowest to highest risk for clinically relevant nonmajor bleeding as follows: apixaban, edoxaban, VKAs, and rivaroxaban. In both treatment and prevention analyses, the bleeding risk was higher for rivaroxaban, which may be related to drug interactions. The four DOACs are all substrates of P-glycoprotein. Among them, apixaban and edoxaban are also substrates of CYP450. Rivaroxaban is not only a substrate of P-glycoprotein and CYP450, but is also a substrate of breast cancer resistance protein. 36 Therefore, the drug interactions of rivaroxaban are more complex, which may affect its pharmacokinetics and pharmacodynamics, thereby increasing the risk of bleeding. Feng et al. 37 showed that the bleeding risk was lower for LMWH, although they were relatively high for apixaban, which is inconsistent with our results. This may be because that study combined the analysis of major bleeding and clinically relevant nonmajor bleeding data; this suggests that separate analysis and combined analysis of different bleeding types may affect the bleeding risk of the drugs. Therefore, independent analysis of clinically relevant nonmajor bleeding is greatly necessary.
For treating VTE, Touma et al. 38 showed that although the risk for minor bleeding was lower for apixaban than for VKAs, the difference was not statistically significant, which is inconsistent with our results. It may be because they did not target VTE patients but included patients with atrial fibrillation. Different disease states may have differences in bleeding. Therefore, independent analysis of different diseases such as VTE and atrial fibrillation is also very important. In their VTE prevention study, Li et al. 39 only discussed the minor bleeding risk of apixaban and LMWH, and found that apixaban had lower bleeding risk than LMWH, which is consistent with our results, but they did not include the other three DOACs, so the bleeding risk ranking of the four DOACs is unknown. Yoshida et al. 40 included three DOACs (apixaban, dabigatran, rivaroxaban) in their study, but because they only included major orthopedics surgical VTE prevention testing and lacked the bleeding data of edoxaban, the safety ranking of the four DOACs could not be determined accurately. Compared with the above studies, our NMA not only includes more comprehensive RCTs of minor bleeding, but also includes bleeding data of four DOACs, which provides newer, more direct, and broader evidence for exploring the risk of minor bleeding in VTE prevention and treatment by DOACs.
In general, apixaban has shown very low bleeding risk in clinically relevant nonmajor bleeding and minor bleeding, but rivaroxaban and edoxaban have relatively higher bleeding risk. This may be explained by Heidbuchel and Vrijens, 41 who found that under the same total daily dose, once-daily dosing has greater peak-to-valley variability than twice-daily dosing, which may be related to the increased risk of bleeding or thrombotic events. Apixaban, rivaroxaban, and edoxaban are all factor X inhibitors, but apixaban is administered twice daily, whereas rivaroxaban and edoxaban are administered once daily. Therefore, in practice, the once-daily doses of rivaroxaban and edoxaban may require vigilance against single missed or additional doses to manage patient compliance more closely.
Our research has some advantages. First, we conducted a comprehensive literature search to provide a detailed summary of the current best evidence, investigated RCTs of all existing DOACs on VTE prevention and treatment, and provide updated evidence of bleeding. Second, as far as we know, this is the first study to use NMA to independently compare the risk of nonmajor bleeding occurring as a result of DOACs being used for preventing and treating VTE. This provides a more comprehensive reference for clinical patients, thereby improving their quality of life. Finally, unlike the previous study, 38 we only studied VTE without combining other indications for anticoagulants, and the results were more accurate. Therefore, it may have more accurate predictive power and a better reference value for the risk of nonmajor bleeding in VTE patients in clinical practice.
Study limitations
Our research has limitations. First, there is a lack of direct comparative evidence for different DOACs in the same RCT, although there are direct comparisons of DOACs with LMWH or VKAs. Second, as the duration of follow-up and treatment varied among the included RCTs, the difference would slightly affect the occurrence of nonmajor bleeding. Therefore, conducting real-world research to confirm our findings would be worthwhile.
Conclusion
Regardless of whether it was used for preventing or treating VTE, apixaban had the lowest risk of nonmajor bleeding. This suggests that apixaban may have a lower risk of bleeding than other anticoagulants and may help provide some clinical reference for choosing a more appropriate drug for the patient.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X221115213 – Supplemental material for Risk of nonmajor bleeding upon use of direct oral anticoagulants for preventing and treating venous thromboembolism: A network meta-analysis
Supplemental material, sj-docx-1-vmj-10.1177_1358863X221115213 for Risk of nonmajor bleeding upon use of direct oral anticoagulants for preventing and treating venous thromboembolism: A network meta-analysis by Jiana Chen, Meina Lv, Shaojun Jiang, Shuyi Wu, Wenlin Xu, Jiafen Qian, Zhiwei Zeng, Mingrong Chen, Zongwei Fang and Jinhua Zhang in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by the Natural Science Foundation of Fujian Province, China (2018Y0037), and Fujian Provincial Health Technology Project, China (2019-CX-19).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
