Abstract
Lower extremity peripheral artery disease (PAD) induces an ischemic pain in the lower limbs and leads to walking impairment. Electrical stimulation has been used in patients with PAD, but no systematic review has been proposed to address the efficacy of the technique as a treatment for walking impairment in PAD. A systematic search was performed to identify trials focused on electrical stimulation for the treatment of walking impairment in patients with PAD in the Cochrane Central Register, PubMed, Embase, and the Web of Science. Studies were included where the primary outcomes were pain-free walking distance and/or maximal walking distance. When appropriate, eligible studies were independently assessed for quality using the Cochrane Collaboration’s tool for assessing risk of bias. Five studies eligible for inclusion were identified, of which only two were randomized controlled studies. Trial heterogeneity prevented the use of the GRADE system and the implementation of a meta-analysis. Three types of electrical stimulation have been used: neuromuscular electrical stimulation (NMES, n = 3), transcutaneous electrical stimulation (n = 1), and functional electrical stimulation (n = 1). The two available randomized controlled studies reported a significant improvement in maximal walking distance (+40 m/+34% and +39 m/+35%, respectively) following a program of NMES. Owing to the low number of eligible studies, small sample size, and the risk of bias, no clear clinical indication can be drawn regarding the efficacy of electrical stimulation for the management of impaired walking function in patients with PAD. Future high-quality studies are required to define objectively the effect of electrical stimulation on walking capacity.
Keywords
Background
Lower extremity peripheral artery disease (PAD) is a significant public health issue with more than 200 million individuals affected worldwide. 1 Atherosclerosis is the main cause of PAD 2 and leads to the chronic narrowing of arteries thereby reducing the blood flow to the lower extremity. 3 This hemodynamic impairment can induce an ischemic pain in the exercising skeletal muscle of the lower limbs, 4 which severely impairs the performance of daily physical activities – such as walking 5 – as well as quality of life. 6 Furthermore, exercise ischemia is followed by reperfusion when the patient stops to recover from walking pain. Over the long term, acute repetitions of this ischemia-reperfusion cycle set up a myriad of pathophysiological responses, which include a worsening of endothelial function and muscle structural and metabolic abnormalities. 7 This contributes to the impairment of the exercising skeletal muscle function, which further adds to the walking limitation. 7
In addition to optimal pharmacotherapy and to lifestyle modification (e.g. smoking cessation), specific interventions designed to improve walking impairment may have a significant impact on the walking capacity of people with PAD. Supervised walking exercise, cilostazol therapy, and endovascular revascularization each improve walking capacity to different degrees through either hemodynamic or skeletal muscle adaptations, or a combination of both according to the intervention.8,9 Supervised walking exercise is recommended as a first line therapy due to its proven efficiency (expected improvement: +204 m for pain-free walking distance (PFWD) and +307 m for maximal walking distance (MWD)). 10 However, it remains underutilized and underprescribed due to a number of barriers. 11 Further, not every patient can engage in regular exercise as some people with PAD can be affected by comorbidities that limit exercise (e.g. exercise-limiting osteoarthritis or severe chronic obstructive pulmonary disease). Importantly, pain symptoms mean that people with PAD can be unable or reluctant to partake in exercise. 12 Cilostazol therapy is also recommended as an effective therapy for treatment of leg symptoms and to improve walking impairment, but the expected improvement (+32 to +43 m for MWD) is lower compared to supervised exercise therapy. 9 Further, owing to side effects, it has been reported that 20% of patients discontinued cilostazol within 3 months. 13 Revascularization procedures improve walking impairment to a similar extend as supervised exercise therapy, and a synergetic effect may occur when combining both interventions.10,14 However, independently of the issue of the long-term patency, revascularization procedures remain invasive, costly, and are associated with a higher complication rate than exercise.15,16
Given the increasing number of people with PAD with related functional impairment, effective alternative or complementary therapies are needed. Limb compression protocols have been shown to be beneficial in improving walking impairment. 17 However, the technique acts only on hemodynamic through passive compression with no active skeletal muscle activation. 18 Lower extremity electrical stimulation is an older technique that relies on different types of electrical stimulations with different expected physiological effects on hemodynamics, skeletal muscle functions or pain pathway.19,20 Three types of lower extremity electrical stimulation have been used in people with PAD: neuromuscular electrical stimulation (NMES), transcutaneous electrical nerve stimulation (TENS), and functional electrical stimulation (FES). Each type relies on different parameters of electrical stimulation and is expected to have the potential to improve walking capacity by acting on vascular and/or nonvascular pathways (Table 1). To date, no systematic review has been proposed to address the efficacy of the technique as a treatment for walking impairment in PAD.
Description of the three types of lower extremity electrical stimulation together with their expected physiological effects.
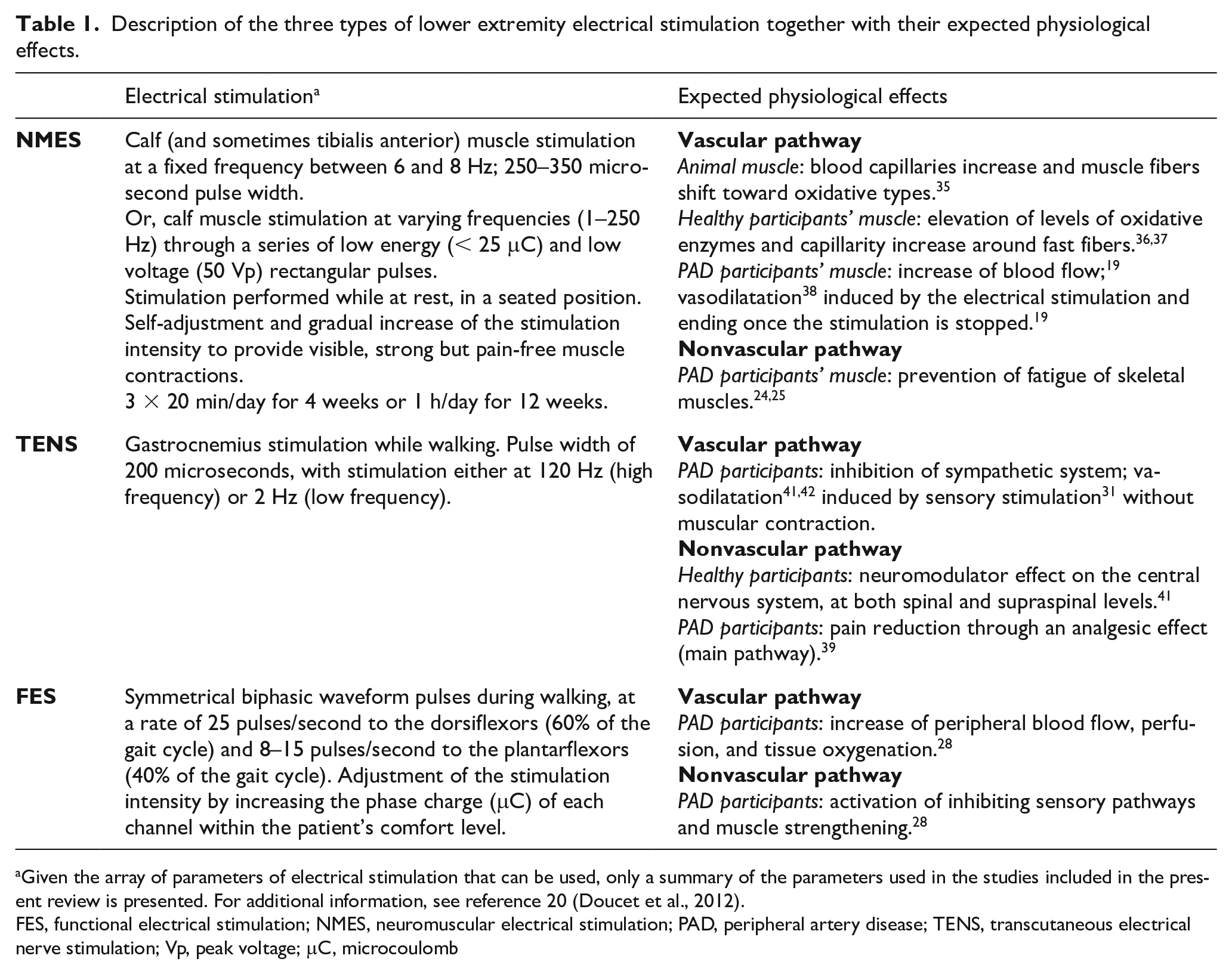
Given the array of parameters of electrical stimulation that can be used, only a summary of the parameters used in the studies included in the present review is presented. For additional information, see reference 20 (Doucet et al., 2012).
FES, functional electrical stimulation; NMES, neuromuscular electrical stimulation; PAD, peripheral artery disease; TENS, transcutaneous electrical nerve stimulation; Vp, peak voltage; μC, microcoulomb
Methods
Search strategy
The review was conducted per the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA). A computerized search of the Cochrane Central Register of Controlled Trials, PubMed (National Library of Medicine), Embase, and the Web of Science for the time period from the inception of each database until May 30, 2019 was performed. Variations of the following search terms were used: peripheral artery disease, electrical stimulation, and walking capacity (Figure 1). The search was limited to the English language and to studies on humans. Bibliographies of relevant articles were screened for additional studies. The full search strategy used for the PubMed database is detailed in Appendix S1 (see the online supplementary material).
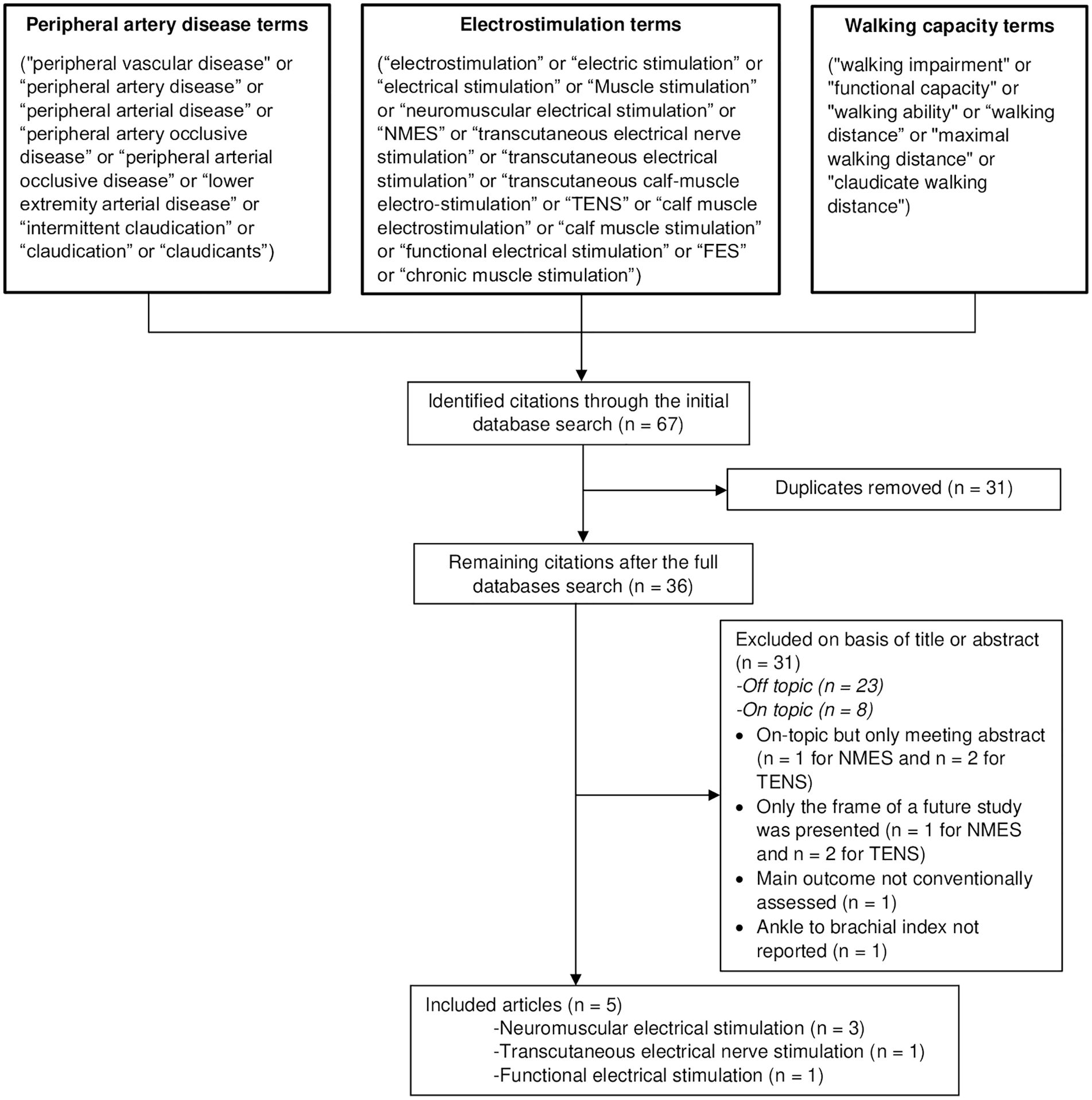
Study flow diagram.
Eligibility criteria
As this systematic review focused on lower extremity electrical stimulation for treatment of walking impairment in PAD, the first eligibility criterion was the presence of PAD defined by an ankle–brachial index (ABI) ⩽ 0.90. A second eligibility criterion was the use of a lower extremity electrical stimulation procedure as a treatment modality applied at the lower limb level only. Third, the main end point of interest for walking impairment was the walking capacity as measured by either treadmill test or 6-minute walk test. These are the two main functional tests used in therapeutic trials of PAD participants.2,21,22 For studies that used treadmill walking tests, the two primary outcome measures to be assessed were the pain-free walking distance or time (PFWD or PFWT) and/or the maximal walking distance or time (MWD or MWT). PFWD, also called claudication walking distance, corresponds to the distance walked before the onset of pain. MWD corresponds to the amount of distance walked before the pain becomes so severe that the patient is forced to stop. PFWD and MWD can also be expressed as a time and this was also considered in the search strategy. For studies that employed the 6-minute walk test, the total distance achieved during the test was the main outcome measure to be assessed.
Selection of studies and data extraction
After the computerized search, the abstract and titles of all retrieved articles were screened by three of the authors (PJ, MC, and ALF). These authors reviewed relevant articles, determined study eligibility, and then extracted data on the study characteristics and end points. The study characteristics recorded were study design, description of intervention, outcome measures, and main findings. Besides the primary end point of interest related to walking capacity, other outcomes assessed were ABI and quality of life changes, and adherence to the treatment protocol.
Assessment of risk of bias
When appropriate, the risk of bias in selected relevant randomized studies was performed using the Cochrane Collaboration’s tool for assessing risk of bias in randomized trials. 23 Two reviewers (PJ and ALF) performed independent assessments of each study. Discrepancies were discussed, and it was arranged that if an agreement could not be reached, a third review author (GM) would be asked to assess the study.
Results
Study characteristics
The search strategy and article selection process identified 13 on-topic studies in which lower extremity electrical stimulation was used as a treatment for walking impairment in PAD, but only five studies24–28 were finally included on the basis of eligibility criteria (Figure 1). These studies included a total of 157 PAD participants, with 116 (73.9%) composing of treatment groups and 41 (26.1%) in the control groups. Among all studies included, three used NMES,24–26 one used TENS, 27 and one used FES. 28 Inclusion criteria varied between studies, with the most common criteria including patients with an ABI ⩽ 0.90, stable intermittent claudication, and a limited walking capacity due to PAD. Inclusion criteria and additional characteristics of each eligible study are presented in Table 2. Among the studies that focused on the topic, eight were excluded. In three studies only the conference abstracts were available, and in another three only the frame of a future study was presented. In one study, an uncommon outcome measure was used to assess treadmill walking capacity (the cumulated walking distance over multiple treadmill walking bouts). In the last study, an inclusion criterion (ABI ⩽ 0.9) was not reported.
Quality of assessment
The Cochrane Collaboration’s tool for assessing risk could be used only for four studies,24,25,27,28 since the other included study did not use a randomized design. 26 The review authors’ judgements about each risk of bias is reported in Figure 2 as percentages across all included studies and detailed in Figure 3 for each study. Three of the four studies were deemed to be at ‘unclear risk’ of selection bias due to inadequate sequence generation and lack of allocation concealment.24,25,27 Performance bias in the four randomized design studies was ranked as ‘unclear risk’ as none of these studies used a double-blind design where both the participants and personnel had no knowledge of the allocated interventions. Two of the four studies followed a single-blinded study design where staff members who performed outcome assessment were blinded – therefore there was a ‘low risk’ of detection bias27,28 – but in the two other studies the risk of detection bias was ‘unclear’.24,25 The dropout level was low for three studies, so attrition bias was low across these studies.24,25,27 Although only the research protocol of one included study was registered in a database of clinical trials, we did not report publication bias because it could reasonably be assumed that the published studies included all expected outcomes (Figures 2 and 3). Trial heterogeneity made it difficult to use the GRADE system and prevented meta-analysis. Hence, a qualitative synthesis was conducted.
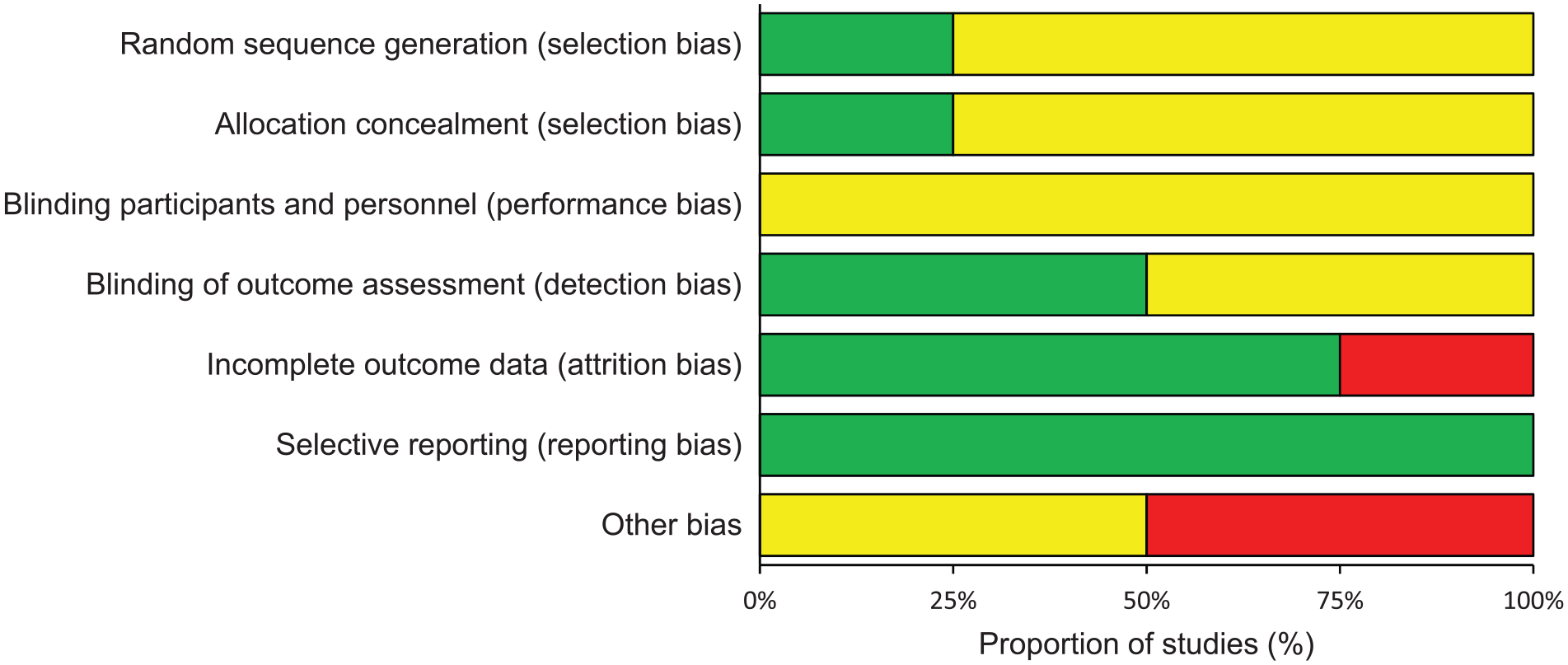
Proportion of studies with each level (‘low risk’, ‘high risk’, and ‘unclear risk’) of bias for each bias item.
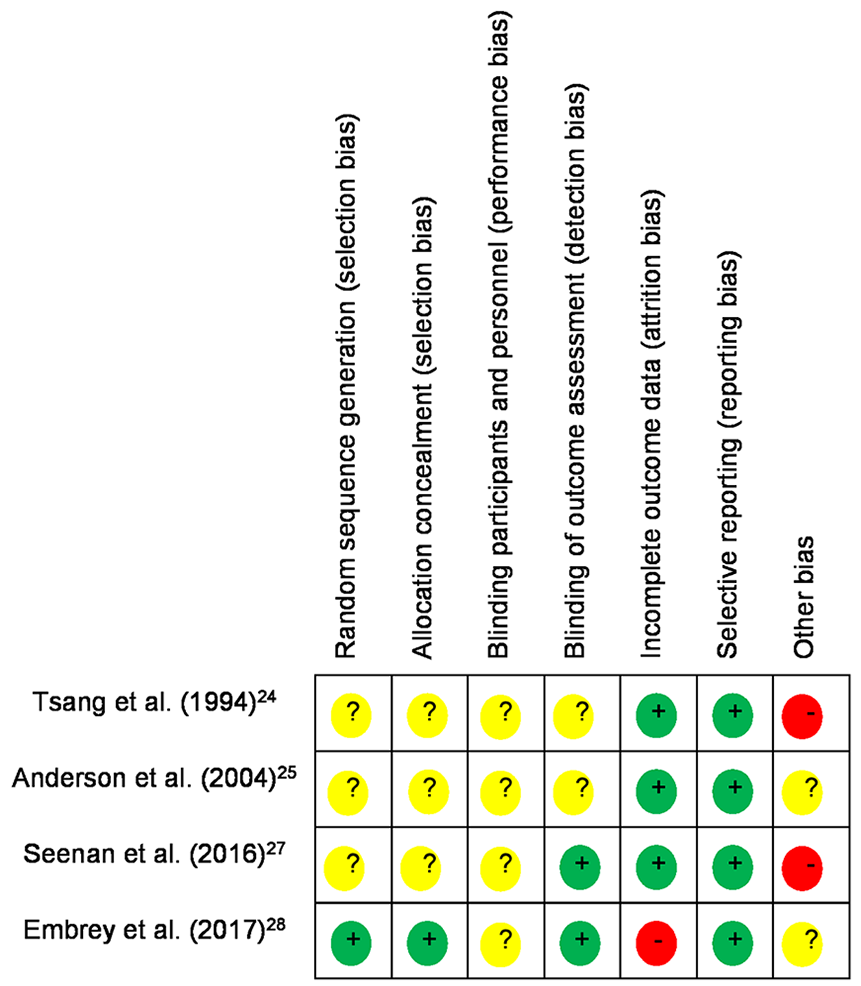
Review of authors’ judgements about each risk of bias item for each included study.
Qualitative synthesis
Table 2 presents an overview of the eligible clinical studies that assessed the effects of lower extremity electrical stimulation on walking capacity in people with PAD.
Studies that assessed the effect of lower extremity electrical stimulation on walking impairment in patients with lower extremity PAD (n = 5 studies).

ABI, ankle–brachial index; ACR, albumin:creatinine ratio; C, control group; FES, functional electrical stimulation; FWD, functional walking distance; HF, high-frequency; IC, intermittent claudication; LF, low-frequency; M/F, male/female; MWD, maximal walking distance; NMES, neuromuscular electrical stimulation; PAD, peripheral artery disease; PADQOL, Peripheral Artery Disease Quality of Life questionnaire; PFWD, pain-free walking distance; T, treatment group; TENS, transcutaneous electrical nerve stimulation.
NMES
The available three studies used NMES as a chronic intervention, with treatment durations ranging from 4 to 12 weeks.24–26 Two of the three studies included a control group, which involved the use of TENS as a placebo.24,25 A significant increase (p < 0.05) in MWD was reported by all studies, with improvements ranging from 39 m to 40 m (34% to 35%) for the two randomized controlled studies and an improvement of 137 m (41%) for the other study (Table 2). PFWD also increased significantly in all studies, from 23 m to 35 m (26% to 60%). Conversely, the two studies that included a control group reported a nonsignificant increase in PFWD and MWD in the control group. With regard to secondary outcomes, there was no significant difference in ABI following the NMES24,25 in two of the studies. However, in one study, a small but significant increase (0.70 ± 0.12 vs 0.74 ± 0.16, p < 0.001) was reported. 26 Quality of life or adherence to protocol was not reported in any study that used NMES.
TENS
The only available study using TENS as a treatment evaluated acute effects by comparing low (i.e. 2 Hz) and high-frequency (i.e. 120 Hz) 27 TENS on PFWD and MWD. Two groups were included, and each group was used as a control for comparison of high and low frequency. With low-frequency TENS, only MWD significantly improved (+49 m, +27%, p < 0.05) whereas with high-frequency TENS only PFWD significantly improved (+21 m, +34%, p < 0.05).
FES
Only one study used FES as their treatment. 28 This study compared two randomized cohorts: FES + Walk group versus Walk group (walk only), with no control group. Chronic effects were measured after a treatment duration of 8 weeks. A significant increase of 43.8 m (6-minute total walking distance) was measured within the FES + Walk group after active treatment of 8 weeks (no significant improvement in the ‘Walk group’) and this increase remained stable after a follow-up of 8 weeks. No data were reported regarding ABI changes following the electrical stimulation. Quality of life measures significantly improved over the duration of the treatment (Table 2). Adherence to protocol was not reported.
Discussion
Several reviews have explored the effects of different treatment modalities on walking capacity in people with lower extremity PAD,9,14,17,29 but to our knowledge this is the first review to focus on the effects of lower extremity electrical stimulation as a treatment for walking impairment.
Because of the low number of eligible studies and the risk of bias among those studies, no clear conclusions can be drawn regarding the efficacy of lower extremity electrical stimulation for the management of impaired walking function in people with PAD. Only two single-blind studies with random allocation of patients to a treatment group or a control group were available, and focused on the NMES effect on walking capacity of PAD participants.24,25 These two studies from the same team showed a significant increase of the MWD (+40 m/+34% and +39 m/+35%, respectively) following a 4-week intervention. By comparison, this increase is close to the one reported from cilostazol therapy 9 or home-base exercise programs; however, it remains lower than the one reported from supervised walking programs 10 or endovascular procedures. 14 Regarding secondary outcomes, the NEMS program tended to have little or no effect on ABI but few data are available to give a definite conclusion. Although statistically significant, the ABI increase reported by Ellul et al. 26 following a 12-week NMES program was small (0.70 ± 0.12 vs 0.74 ± 0.16, p < 0.001) and remains of low clinical relevance considering both the test-retest variability (~10%) of ABI measurement 30 and the inappropriate design of the study (i.e. nonrandomized). Too few studies have also assessed quality of life changes. Finally, although the method of assessment was not always clearly described in the included studies, lower extremity electrical stimulation was only tested on people with PAD and symptoms of intermittent claudication. Whether the intervention would benefit patients with other types of leg symptoms 31 is an interesting perspective that should be addressed in the future.
The present review showed that different types of lower extremity electrical stimulation have been used in people with PAD, and some confusion could rise from the fact that in some studies the PAD participants that were allocated to the control group received one type of lower extremity electrical stimulation (TENS) that was considered as a placebo,24,25 suggesting no or a limited effect of this type of electrical stimulation on walking capacity. The three different types of stimulation (i.e. NMES, TENS, FES) rely on different characteristics in the signals of electrical stimulation used 20 and can target different pathways to potentially increase walking capacity. Regarding NMES, it should be underlined that the three included studies have used the same technique, which relied on an electrical stimulation directly applied at the calf muscle level throughout a stimulator and electrodes. However, a new technique has been recently proposed based on NMES delivered at the foot level using a footplate. 32 The identified three studies32–34 that used this new technique were excluded from the present review since eligibility criteria were not reached (Figure 1). Whether one technique is more efficient than another to improve walking capacity in PAD is unknown. Furthermore, in the included studies, the use of NMES and FES relied on a chronic procedure,24–26,28 whereas TENS was used as an acute procedure. 27 These important distinctions support the choice we made to separately address the potential effect of each type of lower extremity electrical stimulation on walking capacity, although that lowered the number of eligible studies for each category.
The study of mechanisms behind the increase in walking function was beyond the scope of the present review. However, for the reader’s information, Table 1 provides an overview of the vascular and nonvascular pathways that could be involved in the increase of walking capacity. NMES has been tested on humans and animals and has been shown to target both the vascular and the nonvascular pathways. It has been proposed that NMES enhances capillary supply and blood flow while also elevating oxidative enzyme capacity.35–37 In people with PAD, vasodilation occurred and blood flow was increased following an acute session of NMES.19,38 Two studies reported an improvement in skeletal ankle flexors muscle fatigability following a 4-week program of NMES in PAD participants.24,25 TENS may also target both the vascular and nonvascular pathways. The main pathway that could explain the improvement in walking capacity is the nonvascular pathway as this pathway causes an alleviation of pain through an analgesic effect, 39 which is most likely to lead to increased pain-free walking. Finally, using FES, the potential mechanisms involved remains purely speculative and includes an increased perfusion, the promotion of peripheral artery blood flow and an increase in tissue oxygenation. 28
The underlying mechanics potentially involved in the improvement of walking capacity, together with the trends – although limited – that can be observed regarding the effects of NMES on walking capacity, should encourage the development of future studies with an appropriate experimental design. It would be of interest for future studies to include a non-intervention control group. Another option for future studies would be to directly compare the effect of lower extremity electrical stimulation with supervised or home-based walking programs on walking capacity in people with PAD. Interestingly, the study from Lawton and colleagues will assess the potential adjunct effect of NMES when used with supervised walking training or best medical therapy. 34
The lack of an appropriate control group was clearly a major bias in most of the eligible studies for the present review. Interestingly, this highlights the fact that the definition of a control procedure remains an issue when addressing the effects of a given type of lower extremity electrical stimulation on walking capacity in people with PAD. Although the use of a SHAM intervention could be seen as an ideal way to design double-blind studies, it raises issues with regards to the ethics of volunteer deception and the risk of volunteer awareness of the SHAM treatment, with a subsequent risk of unbalanced dropout rates between groups. We would recommend the use of a control group with patients receiving lifestyle advice including walking advice together with best medical therapy, which may be the best solution both at the methodological (intention to treat approach) and ethical levels.
An additional factor to consider is the displacement of physical activity due to the duration of the intervention. In the case of NMES, single or multiple sessions per day are prescribed to achieve a total daily duration of 1 hour or more.24–26 In a population of people with PAD that already exhibit low levels of physical activity, following such a program in the long term may exacerbate their sedentary behavior and worsen their functional impairment. 40
Conclusion
An analysis of the currently available studies indicates that it is not yet possible to draw a conclusion on the efficacy of lower extremity electrical stimulation for the improvement of walking impairment in people with PAD. Future higher quality studies, with lower risk of bias are required, relying on randomized controlled trials with the definition of an appropriate control procedure. The results of such future trials on lower extremity electrical stimulation may improve the management of people with PAD.
Supplemental Material
Appendix_S1 – Supplemental material for A systematic review of lower extremity electrical stimulation for treatment of walking impairment in peripheral artery disease
Supplemental material, Appendix_S1 for A systematic review of lower extremity electrical stimulation for treatment of walking impairment in peripheral artery disease by Pierre Jéhannin, Meghan Craughwell, Loukman Omarjee, Alan Donnelly, Vincent Jaquinandi, Guillaume Mahé and Alexis Le Faucheur in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported in part by a grant from the French Ministry of Health (PHRCI-15-033; IDRCB: 2016-A00971-50). Our team also received Veinoplus® devices from Ad Rem Technology Society to conduct our ongoing research project entitled ELECTROPAD (NCT03795103). Meghan Craughwell benefited from an Erasmus grant and funding from the ENS Rennes.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
