Abstract
Some believe that certain patients with intermittent claudication may be unsuitable for supervised exercise therapy (SET), based on the presence of comorbidities and the possibly increased risks. We conducted a systematic review (MEDLINE, EMBASE and CENTRAL) to summarize evidence on the potential influence of diabetes mellitus (DM) on the response to SET. Randomized and nonrandomized studies that investigated the effect of DM on walking distance after SET in patients with IC were included. Considered outcome measures were maximal, pain-free and functional walking distance (MWD, PFWD and FWD). Three articles met the inclusion criteria (n = 845). In one study, MWD was 111 meters (128%) longer in the non-DM group compared to the DM group after 3 months of follow-up (p = 0.056). In a second study, the non-DM group demonstrated a significant increase in PFWD (114 meters, p ⩽ 0.05) after 3 months of follow-up, whereas there was no statistically significant increase for the DM group (54 meters). On the contrary, the largest study of this review did not demonstrate any adverse effect of DM on MWD and FWD after SET. In conclusion, the data evaluating the effects of DM on SET were inadequate to determine if DM impairs the exercise response. While trends in the data do not suggest an impairment, they are not conclusive. Practitioners should consider this limitation when making clinical decisions.
Keywords
Introduction
Peripheral arterial disease (PAD) is a major manifestation of systemic atherosclerosis, characterized by obstruction of the lower limb arteries. PAD affects over 200 million individuals worldwide. 1 The most common symptom of PAD is intermittent claudication (IC) resulting from poor oxygenation of the lower extremity muscles. IC is typically experienced as pain, cramping, or aching in calves, thighs or buttocks. Symptoms are induced by walking and relieved by a short period of rest. 2
Diabetes mellitus (DM) is known to have a strong association with the development of PAD. 3 The exact prevalence of DM in patients with PAD is unclear. Data from the Framingham Heart Study revealed that 20% of symptomatic PAD patients also had DM. 4 The associated pro-atherogenic changes include increased vascular inflammation, derangements of cellular components of the vasculature, generalized endothelial cell dysfunction as well as alterations in blood cells and hemostatic factors. 5 Moreover, atherosclerotic lesions are more likely to be diffuse and more distal, accounting for an atypical IC presentation in diabetic patients. 6 Patients with PAD and DM also experience worse lower extremity function than individuals with PAD alone. 7 Other studies have shown faster disease progression, high rates of foot ulcers and more lower limb amputations in patients with PAD and DM.8,9
Treatment of IC, irrespective of the presence of DM, should consist of two components. First, cardiovascular risk management aimed to minimize the risk of future cardiovascular and cerebrovascular ischemic events. Second, IC complaints require symptomatic treatment. Recent international guidelines on the management of IC recommend supervised exercise therapy (SET) as the first-line therapy.10–12
Although the benefits of SET for IC have been well documented, these findings may have limited generalizability in patients with both IC and DM. A recent editorial claimed that certain IC patients may be unsuitable for SET, based on the presence of comorbidities and the possibly increased risks. 13 Characteristics specific to DM may influence exercise capacity due to reduced skeletal muscle oxidative capacity and mitochondrial dysfunction.14,15 As a consequence, SET might be less effective in these patients. However, information regarding the influence of DM on SET in contemporary literature is limited. We conducted a review to summarize the literature and to address the lack of evidence on the potential influence of DM on the response to SET in patients with IC.
Methods
Eligibility criteria
A systematic review of the literature was conducted according to the PRISMA guidelines. 16 Randomized and nonrandomized studies investigating SET in PAD patients with IC due to lower limb atherosclerosis were eligible for inclusion. Only original articles comparing the effect of SET in diabetic and nondiabetic patients with IC were included. Studies of patients with asymptomatic lower limb atherosclerosis were excluded. Inclusion of studies was not limited by the content, frequency, duration or intensity of the SET program. Studies of patients who did not receive SET or who received usual care were excluded. Considered outcome measures were maximal walking distance or time, pain-free walking distance or time and functional walking distance or time. The search was limited to studies in humans and articles written in English or Dutch. No limitation to the date of publication was considered.
Search strategy
We performed a search in MEDLINE, EMBASE and CENTRAL in April 2016, using MeSH, Emtree and free terms (Supplementary file – Search strategies). We also hand-searched the bibliographies of included articles. Two authors (DH and LG) independently screened titles and abstracts and selected articles, based on full-text evaluation. Disagreement between reviewers was discussed and resolved by consensus.
Data extraction and management
The same two authors used a standardized data extraction form to extract the following data: details of the study population, description of the SET protocol and outcome measures (walking distance or time, change in walking distance or time, difference in walking distance or time between diabetic and nondiabetic patients with IC, and walking speed).
Quality assessment
Studies were evaluated for methodological quality using a modified version of the Downs and Black checklist for measuring study quality. 17 The checklist consisted of 27 questions with a maximum score of 28 points. Papers were considered as high-quality studies when they scored 20 to 28 points, moderate-quality studies when they scored 10 to 19 points and low-quality studies when they scored 1 to 9 points. The quality of the evidence was evaluated using the GRADE approach. 18
Data analysis
Calculations of outcome measures were made using Microsoft Excel 2010 software (Microsoft, Redmond, WA, USA). Outcome measures were expressed in walking distance (meters), absolute increase (meters) and relative increase (percentage) for the different follow-up moments. If studies reported walking capacity in walking time (seconds), we converted the outcome to walking distance in meters using the information on the treadmill protocol. Moreover, if improvements in walking capacity were reported in increase in walking time (seconds), outcomes were converted to absolute increase in meters and relative increase in percentage. Graphs were created with GraphPad Prism 6 software (GraphPad Software, Inc., La Jolla, CA, USA).
Results
Study selection
The literature search identified 962 potentially relevant and unique articles (Figure 1). After title and abstract screening, 898 articles were excluded. Full-text evaluation of the remaining 64 articles resulted in the exclusion of another 61 articles. Finally, three studies with a total of 845 patients with IC were included. Among them, 268 patients were diabetic (only type 2 DM) and 577 patients were nondiabetic.
Flow diagram of study selection. IC, intermittent claudication; SET, supervised exercise therapy; DM, diabetes mellitus.
Study characteristics and quality assessment
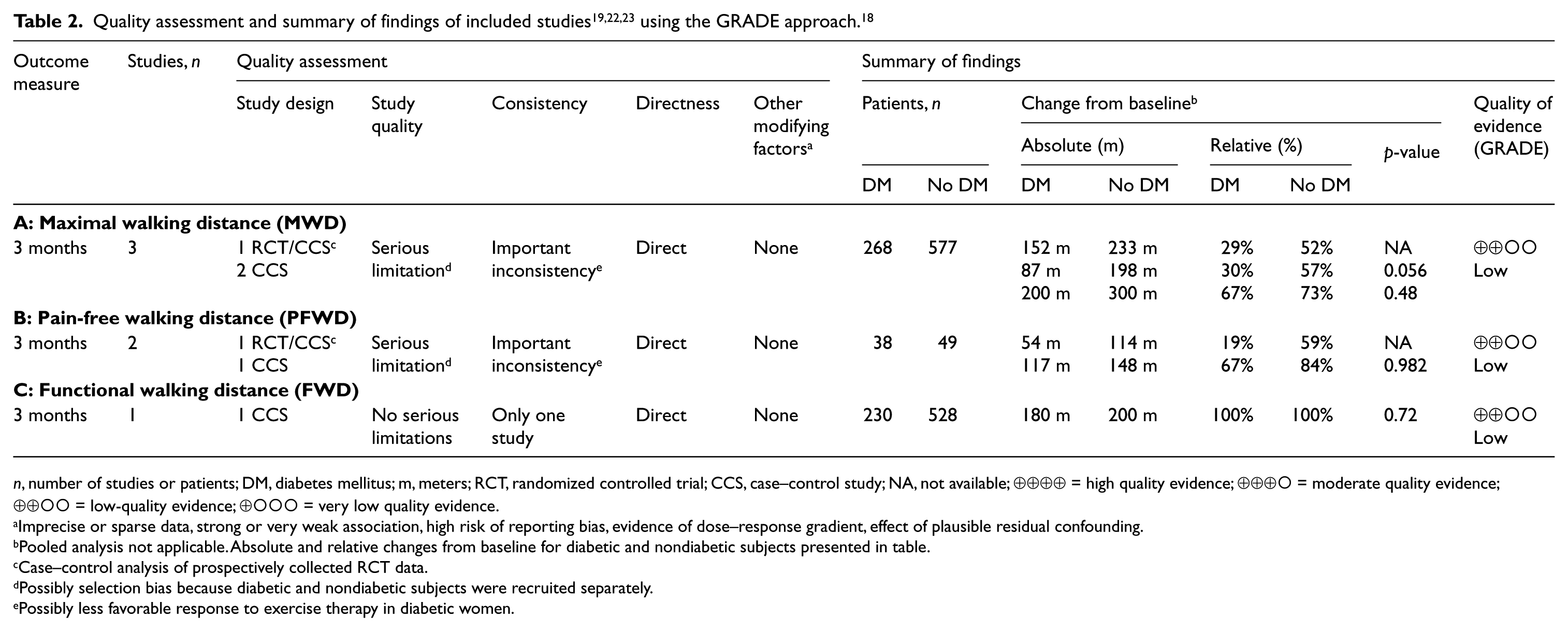
Of the three included studies, two were performed in the USA and one was performed in The Netherlands (Table 1). The mean age of both diabetic patients and nondiabetic patients was 67 years. Overall, more men than women participated in the studies (61% vs 39%). Methodological quality assessment using the modified version of the Downs and Black checklist revealed that there were two high-quality studies and one moderate-quality study (Supplementary file – Assessment). Initially, we planned to conduct a meta-analysis, but given the lack of randomized controlled trials (RCTs) and considerable heterogeneity in study population and methodology, we limited our study to a review to summarize the literature and direct future research. A GRADE analysis rated the quality of the evidence as low (Table 2).
Characteristics of included studies.
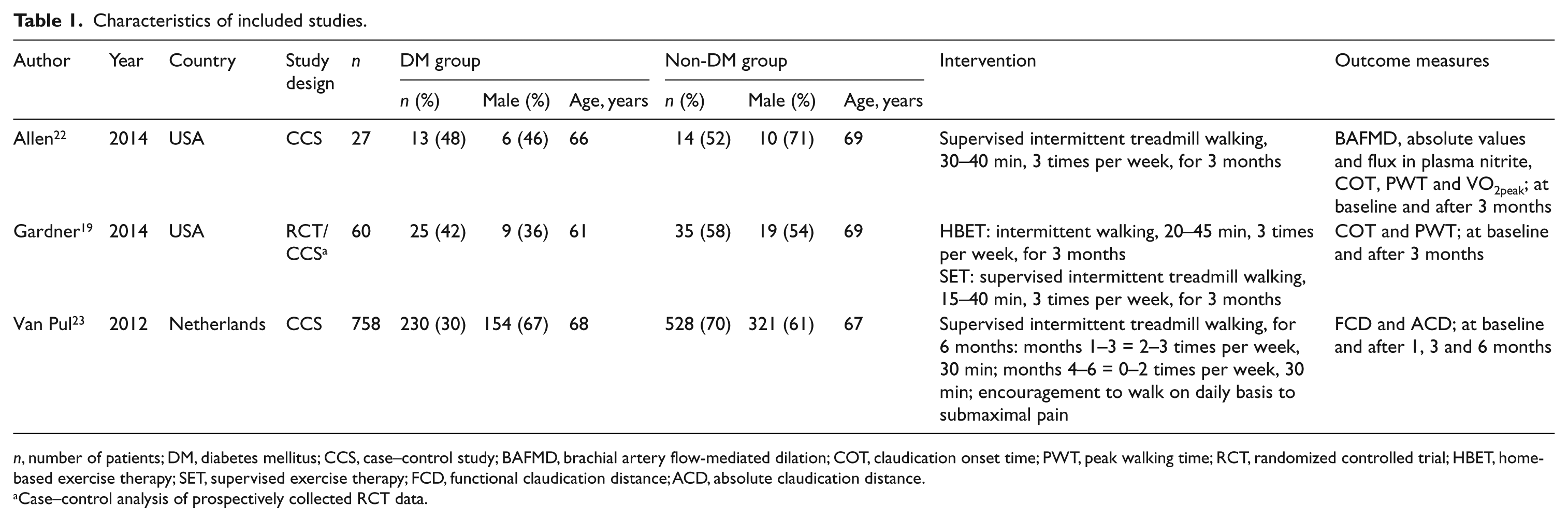
n, number of patients; DM, diabetes mellitus; CCS, case–control study; BAFMD, brachial artery flow-mediated dilation; COT, claudication onset time; PWT, peak walking time; RCT, randomized controlled trial; HBET, home-based exercise therapy; SET, supervised exercise therapy; FCD, functional claudication distance; ACD, absolute claudication distance.
Case–control analysis of prospectively collected RCT data.
n, number of studies or patients; DM, diabetes mellitus; m, meters; RCT, randomized controlled trial; CCS, case–control study; NA, not available; ⊕⊕⊕⊕ = high quality evidence; ⊕⊕⊕○ = moderate quality evidence; ⊕⊕○○ = low-quality evidence; ⊕○○○ = very low quality evidence.
Imprecise or sparse data, strong or very weak association, high risk of reporting bias, evidence of dose–response gradient, effect of plausible residual confounding.
Pooled analysis not applicable. Absolute and relative changes from baseline for diabetic and nondiabetic subjects presented in table.
Case–control analysis of prospectively collected RCT data.
Possibly selection bias because diabetic and nondiabetic subjects were recruited separately.
Possibly less favorable response to exercise therapy in diabetic women.
Interventions
All studies included a SET program that predominantly consisted of treadmill-based walking (Table 1). In one study, patients were randomized to either a home-based exercise therapy (HBET) program or a SET program. 19 The HBET program was designed to be as similar to the SET program as possible. In all studies, SET was performed at least three times per week for at least 3 months.
Outcome measures
To evaluate therapy progress, the maximal walking distance (MWD), pain-free walking distance (PFWD) and/or functional walking distance (FWD) were assessed at baseline and at least after 3 months of follow-up after SET. However, the included studies used different terminology for these three outcome measures. To assess walking distances, all studies used the standardized Gardner maximal treadmill test protocol with a constant speed of 3.2 kph (or 2 mph) and an increasing slope of 2% every 2 minutes to a maximum slope of 10%.20,21
The MWD was defined as the distance that a patient had covered before he/she was forced to stop walking because of intolerable claudication pain. The PFWD was defined as the distance that a patient had covered when he/she first experienced claudication pain. The FWD was defined as the distance that the patient had covered until he/she preferred to stop walking because of a certain level of unacceptable claudication pain.
Effect of DM on MWD
All three studies (n = 845 patients) included outcomes on MWD after SET (Table 2A and Figure 2).19,22,23
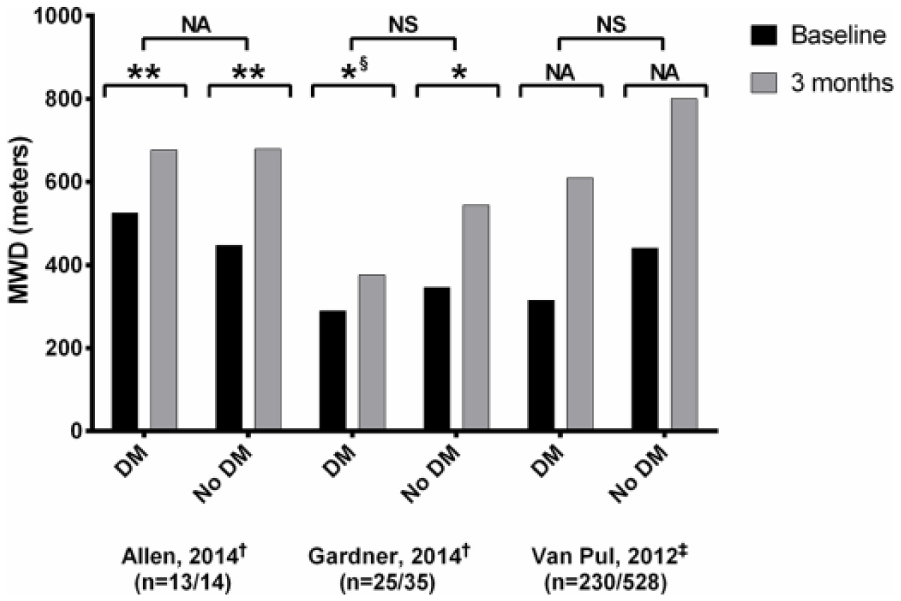
Maximal walking distance (MWD) of included studies19,22,23 (n = 845 patients). MWD, maximal walking distance; DM, diabetic group; No DM, nondiabetic group; n, number of participants for diabetic/nondiabetic groups; †distances are mean values; ‡distances are represented as median values; *significant difference, p < 0.05; **significant difference, p < 0.01; §significant difference in men, but no significant difference in women; NS, no significant difference; NA, no data on significance available.
Following the completion of 3 months of SET as reported by Allen et al., the non-DM group showed a 52% mean increase in MWD (233 meters, p ⩽ 0.01), while the DM group increased by 29% (152 meters, p ⩽ 0.01). 22 Baseline characteristics of diabetic and nondiabetic subjects were not significantly different. Unfortunately, no comparison of change in walking distance between subgroups was provided.
In the study by Gardner et al., patients were classified according to their diabetic status and gender, resulting in four study groups. 19 Three groups demonstrated a significant improvement (p < 0.05) in MWD after 3 months of exercise therapy, except for diabetic women (p > 0.05). The mean MWD of the overall non-DM group increased by 57% (198 meters), compared to a 30% (87 meters) increase for the overall DM group. There was a trend for less improvement in diabetic IC patients (p = 0.056). Furthermore, only 56% of diabetic women had an increase in MWD versus 78% of diabetic men as reported by Gardner et al.
At baseline and at 1, 3, and 6 months of follow-up, Van Pul et al. found that the median MWD in patients with DM was significantly lower compared to patients without DM. 23 However, relative and absolute increases in median MWD were not significantly different between both groups. After 3 months, median MWD increased by 73% in the non-DM group (median increase of 300 meters) compared to 67% in the DM group (median increase of 200 meters, p = 0.48). At 6 months of follow-up, median MWD increased by 100% in the non-DM group (median increase of 400 meters) compared to 91% in the DM group (median increase of 270 meters, p = 0.48).
Effect of DM on PFWD
Of the three included studies, two studies (n = 87 patients) reported on PFWD outcomes (Table 2B and Figure 3).19,22
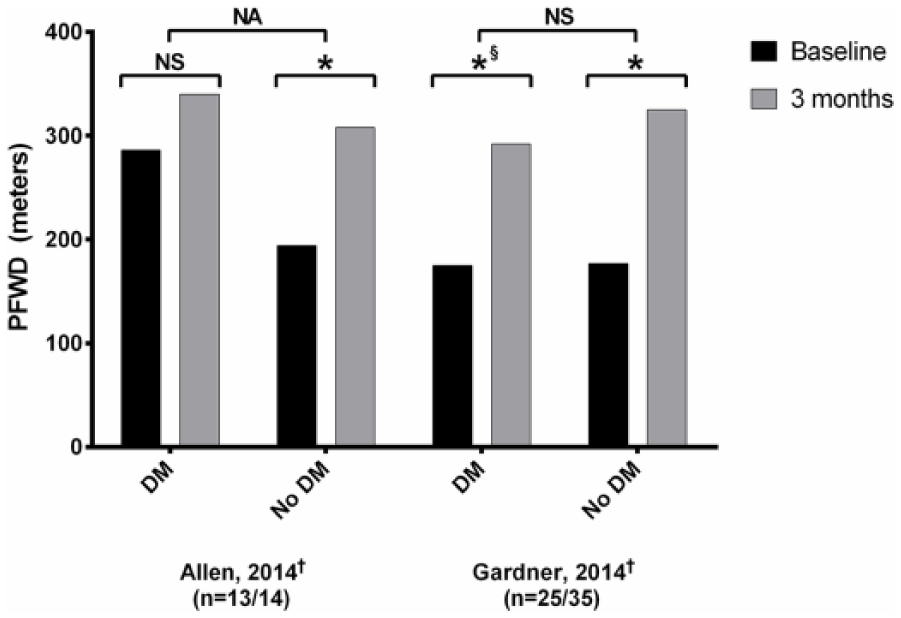
Pain-free walking distance (PFWD) of included studies19,22 (n = 87 patients). PFWD, pain-free walking distance; DM, diabetic group; No DM, nondiabetic group; n, number of participants for, diabetic/nondiabetic groups; †distances are represented as mean values; *significant difference, p < 0.05; §significant difference in men, but no significant difference in women; NS, no significant difference; NA, no data on significance available.
Following the completion of 3 months of SET as reported by Allen et al., the non-DM group demonstrated a 59% mean increase in PFWD (114 meters, p ⩽ 0.05), whereas there was no statistically significant increase in PFWD for the DM group (19%, 54 meters). 22 Again, no comparison of change in walking distance between subgroups was provided.
In the study by Gardner et al., three groups had significant improvement (p < 0.05) in mean and median PFWD after 3 months of exercise therapy, except for diabetic women (p > 0.05). 19 The mean PFWD of the overall non-DM group increased by 84% (148 meters) compared to 67% (117 meters) for the overall DM group (p = 0.982). In patients with DM, the increase in mean PFWD was 425% (196 meters) greater in men than in women (p < 0.05), whereas in those without DM, the increase in mean PFWD was only 35% (44 meters) greater in men (p > 0.05). Similar to the MWD results, diabetic women had the lowest percentage of responders to exercise therapy. Only 37% of women with DM had an increase in PFWD versus 100% of men with DM, as reported by Gardner et al. (p < 0.01).
Effect of DM on FWD
Of the three included studies, one study (n = 758 patients) reported on FWD outcomes (Table 2C and Figure 4). 23
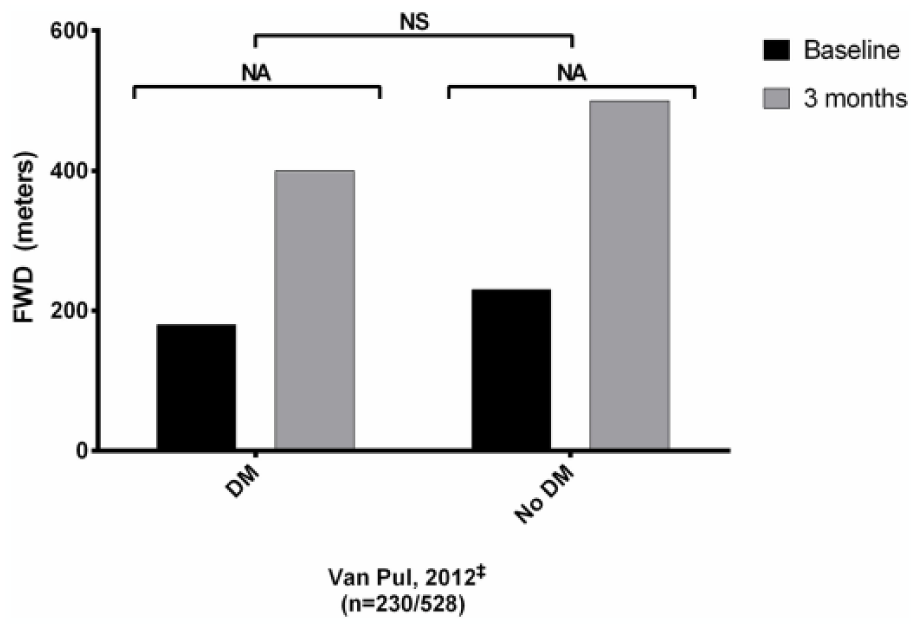
Functional walking distance (FWD) of included study 23 (n = 758 patients). FWD, functional walking distance; DM, diabetic group; No DM, nondiabetic group; n, number of participants for, diabetic/nondiabetic groups; ‡distances are represented as median values; NS, no significant difference; NA, no data on significance available.
At baseline and at 1, 3, and 6 months of follow-up, Van Pul et al. found that the median FWD in patients with DM was significantly lower compared to patients without DM. 23 However, relative and absolute increases in median MWD were not significantly different between both groups. After 3 months of SET, median FWD increased by 100% in both groups, corresponding to a median increase of 200 meters in the non-DM group and a median increase of 180 meters in the DM group (p = 0.72). At 6 months of follow-up, median FWD increased by 148% in the non-DM group (median increase of 332 meters), compared to 140% in the DM group (median increase of 280 meters, p = 0.72).
Discussion
The aim of the present study was to summarize the literature and to address the lack of evidence on the potential influence of DM on the response to SET in patients with IC. The studies included in this review reported improvement in walking distance parameters for both diabetic and nondiabetic IC patients. However, conflicting results were found when comparing the magnitude of effect of SET. One smaller study reported a nearly significant different increase in MWD in DM versus non-DM patients. 19 Another smaller study demonstrated a significantly improved PFWD in nondiabetic IC patients when compared to diabetics. 22 On the contrary, the largest study of this review including 90% of all patients did not find any deleterious effect of DM on improved values of MWD and FWD after SET. 23 Overall, no significant differences in improvement of walking distances between diabetics and nondiabetics were reported. Consequently, it may be concluded from our review that there is no convincing evidence that SET is less effective in IC patients with DM.
One may question the additional value of our review as recently a meta-analysis on the efficacy of exercise in the presence of DM has been published. In this study, Lyu et al. stratified PAD populations according to the prevalence of DM (<25% DM patients vs 25–50% DM patients) and reported greater improvement in MWD among the subgroups with a lower proportion of diabetic patients. 24 There are some important methodological differences between the meta-analysis of Lyu et al. and the present review. First, the meta-analysis by Lyu et al. included RCTs generally comparing the effects of intensive walking exercise and walking advice in patients with PAD and added subgroup analyses on the effect of DM. In contrast, our review included studies specifically focusing on the effects of DM on the response to SET, the first-line treatment in IC. Second, Lyu et al. compared subgroups with different proportions of diabetic patients, whereas our review compared groups with DM and groups without DM, intuitively a fairer comparison.
Some other studies also investigated the effects of DM on the response to exercise in IC, but did not meet our inclusion criteria. Ubels et al. reported that IC patients with DM gained even more from exercise therapy than their counterparts without DM. 25 Although no differences in PFWD or MWD between diabetic and nondiabetic participants were found, the relative gain in MWD was significantly greater in diabetic than in nondiabetic participants at the end of the training program. This study did not qualify for inclusion in the current review since the patients did not receive a true supervised program, just a training schedule that was adapted every 2 months. In a study by Collins et al., only diabetic IC patients were included and no improvement in MWD and PFWD after a 6-month HBET program was observed. 26 In contrast, a study by McDermott et al. found significant improvement in the 6-minute walking distance after 6 months for both diabetic and nondiabetic participants randomized to a HBET program. 27 Unfortunately, no comparison of change in walking distance between subgroups was provided. The apparent inconsistencies between studies call for further investigations to confirm whether the efficacy of walking exercise is compromised in IC patients with DM.
Outcomes of SET in patients with IC appear to be gender-related. Recent insights suggest that female IC patients have greater baseline limitations of IC, less calf muscle hemoglobin oxygen saturation and lower daily ambulatory activity.28–30 Moreover, higher levels of oxidative stress, insulin resistance and inflammation are present in postmenopausal women compared to similarly aged men, which may also contribute to the lower claudication threshold levels. 29 At the same time, differences between men and women regarding motivation, willingness to push through discomfort and pain tolerance might also play a role. 31 The recent gender-related insights coincide with one study reporting that women with IC, particularly those with DM, represent a vulnerable subgroup. 19 Gender predicted exercise response better than DM, as also demonstrated by the significant gender effect regarding mean MWD and mean PFWD. Nevertheless, the less favorable response to exercise therapy in women was only found in those having DM. Thus, an effect of DM itself cannot be excluded yet. Despite these findings, we strongly feel that all patients with IC should undergo SET, even female patients with DM.
DM is associated with increased cardiovascular mortality and morbidity rates in females compared to males. The excess risk of cardiovascular disease is to a certain part due to DM being more commonly accompanied by other cardiovascular risk factors in women. 32 As a consequence, women with IC and DM may have a worse response to SET compared to IC women without DM. Diabetic women with IC may therefore require a higher dose of exercise to elicit improvements in claudication parameters.
Regular exercise is increasingly promoted as an integral part of the therapeutic regimen for adults with DM. Exercise has been found to reduce hyperglycemia, insulin resistance, dyslipidemia and hypertension.33–35 These effects may lead to an improved cardiovascular disease risk profile. Exercise may also aid in weight loss and may improve glycemic and lipemic control in diabetic people. 36 Additionally, exercise has already been shown to improve physical function and quality of life.37,38 Therefore, we encourage all adults with DM, particularly those with a sedentary lifestyle, to perform regular physical activity and to engage in an exercise program.
Limitations
This systematic review harbors limitations. First, to date, just a handful of studies have examined the effect of DM on walking distance parameters after SET in IC patients. There were only three studies included in this review, with two of these having small sample sizes.19,22 Second, included patients were not evenly matched for gender. Since evidence exists that diabetic women with IC respond poorly to an exercise program, bias might have been introduced. 19 However, the underrepresentation of women in clinical research reflects the underrepresentation of women in daily practice as PAD is frequently underdiagnosed in women. 39 Third, the study by Gardner et al. used two different exercise interventions (SET and HBET). 19 In the original article, both programs were considered as one group – we therefore did the same. Nevertheless, the lack of supervision in the HBET group might have led to an underestimation of the response to SET. 40 Fourth, although some authors did not report on comorbidities and presence of diabetic neuropathy, differences between subgroups may have influenced walking distance parameters. However, one study which conducted multivariate analysis on the effect of comorbidities revealed no effect from these variables. 23 Fifth, diabetic patients were less often smokers than nondiabetic patients. However, smoking behavior was also not associated with nonresponse to exercise.19,23 Besides these limitations, the studies did not examine the effect of DM on quality of life after SET, whereas long-term results beyond 6 months of follow-up in diabetic patients are currently lacking. Future studies are warranted to elucidate the precise influence of DM on SET outcomes.
Conclusion
There is limited literature examining the influence of DM on the response to SET. The data evaluating the effects of DM on SET were inadequate to determine if DM impairs the exercise response. While trends in the data do not suggest an impairment, they are not conclusive. Therefore, the efficacy of SET cannot be ascertained merely based on the presence or absence of DM. Practitioners should consider this limitation when making clinical decisions.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.