Abstract
Patients with end-stage renal disease undergoing kidney transplant often have diffuse atherosclerosis and high cardiovascular morbidity and mortality rates. We analyzed the correlation of peripheral arterial disease (PAD), here quantified by an abnormal ankle–brachial index (ABI) measured within the 5 years prior to kidney transplant, with graft failure and mortality rates (primary end points) after adjusting for known cardiovascular risk factors (age, sex, smoking history, hypertension, diabetes, stroke, known coronary artery disease or heart failure, years of dialysis). Of 1055 patients in our transplant population, 819 had arterial studies within the 5 years prior to transplant. Secondary end points included myocardial infarction; cerebrovascular accident; and limb ischemia, gangrene, or amputation. Low ABI was an independent and significant predictor of organ failure (OR, 2.77 (95% CI, 1.68–4.58), p<0.001), secondary end points (HR, 1.39 (95% CI, 0.97–1.99), p<0.076), and death (HR, 1.84 (95% CI, 1.26–2.68), p=0.002). PAD was common in this population: of 819 kidney transplant recipients, 46% had PAD. Low ABI was associated with a threefold greater risk of graft failure, a twofold greater risk of death after transplant, and a threefold greater risk of secondary end points. Screening for PAD is important in this patient population because of the potential impact on long-term outcomes.
Keywords
Introduction
Peripheral arterial disease (PAD) is common in patients who are 70 years or older and who have diabetes, renal failure, or a combination of cardiovascular risk factors. Patients with PAD have a three- to fivefold increased risk of cardiovascular events.1–3 A low ankle–brachial index (ABI) has been associated with a greater risk of coronary artery disease (CAD), cerebrovascular accident (CVA), and transient ischemic attack (TIA), even when age, sex, and other risk factors are taken into account.1,4 PAD is typically diagnosed by measuring the ABI, and studies have shown that ABI correlates well with overall survival.5,6 Patients with end-stage renal disease (ESRD) who have PAD are at a higher risk of cardiovascular mortality.7–10 PAD can also lead to functional impairment in this population. 11
CAD is the major cause of death in patients with ESRD before and after their transplants. 12 Identifying patients at higher risk for cardiovascular mortality and graft failure preoperatively enables risk stratification in terms of predicting who might have worse postoperative cardiovascular outcomes and possibly allow for aggressive risk-factor modifications.13–15
PAD has been associated with poor postoperative results in patients undergoing coronary artery bypass surgery. 16 Perhaps a lower extremity vascular physiologic evaluation via ABI should be part of the pre-transplant evaluation.
To determine whether PAD might predict graft failure and death postoperatively, we studied kidney transplant recipients who underwent vascular evaluation for PAD and correlated the PAD severity to our primary end points of organ failure and death and secondary end points of myocardial infarction, TIA/stroke, and limb ischemia, gangrene, or amputation. Prior studies have suggested a higher rate of PAD in patients awaiting kidney transplantation. 17
Methods
The study protocol was approved by the Mayo Clinic Institutional Review Board. We retrospectively analyzed patient charts of all kidney transplant recipients (1 January 1994 to 1 January 2014) at the Mayo Clinic campuses in Arizona, Florida, and Rochester (6712 subjects) to identify those patients who had undergone arterial physiologic testing – specifically, ABI. Primary and secondary outcomes were analyzed for this population from the time of transplant through July 2015. Only 819 patients who had an ABI test in the 5 years before their transplant were included in the final analysis. We chose this time period for two reasons: 90% of the ABI tests were done within 5 years of the transplant date, and a time period beyond 5 years would have had less relevance for study end points. For patients who had multiple ABI measurements, we used the transplant date as the index date, and included the ABI value closest to the index date. The three Mayo Clinic campuses have a standardized method for performing arterial and venous physiologic studies. These criteria are adopted from published guidelines for ABI measurements, which included the highest ABI per limb and the lowest ABI for the individual. 18 Lower extremity physiologic studies were performed by using the Flo-Lab 2100-SX (Parks Medical Electronics, Inc., Aloha, OR, USA).
The ABI measurements were initially grouped into five categories: normal (0.90–1.39); mild disease (0.70–0.89); moderate disease (0.40–0.69); severe disease (0–0.39); and non-compressible arteries (⩾1.40). The final statistical analysis considered the normal range (ABI 0.90–1.39) to reflect common practice at the time these values were collected. Values for an ABI less than 0.90 were then grouped as ‘low ABI’ and those greater than 1.40 were considered as ‘high ABI’ given the small sample sizes within the initial five categories. 18 Risk factors for cardiovascular disease included in the analysis were age; sex; diabetes mellitus; hypertension; smoking history; years on dialysis (includes hemodialysis and peritoneal dialysis); known congestive heart failure or known CAD; CVA; and limb gangrene, ulcer, or ischemia. 1 Primary end points included graft failure and/or death occurring post-transplant. Graft failure was defined as dialysis or return to transplant. Secondary end points included the occurrence of myocardial infarction; CVA; and limb ischemia, gangrene, or amputation.
Statistical analyses
Predictors of organ failure post-transplant were evaluated first by using the Pearson χ2 test individually. Multivariate logistic regression models were used to estimate the odds ratio (OR) for ABI at the low (0–0.89) and high (⩾1.40) ranges compared with the normal (0.90–1.39) range, after adjusting for the risk factors. The Kaplan–Meier method and log-rank test were used to estimate overall survival and event-free survival for secondary events. Cox proportional hazards regression models for multivariate analysis were used to estimate the hazard ratio (HR) for ABI at the low (0–0.89) and high (⩾1.40) ranges versus the normal range (0.90–1.39), after adjusting for risk factors. In addition, model discrimination was assessed using a receiver operating characteristic (ROC) curve for organ failure and a C statistic for mortality and secondary events. P values less than 0.05 were considered statistically significant. Data were analyzed using SAS software, Version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Demographic characteristics
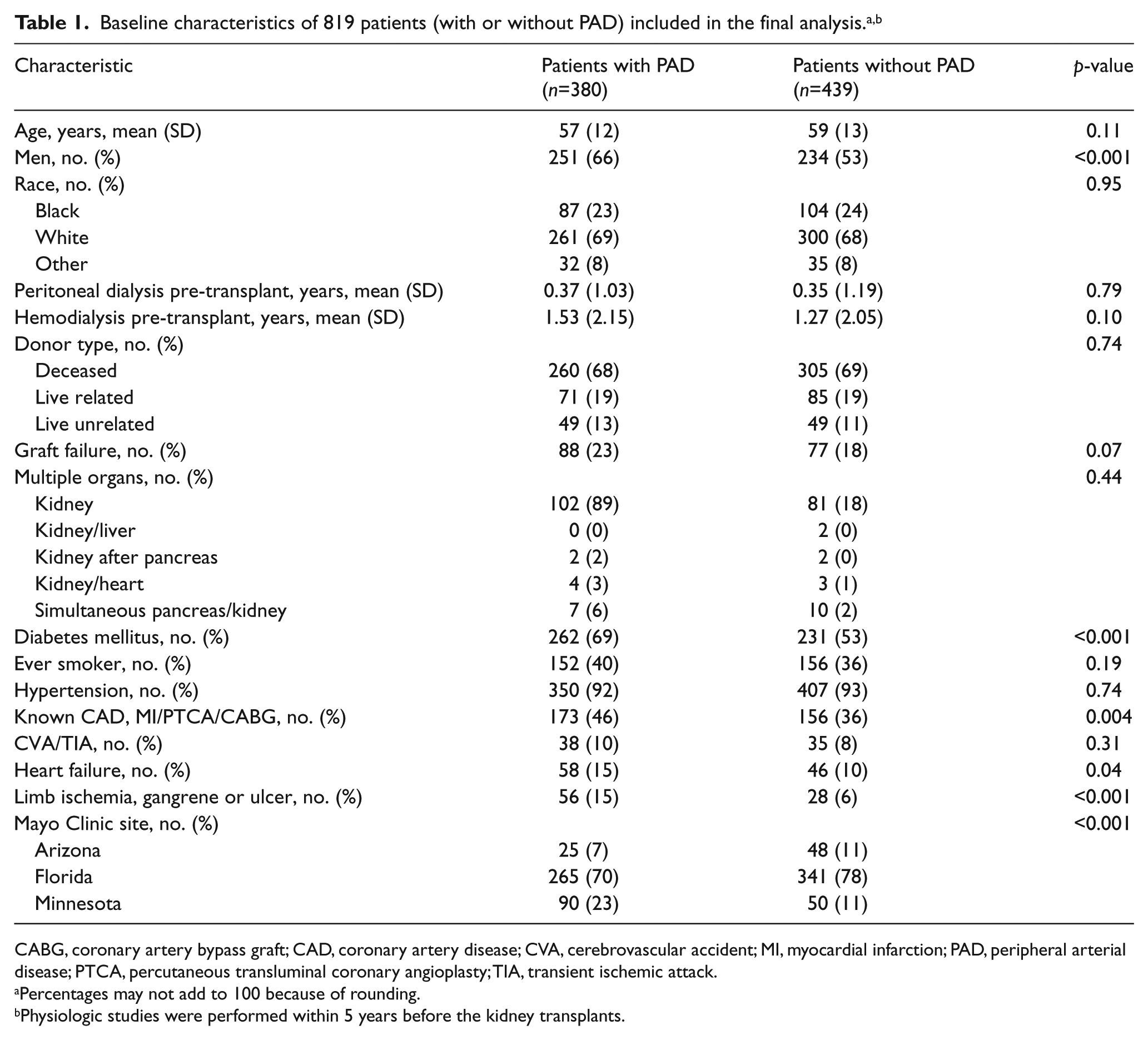
Figure 1 summarizes the overall findings for the study population. Between 1 January 1994 and 1 January 2014, 1055 patients at Mayo Clinic underwent kidney transplantation and had ABI testing for the assessment of lower extremity PAD prior to transplant (mean follow-up, 5.38 years ± 3.58). Of these, 819 patients had ABI testing done within 5 years before transplantation and were included in the analysis. Of the 819 patients, 73 (9%) were in Arizona; 606 (74%) were in Florida; and 140 (17%) were in Rochester. Patients in Arizona and Rochester were selected for lower extremity ABI testing based on the clinician’s clinical suspicion of the presence of substantial PAD. The patients in Florida underwent routine ABI studies as part of the pre-transplant evaluation. Their mean age (SD) was 58 (12) years. Most of the patients were white (68.5%) and male (60%), and most had received kidneys from deceased donors (69%). Graft failure occurred in 165 patients (20%).
Study work flow and results. (ABI, ankle–brachial index; CVA, cerebrovascular accident; PAD, peripheral arterial disease; TIA, transient ischemic attack.)
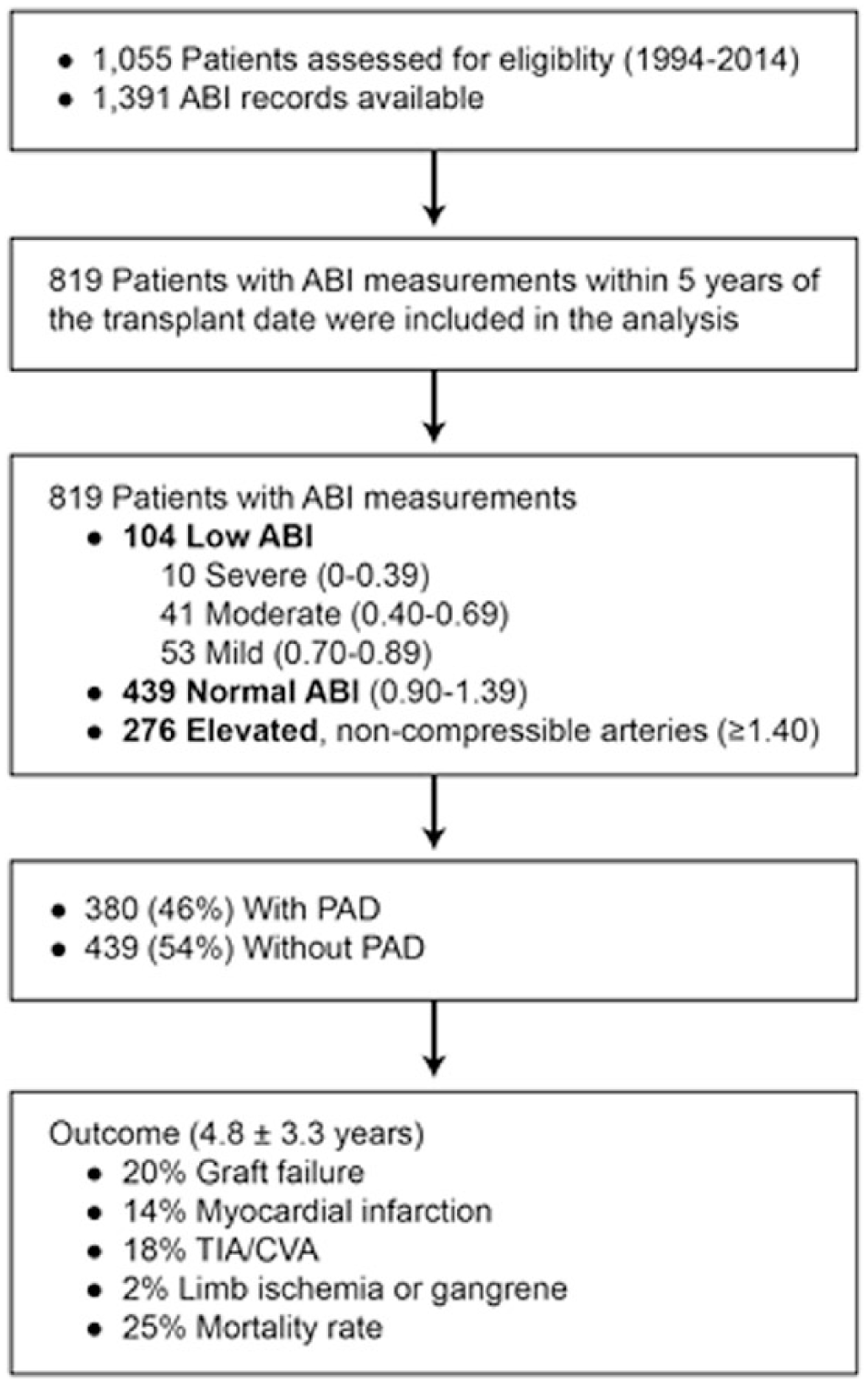
Many of the patients at the time of transplant had several comorbidities: diabetes mellitus (n=493); hypertension (n=757); CAD (n=329); CVA (n=104); smoking history (n=308); heart failure (n=104); and limb ischemia, gangrene, or amputation (n=84). The number of years on peritoneal dialysis (0.36 ± 1.12) or hemodialysis (1.39 ± 2.10) was also considered a comorbidity. Forty-six percent of patients were found to have PAD (low or elevated ABI) before their kidney transplant. Detailed demographic characteristics are shown in Table 1. Of the 819 patients, 276 (34%) had a high ABI and 104 (12%) had a low ABI, ranging from mild (53 patients) to severe (10 patients) (Table 2). The low ABI group included those with mild (6%), moderate (5%) and severe (1%) PAD; however, the sub-categorizations of the low ABI group were not used in additional analyses given the small numbers in each group. Severity of PAD is depicted in Table 2.
CABG, coronary artery bypass graft; CAD, coronary artery disease; CVA, cerebrovascular accident; MI, myocardial infarction; PAD, peripheral arterial disease; PTCA, percutaneous transluminal coronary angioplasty; TIA, transient ischemic attack.
Percentages may not add to 100 because of rounding.
Physiologic studies were performed within 5 years before the kidney transplants.
The severity of peripheral arterial disease (PAD) by ankle–brachial index (ABI). Those with low or elevated ABI are considered subjected with PAD.
Post-transplant outcomes were measured from the time of transplantation through July 2015. During this time, 113 (14%) of the 819 patients had new events of myocardial infarction, 144 (18%) had a TIA or CVA, 15 (2%) had limb ischemia or gangrene, 165 (20%) had graft failure, and 207 (25%) died after transplant.
Outcomes
Predicting graft failure and mortality
By using both multivariate logistic regression and Cox proportional hazard models and accounting for all comorbidities, we found that a low ABI was an independent and significant predictor of organ failure (OR, 2.77 (95% CI, 1.68–4.58), p<0.001), secondary end points (HR, 1.39 (95% CI 0.97–1.99), p<0.076), and death (HR, 1.84 (95% CI, 1.26–2.68), p=0.002). High ABI, on the contrary, was not associated with graft failure (OR, 1.13 (95% CI, 0.76–1.69), p<0.557), and a normal ABI was associated with better survival (HR, 0.729 (95% CI 0.54–0.97), p<0.031). Patients with PAD due to a low ABI were at greater risk for graft failure and death (Figures 2 and 3) than those with a normal or high ABI.
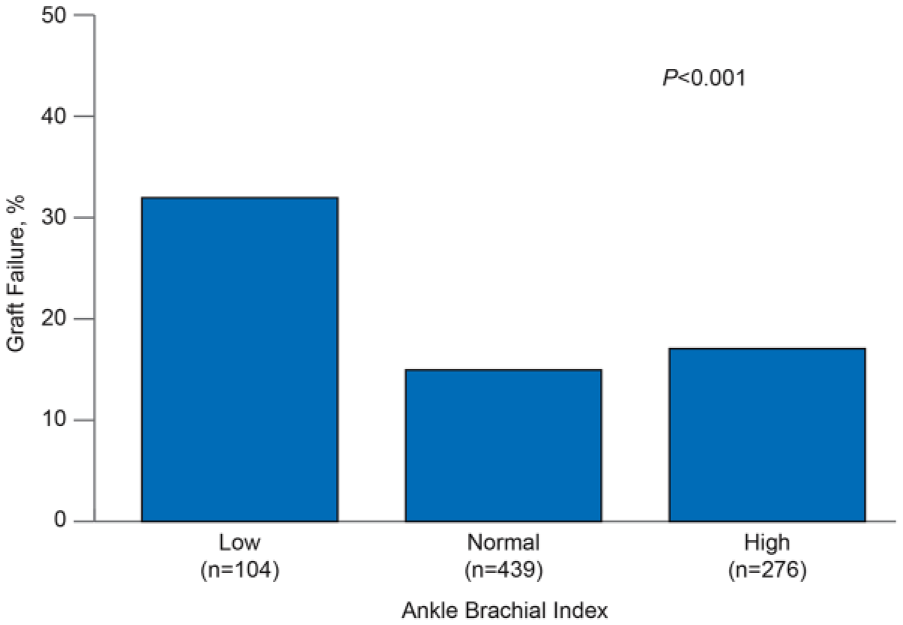
Percentage of graft failure in patients with or without peripheral arterial disease, which was determined by ankle–brachial index (ABI) (n=819). (Normal ABI: 0.9–1.39; elevated ABI: ⩾1.4; low ABI: <0.9.)
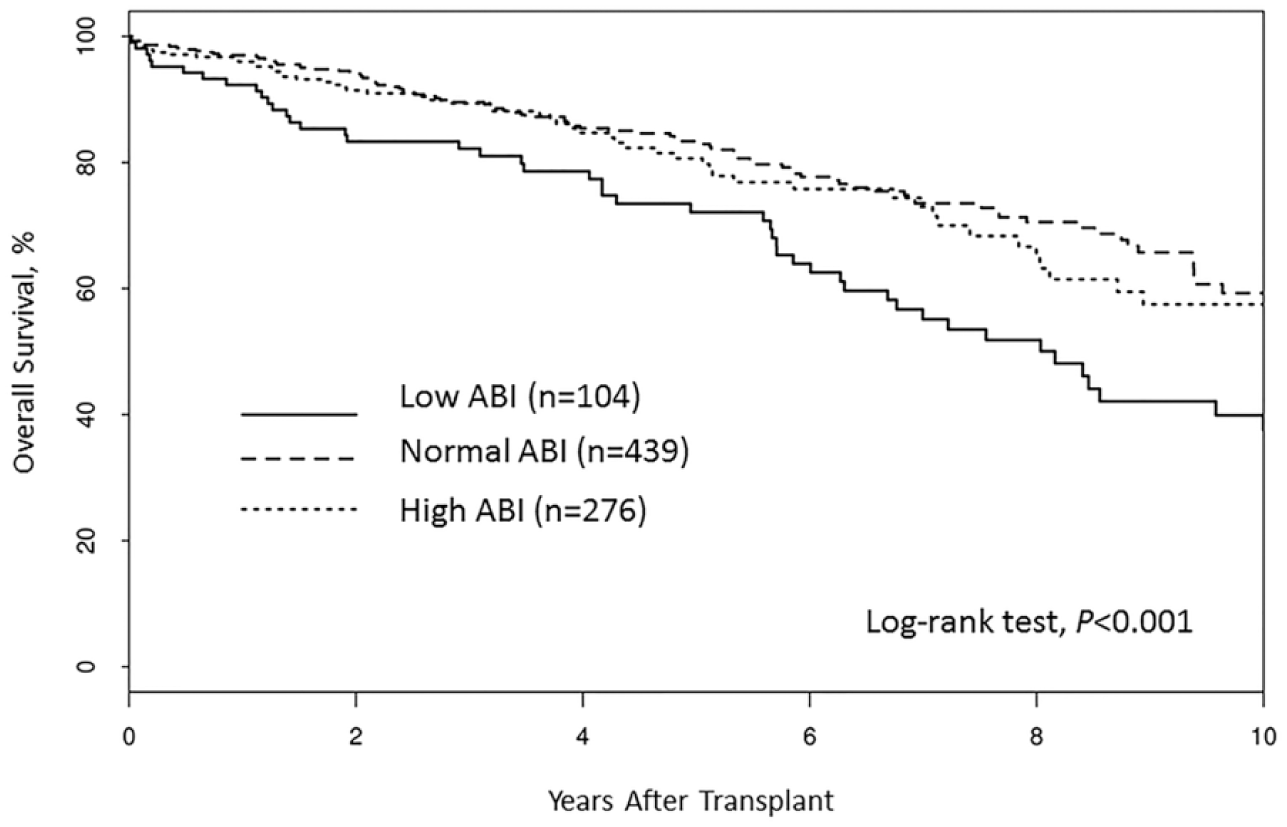
Kaplan–Meier curve showing survival correlated with peripheral arterial disease, as measured by ankle–brachial index (ABI) (n=819). (Normal ABI: 0.9–1.39; elevated ABI: ⩾1.4; low ABI: <0.9.)
Secondary outcomes
Patients with a low ABI are more likely to have myocardial infarction; stroke; and limb ischemia, gangrene, or amputation (OR, 2.49 (95% CI, 2.01–5.28), p<0.001) (Figure 4). By using both multivariate logistic regression and Cox proportional hazard models, we found PAD to be an independent and significant predictor of early myocardial infarction; stroke; or limb ischemia, gangrene, or amputation (OR, 3.26 (95% CI, 2.01–5.28), p<0.001).
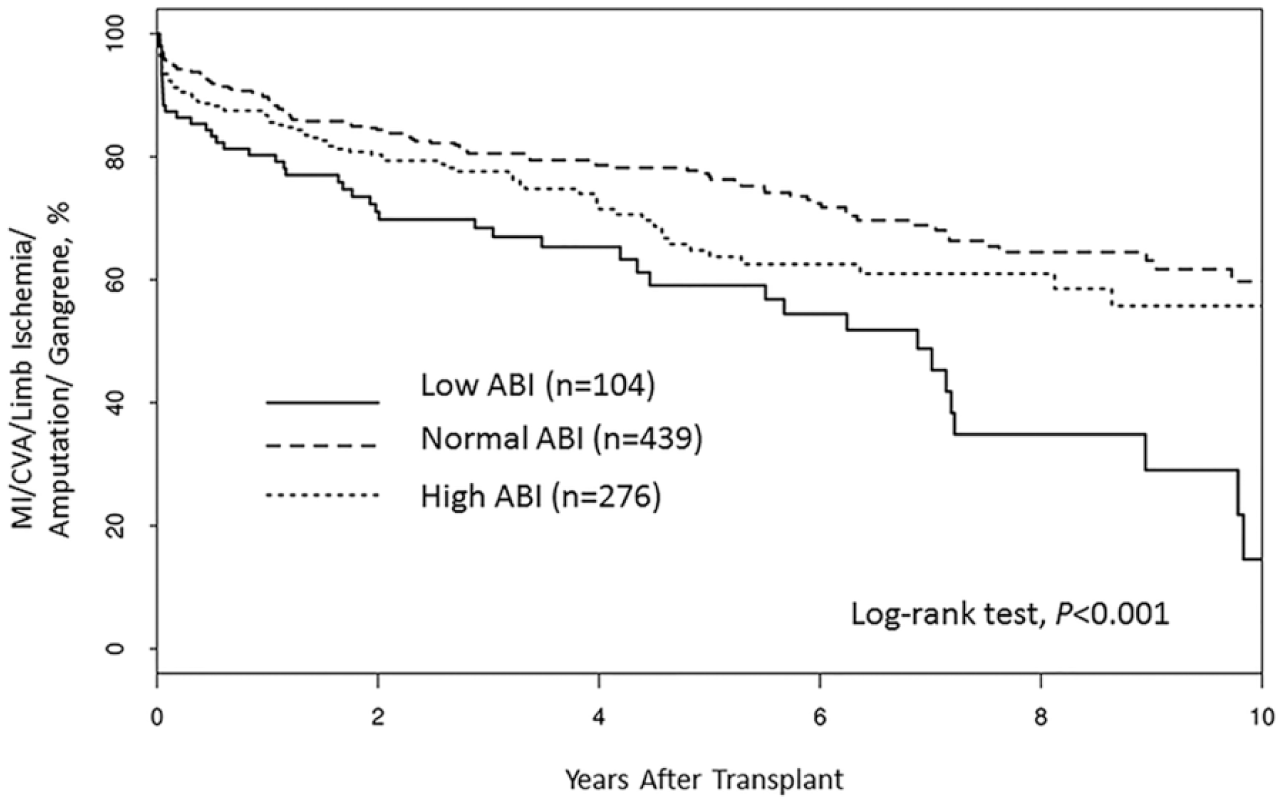
Kaplan–Meier curve showing incidence of myocardial infarction (MI); cerebrovascular accident (CVA); and limb ischemia, amputation, or gangrene by ankle-brachial index (ABI), (Normal ABI: 0.9–1.39: elevated ABI: ⩾1.4; low ABI: <0.9.)
Discussion
Chronic kidney disease is associated with accelerated atherosclerosis including PAD. Patients with PAD have a higher number of cardiovascular events and significant functional impairment. The resting ABI is an accurate method to evaluate the patency of the lower extremity arterial system and is a good indicator for the presence of PAD. In our vascular laboratory we include pulse wave, Doppler wave, and functional symptoms with and without walking as part of a comprehensive physiologic evaluation. Here we sought to address the value of ABI testing in a population of kidney transplant recipients.
In this retrospective study of a cohort of patients who had received kidney transplants and arterial physiologic testing at three different Mayo Clinic sites over approximately 20 years, PAD was diagnosed in almost half of the patients sent for lower extremity physiologic evaluation (46%, mean age 57 years), which is several folds higher than published prevalence in this age group of patients without the need for a kidney transplant. 19 The baseline characteristics for patients included in this study are outlined in Table 1. Diabetes was more common among patients with PAD.
An ABI less than 0.9 was an independent and significant predictor of organ failure (OR, 2.94 (95% CI, 1.75–4.95), p<0.001), mortality (HR, 1.87 (95% CI, 1.26–2.78), p=0.002), and secondary end points (myocardial infarction; stroke; and limb ischemia, gangrene, or amputation; OR, 3.26 (95% CI, 2.01–5.28), p<0.001). These findings occurred regardless of other comorbidities and the number of years that a patient underwent dialysis.
Despite advances in kidney transplantation, patients tend to have a high rate of cardiovascular complications and mortality, 20 which was also seen here. Gowdak et al. demonstrated the importance of screening for major adverse cardiovascular events (MACE) in kidney transplant candidates. 8 Diabetes, PAD and prior myocardial infarction were associated with a higher incidence of significant CAD on angiogram; however, angiography is invasive and costly. 8
ABI testing is fairly inexpensive, especially when the costs are compared with the costs associated with the transplant itself or with the costs of caring for patients with poor transplant outcomes. Therefore, screening for PAD would be beneficial in identifying patients who are at high risk for complications after kidney transplant and tailoring therapy toward modifying risk factors.
Limitations
The study was limited by its retrospective nature and, possibly, by the lack of standardization or differing referral patterns for arterial physiologic testing at the three sites. Some of the vascular studies were done as many as 5 years before transplantation, and have been grouped into normal controls. If their PAD progressed prior to transplant without additional documentation, there may have been PAD at the time of transplant, and this possibility was not accounted for. Ideally, all patients would have had their vascular study within a year of transplant.
The addition of toe–brachial index or Doppler waveform data would have helped to further characterize patients within the ABI >1.4. Furthermore, the sub-group of study patients with both pre-transplant and post-transplant ABI results was too small to analyze further or use to draw conclusions. It is unclear why some patients’ ABI would increase or decrease after transplant, as a pattern of causation was not observed. This question can be further evaluated in future studies. Future studies should also evaluate functional capacity and walking distance and have a large enough sample size to be able to characterize outcomes for the low ABI group based on severity.
Conclusion
ABI evaluation is a meaningful tool for enhancing cardiovascular risk assessment in patients who undergo kidney transplants. Low ABIs predicted higher mortality and cardiovascular outcomes in our patients. Applying this knowledge to patients before transplant may be helpful in prioritizing patients for transplant.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.