Abstract

Keywords
This study describes changes over time in baseline comorbidities, leg symptoms, functional impairment, and medications of people with lower-extremity peripheral artery disease (PAD) randomized into clinical trials designed to improve walking performance.
Baseline data (prior to randomization) were combined from clinical trials conducted between 2004 and 2021 that tested novel interventions to improve walking performance in people with PAD without limb-threatening ischemia. Studies included SILC, GOALS, PROPEL, RESTORE, HONOR, COCOA, LITE, and TELEX.1–8 Additional studies were GIFT II (NCT04377126), NICE (NCT03743636), HI-PAD (NCT03363165), PERMET (NCT03054519), and INTERCEDE (NCT03871075). Individuals who participated in more than one clinical trial were included only once, for the first trial that they participated in.
The institutional review board at all sites approved the protocols. All participants provided written, informed consent. Participants were randomized from Chicago, IL; Minneapolis, MN; New Orleans, LA; Pittsburgh, PA; New York, NY; and Gainesville, FL. Recruitment methods included contacting patients with known PAD, advertisements on public buses and trains, mailing postcards to people over age 50, and newspaper and radio advertisements.
In the SILC trial, the inclusion criterion was an ankle–brachial index (ABI) ⩽ 0.95. In all other trials, the inclusion criterion was ABI ⩽ 0.90. Participants meeting other criteria for PAD, such as noninvasive vascular laboratory evidence of PAD, were potentially eligible.1–8 Exclusion criteria were similar across studies.1–8 Individuals with limb-threatening ischemia were excluded.
Comorbidities and cigarette smoking behavior were assessed using a standard questionnaire administered by trained and certified staff. Height and weight were measured. Body mass index (BMI) was calculated as weight (kg) divided by height (m2). Medications taken by participants at enrollment (prior to randomization) were collected. The Principal Investigator (MMM) categorized medications, blinded to other participant characteristics.
The 6-minute walk test and usual- and fast-paced 4-meter walking velocity were measured using a standard protocol.1–8 The presence of classical symptoms of intermittent claudication (exertional calf pain that did not begin at rest and resolved within 10 minutes of rest) was ascertained using the San Diego Claudication Questionnaire.1–8 Atypical exertional leg symptoms not consistent with intermittent claudication were classified as ‘other exertional leg symptoms’.
Baseline characteristics of participants with PAD were compared across seven time periods (2004–2008, 2008–2012, 2012–2015, 2015–2016, or 2016–2017, 2017–2019, and 2019–2021) using general linear models for trend for continuous variables and logistic regression for remaining variables, adjusting for study site. The SILC trial excluded people with a short physical performance battery (SPPB) score of 12 (i.e., the best functional performance). Therefore, analyses of change in walking performance over time excluded SILC. Statistical analyses used SAS Statistical Software, version 9.4 (SAS Inc., Cary, NC, USA).
A total of 1225 participants with PAD were enrolled between 2004 and 2021, including 45.1% women and 52.6% Black individuals (Table 1). Table 1 shows temporal changes in the characteristics of enrolled participants at baseline, prior to randomization. Over time, the proportion of participants with history of pulmonary disease, cancer, and exertional leg symptoms atypical for classical intermittent claudication significantly increased. Functional performance at baseline significantly declined over time. The proportion of participants taking a statin and the proportion taking opioid medications at baseline significantly increased. Few participants were taking cilostazol throughout the period. The proportion taking pentoxifylline significantly declined.
Temporal trends in the characteristics of people with peripheral artery disease enrolled in clinical trials of walking performance from 2004 to 2021.
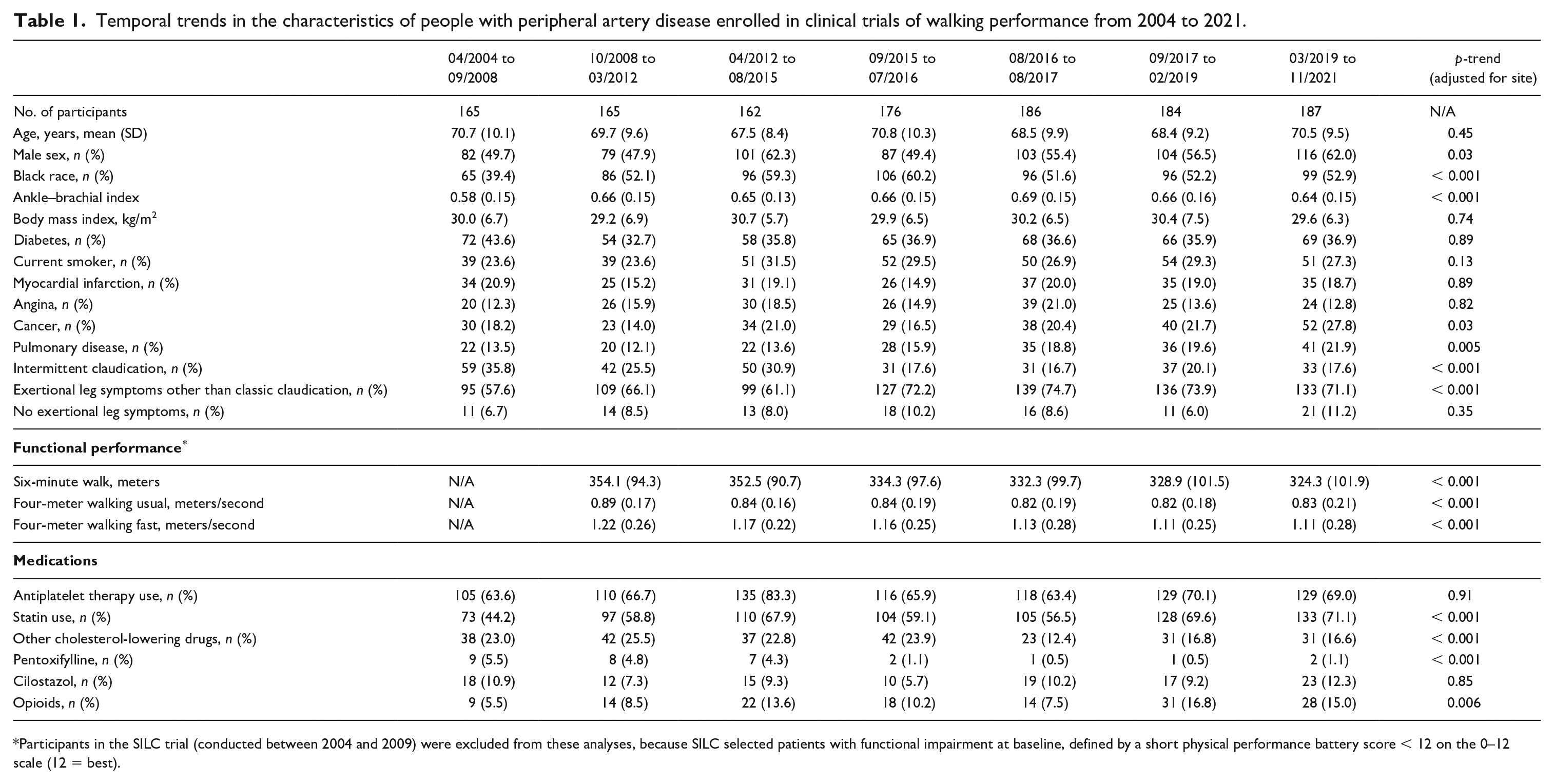
Participants in the SILC trial (conducted between 2004 and 2009) were excluded from these analyses, because SILC selected patients with functional impairment at baseline, defined by a short physical performance battery score < 12 on the 0–12 scale (12 = best).
Among 1225 people with PAD from five states enrolled in randomized clinical trials between 2004 and 2021, the prevalence of cancer, pulmonary disease, and atypical leg symptoms other than classic intermittent claudication significantly increased over time. The higher prevalence of atypical exertional leg symptoms, if also occurring in medical centers or the community, may make PAD more difficult to recognize and diagnose.
Statin use significantly increased over time, to 71.1% in 2019–2021, indicating improved medical therapy over time for people with PAD. Only a small proportion of people with PAD used cilostazol or pentoxifylline during any period, and pentoxifylline use significantly decreased over time, perhaps related to current clinical practice guideline recommendations against use of pentoxifylline, due to lack of efficacy. 9 In contrast, opioid use significantly increased over time to 15% in 2021. It is unclear whether the higher prevalence of opioid use was due to PAD-related pain or due to comorbid conditions causing pain.
This study has limitations. First, these results may not be generalizable to people with PAD who were not eligible or interested in clinical trial participation. Second, the clinical trials were all conducted by the same principal investigator.
In conclusion, more people with PAD may be living with comorbidities. Greater opioid use and declines in 6-minute walk distance over time suggest increased chronic pain and disability in PAD, whereas increases in statin usage may have prolonged lives and enhanced the chronicity of PAD.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr McDermott has received research funding from Helixmith and Regeneron. She has received unrelated research support from Mars, ArtAssist, Chromadex, and ReserveAge.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Heart, Lung, and Blood Institute (R01HL073351, R01HL088589, R01HL107510, R01HL126117, R01HL131771), the National Institute on Aging (R21AG047510, R01AG070033, R01AG072335, R21AG056903, R21AG050897), and by the American Heart Association Strategically Focused Research Network on Peripheral Vascular Disease (18SFRN33900097, 18SFRN33970010).
