Abstract
Background:
The prevalence of peripheral artery disease (PAD) and leg symptoms are higher in Black than White adults. We studied the effects of self-reported lower extremity symptoms and ankle–brachial indices (ABI) groups on outcomes.
Methods:
Black participants in the Jackson Heart Study with baseline ABI and PAD symptom assessments (exertional leg pain by the San Diego Claudication questionnaire) were included. Abnormal ABI was < 0.90 or > 1.40. Participants were divided into (1) normal ABI, asymptomatic, (2) normal ABI, symptomatic, (3) abnormal ABI, asymptomatic, and (4) abnormal ABI, symptomatic to examine their associations with MACE (stroke, myocardial infarction, fatal coronary heart disease) and all-cause mortality, using Kaplan–Meier survival curves and stepwise Cox proportional hazard models adjusting for Framingham risk factors.
Results:
Of 4586 participants, mean age was 54.6 ± 12.6 years, with 63% women. Compared with participants with normal ABI who were asymptomatic, participants with abnormal ABI and leg symptoms had highest risk of MACE (adjusted HR 2.28; 95% CI 1.62, 3.22) and mortality (aHR 1.82; 95% CI 1.32, 2.56). Participants with abnormal ABI without leg symptoms had higher risk for MACE (aHR 1.49; 95% CI 1.06, 2.11) and mortality (aHR 1.44; 95% CI 1.12, 1.99). Participants with normal ABI and no leg symptoms did not have higher risks.
Conclusion:
Among Black adults, the highest risk for adverse outcomes were in symptomatic participants with abnormal ABIs, followed by asymptomatic participants with abnormal ABIs. These findings underscore the need for further studies to screen for PAD and develop preventative approaches in Black adults with asymptomatic disease.
Background
Given the disproportionately higher prevalence of lower extremity peripheral artery disease (PAD) and leg symptom burden in Black compared with White individuals, contemporary risk-stratification methods to predict cardiovascular outcomes are necessary in this participant population.1,2 Studies have found that Black adults have a threefold higher rate of PAD, not explained by traditional and novel risk factors. Participants with PAD are at high risk of developing myocardial infarction, stroke, and overall major adverse cardiac events (MACE),3,4 as well as chronic limb-threatening ischemia (CLTI) and amputation. 5 In Black adults, age-adjusted deaths were 1.2 times more likely to have PAD listed as a contributing cause of death. 6 Social determinants (e.g., socioeconomic status, healthcare access) may help explain this discrepancy, along with delayed recognition and awareness, as well as inequities and access to care barriers.7,8
Black adults have a higher leg symptom burden, 9 but it is unknown how this symptom burden manifests or differs by ankle–brachial index (ABI) risk and whether this incremental leg symptom burden is differentially associated with prognostic outcomes in this population. Previous risk models have found that adding ankle–brachial index (ABI) calculations into the Framingham Risk Score (FRS) calculator improves the prediction of cardiovascular outcomes. 10 Whether ABI, in combination with symptom phenotypes, would be a useful test to improve detection of elevated cardiovascular disease (CVD) risk in Black adults is unclear.
PAD can present with a wide range of symptoms, and not all leg symptoms may be due to PAD. One study found that although no category of leg pain, categorized by the San Diego Claudication Questionnaire, 11 was sufficiently sensitive or specific for routine PAD diagnosis, subjects with legs with abnormal ABIs were more likely to have ischemic leg pain. 12 Many participants with PAD present with no symptoms, and current guidelines do not support screening for asymptomatic participants. 13 However, the correlation of symptoms and ABIs in predicting cardiovascular events in populations with higher baseline PAD risk and a greater burden of leg symptoms, such as in Black adults, remains uncertain.
Therefore, among Black adults enrolled in the Jackson Heart Study (JHS), we aimed to: (1) describe participant characteristics by combinations of ABI categories (normal, abnormal) and PAD leg symptoms (asymptomatic, symptomatic); (2) examine the association between these categories and subsequent MACE and mortality risk, adjusting for confounders. Understanding the impact of combinations of ABI screening results and leg symptom phenotypes can help clinicians understand which Black adults in particular are at risk of future cardiovascular events and screen these people appropriately.
Methods
Our study used data collected in the JHS, which is a large, prospective, longitudinal cohort of 5306 Black participants aged 21–84 years old in Jackson, MS, USA. Study population characteristics and data collection methods have been previously reported.14,15 ABI was recorded at the first visit in the study, with initial study dates from September 2000 to March 2004. For our study, the inclusion criteria were participants who had both an ABI and a questionnaire for PAD symptoms recorded at the first visit. This study was reviewed by the Yale Institutional Research Board, and, given the deidentified nature of the previously collected JHS data, the research was deemed nonhuman subjects research. The data are governed by JHS.
Measures
Medical history was collected at the first visit. 14 Participant demographics included age, sex, insurance status, income, and measured body mass index (BMI). 14 We included clinical variables from the FRS calculator used to predict cardiovascular risk to our model. 16 Comorbidities and medication were self-reported at the first visit. Comorbidities included hypertension (HTN), cardiovascular disease (includes stroke and coronary heart disease), cardiac procedures, diabetes, chronic kidney disease, and smoking status. Medication variables included statin use and blood pressure (BP) medication use. Systolic and diastolic BPs, total cholesterol, low-density lipoprotein (LDL) levels, and high-density lipoprotein (HDL) levels were measured at first visit. The corresponding FRS score was calculated. HTN status was defined as BP > 140/90 mmHg or use of BP-lowering medication. 17 Diabetes was defined using the 2010–2021 American Diabetes Association criteria: (1) glucose-lowering medication use; (2) fasting glucose ⩾ 126 mg/dL; and/or (3) glycated hemoglobin (HbA1c) ⩾ 6.5%. 18 Lipids and BMI were measured at the initial visit, and other measures were collected from a self-reported survey. 14
ABI calculation
To derive the ABI, the systolic BP measurements were obtained twice in the brachial artery on the right arm and at each ankle twice at the first visit (further detailed protocols previously reported). 19 One ABI was calculated for each leg by dividing the mean of the ankle BPs by the mean of the brachial BPs. The lower ABI was recorded as the ABI for each participant. 20
PAD symptom categories
Participants at the first visit filled out a questionnaire on PAD leg symptoms based on the San Diego claudication questionnaire. 21 We categorized the ABI as normal (between 0.9 and 1.4) or abnormal (< 0.9 or > 1.4). Participants were then classified into four groups based on a modified version of McDermott et al.’s (2001) classification system for leg symptoms: (1) normal ABI, asymptomatic; (2) normal ABI, symptomatic; (3) abnormal ABI, asymptomatic; (4) abnormal ABI, symptomatic. 11
Outcomes
Our study examined MACE cardiovascular events (including stroke, myocardial infarction [MI], and fatal coronary heart disease [CHD], whichever occurred first) and all-cause mortality. Stroke, MI, fatal CHD, and all-cause death were defined from medical records and annual follow-up telephone interviews. MI was defined by combined electrocardiogram and self-reported data. 22 Patients were followed from first visit date up to last contact date (latest is December 31, 2018), or death, whichever occurred first.
Statistical analysis
Participant characteristics
We described rates of participant characteristics and comorbidities based on normal ABI and leg symptom categories at the first visit. Data measured at the first visit were presented as means with SD for continuous variables and proportions for categorical variables. Data were compared by the four PAD symptom categories through chi-squared tests for categorical variables or Fischer’s exact test, as appropriate, and ANOVA tests for continuous variables or nonparametric tests, as appropriate. A supplemental analysis further stratifying the groups with abnormal ABI into ABI < 0.90 vs ABI > 1.40 was included for each of the demographic and outcome tables and figures in this study, creating six groups: (1) normal ABI, asymptomatic; (2) normal ABI, symptomatic; (3) ABI < 0.90, asymptomatic; (4) ABI < 0.90, symptomatic; (5) ABI > 1.40, asymptomatic; (6) ABI > 1.40, symptomatic.
Outcome analyses
The crude event rates for each of our two primary endpoints (MACE, all-cause mortality) were reported by each of the four categories of ABI/leg symptoms. Kaplan–Meier curves for MACE and all-cause mortality were created by ABI/symptom group. Cox proportional hazard models were constructed to estimate the hazard ratio (HR) with 95% CI of the risk of MACE and all-cause mortality. We added FRS risk factors into a Cox regression model in four steps: 16 Step 1: ABI/leg symptoms; Step 2: adding age and sex; Step 3: adding systolic BP, BP medication use, and diabetes, smoking, coronary heart disease, and stroke history; Step 4: adding total cholesterol level, LDL, and HDL. Missing data were excluded (n = 502 having incomplete data) and were not at random (Little’s Missing Completely at Random test, p < 0.001). We used Stata Statistical Software, Release 16 (StataCorp LLC, College Station, TX, USA).
Results
Out of 5306 participants in the database, 4586 participants who had information on ABI and PAD symptomatology recorded during the first visit were included. The mean age was 54.6 ± 12.6 years, and 63% were women.
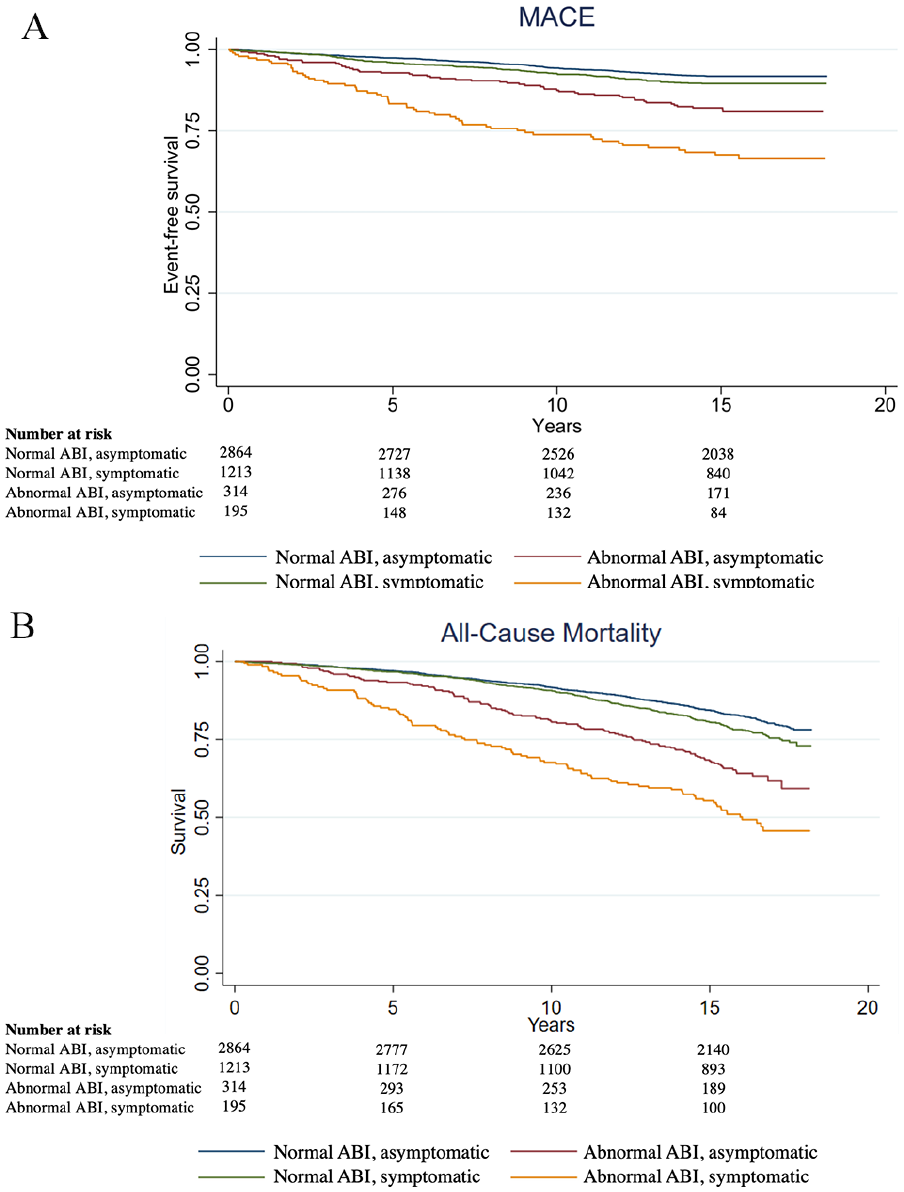
The proportional hazards assumptions were met according to Schoenfeld’s global test (p-value > 0.05) and based on comparison of Kaplan–Meier survival curves. We found that the risks of MACE and death were different in asymptomatic versus symptomatic participants conditional on ABI. The p-value of the interaction between ABI and leg symptom was equal to 0.056 for MACE risk and 0.093 for mortality. Considering two-sided p-value < 0.10 as significant, these results supported our approach to include ABI and leg symptoms as one variable with four categories in our models (normal ABI/asymptomatic leg as reference). By category, 62% had a normal ABI and were asymptomatic (n = 2864) (mean of ABI 1.15, SD 0.11), 26% had a normal ABI and were symptomatic (n = 1213) (mean of ABI 1.14, SD 0.11), 7% had an abnormal ABI and were asymptomatic (n = 314) (< 0.9: 59%, > 1.4: 41%), and 4% had an abnormal ABI and were symptomatic (n = 195) (< 0.9: 70%, > 1.4: 30%).
Participants who had an abnormal ABI and were symptomatic were more likely to be older, had higher rates of Medicaid and Medicare insurance, and were from poor or lower middle class income categories (Table 1). They also had higher rates of hypertension, cardiovascular disease, diabetes, CKD, and were more likely to smoke and to be prescribed a statin.
Participant characteristic by ankle–brachial index/leg symptom categories.
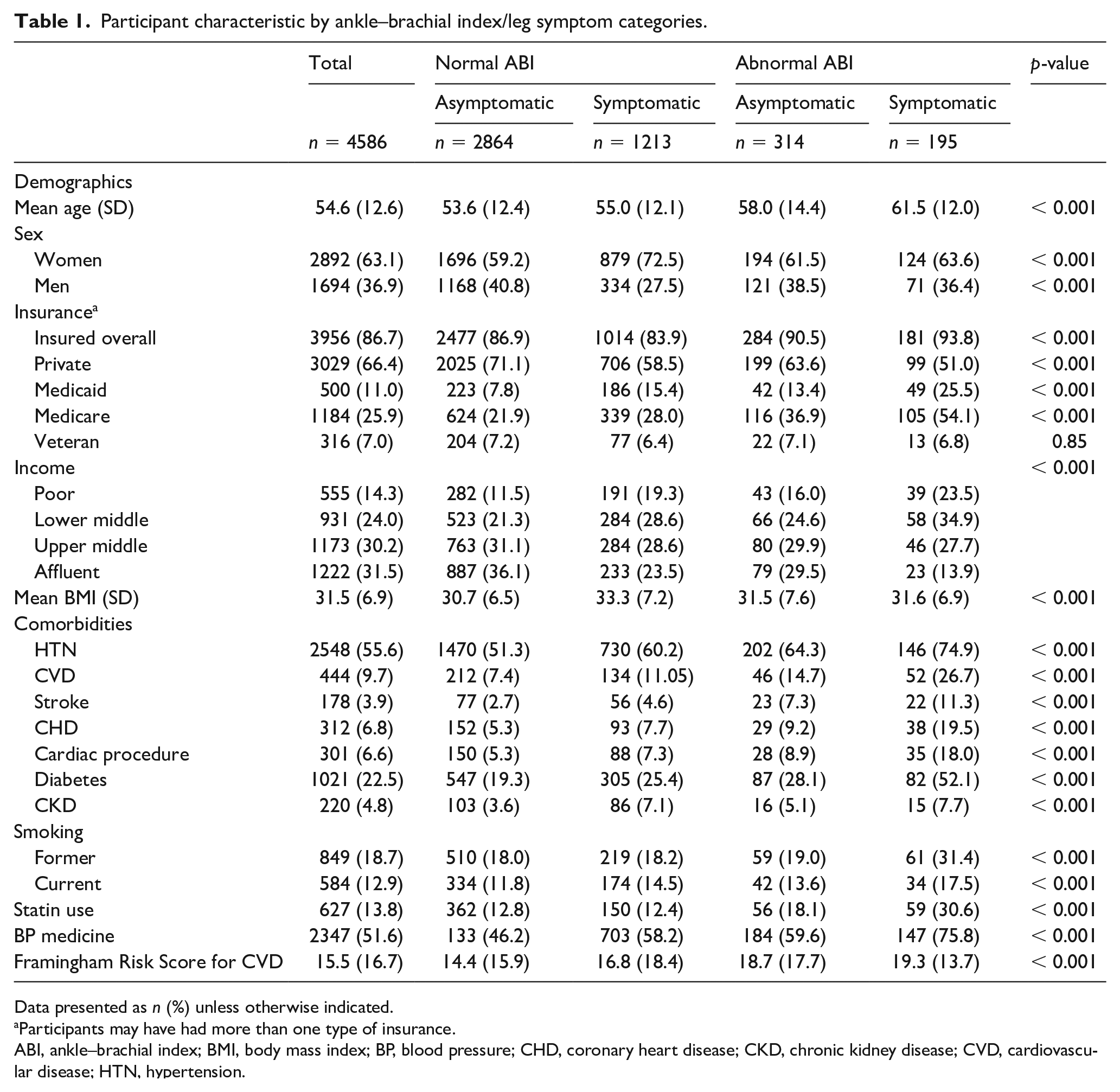
Data presented as n (%) unless otherwise indicated.
Participants may have had more than one type of insurance.
ABI, ankle–brachial index; BMI, body mass index; BP, blood pressure; CHD, coronary heart disease; CKD, chronic kidney disease; CVD, cardiovascular disease; HTN, hypertension.
Out of the total 4586 participants, 452 (9.9%) had MACE (fatal CHD: 105, MI: 219, stroke: 216), and overall 1007 died (22.0%) (Table 2). Participants may have had more than one outcome event. There was a higher rate of MACE in people with an abnormal ABI and who were symptomatic (29%), followed by abnormal ABI and asymptomatic (17%), normal ABI and symptomatic (10%), and normal ABI and asymptomatic (8%) (Table 2). The highest rates of all-cause mortality were in people with abnormal ABI who were symptomatic (51%), followed by those with an abnormal ABI and who were asymptomatic (36%), those who had a normal ABI and were symptomatic (23%), and those who had a normal ABI and were asymptomatic (18%) (Table 2). Median follow-up time was 15.8 years (IQR: 14.9, 16.7 years). Kaplan–Meier curves are shown in Figure 1.
Number of participants with primary outcome events by ankle–brachial index/leg symptom categories.
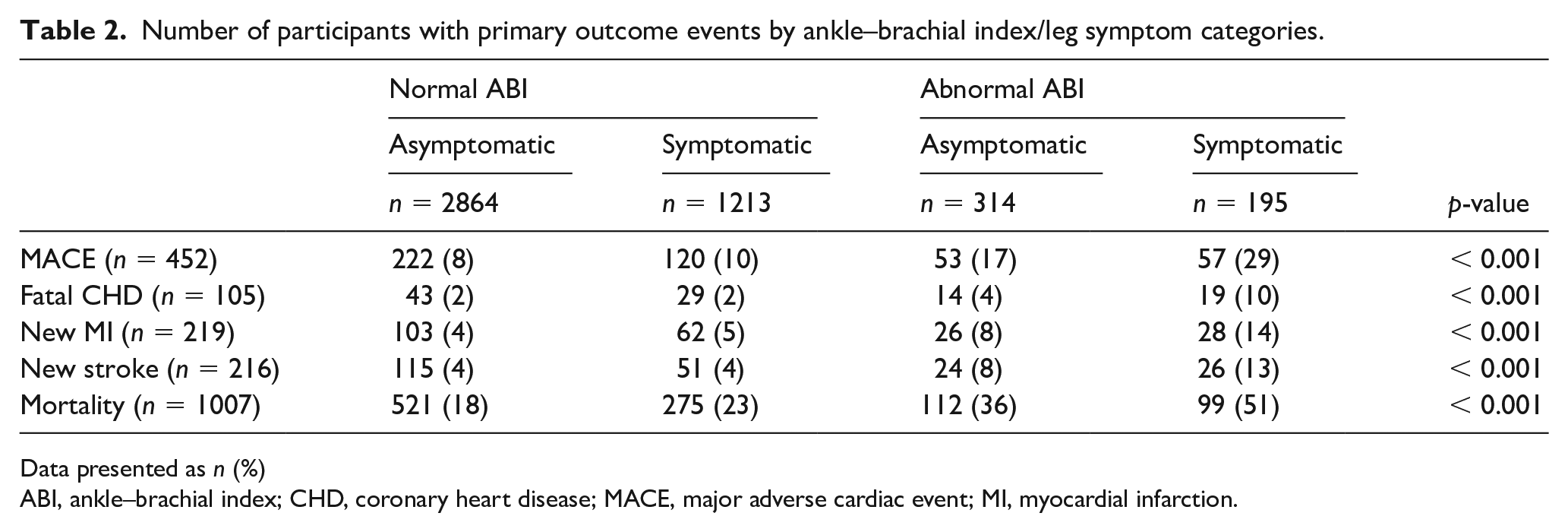
Data presented as n (%)
ABI, ankle–brachial index; CHD, coronary heart disease; MACE, major adverse cardiac event; MI, myocardial infarction.
Participants with an abnormal ABI who were symptomatic had a higher HR for MACE (HR 2.28; 95% CI 1.62, 3.22) and mortality (HR 1.82; 95% CI 1.32, 2.56) than asymptomatic participants with normal ABI (Figure 2) (online Supplemental Tables 1 and 2). Similarly, participants with abnormal ABIs who were asymptomatic also had a higher HR for MACE (HR 1.49; 95% CI 1.06, 2.11) and mortality (HR 1.44; 95% CI 1.12, 1.99). The normal ABI, symptomatic cohort did not have significantly higher all-cause mortality and MACE compared with the reference group.
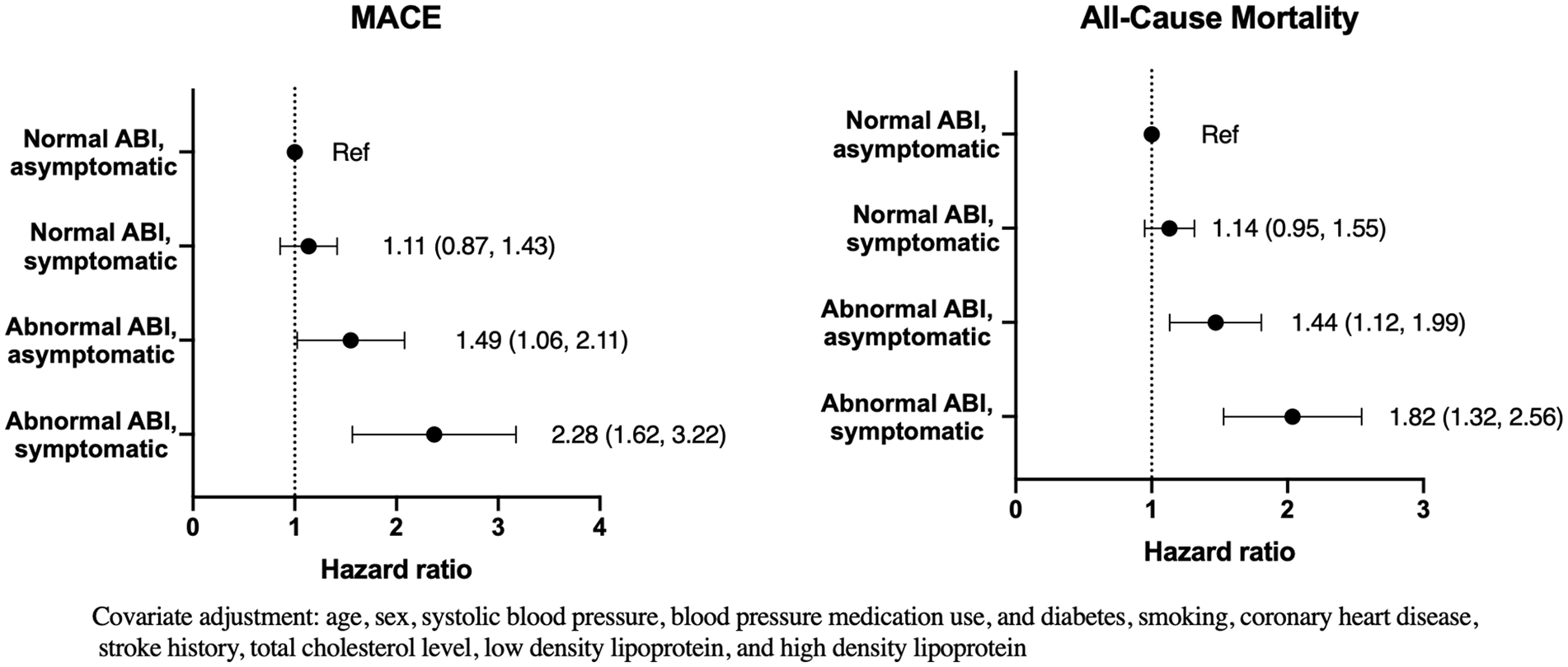
Hazard ratios for ABI/leg symptoms for MACE and all-cause mortality, adjusted for Framingham risk factors. Abnormal ABI was < 0.90 or > 1.40.
These analyses were calculated for groups further stratified by ABI < 0.90 vs ABI > 1.40. Online Supplemental Table 3 shows participant demographics, Supplemental Table 4 shows the number of patients with outcome events, and Supplemental Figure 1 shows Kaplan–Meier curves for each of the six groups. Compared to participants with normal ABI and who were asymptomatic (reference), participants had a higher risk of MACE if they had an ABI < 0.90 and were asymptomatic (aHR 1.72; 95% CI 1.17, 2.53), an ABI < 0.90 and were symptomatic (aHR 2.29; 95% CI 1.56, 3.32), and an ABI > 1.40 and were symptomatic (aHR 2.29; 95% CI 1.07, 2.91) (online Supplemental Figure 2). Compared to the reference group, participants had a higher risk of mortality if they had an ABI < 0.90 and were asymptomatic (aHR 1.94; 95% CI 1.48, 2.56), an ABI < 0.90 and were symptomatic (aHR 1.51; 95% CI 1.60, 1.96), and an ABI > 1.40 and were symptomatic (aHR 2.25; 95% CI 1.32, 3.84). Participants with an ABI > 1.40 and who were asymptomatic did not have higher rates of either outcome.
Discussion
Our study showed that for Black individuals, those who had both an abnormal ABI and lower extremity symptoms had the highest rates of MACE and mortality in a median follow-up of 16 years, with around one-third experiencing MACE and one half dying. Furthermore, participants with abnormal ABI and symptomatic disease were more likely to have a lower socioeconomic status and more cardiovascular comorbidities. As for magnitude of risk, this group was followed by asymptomatic individuals who had abnormal ABIs, with a > 40% increased risk of MACE and mortality as compared with asymptomatic participants who had normal ABIs, even after adjusting for cardiovascular risk factors. Upon sensitivity analysis, participants with an ABI < 0.90 with and without leg symptoms, as well as participants with an ABI > 1.40 who were symptomatic, carried a higher risk of MACE and mortality.
Our study builds on previous literature showing a high risk and severity of PAD in Black participants.23–26 Previous studies using the JHS cohort have shown that the American College of Cardiology/American Heart Association (ACC/AHA) CVD risk algorithm and the FRS algorithms predict outcomes reliably in Black adults, 27 but our study is the first to include the prognostic value of ABI along with symptomatology for cardiovascular risk-stratification in this population.
Our study demonstrates that in asymptomatic Black participants, the risk of adverse outcomes was higher in participants with abnormal ABIs. This highlights the prognostic value of ABI measurements, in combination with PAD symptom assessment, for adverse outcomes in Black adults in our study. The literature surrounding the prognostic value of subclinical PAD in other populations has been mixed. In contrast to the Fowkes et al. (2008) paper showing that ABI may improve the cardiovascular risk prediction of the FRS, 10 one 2013 systematic review of ABI screening for asymptomatic adults suggested that adding ABI to the FRS likely had a small risk reclassification effect and would only be beneficial for participants at the thresholds between risk categories. 28 However, the German Epidemiological Trial on Ankle Brachial Index (getABI) showed that asymptomatic PAD diagnosed at primary care clinics carried the same mortality risk as symptomatic PAD, 29 and one study that looked specifically at subclinical PAD found a 65% increased risk in mortality compared with those who did not have PAD. 30
Health disparities for Black adults are important to uncover and address, especially as Black Americans have a higher prevalence of PAD but also experience worse outcomes than the White participants.9,31,32 Not only do Black Americans have a higher rate of comorbidities such as diabetes, hypertension, and obesity,33,34 they are also less likely to be treated for hypertension and hyperlipidemia.35,36 Though treating asymptomatic PAD with antiplatelet agents is reasonable (Class IIa), all participants with PAD should take a statin (Class Ia), 37 but Black participants are less frequently treated with statins.38,39 One study from 1995 to 1998 showed that Black adults with PAD were at a 2.8-fold increased risk of limb events than White individuals, but the risk was no longer significant if blood glucose was well controlled. 40 These findings suggest that the higher risk of poor outcomes within the Black population may be explained by access and treatment disparities.
Furthermore, many studies have found that clinical risk factors do not fully explain the increased prevalence and morbidity of PAD in Black Americans. In an analysis of the San Diego population study, Criqui et al. found that Black race was a strong and independent risk factor for PAD as compared to nonHispanic White individuals with no evidence of greater susceptibility to cardiovascular risk factors. 2 Follow-up studies showed that novel risk factors, such as fibrinogen and lipoprotein (a), only partially explained this risk.7,31 Thus, social determinants of health may compound access to care issues that lead to poor outcomes in Black Americans. Black Americans have been historically underrepresented and marginalized, and have faced systemic and contextual barriers contributing to inequities and disparities, including access to care and early disease detection. The disproportionately high rates of low socioeconomic status among Black adults may further explain the worse PAD outcomes,8,41,42 as low income is associated with increased rates of PAD as well as higher rates of major amputation for CLI.23,43 Similarly, our study showed an increased rate of many comorbidities and lower income status in Black participants with abnormal ABIs and who were symptomatic.
Therefore, it remains critical to develop interventions to address these inequities, which may include early identification, high rates of guideline-directed therapy, increased access to healthcare, and educational efforts to improve health literacy. 43 Access to comprehensive vascular rehabilitation, including access to supervised exercise programs,44,45 as well as educational support such as smoking cessation counseling, remains especially important for Black Americans. Detection of risk by phenotypes described here may add to the detection of populations at risk.
By the 2016 AHA/ACC screening guidelines, ABIs for PAD among asymptomatic participants is only reasonable for participants with risk factors for PAD (Class IIa). 37 Our study suggests that research should further evaluate when ABIs may be useful to screen Black adults with no leg symptoms as this population experiences a high rate of cardiovascular outcomes. In the JHS cohort, 7% of subjects were asymptomatic with abnormal ABIs. Among the 12% of JHS participants with abnormal ABIs, 62% were asymptomatic. Hence, the majority of JHS participants with abnormal ABIs and thus likely prognostically significant CVD, would probably not be detected relying upon LE symptom status alone. Earlier and more complete identification of asymptomatic individuals at higher CVD risk within high-risk groups might be a path to reduce health disparities. Given the higher risk of PAD in Black adults, a default strategy of objective noninvasive testing with ABIs/toe pressures in participants without LE symptoms is reasonable, but other high-risk features should be further evaluated for clinical utility and cost-effectiveness.
Study limitations
Limitations to this study include the retrospective nature of our review and that our population consisted of one community of Black adults without a comparator group. Furthermore, the number of participants in each category was too low to further stratify risk by different types of leg symptoms (e.g., intermittent claudication, atypical exertional leg pain). 11 Although extensively validated across settings, the San Diego Claudication questionnaire used is only one way to measure symptom severity, and misclassification may have occurred. Next, our findings were associations, not revealing information on causality, and adjusted analyses do not represent the true toll of suffering by patients. Strengths of our study include the size of the population, the long follow-up period, and the goodness-of-fit of the models. Our study provides evidence that Black adults with leg symptoms and abnormal ABIs are at highest risk of cardiovascular outcomes and mortality, but asymptomatic participants with abnormal ABIs also carry a significant risk.
Conclusion
Among Black adults, the highest risk for adverse outcomes was in symptomatic participants with abnormal ABIs, followed by asymptomatic participants with abnormal ABIs, even after adjusting for cardiovascular risk factors. These findings underscore the need for further studies to screen for PAD and to develop preventative approaches in Black adults with asymptomatic disease.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231151729 – Supplemental material for Lower extremity symptoms and ankle–brachial index screening as predictors of cardiovascular outcomes in Black adults
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231151729 for Lower extremity symptoms and ankle–brachial index screening as predictors of cardiovascular outcomes in Black adults by Megan Lee, Kim G Smolderen, Costin Ionescu, William B Hillegass, Gaelle Romain and Carlos Mena-Hurtado in Vascular Medicine
Footnotes
Acknowledgements
The authors wish to thank the staff and participants of the Jackson Heart Study.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Mena-Hurtado is a consultant for Cook Medical, Medtronic, Abbott, Cardinal Health, and Optum Labs. Dr Smolderen reports grant funding from Abbott, Gore, Johnson & Johnson, Cardiva, and Merck, and she is a consultant for Optum Labs and Abbott. William Hillegass is supported by the IDeA grant U54GM115428. The other authors report no disclosures.
Funding
The Jackson Heart Study is supported and conducted in collaboration with Jackson State University (HHSN268201800013I), Tougaloo College (HHSN268201800014I), the Mississippi State Department of Health (HHSN268201800015I), and the University of Mississippi Medical Center (HHSN268201800010I, HHSN268201800011I, and HHSN268201800012I) contracts from the National Heart, Lung, and Blood Institute (NHLBI) and the National Institute on Minority Health and Health Disparities (NIMHD).
Disclaimer
The views expressed in this manuscript are those of the authors and do not necessarily represent the views of the National Heart, Lung, and Blood Institute; the National Institutes of Health; or the US Department of Health and Human Services.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
