Abstract
Background:
We aimed to understand the effects of aspirin dose on outcomes in patients with peripheral artery disease (PAD) as well as their participation in a pragmatic randomized controlled trial.
Methods:
In a subanalysis of the Aspirin Dosing: A Patient-Centric Trial Assessing Benefits and Long-Term Effectiveness (ADAPTABLE) study, we compared aspirin doses (81 vs 325 mg) among participants with PAD and study participation metrics in patients with and without PAD. The primary outcome composite was all-cause mortality, nonfatal myocardial infarction, and nonfatal stroke.
Results:
Among 14,662 participants enrolled in ADAPTABLE with PAD status available, 3493 (23.8%) had PAD. Participants with PAD were more likely to experience the primary composite (13.76% vs 5.31%, p < 0.001), all-cause mortality (7.55% vs 3.01%, p < 0.001), myocardial infarction (5.71% vs 2.09%, p < 0.001), stroke (2.45% vs 0.86%, p < 0.001), and major bleeding (1.19% vs 0.44%, p < 0.001). A higher aspirin dose did not reduce the primary outcome in patients with PAD (13.68% vs 13.84% in 81 mg and 325 mg groups; OR 1.05, 95% CI 0.88–1.25). Participants with PAD were less likely to enroll via email (33.0% vs 41.9%, p < 0.0001), less likely to choose internet follow-up (79.2% vs 89.5%, p < 0.0001), and were more likely to change their aspirin doses (39.7% vs 30.7%, p < 0.0001).
Conclusions:
ADAPTABLE participants with PAD did not benefit from a higher dose of aspirin and participated in the study differently from those without PAD. These results reinforce the need for additional PAD-specific research and suggest that different trial strategies may be needed for optimal engagement of patients with PAD.
Keywords
Background
It has been well established that patients with both peripheral artery disease (PAD) and coronary artery disease (CAD) suffer more cardiovascular and limb events than patients with CAD alone. 1 Current guidelines recommend treatment with at least one antiplatelet drug (aspirin or a P2Y12 inhibitor) for secondary prevention in patients with PAD, but no major studies address the optimal aspirin dose for persons with PAD.2–4 The Aspirin Dosing: A Patient-Centric Trial Assessing Benefits and Long-Term Effectiveness (ADAPTABLE) study randomized persons with established atherosclerotic cardiovascular disease to 81 mg or 325 mg to determine which dose better protected against thrombotic and bleeding events. 5 Since a significant number of ADAPTABLE participants had PAD, it offers an opportunity to inform the question of what is the optimal aspirin dose in persons with PAD.
ADAPTABLE relied on electronic health record (EHR) data, patient self-report, health insurance claims, and a remote call center to make patient identification/enrollment and outcome ascertainment more pragmatic.6,7 ADAPTABLE represents an evolution in the conduct of clinical trials towards increased flexibility and efficiency in enrolling and following participants while also posing potential challenges in regards to protocol adherence and data quality. Patients with PAD are an understudied group who may particularly benefit from the increased flexibility and decentralization of pragmatic trials, in part because patients with PAD, are disproportionately sicker, and typically less educated, less socioeconomically secure, and more likely to be Black and from rural regions, all characteristics associated with lower likelihood of trial recruitment and enrollment in conventional contexts.8–15 Many of these same characteristics have also been associated with lower medication adherence in cardiometabolic and atherosclerotic conditions similar to PAD, suggesting that patients with PAD may be more likely to have protocol deviations.16–20 Thus, though pragmatic trials may offer an opportunity to enroll more, and (crucially) more representative, patients with PAD, a better understanding of the benefits and drawbacks of pragmatic studies in these patients is needed. Therefore, we sought to use data from ADAPTABLE both to assess the effect of aspirin dose on cardiovascular events among patients with PAD and to compare features of protocol adherence (completion of eligible study visits, duration on assigned dose, dose switching, and aspirin discontinuation) between patients with and without PAD. We hypothesized that patients with PAD would derive greater benefit from a higher dose of aspirin, and that patients with PAD would be less protocol adherent in a pragmatic trial setting.
Methods
Study design and population
The design and primary results of ADAPTABLE (ClinicalTrials.gov Identifier: NCT02697916) have previously been published.5,7 ADAPTABLE was an open-label, pragmatic, randomized controlled trial comparing the efficacy and safety of an aspirin dose of 81 mg a day with 325 mg a day for the prevention of cardiovascular events in patients with established atherosclerotic disease. Established atherosclerotic disease was defined as a history of myocardial infarction, coronary revascularization, coronary angiography demonstrating ≥ 75% stenosis of at least one epicardial coronary artery, or chronic CAD with at least one enrichment factor. Key exclusion criteria centered around contraindications to aspirin therapy, including current/planned treatment with oral anticoagulants or ticagrelor (clopidogrel and prasugrel use was permitted). For the purposes of this analysis, the presence of PAD (which was an enrichment criterion) in addition to CAD was defined as a documented history of PAD in the EHR (codes used in query available in online Supplemental Table 1). Data on PAD severity (i.e., asymptomatic, claudication, chronic limb-threatening ischemia) and on prior PAD-related interventions such as revascularization were not collected. Eligible patients were identified via computable phenotype queries at participating centers and recruitment proceeded via a combination of in-person and remote methods (e.g., letters, e-mails). Most participants completed follow-up procedures using an online portal in which they reported medication adherence and clinical events (which were subsequently confirmed). A limited number of participants were allowed to elect noninternet follow-up and were called by the Duke Clinical Research Institute call center instead (initially capped at 1000 participants, eventually expanded to 1904 participants). Internet follow-up participants who failed to complete follow-up visits could be ‘rescued’ with calls from the call center. All patients provided electronic informed consent and the institutional review boards at all participating centers approved the study.
Endpoints
The primary endpoint of ADAPTABLE was time to a composite of all-cause mortality, hospitalization for nonfatal myocardial infarction (MI), or hospitalization for nonfatal stroke. Secondary outcomes included the individual components of the primary endpoint. The primary safety outcome was major bleeding (hospitalization for bleeding requiring blood transfusion). Endpoints were ascertained via the online or telephone study follow-up contacts every 3 or 6 months (follow-up interval was a secondary randomization), EHR data, and public and private insurance claims data. For the purposes of this analysis, we also analyzed features of study conduct including completion of eligible study visits, duration on assigned dose, dose switching, and aspirin discontinuation. Trial medication adherence is based on patient report at visits every 3 or 6 months. The end of study visit was excluded from defining adherence because an observed spike in reports of dose switching at the End of Study visit seemed to reflect participants’ intended dose after trial participation. Dose switching was defined as reporting a dose of aspirin different from the randomized dose at one or more postrandomization trial encounters. Discontinuation was defined as reporting ‘No’ to the trial question ‘Are you regularly taking aspirin?’ at one or more postrandomization trial encounters or never reporting aspirin information during follow-up (missing visits after randomization).
Statistical methods
Patients were grouped by PAD status at baseline. Baseline characteristics included age, sex, race, ethnicity, and comorbidities. These characteristics were described using medians (IQR), means (SDs), and counts (proportions) as relevant. Continuous variables were compared using t-tests or Wilcoxon rank-sum and categorical variables using chi-squared tests.
Event rates were calculated at median follow-up (26.2 months) using the Kalbfleisch and Prentice cumulative incidence function estimator for patients with and without PAD by aspirin dose. 21 Endpoints were compared between patients with and without PAD using Cox proportional hazards models. Adjusted models for endpoints were constructed using candidate variables prespecified on the basis of clinical feasibility and included demographics, comorbidities, bleeding history, and prior aspirin and P2Y12 inhibitor use (online Supplemental Table 2).
The unadjusted and multivariable models were performed to assess the effect of dose on the primary endpoint and all-cause mortality among patients with PAD and the interaction between PAD status and aspirin dose. Covariates included in risk-adjusted models included age, ethnicity, strata, sex, race, prior aspirin use, P2Y12 use at baseline, smoking status, internet use at randomization, history of atrial fibrillation, history of bleeding, CAD, congestive heart failure (CHF), cerebrovascular disease (CVD), diabetes, hypertension, hyperlipidemia, and prior MI. These covariates were included based on increasing risk for the endpoint. Modeling assumptions of linearity and proportional hazards were tested. Transformations were included when necessary.
Trial participation metrics included recruitment approach, internet use, study withdrawal, dose adherence, and follow-up complete, and were compared between patients with and without PAD using t-tests or Wilcoxon rank-sum (continuous variables) and chi-squared tests (categorical variables).
Finally, time-dependent proportional hazard models were performed to examine if PAD modified the effect of self-reported aspirin dose on outcomes. Covariates were measured at baseline and included age, sex, race, ethnicity, prior aspirin dose, prior MI, prior percutaneous coronary intervention (PCI), history of atrial fibrillation, internet used at randomization, history of bleeds, and baseline P2Y12. Aspirin dose (self-reported at 81 mg, 325 mg, not taking, or other) was modeled as a time-varying exposure. Hazard ratios (HR) are interpreted as the effect of current aspirin dose on outcomes, with an assumption that the decision to switch doses is made for reasons unrelated to the outcome of interest, beyond the baseline adjustment measures. All hypothesis tests were two-sided, and p < 0.05 is interpreted as statistically significant without adjustment for multiple comparisons in this exploratory analysis. Analyses were performed using SAS software, Version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Study cohort and baseline characteristics
Of 14,662 participants enrolled in ADAPTABLE with EHR data available to inform PAD status, 3493 (23.8%) had a documented history of PAD and 11,169 (76.2%) did not. Randomization did not differ between participants with and without PAD (48.8%, n = 1706 of PAD randomized to 81 mg vs 50.3%, n = 5623 of non-PAD, p = 0.121). There were significant baseline differences between participants with and without PAD (Table 1). Participants with PAD were older (68.1 ± 9.7 vs 66.3 ± 9.8 years, p < 0.0001) and more likely to be Black (12.3% vs 7.7%, p < 0.0001), current smokers (13.1% vs 8.8%, p < 0.0001), and have each of the major comorbidities collected (p < 0.0001 for all). Participants with PAD were more likely to take a P2Y12 inhibitor at baseline (29.8%, n = 965 of PAD vs 19.7%, n = 2025 of non-PAD, p < 0.0001) and were slightly more likely to take aspirin at baseline (97.2%, n = 3209 of PAD vs 95.7%, n = 10,023 of non-PAD, p = 0.0002), without any difference in aspirin dose prior to randomization (p = 0.308).
Demographic and presenting characteristics, medical history, and medications.
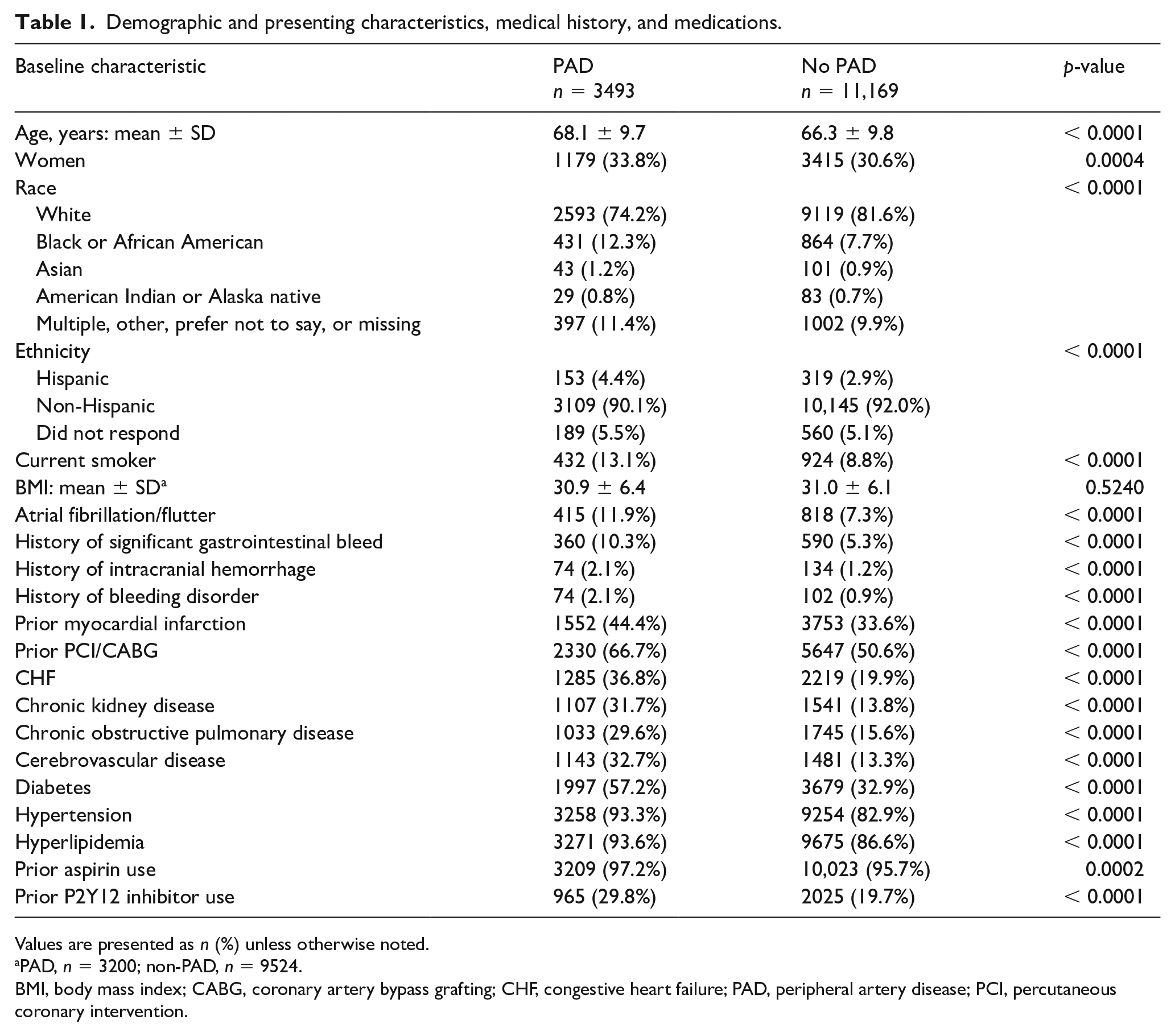
Values are presented as n (%) unless otherwise noted.
PAD, n = 3200; non-PAD, n = 9524.
BMI, body mass index; CABG, coronary artery bypass grafting; CHF, congestive heart failure; PAD, peripheral artery disease; PCI, percutaneous coronary intervention.
Clinical endpoints and interaction with aspirin dose
Over 26.2 months, participants with PAD were more likely to experience every endpoint, including the primary composite (13.76% vs 5.31%, p < 0.001), all-cause mortality (7.55% vs 3.01%, p < 0.001), MI (5.71% vs 2.09%, p < 0.001), and stroke (2.45% vs 0.86%, p < 0.001) (Table 2). These differences remained significant even after adjustment for demographics, comorbidities, and baseline P2Y12 inhibitor and aspirin use. After adjustment, participants with PAD had a 69% greater likelihood of experiencing the primary endpoint (adjusted hazard ratio (aHR) 1.69, 95% CI 1.48–1.93, p < 0.001), 77% greater likelihood of death (aHR 1.77, 95% CI 1.49–2.10, p < 0.001), 66% greater likelihood of having a MI (aHR 1.66, 95% CI 1.34–2.04), 48% greater likelihood of having a stroke (aHR 1.48, 95% CI 1.06–2.07, p = 0.02), and 132% greater likelihood of having major bleeding (aHR 2.32, 95% CI 1.53–3.53, p < 0.001).
Cumulative incidence of clinical endpoints among patients with and without PAD with unadjusted and adjusted hazard ratios.
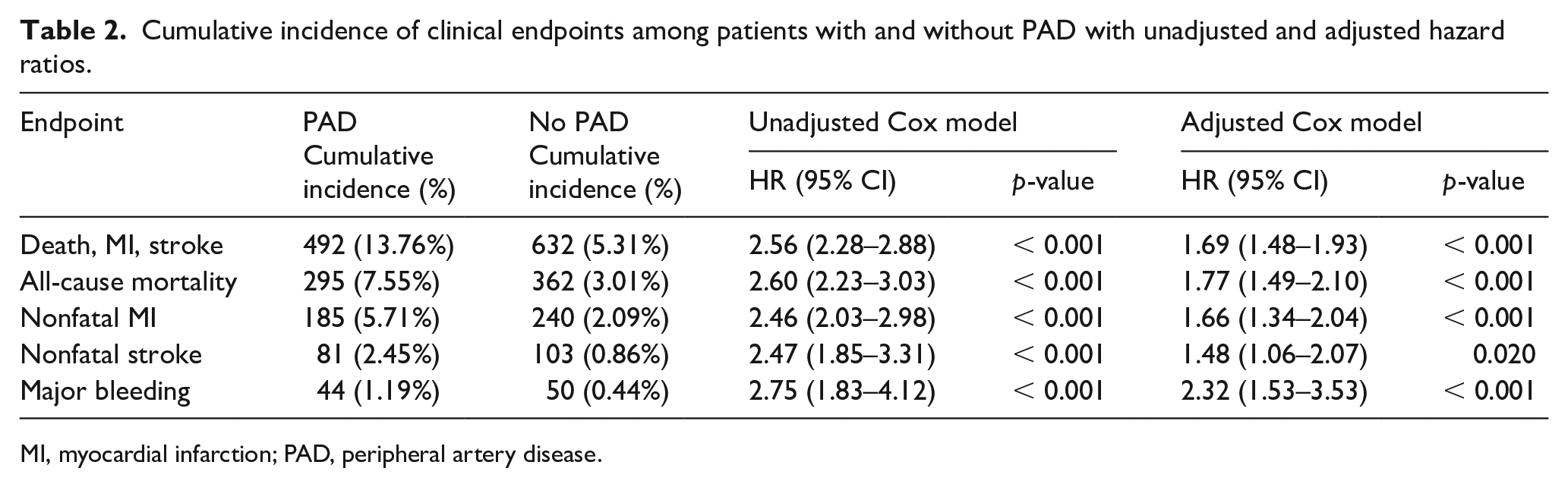
MI, myocardial infarction; PAD, peripheral artery disease.
Randomization to 325 mg of aspirin did not reduce occurrence of the primary composite endpoint or all-cause mortality among patients with PAD (Table 3). Among patients with PAD, the rate of the primary endpoint was 13.68% among participants randomized to 81 mg and 13.84% among those randomized to 325 mg (HR 1.05, 95% CI 0.88–1.25). The rate of all-cause mortality was 7.25% among those randomized to 81 mg versus 7.84% among those randomized to 325 mg (HR 0.94, 95% CI 0.75–1.18). The rate of major bleeding was 1.53% among those randomized to 81 mg and 0.80% among those randomized to 325 mg (HR 1.66, 95% CI 0.90–3.04). There was no significant interaction between PAD status and aspirin dose in regards to the primary composite, all-cause mortality, or major bleeding on either adjusted or unadjusted analyses.
Unadjusted and adjusted association between randomized aspirin dose and cumulative incidence of clinical endpoints in patients with and without PAD, with interaction between peripheral artery disease status and dose.
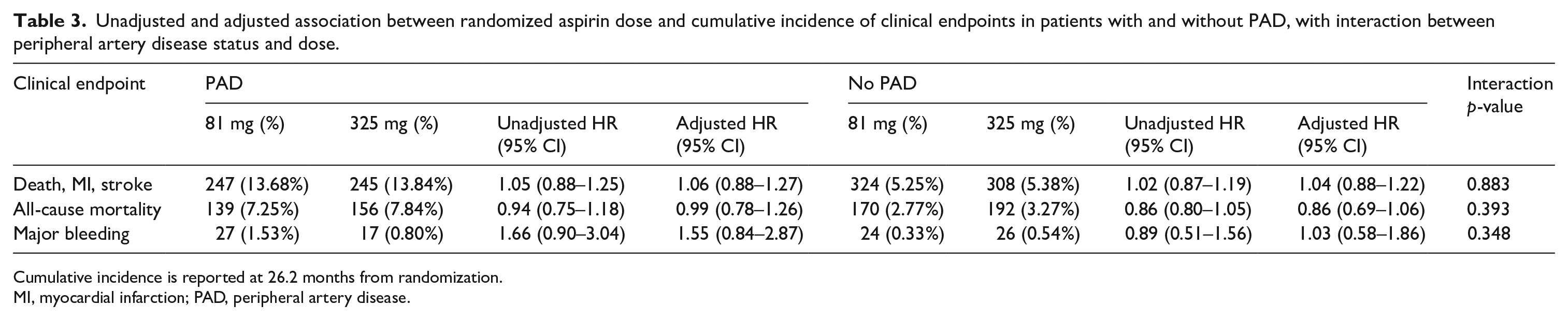
Cumulative incidence is reported at 26.2 months from randomization.
MI, myocardial infarction; PAD, peripheral artery disease.
ADAPTABLE study participation metrics
There were significant differences in the ways in which participants with and without PAD were recruited into and continued through ADAPTABLE. Participants with PAD were more likely to be recruited in a clinical setting (31.1% vs 23.6%, p < 0.0001) (Table 4) and less likely to have enrolled via email invitation (33.0% vs 41.9%, p < 0.0001) when compared with patients without PAD. Fewer participants with PAD elected for internet follow-up at baseline (79.2% vs 89.5%, p < 0.0001) and this proportion declined throughout the study such that by the last study visit, the gap in internet participation between PAD and non-PAD patients had widened (66.5% vs 78.7%, p < 0.0001). Participants with PAD completed a slightly lower proportion of study visits (PAD median 75.0% [IQR 57.1, 85.7] vs non-PAD median 77.8% [IQR 60.0, 85.7], p = 0.0175).
Study participation metrics.
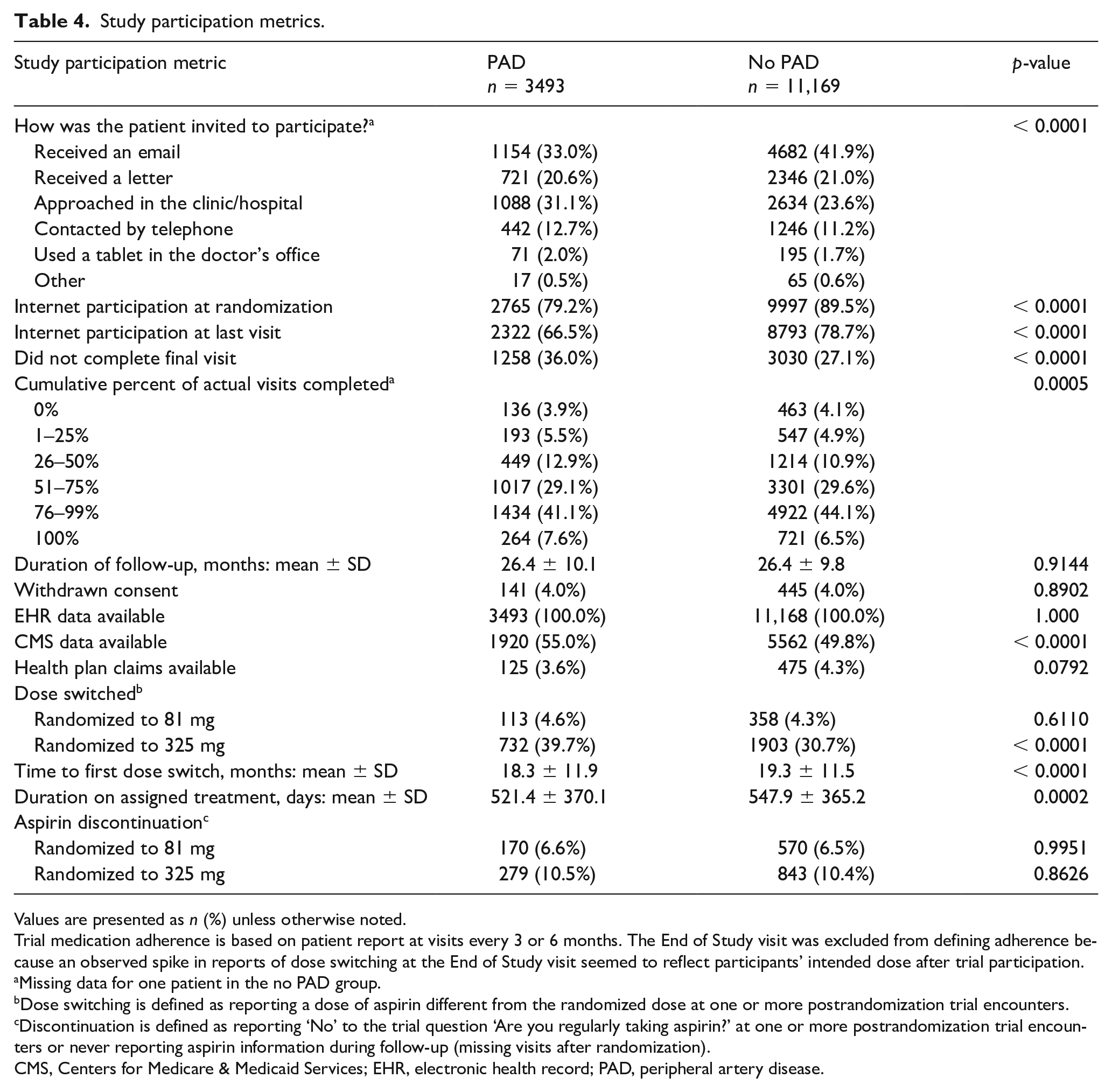
Values are presented as n (%) unless otherwise noted.
Trial medication adherence is based on patient report at visits every 3 or 6 months. The End of Study visit was excluded from defining adherence because an observed spike in reports of dose switching at the End of Study visit seemed to reflect participants’ intended dose after trial participation.
Missing data for one patient in the no PAD group.
Dose switching is defined as reporting a dose of aspirin different from the randomized dose at one or more postrandomization trial encounters.
Discontinuation is defined as reporting ‘No’ to the trial question ‘Are you regularly taking aspirin?’ at one or more postrandomization trial encounters or never reporting aspirin information during follow-up (missing visits after randomization).
CMS, Centers for Medicare & Medicaid Services; EHR, electronic health record; PAD, peripheral artery disease.
Participants with PAD were more likely to switch aspirin dose when randomized to 325 mg than were patients without PAD (39.7% vs 30.7%, p < 0.0001) and spent less time on their assigned treatment (521.4 ± 370.1 vs 547.9 ± 365.2 days, p = 0.0002), but were not any more likely to discontinue aspirin altogether, regardless of assigned dose. The time-varying effect of self-reported aspirin dose switching on event rates was not modified by the presence of PAD in either unadjusted or adjusted analysis (Table 5).
Effect of time-varying self-reported aspirin dose on outcomes, by PAD status.
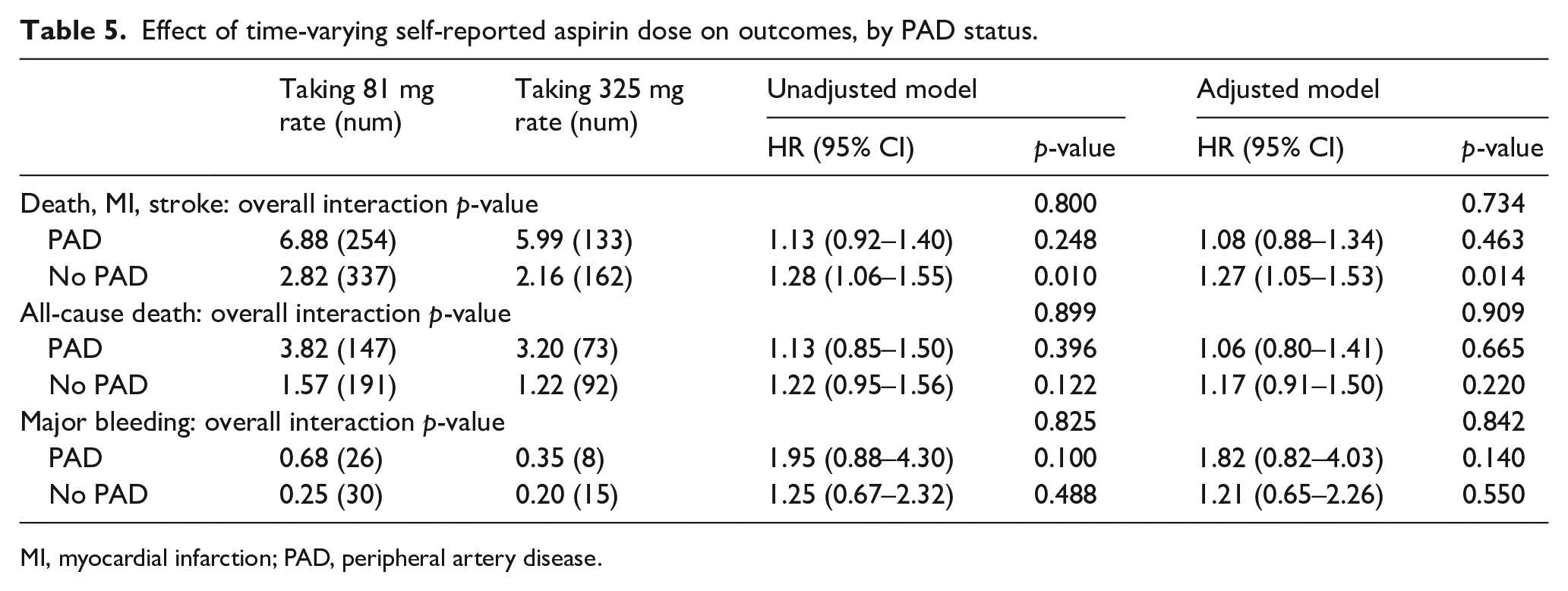
MI, myocardial infarction; PAD, peripheral artery disease.
Discussion
In this study, we compared the clinical effectiveness of aspirin doses among patients with PAD and metrics of participation in a pragmatic, randomized controlled trial between patients with CAD with and without concomitant PAD. We found that a higher aspirin dose did not reduce the occurrence of cardiovascular events among people with PAD, who had higher event rates than participants with CAD alone. We also demonstrated for the first time that participants with PAD are different in terms of their recruitment and retention in pragmatic trials, a finding with significant implications for future research in this population of patients with polyvascular disease.
Prior studies have demonstrated that participants with both CAD and PAD have more comorbidities and are more likely to suffer cardiovascular or limb events than are patients with CAD alone. 1 In the REACH and SMART registries, patients with disease in multiple territories were 50–75% more likely to experience cardiovascular mortality, stroke, or MI.22,23 Despite the increased risk of ischemic events in participants with PAD, we found no benefit of a higher aspirin dose when compared to low-dose aspirin, consistent with the overall findings in ADAPTABLE. At face value, this runs counter to subgroup analyses from other antithrombotic trials (such as PEGASUS, CHARISMA, PLATO, etc.) that suggested that patients with polyvascular disease randomized to increased-intensity antithrombotic therapy experienced similar or greater relative risk reductions compared to the trial cohorts more broadly, leading to greater absolute risk reductions. 1 However, those studies generally compared two antithrombotic medications to each other, rather than two doses of a single antithrombotic agent. 1
Although it is tempting to point to the greater comorbidity burden among participants with PAD as the reason for their worse outcomes, participants with PAD, remained at statistically and clinically significant increased risk of cardiovascular events even after adjustment for comorbidities. This suggests that other factors contributed to the observed differences in event rates. Severity and control of comorbidities is one possibility, as we controlled for the binary presence/absence of conditions rather than their severity. Differences in the adequacy of comorbidity management may also have contributed, as risk factor control may be worse in patients with PAD compared to those with CAD alone.24,25 Unfortunately, we were unable to control for social determinants of health, another set of factors strongly associated with outcomes that prior research suggests may be disproportionately disadvantageous among patients with PAD.15,26–28
Our findings support a view of PAD in addition to CAD as a condition different in both degree and kind from CAD alone, with different medical and socioeconomic stressors, less adequate treatment, and worse outcomes than CAD alone. Yet, PAD is relatively understudied and few data to support PAD guidelines come from high-quality PAD-specific studies, with much coming from subanalyses of CAD trials instead.8,29,30 When PAD-specific studies do exist, the study cohorts are not well representative of patients with PAD more broadly, under-enrolling women and non-White patients.31–34 This may be due, in part, to studies’ reliance on conventional recruitment and enrollment procedures that are centered around in-person medical visits, though women and non-White patients have been shown to have worse access to care.35,36 Therefore, decentralizing study conduct by directly contacting patients, as was done in ADAPTABLE, offers the possibility of reaching and enrolling some patients who might otherwise be overlooked. Further pragmatizing study conduct by reducing the burden of in-person study visits, another feature of ADAPTABLE, could also improve patients’ willingness and ability to participate.37–39 This idea has gained even more currency during the COVID-19 pandemic, during which many study activities shifted remotely as a matter of necessity.40–42
However, this analysis of ADAPTABLE suggests that a one-size strategy may not fit all. A greater proportion of participants with PAD were recruited in a clinic/hospital setting, likely related both to their greater comorbidity burden as well as to their lower internet use. Given well-established disparities in access to care, this finding raises questions about the patients who both did not make it to clinic/hospital settings and who do not have/use the internet.35,36 Even with a multipronged approach to recruitment, patients with PAD enrolled in ADAPTABLE were less commonly women and less commonly Black than the United States PAD population.15,28,33 Participants with PAD were also less likely to choose internet-based follow-up, though it is difficult to know whether this was due to disparities in internet access or to personal preference. In addition, noninternet participation was initially capped before being expanded due to high demand, which may have either discouraged some eligible patients from enrolling or pushed some participants to internet follow-up despite discomfort with using the web. This suggests that more decentralized study designs relying more heavily on the internet may require modification to be successful in cohorts of patients with demographics and comorbidities similar to patients with PAD – or that patients recruited into heavily internet-reliant studies might not be representative of broader cohorts – because of the overlap between people more likely to have PAD and people less likely to have internet access. 43 Called ‘the digital divide’, this disparity in access to the internet has been attributed to discriminatory infrastructure decision-making and high prices for internet access, among other contributing factors. 44
At the same time, participants with PAD completed a slightly lower proportion of study follow-up and were somewhat more likely to switch their aspirin dose when randomized to 325 mg than patients with CAD alone. These observations may be due to other stressors, events (hospitalizations, revascularizations), or medication changes (addition of other antiplatelet/anticoagulant agents) experienced more commonly by patients with PAD than those without. It was reassuring to note that patients with PAD appeared no more likely to discontinue aspirin than patients without PAD, though for both groups, discontinuation was more common among patients randomized to 325 mg. Unfortunately, reasons for dose switching or discontinuation were not available for analysis; this is a facet of optimal medical therapy in need of further exploration.
There are few data comparing explanatory versus pragmatic trial methodologies in patients with cardiovascular disease (and none in PAD),45,46 but there are data suggesting that pragmatic features of study design are gaining popularity. 47 Certainly, efforts to overcome barriers to recruitment, retention, and representation through multipronged recruitment strategies and reducing participant burden are to be lauded, but our analysis of ADAPTABLE shows that these efforts must be tailored for each patient population. 48 In the absence of evidence to guide implementation of pragmatic study designs, patient input is necessary. 48 ADAPTABLE was innovative in its robust inclusion of a group of patient advisors who provided input to modify study recruitment materials and policies, but their role was to guide implementation of established protocols rather than to contribute to creation of those protocols. 49 Ideally, future studies should build on the lessons from ADAPTABLE in regards to pragmatic study design and patient engagement in order to be even more inclusive and relevant for patients.
Study limitations
There were several limitations of this analysis, predominantly related to difficulties with fully characterizing patients with PAD and their care within a study designed to capture data about aspirin in atherosclerotic disease more broadly. For instance, we were not able to incorporate PAD severity due to the manner in which data were ascertained from participating health systems (i.e., via EHR data queries in the form of the PCORnet Common Data Model). As previously noted by other authors, the most sensitive diagnostic codes for identifying PAD in structured data are the most general ones, which do not specify subtype of PAD. 50 It may be that patients with intermittent claudication were more likely to be recruited via email or that dose switching was disproportionately seen among patients with chronic limb-threatening ischemia but we were not able to capture any such patterns. Similarly, we were not able to report rates of limb events or analyze the effect of aspirin on limb events. Also, we were not able to fully characterize the relationship between baseline P2Y12-inhibitor use, event rates, and aspirin dose – though we controlled for P2Y12-inhibitor use in our analyses, we did not look for interactions with aspirin dose. Finally, we were not able to report subtypes of bleeding events to better understand the increased risk for them among patients with PAD, though the absolute number of such events was low.
Conclusions
This analysis demonstrated that patients with both CAD and PAD do not benefit from a higher dose of aspirin and that they are different from those with CAD alone in regards to their participation in clinical trials. These differences are interconnected, as sociodemographic and comorbid factors likely affect clinical outcomes, and all together influence patients’ abilities to join and continue in research studies. Pragmatic elements of clinical trials such as direct-to-participant recruitment and remote follow-up hold potential to improve study cohort representativeness and reduce study burden, but our analysis suggests that different patient subgroups may experience these pragmatic innovations in different ways. Though ADAPTABLE robustly engaged patients as partners during study conduct, future trials may benefit from patient guidance in earlier phases, including study design, in order to ensure that features intended to improve cohort diversity and reduce participant burden have the intended effects.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231154951 – Supplemental material for Outcomes among patients with peripheral artery disease in the Aspirin Dosing: A Patient-Centric Trial Assessing Benefits and Long-Term Effectiveness (ADAPTABLE) study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231154951 for Outcomes among patients with peripheral artery disease in the Aspirin Dosing: A Patient-Centric Trial Assessing Benefits and Long-Term Effectiveness (ADAPTABLE) study by E Hope Weissler, Amanda Stebbins, Lisa Wruck, Daniel Muñoz, Kamal Gupta, Saket Girotra, Jeff Whittle, Catherine P Benziger, Tamar S Polonsky, Steven M Bradley, Bradley G Hammill, James G Merritt, Doris N Zemon, Adrian F Hernandez and W Schuyler Jones in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
E Hope Weissler reports research support from National Institutes of Health (NIH) and National Heart, Lung, and Blood Institute (F32HL151181). Saket Girotra reports research support from NIH (R56HL158803 and R01HL160734) and funding support from the American Heart Association for editorial work. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
