Abstract
Patients with chronic kidney disease may develop new or more severe anemia when treated with antiplatelet agents due to blood loss in conjunction with impaired erythropoiesis. Because anemia independently predicts limb amputation and mortality among patients with peripheral artery disease (PAD), we evaluated the relationship between estimated glomerular filtration rate (eGFR) and hemoglobin (Hb) levels in the EUCLID trial in which patients with symptomatic PAD were randomized to ticagrelor or clopidogrel. At baseline, 9025, 1870, and 1000 patients had eGFR ⩾ 60, 45–59, and < 45 mL/min/1.73 m2, respectively. The mean fall in Hb during the trial was 0.46 ± 1.68 g/dL and did not differ by baseline eGFR category, although Hb fall ⩾ 10% was more frequent among patients with lower eGFR (p for trend < 0.0001). On-study treatment with iron, erythropoiesis-stimulating agents, and/or red blood cell transfusion was reported for 479 (5.3%), 165 (8.8%), and 129 (12.9%) patients in the three eGFR categories, respectively (p for trend < 0.0001). After adjustment for baseline and post-randomization effects, those not receiving anemia treatment had a smaller reduction in Hb from baseline than those receiving anemia treatment (p < 0.0001). Other determinants of Hb reduction included absence of on-study myocardial infarction, coronary or peripheral revascularization, residence outside North America, male sex, and baseline eGFR. We conclude that among patients with PAD treated with P2Y12 inhibitors, lower baseline eGFR was associated with a greater reduction in Hb.
Keywords
Introduction
Antiplatelet monotherapy is recommended for prevention of thrombotic cardiovascular (CV) events in patients with symptomatic peripheral artery disease (PAD). 1 More intense antiplatelet therapy has been associated with increased bleeding risk; hemoglobin (Hb) decrease is clinically meaningful in that anemia independently predicts amputation and death among patients with PAD. 2 PAD in conjunction with renal dysfunction confers even greater risk as anemia is more frequent among patients with impaired renal function taking P2Y12 inhibitors. 3
The Effects of Ticagrelor and Clopidogrel in Patients with Peripheral Artery Disease (EUCLID) trial evaluated CV events and bleeding in patients with symptomatic PAD randomized to ticagrelor or clopidogrel. 4 The event rate for the composite endpoint of CV death, myocardial infarction (MI) or ischemic stroke was 6.75/100 patient-years among patients with estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2 compared with 3.72/100 patient-years for those with eGFR ⩾ 60. 5 Minor bleeding was also more frequent in the group with chronic kidney disease (CKD) (adjusted hazard ratio (HR) 1.51, 95% CI: 1.07–2.15; p = 0.02), although major bleeding did not differ significantly (adjusted HR 1.21, 95% CI: 0.89–1.64; p = 0.22). EUCLID participants with CKD might be expected to have lower Hb resulting from impaired erythropoiesis and increased bleeding. Because of the prognostic implications of anemia in patients with PAD, we evaluated the hypothesis that patients with lower eGFR would have a greater fall in Hb level during long-term antiplatelet therapy in EUCLID.
Methods
This is a post hoc analysis of the EUCLID trial (ClinicalTrials.gov identifier: NCT01732822). The study design and primary results have been previously reported. 6 Briefly, EUCLID was a randomized, double-blind trial comparing ticagrelor versus clopidogrel in patients with symptomatic PAD. Between December 2012 and March 2014, patients were randomized (1:1) to receive ticagrelor, 90 mg twice daily, or clopidogrel, 75 mg once daily; follow-up continued until May 2016. Eligible patients were aged 50 years or older and had symptomatic PAD. Exclusion criteria included current or planned use of dual antiplatelet therapy, two loss-of-function cytochrome P450 enzyme 2C19 allele variants, need for long-term anticoagulation, elevated bleeding risk, and active malignancy other than basal or squamous cell skin cancer. All patients provided written informed consent, and the trial protocol was reviewed and approved by appropriate ethics committees at participating sites. The local laboratory Hb value was recorded at baseline and end of study. The EUCLID publication committee approved this post hoc analysis.
Statistical analysis
The analysis dataset included all randomized patients with baseline eGFR and Hb values and an end of study Hb value. Ticagrelor and clopidogrel treatment were combined for analysis unless otherwise specified. Patients were categorized by baseline eGFR (⩾ 60, 45–59, and < 45 mL/min/1.73 m2) using the Modification of Diet in Renal Disease equation. 7
Continuous variables are presented as means with SDs. Categorical variables are reported as counts and percentages. Analyses were performed according to the intention-to-treat principle. A one-way analysis of variance (ANOVA) was used to assess if there was a significant difference between the mean change from baseline to end of study Hb values across the three eGFR groups. Tests for trends across the three eGFR groups for continuous variables were conducted by testing the slope; categorical variables were conducted using the Cochran–Armitage trend test. Anemia treatment was defined as treatment with therapeutic doses of iron or with an erythropoiesis-stimulating agent or red blood cell transfusion recorded on the case report form. The difference in the proportion of patients receiving anemia treatment across the three eGFR categories was assessed by chi-squared test.
An ANCOVA model was used to assess the relationship between categorized baseline eGFR and Hb change from baseline. The model included the change from baseline to end of study Hb value as the dependent variable, and the following variables as covariates: baseline Hb, baseline eGFR, sex, age, number of days between baseline and end of study Hb values, occurrence of an on-study MI, coronary or peripheral revascularization procedure (yes/no), a variable reflecting whether the subject received concomitant iron therapy, erythropoiesis-stimulating agent therapy and/or a transfusion during the study, history of cancer (yes/no), an adjudicated Thrombolysis in Myocardial Infarction (TIMI) major bleeding event (yes/no), randomized treatment assignment, and geographic region.
All statistical analyses were performed by the CPC Clinical Research using SAS software, version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
The EUCLID trial randomized 13,885 participants with symptomatic PAD; this analysis includes the 11,895 participants with recorded baseline eGFR and Hb levels and an end of study Hb value. The interval between baseline and final Hb values was 2.4 ± 0.7 years (mean ± SD). Baseline characteristics of subjects included in this analysis are summarized (Table 1). Participants were categorized by eGFR at baseline: 1000 had an eGFR < 45 mL/min/1.73 m2, 1870 had an eGFR 45–59, and 9025 had an eGFR ⩾ 60. Those with lower eGFR were older, more often female, less likely to be current smokers, and had higher prevalence of diabetes mellitus, hypertension, and prior MI/peripheral or coronary revascularization (all p < 0.0001). The baseline Hb was lower among patients with lower eGFR at baseline.
Baseline characteristics.
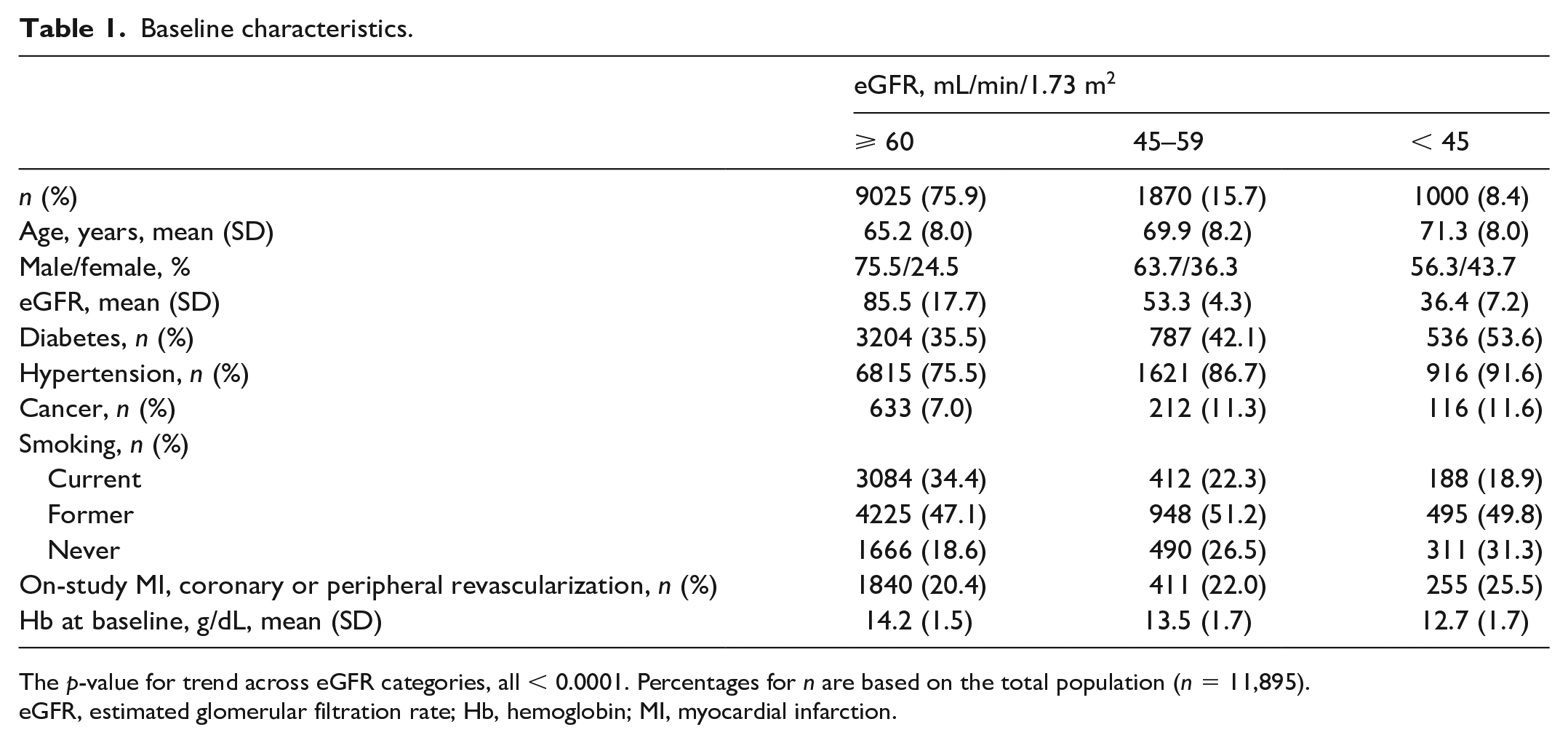
The p-value for trend across eGFR categories, all < 0.0001. Percentages for n are based on the total population (n = 11,895).
eGFR, estimated glomerular filtration rate; Hb, hemoglobin; MI, myocardial infarction.
Mean Hb change during follow-up did not differ across eGFR categories, −0.44 ± 1.66, −0.50 ± 1.67, and −0.48 ± 1.84 g/dL for eGFR ⩾ 60, 45–59, and < 45 mL/min/1.73 m2, respectively (mean ± SD, p for trend = 0.25). Hb at the end of the study was lower than at baseline in 59% of participants; the proportion with a < 10% fall in Hb during follow-up was similar across baseline eGFR categories. However, a fall in Hb ⩾ 10% was more frequently observed among participants with lower baseline eGFR (p for trend < 0.0001; Figure 1 and online supplementary Table).
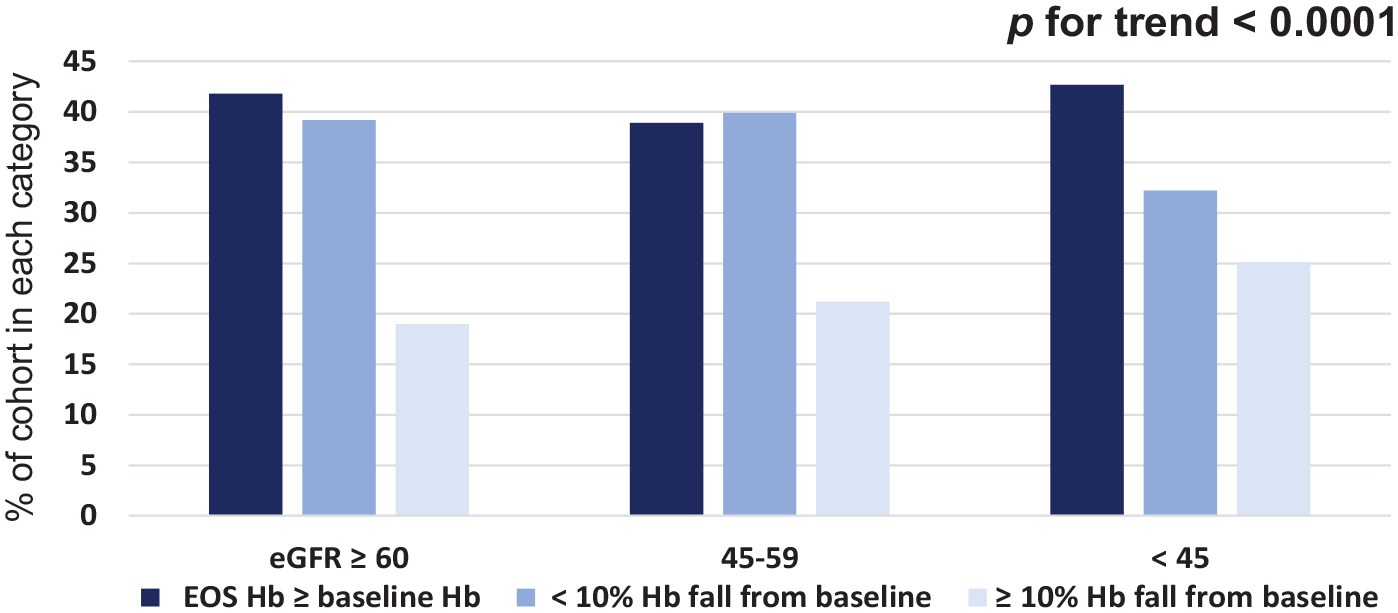
Hemoglobin change (unadjusted) from baseline by eGFR category.
Anemia treatment was recorded for 6.5% of the overall cohort; 5.0% received a red blood cell transfusion, 2.3% received a therapeutic dose of iron therapy, and 0.2% an erythropoiesis-stimulating agent. The proportion of patients receiving each type of anemia treatment increased with lower eGFR (Figure 2). Of the 600 participants receiving a red blood cell transfusion, 20% reported a TIMI major bleeding event and 80% did not. The duration of iron therapy was 824 ± 1156 days (mean ± SD), including treatment initiated before study enrollment; the duration of the erythropoiesis-stimulating agent therapy was 317 ± 379 days.
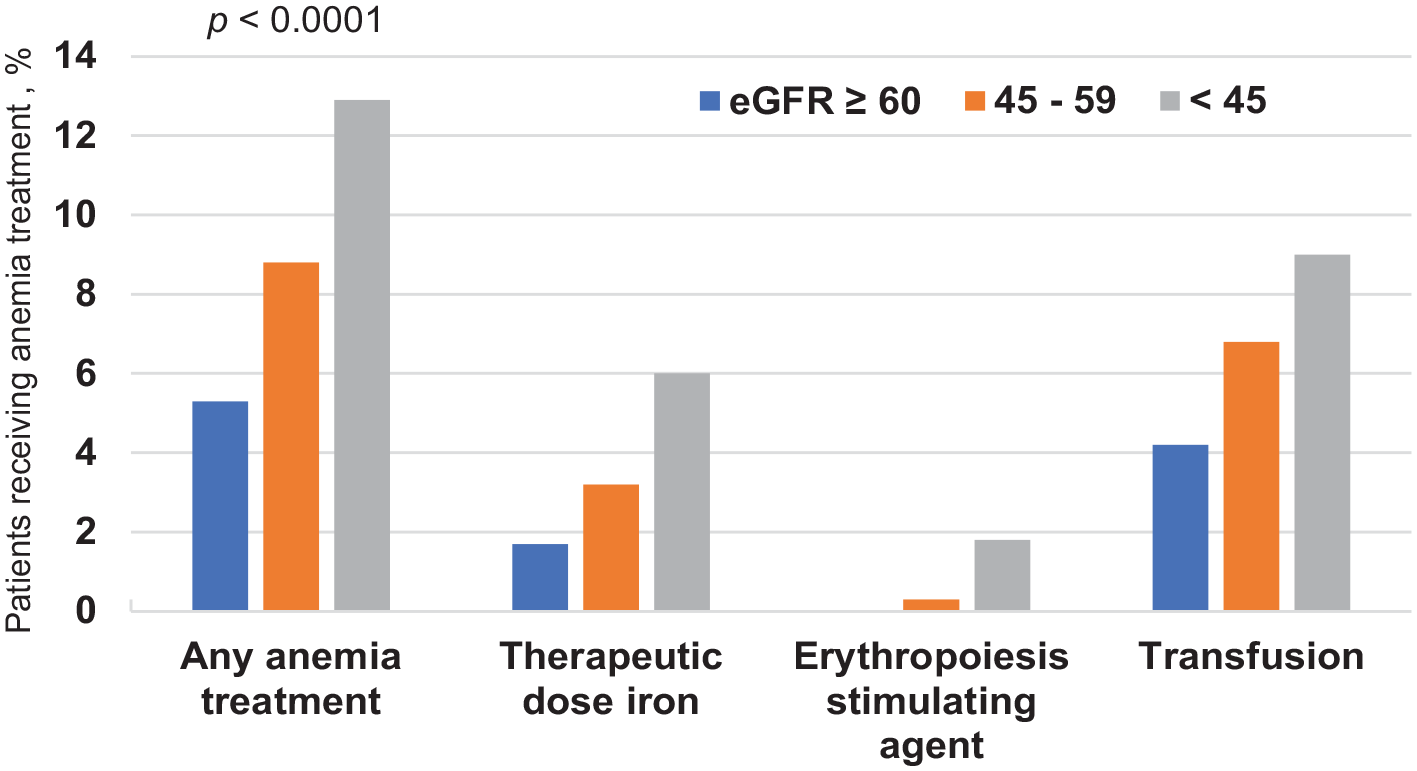
Anemia treatment by eGFR category.
After adjustment for baseline and post-randomization effects, those not receiving anemia treatment had a smaller reduction in Hb from baseline than those receiving anemia treatment (p < 0.0001). Other independent but more moderate associations with change in Hb included absence of on-study MI, coronary or peripheral revascularization, residence outside North America, and male sex. Participants without an on-study clinical event, those residing outside North America, and men had a smaller reduction in Hb compared to those with an on-study clinical event, those residing in North America, and women. Lower eGFR at baseline had an independent, albeit modest association with Hb fall, whereby participants with a lower baseline eGFR had a greater reduction in Hb compared to those with a higher baseline eGFR. Randomization to clopidogrel, the number of days from baseline to end of study Hb, age, absence of on-study major bleeding events, and absence of cancer history were not observed to be associated with a meaningful change in Hb (Table 2).
Determinants of Hb change from baseline.
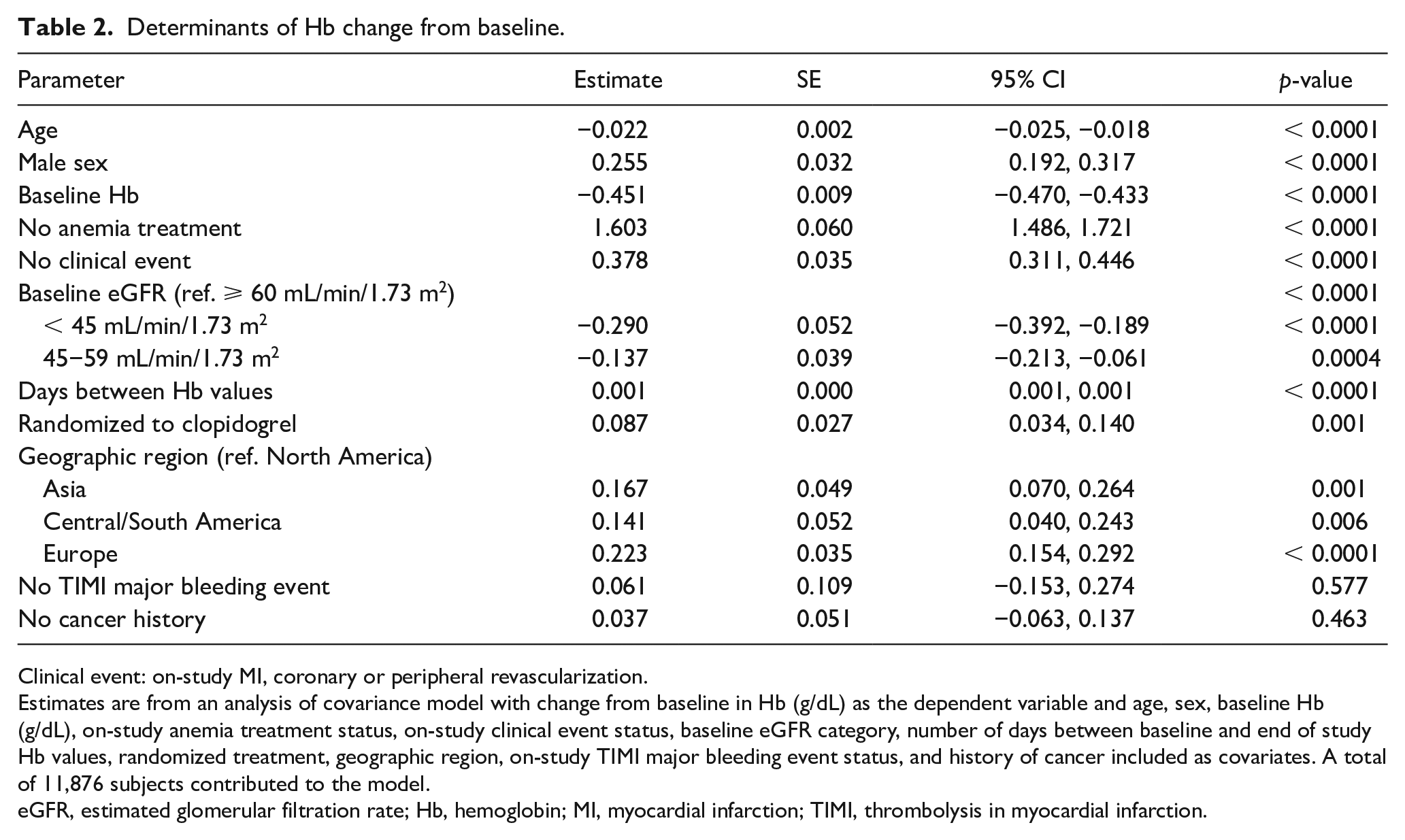
Clinical event: on-study MI, coronary or peripheral revascularization.
Estimates are from an analysis of covariance model with change from baseline in Hb (g/dL) as the dependent variable and age, sex, baseline Hb (g/dL), on-study anemia treatment status, on-study clinical event status, baseline eGFR category, number of days between baseline and end of study Hb values, randomized treatment, geographic region, on-study TIMI major bleeding event status, and history of cancer included as covariates. A total of 11,876 subjects contributed to the model.
eGFR, estimated glomerular filtration rate; Hb, hemoglobin; MI, myocardial infarction; TIMI, thrombolysis in myocardial infarction.
Discussion
Among patients with symptomatic PAD taking antiplatelet therapy in the EUCLID trial, those with lower eGFR had lower Hb levels at baseline: 14.2 versus 12.7 g/dL for patients with eGFR ⩾ 60 versus < 45 mL/min/1.73 m2. The magnitude of Hb reduction observed during follow-up was similar across baseline eGFR categories; however, an Hb fall ⩾ 10% and anemia treatment were more frequent among those with a lower baseline eGFR. In multivariable analysis, reductions in Hb were associated with absence of anemia treatment, on-study MI or revascularization, residence outside North America, male sex, and baseline eGFR.
This is a post hoc analysis from a large clinical outcomes trial in which bleeding was a safety focus. Major bleeding was assessed using the TIMI, PLATelet inhibition and patient Outcomes (PLATO), Bleeding Academic Research Consortium (BARC), and International Society on Thrombosis and Haemostasis (ISTH) criteria 8 ; for each of which the magnitude of Hb decrease can define a major bleeding event. 9 Some definitions also include red blood cell transfusion as a criterion for major bleeding. Consequently, baseline and end of study Hb as well as transfusion and concomitant medications were collected as protocol-specified variables in EUCLID.
The mean Hb fall during EUCLID approximated half a unit of blood per participant. Overall, 280 EUCLID participants (2.4%) reported TIMI major or minor bleeding events, 6 of which 218 (78%) occurred among the 2661 patients who underwent revascularization or lower extremity amputation procedures. 10 Blood sampling for the study was minimal. The magnitude of Hb loss (0.46 g/dL) was consistent with that observed in a cohort of patients with vascular disease initiating antiplatelet therapy, in whom Hb fell 0.31 g/dL during the 35-day mean follow-up. 11 Among patients with PAD in the control groups of studies which included 6-minute walk testing, walking distance fell in all four studies during the 6-month follow-up. 12 Hb loss could be speculated to contribute to the functional deterioration along with disease progression and aging. Taken together with the established relationship between Hb fall, amputation, and death, 2 our observation that Hb fell by at least 10% in one-fifth of patients in EUCLID suggests that systematic anemia identification and treatment could favorably impact clinical and functional outcomes in patients with PAD.
A consistent pattern of anemia treatment was observed in which treatment was more common among patients with lower eGFR for transfusion, iron replacement and erythropoiesis-stimulating agent therapy, and for the composite of any anemia treatment. Our observation that patients not receiving anemia treatment had a smaller reduction in Hb from baseline than those receiving anemia treatment may reflect underlying impaired erythropoiesis among those requiring anemia treatment. Anemia is associated with poorer outcomes in patients with PAD,2,13 although it is uncertain whether anemia treatment improves outcomes in this population. Iron replacement to treat iron deficiency anemia due to blood loss is broadly recommended for patients with or without CKD,14,15 whereas use of erythropoiesis-stimulating agents and transfusion requires individualized assessment of risks and benefits. 16
Limitations of this analysis include the small proportion of participants with CKD stage 4 or 5 (n = 237, 2%) and the fact that albuminuria was not systematically recorded and therefore is not included in the analysis. In addition, this analysis excluded 14% of EUCLID participants who had missing eGFR or Hb values. Among the 1900 patients excluded from the analysis population, 532 died during the study, all of whom were missing either baseline Hb or eGFR, so the analysis was not confounded by missing end of study Hb due to death. Demographics and CV risk characteristics of the analysis population were similar to the entire cohort.
Among patients with PAD taking antiplatelet therapy in the EUCLID trial, those with lower eGFR had lower Hb levels at baseline. In multivariable analysis, lower eGFR was independently associated with a greater Hb fall during the trial. Because anemia is associated with poorer outcomes, including amputation and death, in this population, monitoring of Hb should be considered. Studies are needed to assess whether anemia treatment and which types of treatment affect clinical event rates in this high-risk population.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X211017641 – Supplemental material for Impact of chronic kidney disease on hemoglobin among patients with peripheral artery disease treated with P2Y12 inhibitors: Insights from the EUCLID trial
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X211017641 for Impact of chronic kidney disease on hemoglobin among patients with peripheral artery disease treated with P2Y12 inhibitors: Insights from the EUCLID trial by Judith Hsia, Sarah T Kavanagh, Charles W Hopley, Iris Baumgartner, Jeffrey S Berger, Gerry R Fowkes, W Schuyler Jones, Kenneth W Mahaffey, Lars Norgren, Manesh R Patel, Frank Rockhold, Juuso Blomster, Brian G Katona, William R Hiatt and Marc P Bonaca in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs Bonaca, Hiatt, Patel, Rockhold, Fowkes, and Jones received grant support from AstraZeneca. Drs Mahaffey, Berger, Baumgartner, and Norgren received fees for consulting or other services from AstraZeneca. Dr Hsia owns AstraZeneca stock. Drs Katona and Blomster were employed by AstraZeneca during the conduct of the study. Ms Kavanagh receives consulting fees from UCB Pharma, AveXis, Karuna Therapeutics, Zosano Pharma, and Worldwide Clinical Trials.
Funding
The EUCLID trial was sponsored by AstraZeneca. The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
