Abstract

Keywords
Cardiovascular disease remains the leading cause of death worldwide. Recognition of the burden of cardiovascular risk has driven research into therapies that can reduce ischemic outcomes. A key axis of prevention for cardiovascular risk is antithrombotic therapies, which reduce risk of atherothrombosis at the cost of increased risk of bleeding. The associated risks as well as costs of these therapies limit broad application, with use of more intensive strategies limited to secondary prevention populations. Even within these populations, however, there is growing recognition that further risk stratification may allow for greater personalization of treatment strategies, with prescription for those patients at the highest risk who have the most favorable risk/benefit ratio. 1 It is within this framework that the analysis presented by Berger and colleagues of outcomes of patients with peripheral artery disease (PAD) with concomitant coronary artery disease (CAD) from the EUCLID trial adds important evidence of the heterogeneity of cardiovascular risk among patients with symptomatic lower extremity PAD. 2
A growing body of literature has described the malignant atherothrombotic phenotype associated with symptomatic atherosclerotic disease in more than one territory (e.g. coronary, peripheral, cerebrovascular) called polyvascular disease. 3 More recently, randomized clinical trials of long-term secondary prevention with more potent antithrombotic therapies in patients with CAD have shown heightened risk of major adverse cardiovascular events (MACE) in subgroups with concomitant PAD (CAD+PAD).4,5 Even after adjustment for baseline differences, patients with CAD+PAD are at ~60% increased risk of MACE relative to patients with CAD alone over 3–4 years of follow up. This greater risk has been shown to translate into even greater absolute risk reductions with more intensive antithrombotic therapy using long-term dual-antiplatelet therapy (DAPT), including consistent patterns for cardiovascular and all-cause mortality.4,5 This concept has come to the forefront with the results of the COMPASS trial, which enrolled high-risk patients with CAD or PAD and enriched for polyvascular disease by nature of inclusion criteria that required either increased age, revascularization in more than one vascular territory, or at least two other high-risk features. In this cohort, the addition of rivaroxaban 2.5 mg twice daily added to aspirin 100 mg daily significantly reduced MACE with consistency for cardiovascular and all-cause mortality. 6 A rivaroxaban 5 mg twice daily monotherapy arm was also included but was not superior to aspirin.
Until recently, few studies have explored whether the concept of polyvascular disease further stratifies risk of MACE among patients with symptomatic PAD. 7 Classic teaching is that all patients with PAD essentially have CAD even if they do not know it; however, whether the presence of clinically evident CAD is independently associated with higher risk of MACE is unclear. The lack of robust data regarding the associated risk of CAD among patients with PAD may, in part, be due to the lack of large PAD trials well powered for subgroup analyses. The EUCLID trial presents an optimal dataset for this exploration by nature of its size and well-characterized population of patients with PAD. In this analysis of patients enrolled in EUCLID, Berger et al. show that concomitant CAD (prior myocardial infarction (MI), prior percutaneous coronary intervention, or prior coronary artery bypass grafting surgery) was present in 29% of the 13,885 patients randomized. 2 Patients with CAD+PAD were at markedly higher risk of MACE, with an event rate of 17.8% at 3 years versus 10.3% in those with PAD alone. After adjustment for baseline differences, CAD+PAD was associated with a 50% excess risk of MACE. The relative and absolute differences in risk observed were consistent with those seen in PEGASUS-TIMI 54, which evaluated patients with CAD+PAD versus CAD alone, suggesting that polyvascular disease is a useful marker of risk in either a primary PAD or CAD population (Figure 1). 4 The observations stand in contrast to the relationship of polyvascular disease and the risk of acute limb ischemia (ALI), which was similar regardless of concomitant CAD and underscores that the predictors of MACE and ALI in patients with PAD are likely different.
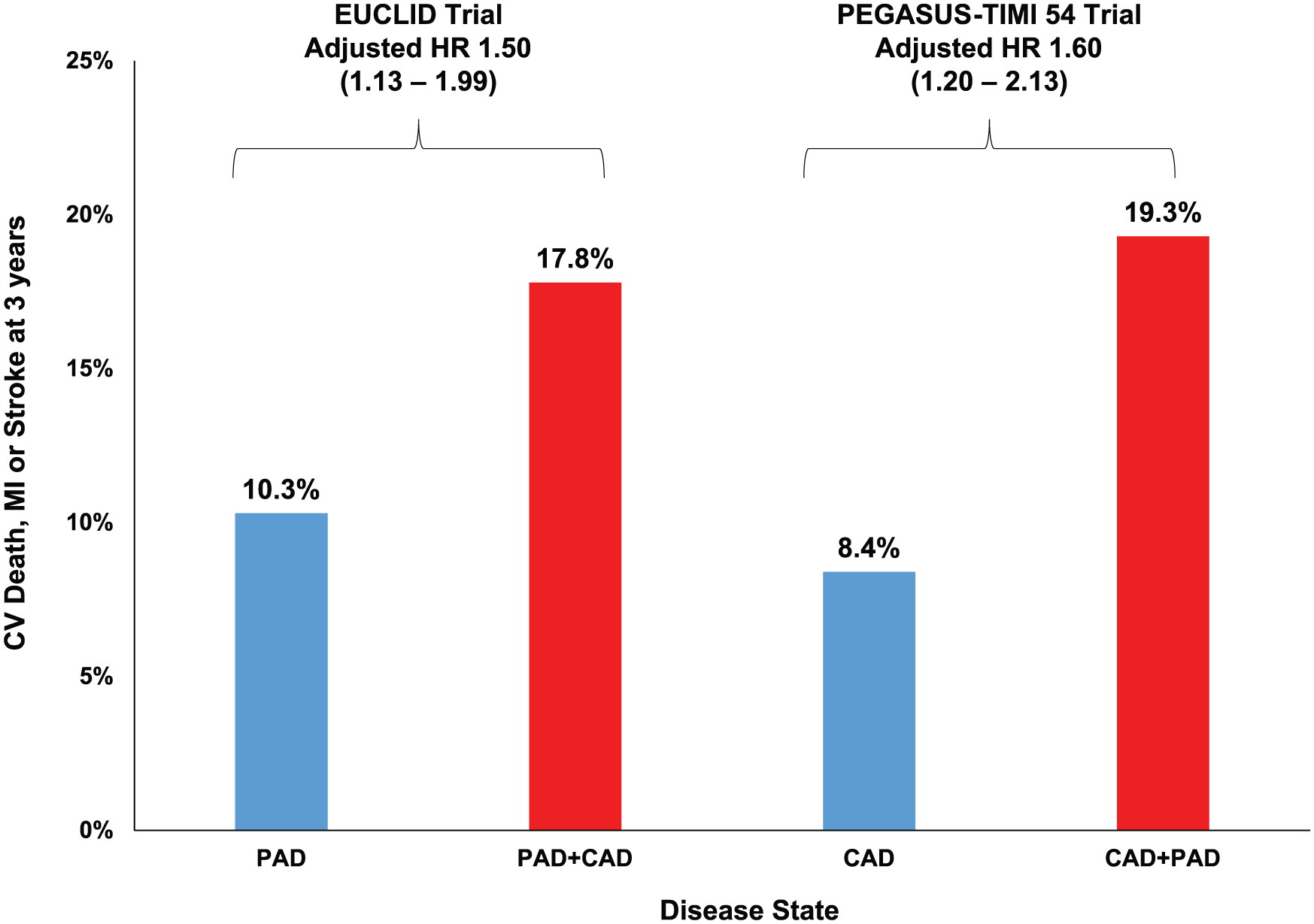
Although patients in EUCLID with CAD+PAD were at higher risk of MACE relative to those with PAD alone, there was no effect modification based on the presence of CAD at baseline and the effect of ticagrelor versus clopidogrel for MACE. Overall the study showed similar outcomes with ticagrelor compared to clopidogrel and was neutral for superiority. The consistent lack of superiority in this high-risk subgroup is unexpected given the clear benefit of ticagrelor versus clopidogrel in the PLATO trial. 8 It may be that patients randomized in PLATO derived greater benefit from ticagrelor relative to clopidogrel due to the fact they were presenting with an acute coronary syndrome (ACS). It is also possible that the CAD subgroup of EUCLID is less specific for patients with a history of spontaneous atherothrombotic MI and could include patients with non-thrombotic MI (e.g. Type 2) as a primary etiology who are less likely to benefit from more potent antithrombotic therapy. The observation that effect modification was present based on prior coronary stenting (p-interaction = 0.03) suggests that in those patients with clear atherosclerotic CAD, there may have been a benefit of ticagrelor versus clopidogrel. This observation is consistent with that of TRILOGY, a trial of prasugrel versus clopidogrel in patients with non-ST-segment elevation ACS managed medically. 9 Although the overall trial was neutral, there was effect modification with benefit of prasugrel versus clopidogrel in those with angiographically confirmed CAD. 9
The observations from Berger et al. from the EUCLID trial may be most useful in the clinic, where clinicians caring for patients with PAD may want practical and useful ways of identifying patients at the highest risk of MACE who may benefit more from intensive antithrombotic therapy. The results may also be useful in clinical translation of the COMPASS trial, where although the relative benefit of rivaroxaban would be expected to be consistent in all patients with PAD, the absolute benefit is likely to be greater in those with CAD+PAD compared to PAD alone. Future guidelines may incorporate risk stratification, including clinical markers such as polyvascular disease, as the list of effective therapies in PAD grows and the need for personalization increases.
Footnotes
Declaration of conflicting interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: consulting for Amgen, Aralez, AstraZeneca, Bayer, Janssen, MedImmune, and Merck.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: grant support to Brigham and Women’s Hospital from Amgen, AstraZeneca, MedImmune, and Merck.
