Abstract
Controversy exists regarding the use of dose capping of weight-based unfractionated heparin (UFH) infusions in obese and morbidly obese patients. The primary objective of this study was to compare time to first therapeutic activated partial thromboplastin time (aPTT) in hospitalized patients receiving UFH for acute venous thromboembolism (VTE) among three body mass index (BMI) cohorts: non-obese (< 30 kg/m2), obese (30–39.9 kg/m2), and morbidly obese (⩾ 40 kg/m2). In this single-center, retrospective cohort study, patients were included if they ⩾ 18 years of age, had a documented VTE, and were on an infusion of UFH for at least 24 hours. Weight-based UFH doses were calculated using actual body weight. A total of 423 patients met the inclusion criteria, with 230 (54.4%), 146 (34.5%), and 47 (11.1%) patients in the non-obese, obese, and morbidly obese cohorts, respectively. Median times to therapeutic aPTT were 16.4, 16.6, and 17.1 hours in each cohort. Within 24 hours, the cumulative incidence rates for therapeutic aPTT were 70.7% for the non-obese group, 69.9% for the obese group, and 61.7% for the morbidly obese group (obese vs non-obese: HR = 1.02, 95% CI: 0.82–1.26, p = 0.88; morbidly obese vs non-obese: HR = 0.87, 95% CI: 0.62–1.21, p = 0.41). There was no significant difference in major bleeding events between BMI groups (obese vs non-obese, p = 0.91; morbidly obese vs non-obese, p = 0.98). Based on our study, heparin dosing based on actual body weight without a dose cap is safe and effective.
Introduction
Controversy exists regarding the use of dose capping of weight-based unfractionated heparin (UFH) infusions in obese and morbidly obese patients for the acute treatment of venous thromboembolism (VTE). Based on the Raschke et al. trial, the gold standard for acute treatment of VTE is weight-based UFH dosed as an 80 units/kg bolus followed by an initial infusion rate of 18 units/kg/hour, titrated to a goal activated partial thromboplastin time (aPTT) of 46–70 seconds. 1 Although this study used actual body weight (ABW) for all patients, clinicians have hypothesized that obese and/or morbidly obese patients may benefit from utilization of a modified weight-based approach to achieve therapeutic levels. Conversely, a delay in achievement of goal aPTT in the setting of VTE may result in poor outcomes, such as thrombus expansion. Several studies have shown a long-term decrease in recurrent VTE when patients achieve therapeutic aPTT within 24 hours of initiating intravenous UFH.1–3
Even though several case reports and studies have been published exploring outcomes with weight-based dosing of UFH in obese and morbidly obese patients, the methodologies, indications for use, and results are highly mixed, making consensus difficult.4–16 The largest study to date, which consisted of 1054 patients who received UFH dosed using ABW without a dose cap, demonstrated no differences in percent of initial aPTTs within goal range. Because this study was conducted in patients with non-VTE indications, such as acute coronary syndrome and atrial fibrillation, where a lower UFH bolus, rate, and aPTT were utilized, applicability to patients with VTE is unclear. 15 A matched analysis of 273 patients on UFH for any indication showed a significant difference in mean UFH infusion rate required to achieve therapeutic aPTT among three body mass index (BMI) cohorts, where higher BMI patients required lower weight-based rates. However, in a subanalysis, the difference was only upheld in patients who did not receive an initial bolus. 8 Another study evaluated outcomes of a modified weight-based approach in 298 patients, utilizing a dosing weight for patients 20% over their ideal body weight. Although no difference in time to therapeutic aPTT among three BMI cohorts was observed, the severely obese cohort (BMI ⩾ 40 kg/m2) experienced a higher percentage of subtherapeutic aPTTs (p-value not calculated). 13 All three studies reported no differences in bleeding events, albeit the sample sizes to assess this outcome were small.
The purpose of this study was to compare the outcomes of non-obese, obese, and morbidly obese patients who received weight-based UFH using ABW without a dose cap for treatment of an acute VTE. The primary objective was to compare time to first therapeutic aPTT. The secondary objectives were to compare: (1) achievement of a therapeutic aPTT, subtherapeutic aPTT, and supratherapeutic aPTT at 10 hours and 24 hours; (2) bleeding and thrombotic complications; and (3) the number of dose adjustments needed to achieve therapeutic aPTT.
Methods
Subjects
This study is a retrospective cohort, single-center study that included patients who were admitted to Mayo Clinic Hospital in Rochester, Minnesota, from January 1, 2010 through December 31, 2016. All patients initiated on the institutional high-intensity heparin nomogram (HIHN) during this timeframe were identified (Table 1, Figure 1). Patients were included if they were ⩾ 18 years of age, had documented VTE (identified electronically via the International Classification of Diseases, Ninth Revision (ICD-9) and Tenth Revision (ICD-10) codes) as the indication for heparin, and were on the HIHN for at least 24 hours. Patients were included even if the heparin drip was paused for a procedure. Patients were excluded if they did not authorize review of their medical records for research, received a thrombectomy (due to increased risk of bleeding), or received a fibrinolytic agent. Under the Minnesota Research Authorization law, patients have the right to deny access to their Minnesota medical records for research purposes. Included patients were divided into three groups based on body mass index (BMI) at the time of heparin initiation: non-obese (BMI < 30 kg/m2), obese (BMI 30–39.9 kg/m2), and morbidly obese (BMI ⩾ 40 kg/m2). This was approved by the Mayo Clinic Institutional Review Board.
High-intensity heparin dosing nomogram parameters.
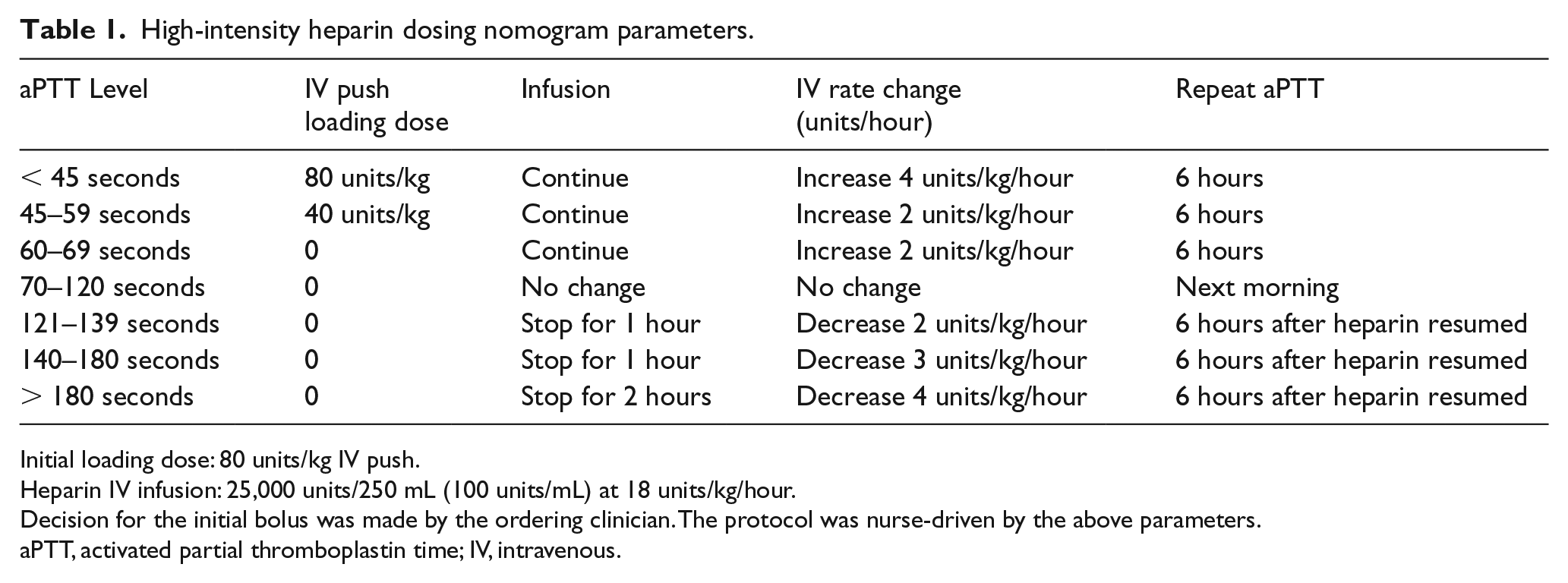
Initial loading dose: 80 units/kg IV push.
Heparin IV infusion: 25,000 units/250 mL (100 units/mL) at 18 units/kg/hour.
Decision for the initial bolus was made by the ordering clinician. The protocol was nurse-driven by the above parameters.
aPTT, activated partial thromboplastin time; IV, intravenous.
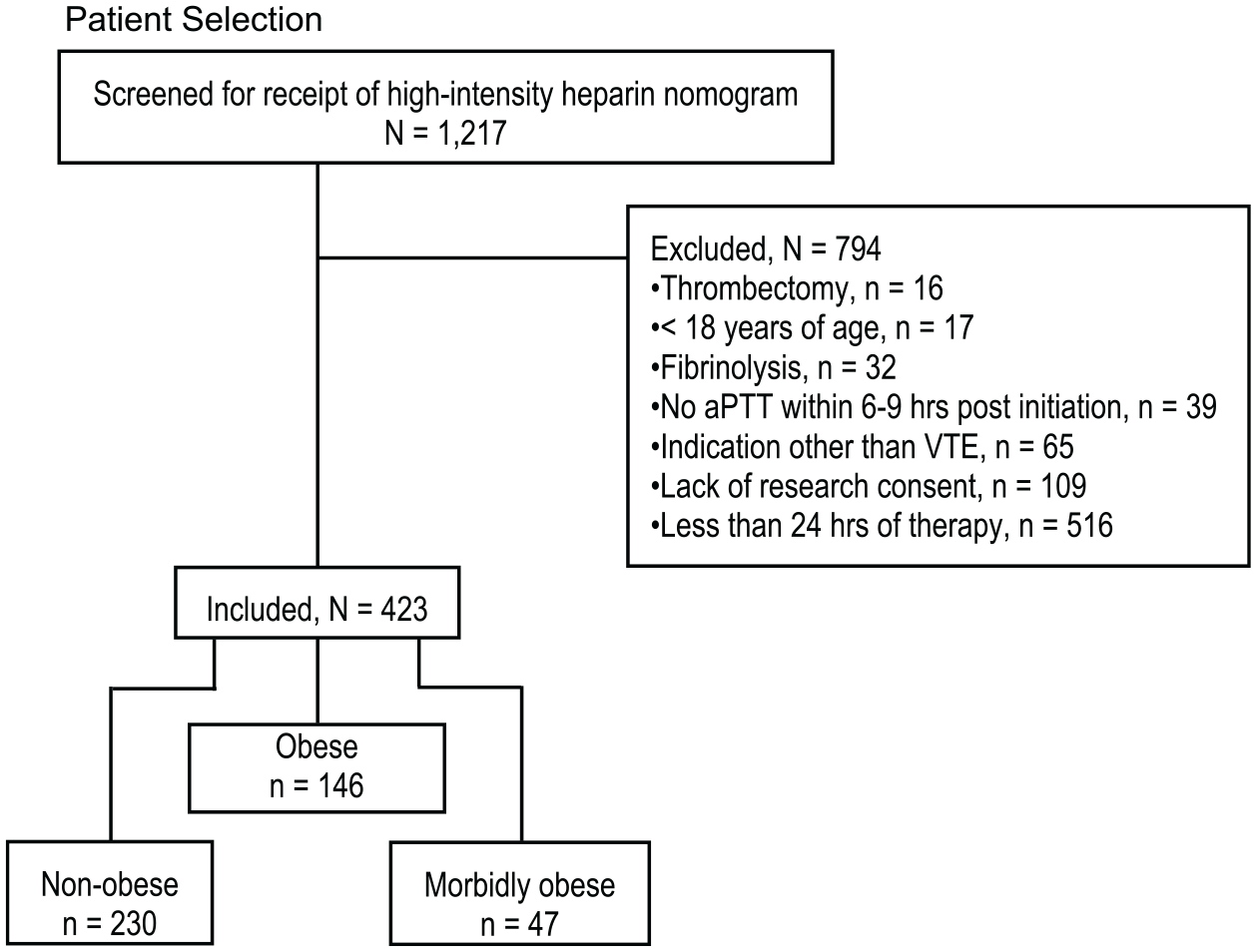
Use of inclusion and exclusion criteria.
Procedures
The HIHN is a nurse-driven protocol with parameters defined in Table 1. Upon initiation, patients received an optional 80 units/kg bolus of UFH followed by a continuous infusion starting at 18 units/kg/hour. The decision to give the initial bolus was made by the ordering clinician. Doses were calculated using ABW (charted on admission during the hospitalization included in the study) regardless of body size. The first aPTT was to be checked 6 hours following the infusion start per protocol. Based on aPTT values, further dose adjustments were made by registered nurses using the protocol directions.
Included patients were identified electronically from the Heparin Nomogram System, an electronic system utilized at Mayo Clinic Hospital – Rochester to capture all patients on a heparin infusion while hospitalized and data points such as weight, heparin start and stop time, aPTT results, and heparin rate adjustments.17,18 Type of VTE (deep vein thrombosis or pulmonary embolism) was identified electronically by cross-referencing admission dates of the respective hospitalization with ICD-9 and ICD-10 codes.
Endpoints and definitions
The primary endpoint of the study was time to first therapeutic aPTT, defined as 70–120 seconds per the HIHN (correlated to a heparin anti-Xa 0.3–0.7 units/mL). Secondary endpoints were to compare: (1) incidence of patients with a subtherapeutic, therapeutic, and supratherapeutic aPTT at 10 hours and 24 hours after starting the heparin nomogram; (2) bleeding and thrombotic complications related to heparin infusion during hospitalization; and (3) the number of dose adjustments necessary to achieve therapeutic aPTT. The definition of major bleeding was any fatal bleeding and/or symptomatic bleeding in a critical area or organ, such as intracranial, intraspinal, intraocular, retroperitoneal, intra-articular or pericardial, or intramuscular with compartment syndrome, and/or bleeding causing a fall in hemoglobin level of 2 g/dL or more, or leading to transfusion of ⩾ 2 units of whole blood or red cells. 19 Clinically relevant non-major bleeding was defined as having any sign or symptom of hemorrhage that does not fit the criteria for major bleeding but meets at least one of the following criteria: requiring medical intervention by a healthcare professional, leading to hospitalization or increased level of care, or prompting face-to-face evaluation. 20 Thrombotic complications were defined as new or worsening VTE, or development of transient ischemic attack (TIA), cerebrovascular accident (CVA), or arterial embolism. 21 Both bleeding and thrombotic complications were assessed from the start of the UFH infusion through hospital discharge.
Data collection
For each included patient, demographic data including age, sex, race, height, and weight were collected. BMI was calculated using the patient’s height and weight documented on admission during which they received the heparin nomogram. Data related to the heparin infusion (start and stop date/time, aPTT values and their respective date/time, initial heparin bolus dose (if given), infusion rates utilized (units/hour), number of dose adjustments while on the HIHN, duration (hours)) were also collected. Bleeding and thrombotic complications were assessed by chart review for the aforementioned criteria; mention of bleeding and thrombotic events was reviewed within the dismissal summary. Concurrent use of antiplatelet agents, anticoagulants, and nonsteroidal anti-inflammatory drugs (NSAIDs) was collected as well. The severity and age weighted Charlson Comorbidity Index was computed based on the date of hospital admission for all included patients to assess severity of illness. 22 Study data were collected and managed using Microsoft Excel (Redmond, WA, USA) and REDCap (Research Electronic Data Capture). REDCap is a secure, web-based application designed to support data capture for research studies. 23
Statistical analysis
Statistical analyses were performed using SAS software, Version 9.4 (SAS Institute, Inc., Cary, NC, USA) and R version 3.4.2 (R Core Team, R Foundation for Statistical Computing, Vienna, Austria).
Descriptive statistics are reported as mean and SD or median and range for continuous variables, or frequencies and percentages for categorical variables. Baseline characteristics were compared among the three BMI groups using either a t-test or Kruskal–Wallis test for continuous variables, and a chi-squared test for categorical variables. Kaplan–Meier curves were constructed to compare the time to first therapeutic aPTT while on heparin among the three BMI groups. Cox proportional hazards regression models were used to estimate the association among BMI groups and the time to first therapeutic aPTT. The number of dose adjustments was treated as a time-dependent covariate in these Cox models due to the value changing over the same time frame as when the therapeutic aPTT event is measured. An interaction term between BMI and number of dose adjustments was included to test whether the relationship between BMI and time to a therapeutic aPTT depends on the number of dose adjustments, and vice versa. Similarly, Kaplan–Meier curves and Cox models were used to analyze the outcomes of time to a subtherapeutic aPTT and the time to a supratherapeutic aPTT while on heparin in addition to the outcomes of a bleed and a thrombotic complication through hospital discharge.
We anticipated 80–90% of patients achieving a therapeutic aPTT. Using this and the assumption that the ratio of morbidly obese to non-obese patients was 1:4, at least 235 events were needed to detect a hazard ratio (HR) of 1.58 (58% increased chance of achieving a therapeutic aPTT) with 80% power (two-sided, alpha = 0.05 test).
Results
During the study period, 1217 patients who had received the HIHN were identified. Of these, 794 were excluded, with the most common reason being less than 24 hours of heparin therapy administered. The remaining 423 patients were included (Figure 1).
Of the 423 patients included, 230 (54.4%) were non-obese, 146 (34.5%) were obese, and 47 (11.1%) were morbidly obese. Patient demographics are listed in Table 2. Significant differences were noted with respect to age, sex, Charlson Comorbidity Index, admission location, and receipt of concurrent antiplatelet therapy. The morbidly obese cohort had a lower mean age and mean Charlson score, as well as a higher rate of ICU admission. In the overall study cohort, the average BMI (kg/m2) was 30.2 (SD = 8.4), 98.1% were Caucasian, 62.7% had a diagnosis of pulmonary embolism, and 62.4% received an initial heparin bolus. The median time to first aPTT drawn was 7.4 hours after heparin initiation.
Baseline characteristics.
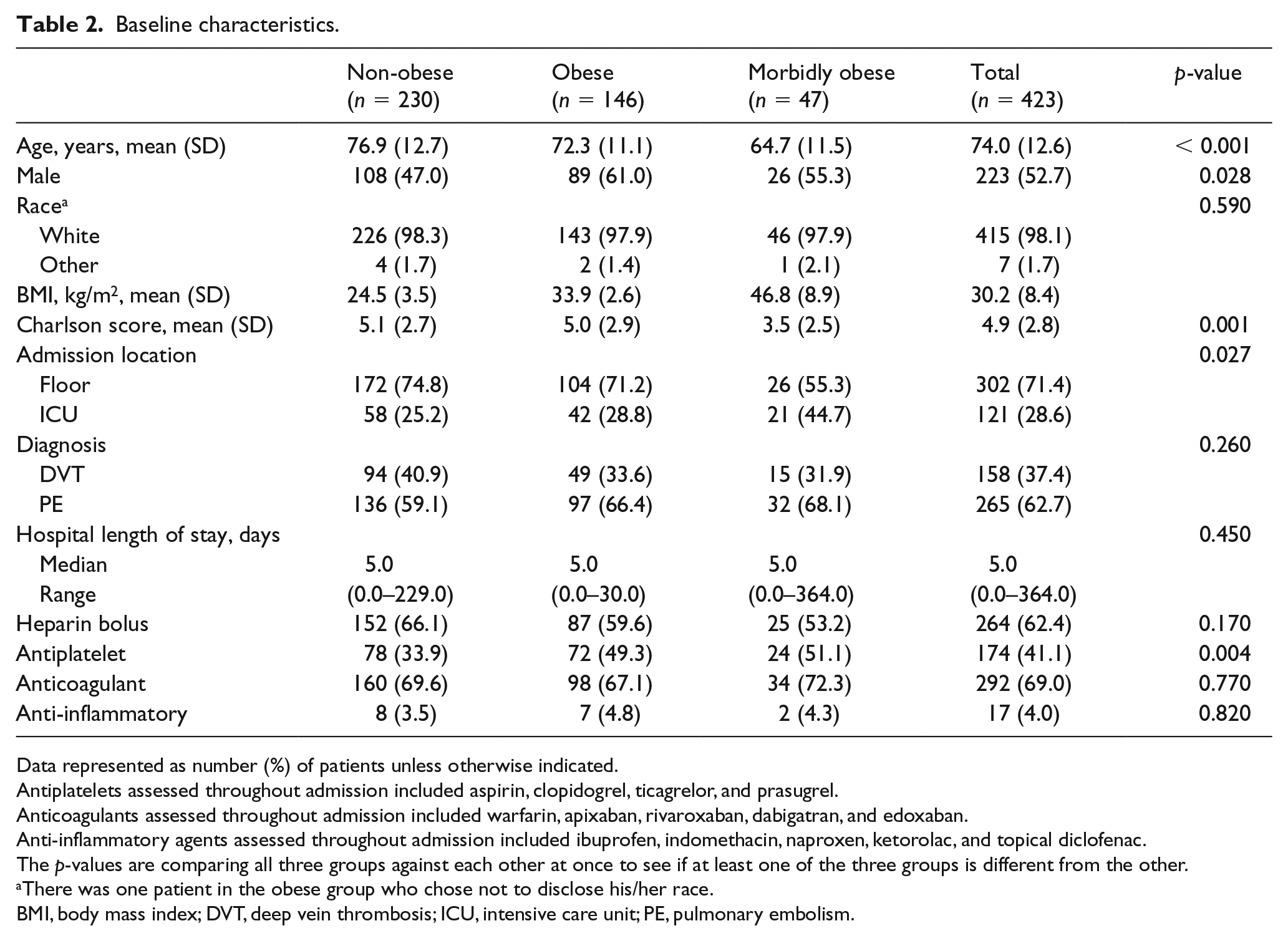
Data represented as number (%) of patients unless otherwise indicated.
Antiplatelets assessed throughout admission included aspirin, clopidogrel, ticagrelor, and prasugrel.
Anticoagulants assessed throughout admission included warfarin, apixaban, rivaroxaban, dabigatran, and edoxaban.
Anti-inflammatory agents assessed throughout admission included ibuprofen, indomethacin, naproxen, ketorolac, and topical diclofenac.
The p-values are comparing all three groups against each other at once to see if at least one of the three groups is different from the other.
There was one patient in the obese group who chose not to disclose his/her race.
BMI, body mass index; DVT, deep vein thrombosis; ICU, intensive care unit; PE, pulmonary embolism.
For the primary outcome, time to first therapeutic aPTT, median times to achieving a therapeutic aPTT were 16.4 hours in the non-obese group, 16.6 hours in the obese group, and 17.1 hours in the morbidly obese group (Figure 2). For achievement of therapeutic aPTT within 10 hours, the cumulative incidence rates were 32.2% for the non-obese group, 32.9% for the obese group, and 36.2% for the morbidly obese group. For achievement of therapeutic aPTT within 24 hours, the cumulative incidence rates were 70.7% for the non-obese group, 69.9% for the obese group, and 61.7% for the morbidly obese group (Table 3). There were no significant differences in achieving therapeutic aPTT between the groups (obese vs non-obese: HR = 1.02, 95% CI: 0.82–1.26, p = 0.88; morbidly obese vs non-obese: HR = 0.87, 95% CI: 0.62–1.21, p = 0.41).
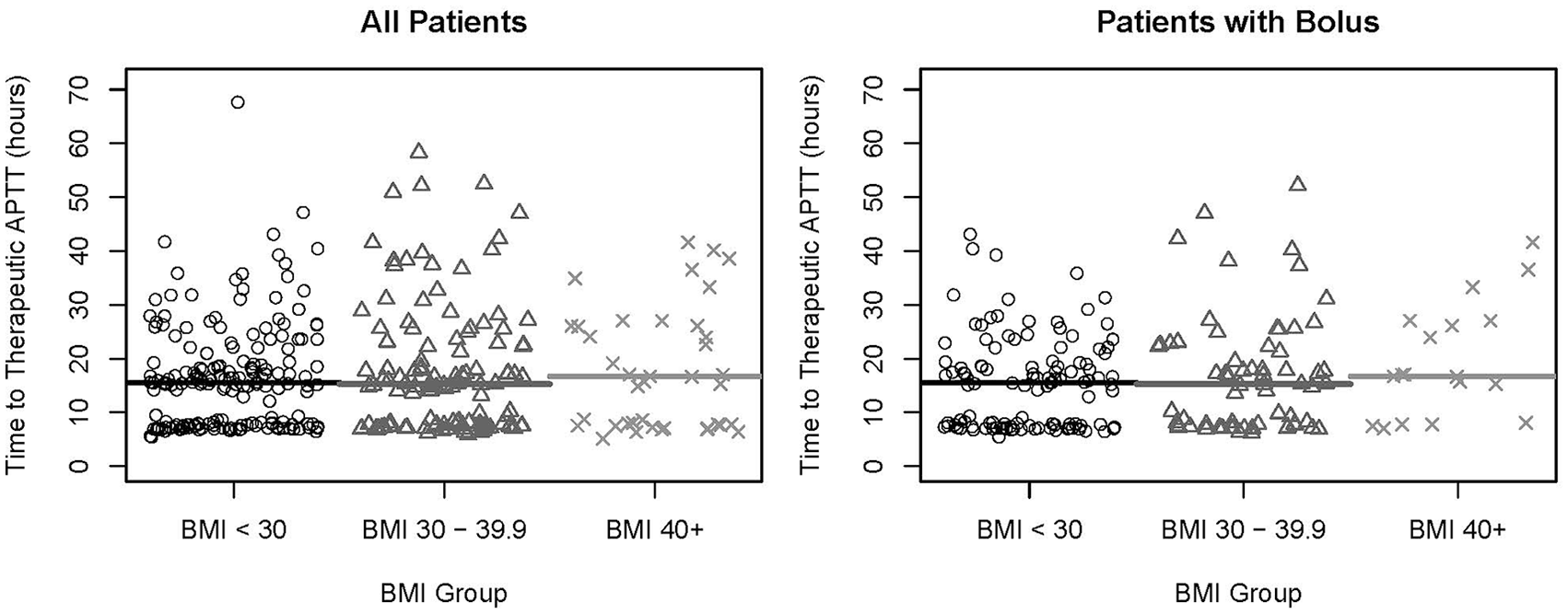
Time to therapeutic aPTT.
Cumulative incidence of aPTT values within 10–72 hours of starting the high-intensity heparin nomogram.
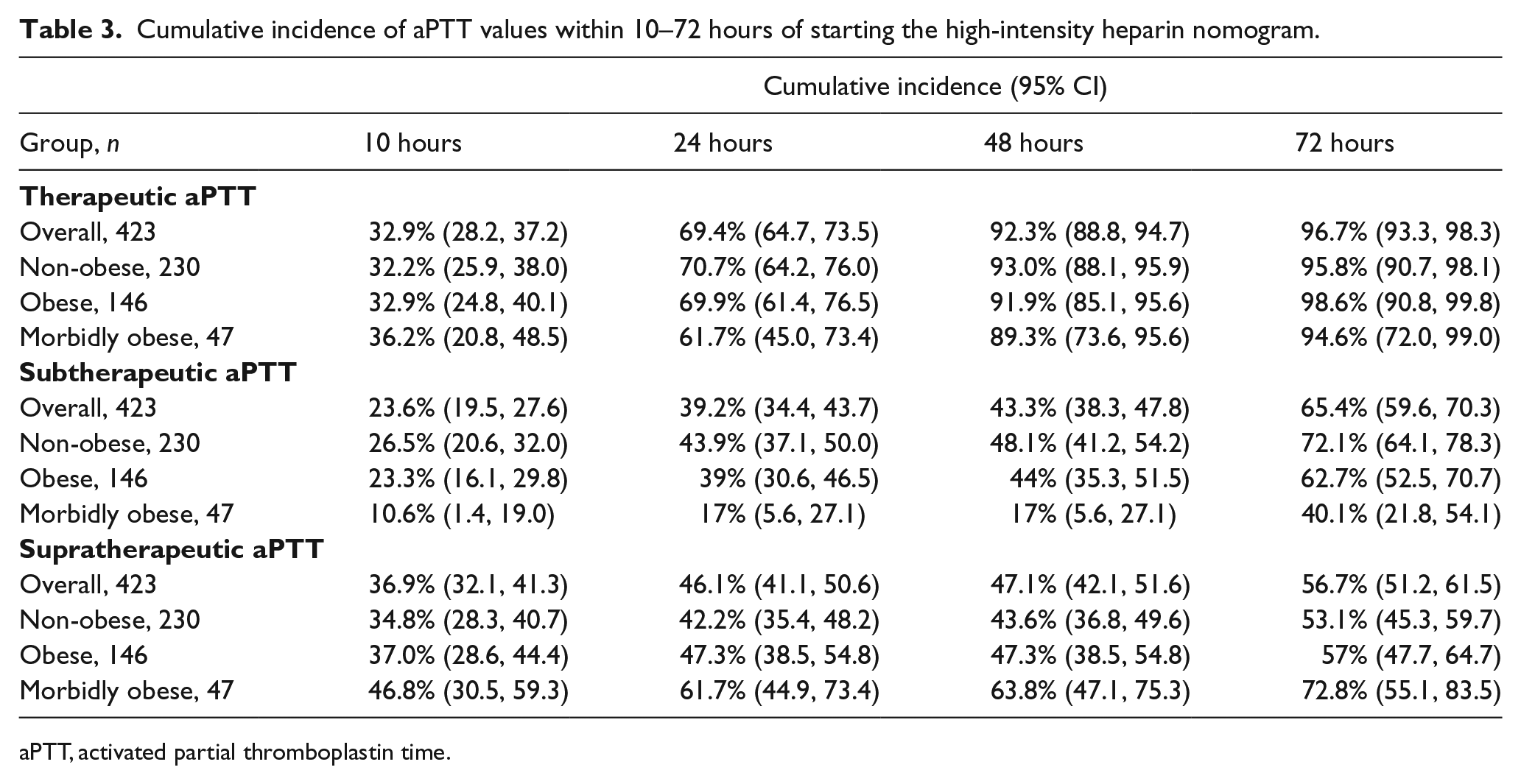
aPTT, activated partial thromboplastin time.
The cumulative incidence for achievement of subtherapeutic aPTT within 10 hours was 26.5% for the non-obese group, 23.3% for the obese group, and 10.6% for the morbidly obese group. For achievement of subtherapeutic aPTT within 24 hours, the cumulative incidence rates were 43.9% for the non-obese group, 39% for the obese group, and 17% for the morbidly obese group (Table 3). This difference was statistically significant between the morbidly obese and non-obese groups (HR = 0.78, 95% CI: 0.60–1.02, p < 0.001) but not between the obese and the non-obese groups (HR = 0.39, 95% CI: 0.24–0.64, p = 0.065).
For achievement of supratherapeutic aPTT within 10 hours, the cumulative incidence rates were 34.8% for the non-obese group, 37% for the obese group, and 46.8% for the morbidly obese group. For achievement of supratherapeutic aPTT within 24 hours, the cumulative incidence rates were 42.2% for the non-obese group, 47.3% for the obese group, and 61.7% for the morbidly obese group (Table 3). In this instance, there was also a significant difference in achieving supratherapeutic aPTT between the morbidly obese and the non-obese groups (HR = 1.60, 95% CI: 1.11–2.33, p = 0.013). There was no significant difference in achieving supratherapeutic aPTT between the obese and non-obese groups (HR = 1.08, 95% CI: 0.82–1.42, p = 0.59). Figure 3 graphically depicts mean aPTT values for all three groups across the study period.
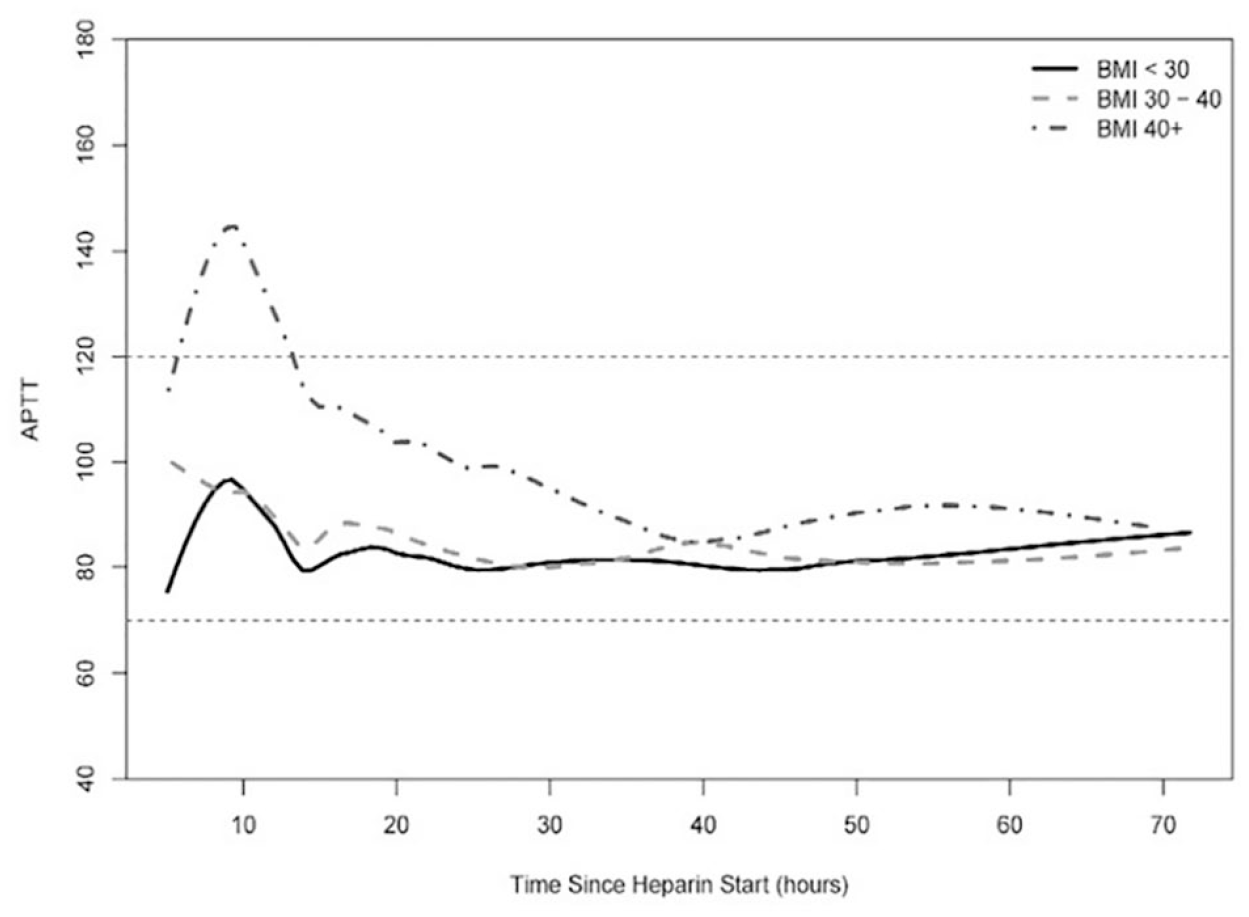
Average aPTT values, per BMI group, over given time.
For the secondary endpoint of bleeding, there were a total of 27 (16.9%) major bleeding events within 5 days of heparin initiation: 14 (15%) in the non-obese group, 10 (18.6%) in the obese group, and 3 (16.4%) in the morbidly obese group. Since patients had variable durations of heparin exposure and follow-up, these rates are reported using Kaplan–Meier rates, rather than raw rates. There was no significant difference in major bleeding events among the BMI groups (obese vs non-obese, p = 0.91; morbidly obese vs non-obese, p = 0.98). Of these 27 events, eight non-obese patients, seven obese patients, and three morbidly obese patients had a supratherapeutic aPTT value before and/or after their bleeding event. There was also no significant difference in clinically relevant non-major bleeding rates (obese vs non-obese, p = 0.66; morbidly obese vs non-obese, p = 0.48). Only two thrombotic complications occurred overall, both at 72 hours post-initiation of heparin: one in the non-obese group and one in the obese group.
Among those who achieved a therapeutic aPTT, the average number of dose adjustments necessary to achieve therapeutic aPTT for each BMI group was as follows: non-obese 2.54 (SD = 1.94), obese 2.45 (SD = 1.97), and morbidly obese 2.55 (SD = 1.78). No significant interaction was found between BMI and number of dose adjustments for therapeutic aPTT achievement (p = 0.14).
Since 37.6% of the study cohort did not receive an initial heparin bolus, comparisons were made to determine if the outcomes changed based on bolus receipt. Risk of a supratherapeutic aPTT within 10 hours of infusion start was similar for morbidly obese and non-obese patients who received a bolus (HR 1.79, 95% CI: 1.00–3.22, p = 0.052). For those who received a bolus, obese patients were at a higher risk than non-obese patients of having a major bleed (HR = 3.25, 95% CI: 1.07–11.29, p = 0.037) or any bleed (HR = 2.85, 95% CI: 1.04–7.85, p = 0.043). There was no difference in risk of bleeding (major or any) between morbidly obese and non-obese patients who received a bolus. In addition, the median time to first therapeutic aPTT was 17 hours for those who received a bolus and 16 hours for those who did not receive a bolus (p = 0.71). Comparisons were also made within each BMI cohort (non-obese, obese, morbidly obese) and there were no differences in median time to therapeutic aPTT for patients who did and did not receive a bolus.
Discussion
In this retrospective study, we included 423 episodes of administration of our HIHN for treatment of VTE. Our study found no significant difference in median time to therapeutic aPTT among the different BMI groups while dosing UFH using ABW without a dose cap. Although a higher percentage of patients in the morbidly obese group had a higher incidence of a supratherapeutic aPTT, this was not associated with an increased rate of bleeding at any point in the study period. Moreover, the morbidly obese group had a statistically lower incidence of a subtherapeutic aPTT compared to the non-obese group.
When considered within the context of heparin pharmacokinetics, the results of this study are a bit perplexing. Heparin binds a number of plasma proteins allowing for its distribution to be limited to blood volume.24,25 Based on this, accumulation in fat or muscle tissue is believed to be low. Blood volume does not scale linearly with weight in obese and morbidly obese patients; 26 therefore they should require a smaller weight-based heparin dose in theory. However, our study suggests there may be another pharmacokinetic phenomenon to consider. Regardless, the pharmacokinetic differences do not appear to be clinically significant based on our findings.
The results of this study are difficult to compare with studies of similar intent due to a wide variety of methodologies utilized. We found only one other study that compared patients who received UFH for VTE and used ABW for all subjects. 4 The primary outcome, which differed from ours, was mean aPTT at 6 and 12 hours. Significant differences were observed at both time points. Of the four bleeding events reported, only two had supratherapeutic aPTTs. Despite this, the authors recommended consideration of alternative dosing strategies for morbidly obese patients. 4 Even though our study found more supratherapeutic aPTTs within the first 24 hours of UFH initiation in the morbidly obese group, we also observed less subtherapeutic aPTTs within the first 24 hours in this cohort. Therefore, we do not recommend utilization of an alternative weight or a dose cap which is based on the lack of correlation to bleeding events observed. Similarly, the Raschke et al. trial demonstrated the benefit of achieving an aPTT within therapeutic range and higher than the therapeutic threshold within the first 24 hours: no increase in bleeding and a fivefold decrease in recurrent VTE. 1
Four other studies utilized ABW for all their patients. Three were inclusive of all indications for UFH.5,8,12 Shin et al. found a significant difference in mean time to first therapeutic aPTT, where patients > 150 kg demonstrated a higher time (23.8 ± 27.9 hours) than patients of lower weight groups, which is not in concert with our findings. No differences in bleeding were observed. 12 Spruill et al. did not find a difference between mean initial and final heparin infusion rates for non-obese and obese patients. The mean times to targeted aPTT were 25.18 ± 14.76 hours (non-obese cohort) versus 25.86 ± 12.83 hours (obese cohort), which were longer than the times we reported. 5 Riney et al found a significant difference in mean infusion rate needed to achieve first therapeutic aPTT among three BMI cohorts. These rates were 13.5 units/kg/hour (BMI < 25 kg/m2), 12.5 units/kg/hour (BMI 25–39.9 kg/m2), and 11.5 units/kg/hour (BMI ⩾ 40 kg/m2; p = 0.001). Similar to our study, there was no difference in mean time to first therapeutic aPTT: 21.3 hours, 22.1 hours, and 29.9 hours, respectively (p = 0.421). Also, no significant differences in bleeding were observed. Of the 10 patients who bled, only one had a supratherapeutic aPTT. Despite this, the authors recommended use of a maximum infusion rate of 14 units/kg/hour in patients with BMI ⩾ 40 kg/m2. 8 Bauer et al. utilized ABW for patients with cardiac indications and report the largest study to date comparing weight-based dosing of UFH among different BMI cohorts. For their primary outcome, rate of first aPTT within goal (measured at 6 hours after UFH initiation), they reported similar rates of 31% (BMI 15.9–25.9 kg/m2), 39% (BMI 26–29.4 kg/m2), 31% (BMI 29.5–34.2 kg/m2), and 29% (BMI > 34.2 kg/m2; p = 0.27). Like our study, there was no difference in bleeding events among the groups. 15
Based on the observation of a higher rate of supratherapeutic aPTT levels in patients of high body weights, some institutions have implemented modified weight-based heparin dosing approaches and report their outcomes. A challenge with making comparisons is that multiple modification types were utilized. Examples include use of a dose cap, 16 use of an adjusted initial rate for patients > 50% above their ideal body weight, 10 use of a dosing weight if > 20% above their ideal body weight, 13 and use of a dosing weight if > 30% above their ideal body weight. 14 Myzienski et al. report a case of a patient with an acute pulmonary embolism and BMI of 134 kg/m2. The maximum heparin infusion rate allowed according to the institution’s protocol was 1500 units/hour, which translated to 3.9 units/kg/hour using ABW. Subsequently, it took 55 hours for the patient to achieve a therapeutic aPTT at a rate of 9.4 units/kg/hour. 16 Given lack of supportive data, clinicians should refrain from using an arbitrary dose cap. The data published by Dee et al., 10 Hosch et al., 13 and Fan et al. 14 that evaluated use of a dosing weight in patients above a specific weight threshold reported no difference in the primary outcomes. However, results from these studies should be applied cautiously given their retrospective nature and lack of published adverse clinical outcomes associated with use of ABW in obese and morbidly obese patients with VTE.
Limitations
Our study is not without limitations. Aside from its retrospective design, we had significantly fewer patients in our study who were morbidly obese compared to the other two groups. However, the proportions of patients are similar to other studies that compare patient populations based on BMI. Additionally, some patients were transitioned from our intermediate-intensity heparin nomogram to the high-intensity nomogram. The specific limitation with this is that patients did not receive the 80 units/kg bolus at the start of the high-intensity heparin infusion; nonetheless, they were included to reflect real-world practice. Finally, diagnosis of a bleeding or thrombotic complication was determined via the hospital discharge summary and not via ICD-9 and ICD-10 codes. Therefore, if there was no documentation of it in the discharge summary, our incidence rate may be underestimated.
Conclusion
Overall, our retrospective study did not show a significant difference in median time to achieve therapeutic aPTT between non-obese, obese, and morbidly obese patients with VTE, where the majority received an 80 units/kg heparin bolus followed by an 18 units/kg/hour infusion dosed using ABW without a dose cap. Additionally, there were no differences in rates of bleeding among all three groups. Based on our study, heparin dosing based on ABW without a dose cap is safe and effective. Prospective trials to compare this strategy with capped or adjusted weight dosing are needed.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
