Abstract

Keywords
Patients with established atherosclerosis, particularly peripheral artery disease (PAD), are at heightened risk of atherothrombotic events including acute myocardial infarction (MI), stroke or cardiovascular death, as well as risk of major adverse limb events such as acute limb ischemia (ALI).1–3 Aging is a well-established risk factor for atherothrombotic complications. 4 However, advanced age is also associated with an increased risk of bleeding, including antiplatelet-related intracranial hemorrhage (ICH), and there is an incremental risk of bleeding with each additional antithrombotic agent utilized. 5 Vorapaxar, a novel oral antiplatelet agent that antagonizes activation of the protease-activated-receptor-1 (PAR-1) by thrombin, reduced major adverse cardiovascular and limb events in patients with established atherosclerosis in the Thrombin Receptor Antagonist in Secondary Prevention of Atherothrombotic Ischemic Events (TRA 2°P) TIMI 50 multinational, randomized, double-blind, placebo-controlled trial (NCT00526474).6–8 Vorapaxar is approved for secondary prevention of atherothrombosis in patients with symptomatic PAD and is one of few options labeled for use in this population.6,7 Current guidelines give vorapaxar a class IIb recommendation noting the importance of weighing ischemic benefit against bleeding risk in selecting patients. 9
In the present analysis, we analyzed the ischemic benefits and bleeding risks with vorapaxar in TRA 2°P-TIMI 50 in patients with PAD with respect to age. The details of the trial design and primary results have been previously reported.8,10 This analysis included all patients with PAD inclusive of those with either known symptomatic disease (history of claudication, prior revascularization or amputation) and asymptomatic patients with an abnormal ankle–brachial index (< 0.90). As vorapaxar is contraindicated in patients with a history of TIA or stroke, our analysis included only patients without a known cerebrovascular event prior to randomization. Given the previously reported interaction with body weight, this analysis was also restricted to patients with a body weight ⩾ 60 kg.8,11 All end points were adjudicated according to prespecified standard definitions by members of an independent Clinical Endpoints Committee (CEC) who were blinded to treatment assignment. Major adverse cardiovascular events (MACE) were comprised of cardiovascular death, myocardial infarction, or stroke. Major adverse limb events (MALE) were comprised of hospitalization for acute limb ischemia, peripheral revascularization or amputation. Bleeding was assessed using the Global Use of Strategies to Open Occluded Coronary Arteries (GUSTO) and the International Society on Thrombosis and Haemostasis (ISTH) classification systems. Net clinical outcome (NCO) for the PAD analysis was comprised of MACE, MALE, and GUSTO severe bleeding. 6 Event rates were calculated as Kaplan–Meier estimates at 3 years.
Of 5608 patients eligible for this analysis, 2958 (52.7%) were < 65 years old and > 2500 were ⩾ 65 years (age 65 to < 75, 32.4%; 75–80, 10.7%; > 80, 4.1%). Patients aged ⩾ 75 years had a higher prevalence of hypertension (82.7% vs 75.6%), prior known symptomatic PAD (81.5% vs 74.9%) and cerebrovascular disease (18.8% vs 11.0%); they were less likely to be current smokers (11.8% vs 31.0%). Nearly all patients (93.1%) were treated with aspirin. Thienopyridine use was less common in patients ⩾ 75 years old (47.7% vs 56.2%). The rate of MACE or MALE for patients randomized to placebo was 13.7%, 17.3%, and 23.2% in patients aged < 65, 65 to < 75, and ⩾ 75, respectively. The relative risk reduction with vorapaxar versus placebo for MACE and MALE was consistent across age groups (hazard ratio (HR) 0.85, HR 0.74, and HR 0.80; p-interaction = 0.74; Figure 1C). However, given the increased absolute risk with increasing age, the absolute risk reduction tended to be greater for older patients (2.4%, 4.4%, and 4.1%). Bleeding tended toward an increase with vorapaxar for GUSTO moderate to severe bleeds (3.9% vs 2.0%, HR 1.98 [1.22–3.21] [95% CL]; 6.7% vs 5.8%, HR 1.23 [0.82–1.86]; 9.7% vs 8.2%, HR 1.22 [0.73–2.06]; p-interaction = 0.28), GUSTO severe bleeds (Figure 1B), and ISTH major bleeds (Figure 1A). However, the net clinical outcome comprised of MACE, MALE, and GUSTO severe bleeding was favorable across the entire range of ages (Figure 1D). ICH was uncommon across all age groups (0.5% vs 0.3%, HR 1.56 [0.44–5.52]; 1.1% vs 1.0%, HR 1.11 [0.40–3.06]; 0.3% vs 0.6%, HR 0.55 [0.05–6.01]; p-interaction = 0.74).
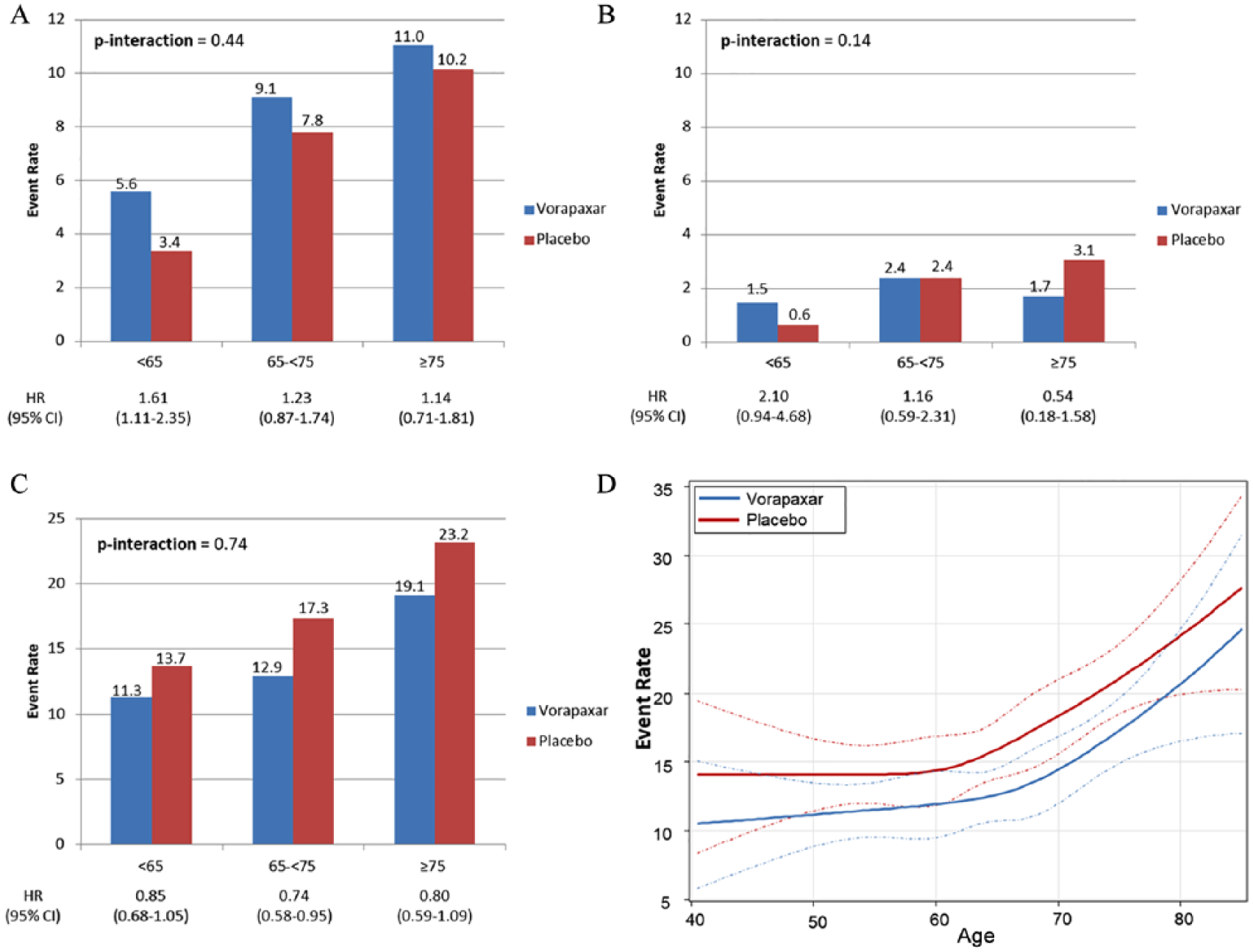
Kaplan–Meier event rates stratified by age for (A) ISTH major bleeding; (B) GUSTO severe bleeding; (C) composite of MACE or MALE: cardiovascular death, myocardial infraction, stroke, acute limb ischemia, peripheral revascularization, or amputation; (D) net clinical outcome comprised of MACE, MALE or GUSTO severe bleeding.
In conclusion, age is associated with an increased risk of ischemic events and bleeding. Vorapaxar had a favorable net clinical outcome across all age groups in patients with PAD.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Morrow – consulting fees from Abbott Laboratories, Aralez Pharmaceuticals, Bayer Pharmaceuticals, GlaxoSmithKline, Merck, Peloton, Roche Diagnostics, and Verseon; grants from Abbott, Amgen, AstraZeneca, Eisai, GlaxoSmithKline, Johnson & Johnson, The Medicines Company, Merck, Novartis, Pfizer, Roche Diagnostics, and Takeda. Dr Scirica – personal fees from AstraZeneca, Biogen Idec, Boehringer-Ingelheim (BI), Covance, Dr. Reddy’s Laboratories, Eisai, Elsevier Practice Update Cardiology, GlaxoSmithKline, Health [at] Scale, Lexicon, Merck, NovoNordisk, Sanofi, and St Jude’s Medical; grants from AstraZeneca, Eisai, Gilead, Novartis, and Poxel. Dr Bonaca – consulting fees from Aralez, AstraZeneca, Bayer, Janssen, Johnson & Johnson, and Merck; grants from AstraZeneca, MedImmune, Merck, and Pfizer. Remaining authors – none.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the TRA 2°P-TIMI 50 trial was supported by a grant from Merck & Co. to the TIMI Study Group. The study sponsor had no role in the analysis/interpretation of these data or in manuscript preparation.
