Abstract
Introduction:
There are no randomized trials studying the outcomes of mechanical aspiration thrombectomy (MAT) for management of pulmonary embolism (PE).
Methods:
We performed a systematic review and meta-analysis of existing literature to evaluate the safety and efficacy of MAT in the setting of PE. Inclusion criteria were as follows: studies reporting more than five patients, study involved MAT, and reported clinical outcomes and pulmonary artery pressures. Studies were excluded if they failed to separate thrombectomy data from catheter-directed thrombolysis data. Databases searched include PubMed, EMBASE, Web of Science until April, 2021.
Results:
Fourteen case series were identified, consisting of 516 total patients (mean age 58.4 ± 13.6 years). Three studies had only high-risk PE, two studies had only intermediate-risk PE, and the remaining nine studies had a combination of both high-risk and intermediate-risk PE. Six studies used the Inari FlowTriever device, five studies used the Indigo Aspiration system, and the remaining three studies used the Rotarex or Aspirex suction thrombectomy system. Four total studies employed thrombolytics in a patient-specific manner, with seven receiving local lysis and 17 receiving systemic lysis, and 40 receiving both. A random-effects meta-analyses of proportions of in-hospital mortality, major bleeding, technical success, and clinical success were calculated, which yielded estimate pooled percentages [95% CI] of 3.6% [0.7%, 7.9%], 0.5% [0.0%, 1.8%], 97.1% [94.8%, 98.4%], and 90.7% [85.5%, 94.3%].
Conclusion:
There is significant heterogeneity in clinical, physiologic, and angiographic data in the currently available data on MAT. RCTs with consistent parameters and outcomes measures are still needed.
Introduction
Pulmonary embolism (PE) is the third most common cause of cardiovascular death and is categorized as low-, intermediate-, and high-risk based on short-term mortality.1–3 Beyond prompt therapeutic dose anticoagulation, systemic thrombolysis is the recommended treatment for high-risk PE.4,5 Catheter-directed thrombolysis (CDT) is infusion of a thrombolytic agent directly into a pulmonary embolus. CDT carries risk of major bleeding and may not be an option in patients with contraindications to thrombolytic medications. 6
Percutaneous mechanical thrombectomy (PMT) includes techniques to treat intermediate- and high-risk PE that do not require thrombolytic drugs. There are three broad categories of PMT: (1) catheter-directed fragmentation (CDF), (2) rheolytic thrombectomy (RT), and (3) mechanical aspiration thrombectomy (MAT).2,7–9
To date, there has been no comparison of the technical results, clinical effectiveness, or safety of MAT devices. The purpose of this study is to systematically review the current literature on the use of MAT for the management of PE.
Methods
A systematic review and meta-analysis of the published literature on MAT for high-risk and intermediate-risk PE was performed. This study was exempt from formal review by our institutional review board. PRISMA guidelines were used while conducting this systematic review. 10 Article selection and data extraction were performed with the online Covidence systematic review software (Veritas Health Innovation, Melbourne, Australia). This study comprises peer-reviewed articles written in English only.
Literature search strategy
The PubMed, EMBASE, and Web of Science databases were searched until April 30, 2021. The search was performed using varying combinations of the multiple keywords, as detailed in the online Supplementary Materials.
Eligibility criteria
After removal of duplicates, each title and abstract were evaluated for inclusion by two authors (VC and MK). Inter-author disagreement was discussed and ultimately decided upon by the corresponding senior author if an agreement could not be reached. Inclusion criteria during the abstract review phase included any English language study which presented data on PMT for management of PE. The study included all patients for whom original intent was to use MAT and included patients who subsequently received CDT.
Exclusion criteria during the abstract review phase are outlined in the flow diagram (Figure 1) and online Supplementary Materials. All studies involving the performance of PMT for high- and intermediate-risk PE, with measured outcomes of effectiveness and safety, were moved into full-text review. Exclusion criteria for full-text review are detailed in the flow diagram (Figure 1) and online Supplementary Materials. If a study met the inclusion criteria above but did not report pulmonary artery pressures (PAP), the corresponding authors were contacted to provide these data. However, none of the authors was able to provide previously nonreported PAPs.
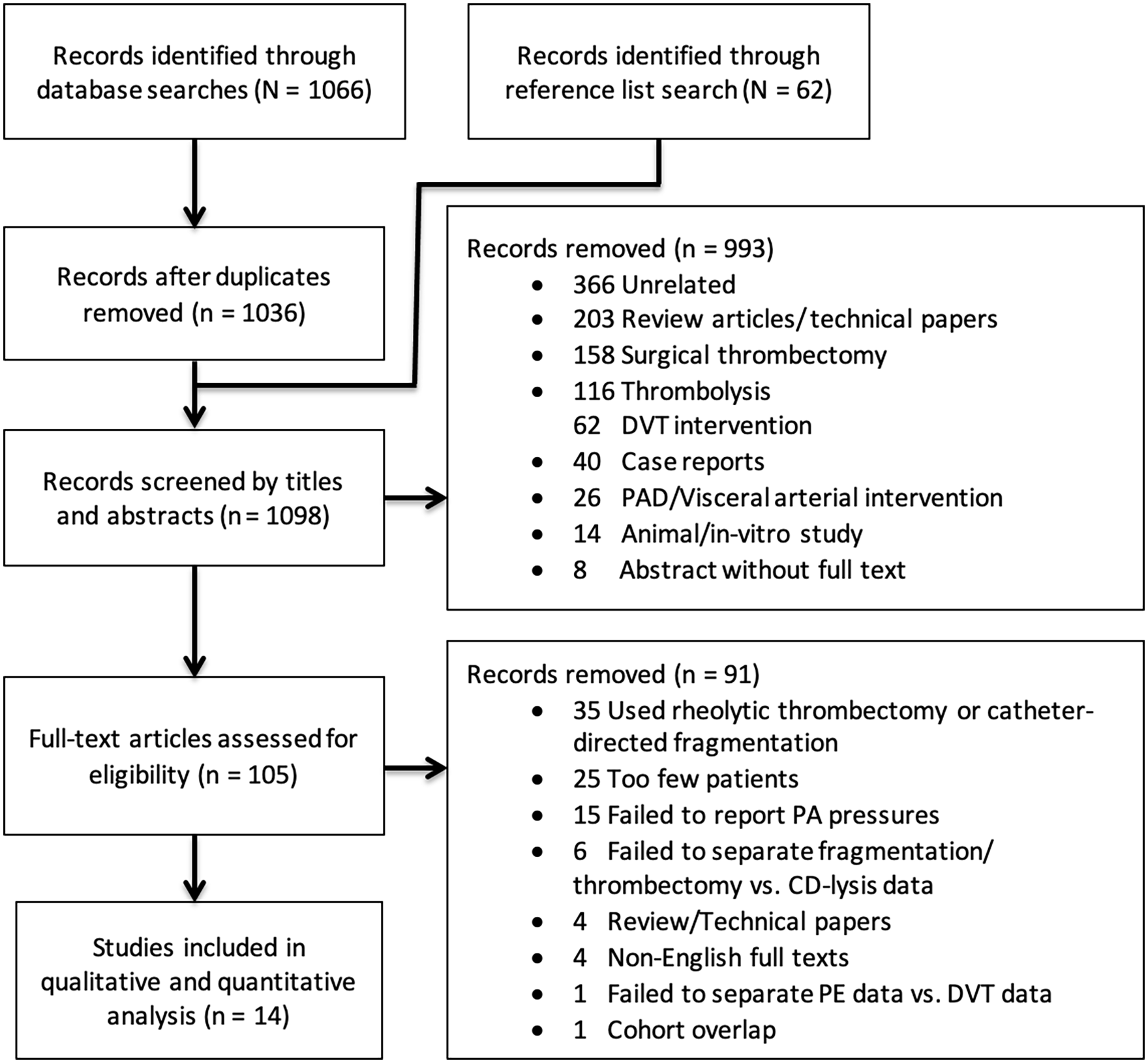
Flow diagram documenting the selection process for articles included in the systematic review and meta-analysis.
Study periods and patient selection methods were reviewed to prevent the inclusion of duplicate patients in separate publications. If there was a concern for duplicated cohorts, the study with the largest patient cohort was included. Reference lists of full-text studies were reviewed, and 62 additional abstracts were identified for abstract review.
Data extraction and risk of bias assessment
Demographic and independent variables along with outcome (dependent) measures that were recorded by two authors (VC and MK) independently are detailed in Tables 1 and 2, respectively. Risk of bias for individual studies were performed by two authors (VC and MK) independently using the Joanna Briggs Institute’s ‘Critical Appraisal Checklist for Case Series’. 11 Overall quality of evidence was assessed using the GRADE approach (Grading of Recommendations, Assessment, Development and Evaluation). 12
Demographics of patients undergoing mechanical aspiration thrombectomy.
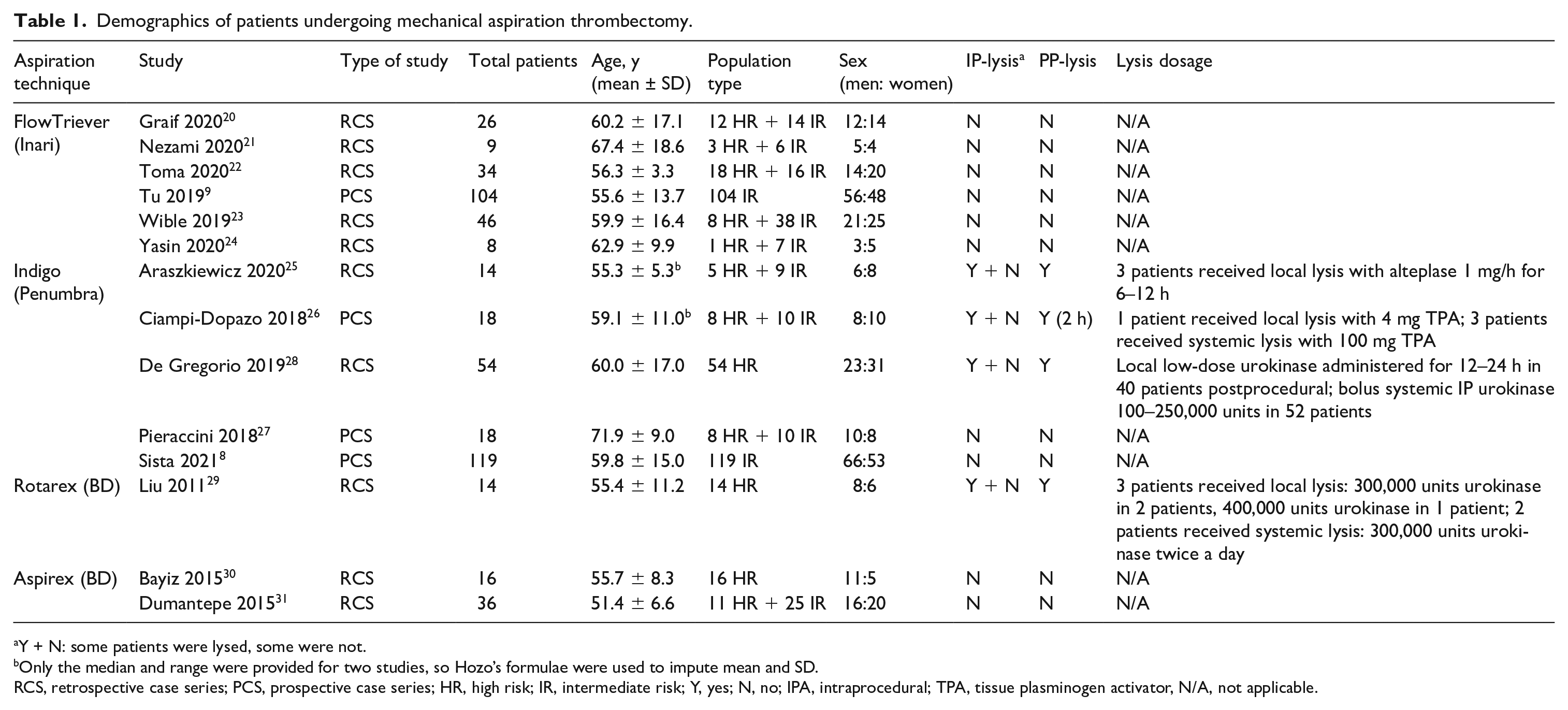
Y + N: some patients were lysed, some were not.
Only the median and range were provided for two studies, so Hozo’s formulae were used to impute mean and SD.
RCS, retrospective case series; PCS, prospective case series; HR, high risk; IR, intermediate risk; Y, yes; N, no; IPA, intraprocedural; TPA, tissue plasminogen activator, N/A, not applicable.
Procedural outcomes for patients undergoing mechanical aspiration technique.
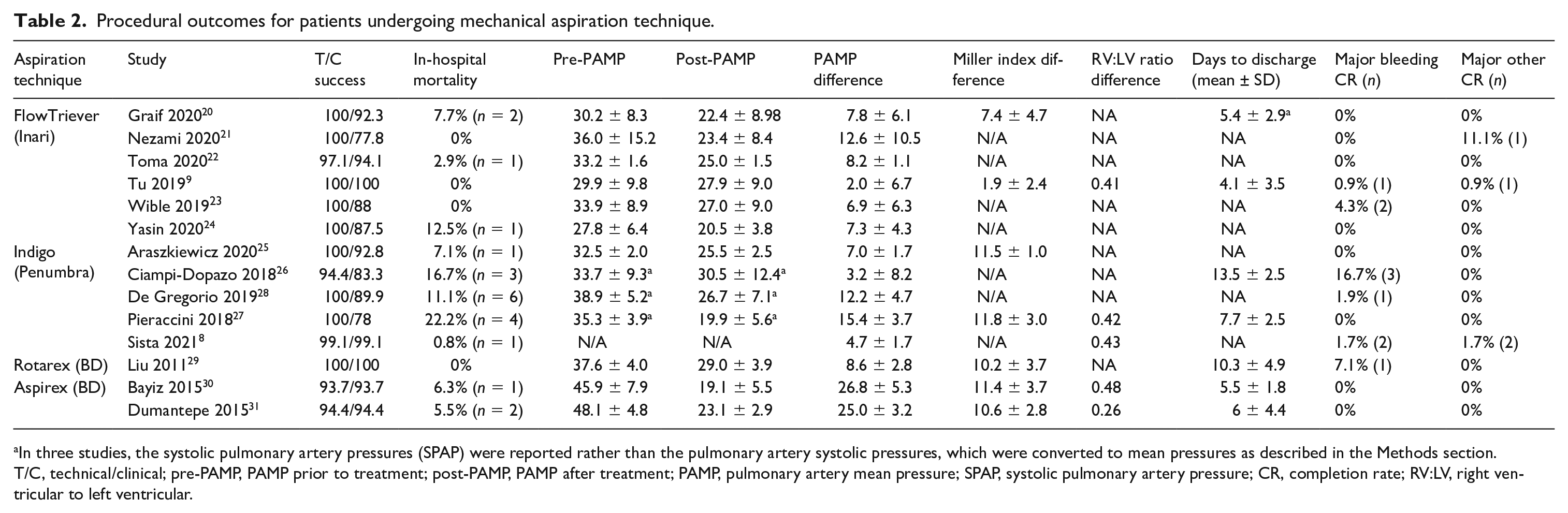
In three studies, the systolic pulmonary artery pressures (SPAP) were reported rather than the pulmonary artery systolic pressures, which were converted to mean pressures as described in the Methods section.
T/C, technical/clinical; pre-PAMP, PAMP prior to treatment; post-PAMP, PAMP after treatment; PAMP, pulmonary artery mean pressure; SPAP, systolic pulmonary artery pressure; CR, completion rate; RV:LV, right ventricular to left ventricular.
Data synthesis
Technical success was defined according to the Society of Interventional Radiology (SIR) guidelines as the device’s successful delivery to the pulmonary artery, operation of the device, and removal of the device without complication. 13 Based on SIR quality improvement guidelines for endovascular treatment of PE, cases were considered clinically successful if the therapeutic goal was achieved. 13 This was defined in this study as stabilization of hemodynamic parameters and survival to hospital discharge. This is a broad and nonspecific definition, but more inclusive and one that most studies used.
Major bleeding was defined as per the International Society on Thrombosis and Hemostasis (ISTH) bleeding scale, with major bleeding being that in a critical organ (e.g. intracerebral hemorrhage), causing hemodynamic compromise, requiring blood transfusion but not causing hemodynamic compromise, requiring a secondary intervention. 14 Nonbleeding complications were classified as major or minor according to the NIH Common Terminology Criteria for Adverse Events (CTCAE) based on the outcome, with major complications being grade 3 (severe or medically significant having permanent adverse sequelae but not immediately life-threatening), grade 4 (life-threatening consequences; urgent intervention indicated) or grade 5 (death related to adverse event). 15
Studies in which the pulmonary artery systolic pressures (PASP) were reported rather than the pulmonary artery mean pressures (PAMP), PASP were converted to PAMP using the following formula: PAMP = 0.61 * (PASP) + 2.16,17 PAMP difference was calculated as: (preintervention PAMP) – (postintervention PAMP). Standard deviation for measures of changes from baseline (PAMP difference and Miller index difference) were imputed using a correlation coefficient of 0.75 and 0.6, based on previous studies and Cochrane guidelines. 18
Weighted averages were calculated to determine overall mean age of the combined cohort. If an individual study did not report mean and SD values for any of the numeric demographic or outcome measures but reported patient-level data, mean and SD values were calculated using this patient-level data. If the median and range of a value were provided, the mean and SD was imputed using Hozo’s formulae. 19
Meta-analysis methods
Owing to the varied nature of the PE thrombectomy and population studies, overall pooled proportions of in-hospital mortality, major bleeding, clinical success, and technical success were calculated using a random-effects meta-analysis model based on individual study incidence data for each variable (see above regarding definitions for each variable). A transformation of proportions was performed for each variable. A logit transformation was used for technical and clinical success since the expected proportions for these values was expected to be between 0.1 to 0.9. A Freeman-Turkey double arcsine transformation was used for mortality, major bleeding, and major complications since the expected value was less than 0.1.
The R software (R Foundation for Statistical Computing, Vienna, Austria) was used for analysis. Subgroup analysis was performed to investigate the effect of use of intraprocedural (IP) thrombolysis (Yes vs No) on effect size. Additionally, meta-regression was performed using the following numerical independent variables: (1) percent of PEs that were high-risk within the study group, and (2) mean age of patients within each study group.
Results
Studies meeting inclusion criteria
Among 1098 unique abstracts reviewed, 993 were removed for reasons highlighted in the flow diagram (Figure 1). A total of 105 full-text articles were reviewed. There were no randomized control trials (RCTs) comparing one type of fragmentation or thrombectomy technique to another. After full-text review, 14 studies met the inclusion criteria8,9,20–31 (Figure 1). Among the 14 included studies, four were prospective case series and 10 were retrospective case series. Included studies were considered level 4 evidence, based on criteria developed by the Center for Evidence-Based Medicine. 32 The included studies, along with their patient and study demographic information, are summarized in Table 1. All 14 studies were included for the systematic review and meta-analysis.
Patient population
A total of 516 patients (299 men and 217 women) underwent mechanical aspiration thrombectomy procedures for the management of their PE. The average age of the included patients was 58.4 ± 13.6 years (weighted mean and Cohen’s pooled SD). Three studies strictly reported treatment of high-risk PE; two studies strictly reported treatment of intermediate-risk PE. The remaining nine studies treated a combination of high-risk and intermediate-risk PE (Table 1). Out of 14 total studies, six studies used the Inari FlowTriever (Inari Medical, Irvine, CA, USA); five studies used the Indigo Aspiration System (Penumbra, Alameda, CA, USA); and three studies used the Rotarex or Aspirex suction thrombectomy system (BD, Franklin Lakes, NJ, USA). Four studies reported using IP-thrombolytics in some of their patients in a patient-specific manner, with three of these four studies using the Indigo Aspiration System and the last study using Rotarex/Aspirex. The remaining 10 studies did not use IP-thrombolysis on any of their patients. All four of these studies reported the use of postprocedural (PP)-thrombolysis in a patient-specific manner as well. This equated to seven patients receiving local lysis only and 17 receiving systemic lysis only, and 40 receiving both. The details of this and dosages for IP- and PP-thrombolysis are shown in Table 1. The assessment of the risk of bias is shown in online Supplementary Figure 1.
Technical/clinical success and complications
Table 2 reports the procedural outcomes for each individual study included in the analysis. Out of 516 MATs for PE, the device was successfully delivered to the pulmonary artery, operated, and removed in 510 cases, equivalent to a pooled percentage of 97.1% [95% CI: 94.8%, 98.4%]. Clinical success was achieved in 483 of 516 cases, corresponding to a pooled percentage of clinical success of 90.8% [85.5%, 94.3%]. Subgroup analysis and meta-regression showed that a higher mean age of patients within a study was associated with lower clinical success (p = 0.0218). The type of device used, use of IP-thrombolysis, and percent of patients with high-risk PE were not associated with clinical success or technical success (Table 3).
Summary of subgroup and meta-regression analysis.
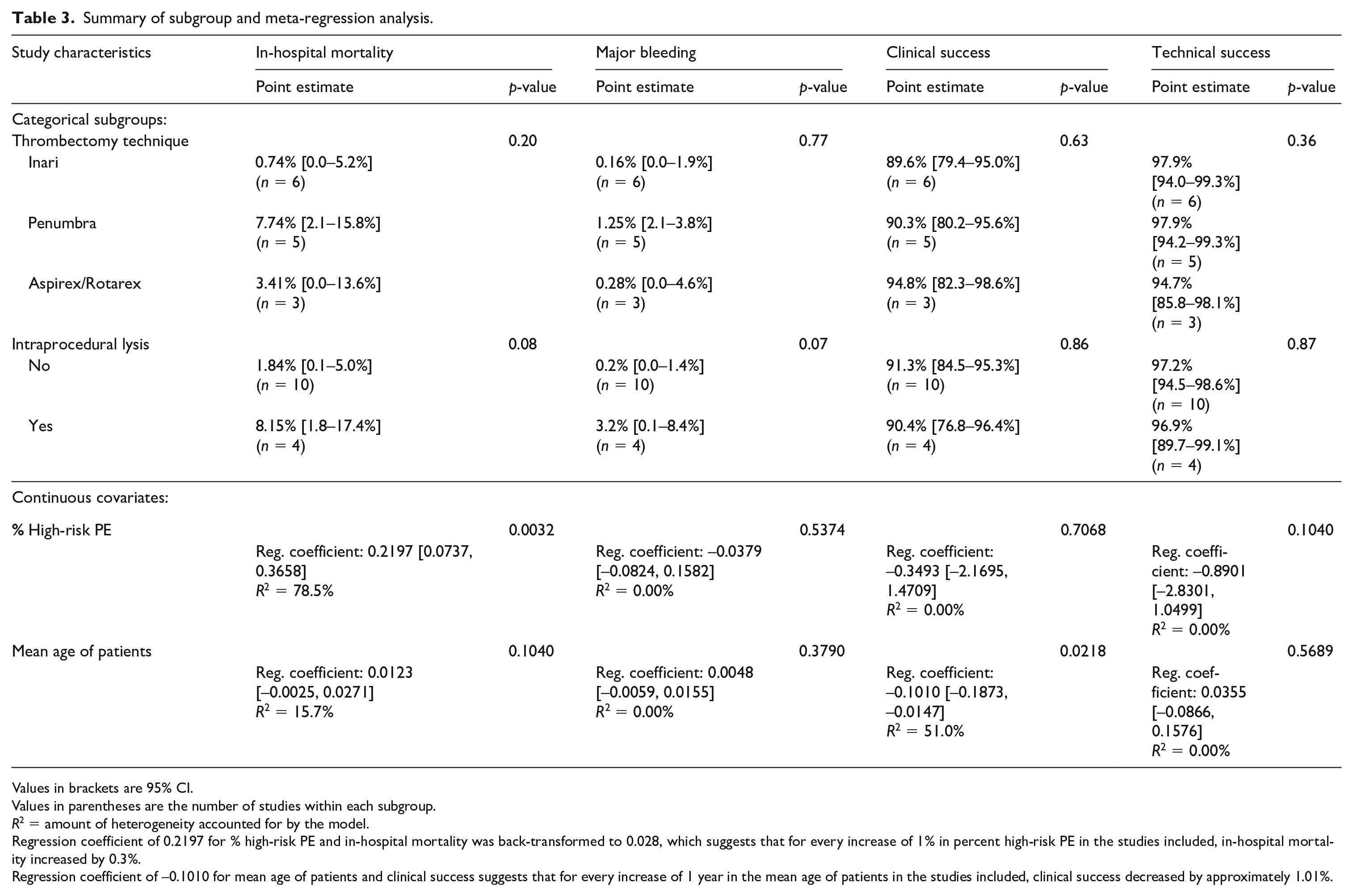
Values in brackets are 95% CI.
Values in parentheses are the number of studies within each subgroup.
R2 = amount of heterogeneity accounted for by the model.
Regression coefficient of 0.2197 for % high-risk PE and in-hospital mortality was back-transformed to 0.028, which suggests that for every increase of 1% in percent high-risk PE in the studies included, in-hospital mortality increased by 0.3%.
Regression coefficient of −0.1010 for mean age of patients and clinical success suggests that for every increase of 1 year in the mean age of patients in the studies included, clinical success decreased by approximately 1.01%.
There were four major nonbleeding complications in total, which included two incidences of cerebral ischemic infarct (one FlowTriever, one Indigo), one sustained ventricular tachycardia (Indigo), and one ventricular fibrillation requiring emergency coronary angioplasty and stenting (FlowTriever). There were 10 episodes of major bleeding, including three access site hematomas requiring intervention (two Indigo, one FlowTriever), one retroperitoneal/rectus sheath hematomas (Indigo), three intracranial hemorrhage (ICH) (three Indigo), two episodes of severe hemoptysis (one FlowTriever, one Aspirex), and one pulmonary bleed requiring lobectomy (FlowTriever).
In-hospital mortality and major bleeding
There was moderate heterogeneity across all 14 studies reporting in-hospital mortality data (p = 0.0038, I2 = 57.3%). A random-effects model yielded an estimated mortality of 3.6% [95% CI: 0.7%, 7.9%], as shown in the forest plot in Figure 2. Subgroup analysis and meta-regression showed that the percent of patients with high-risk PE within a study was associated with higher in-hospital mortality (p = 0.0032). Studies with only high-risk PE had an estimate mortality of 8.7% [3.2%, 16.1%] (n = 3 studies, 84 patients) compared to 0.1% [0%, 1.4%] (n = 2 studies, 223 patients) in studies with only intermediate-risk PE (p = 0.0005). The type of device used (p = 0.20), use of IP-thrombolysis (p = 0.08), and mean age of included patients in the study (p = 0.1040) were not associated with in-hospital mortality.
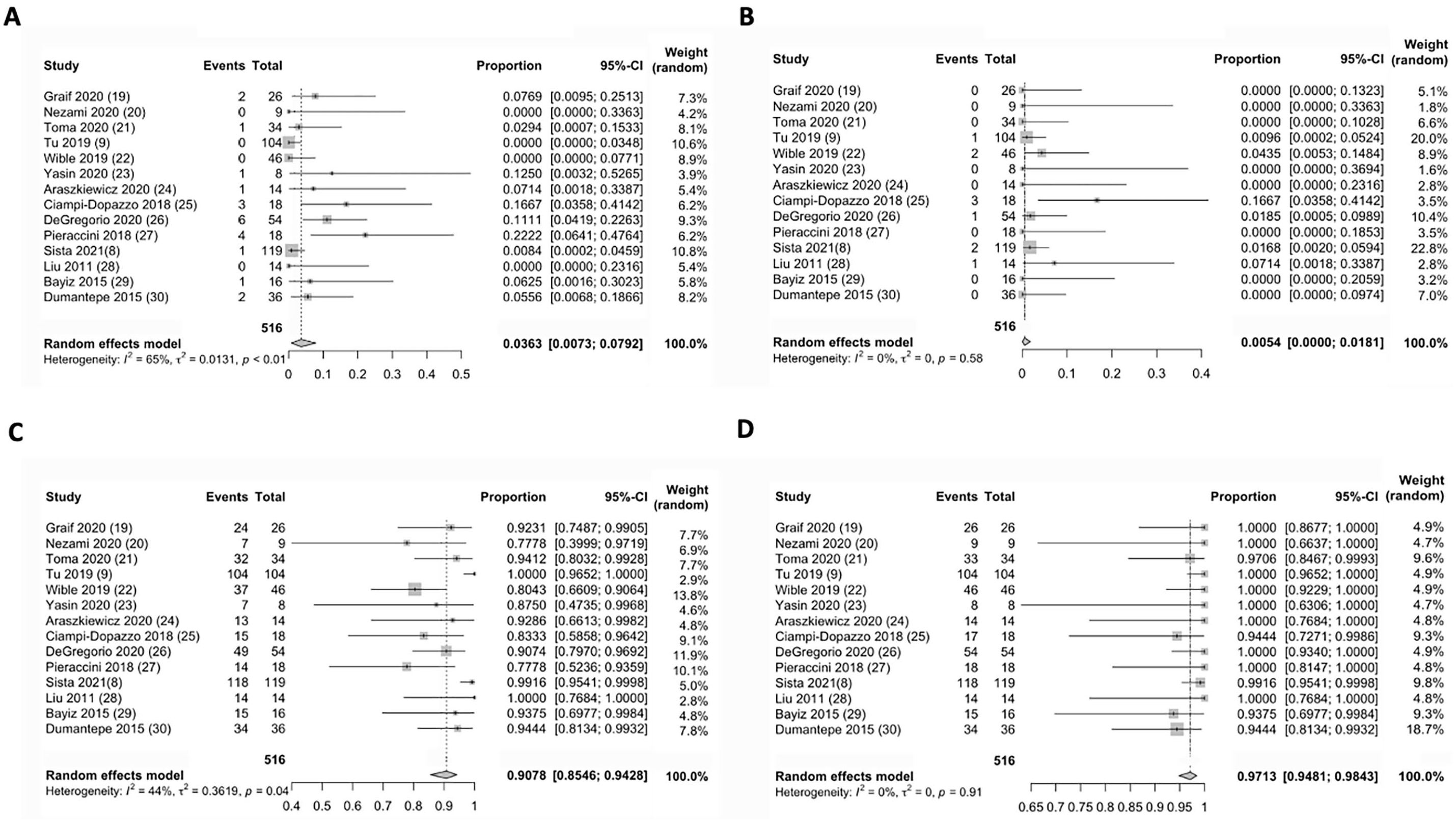
Forest plot of random effects meta-analysis of
There was much lower heterogeneity for major bleeding data across the same 14 included studies (p = 0.577, I2 = 0%). A random-effects model of major bleeding data yielded an estimate major bleeding percentage of 0.5% [95% CI: 0.0%, 1.8%] (Figure 2). Subgroup analysis and meta-regression demonstrated that none of the moderator variables were associated with major bleeding (Table 3).
Pulmonary artery mean pressures, RV/LV ratio, Miller indices, and days to discharge
Thirteen studies provided both pre- and posttreatment PAMP, representing 397 patients. Mean pre-PAMP changed from 35.1 ± 7.7 to a mean post-PAMP 25.7 ± 7.5 among these patients. The last study by Sista et al. 8 provided a mean PAMP difference of 4.7 ± 1.7 among 119 patients. Only five studies, representing 293 patients, reported both pre- and post-RV/LV ratios. A mean pre-RV/LV ratio of 1.44 improved to a post-RV/LV ratio of 1.04. Seven studies provided values for pre- and posttreatment Miller indices representing 228 patients. The Miller index improved from 20.6 ± 3.2 to 14.1 ± 3.4 following intervention in these patients. Seven studies (n = 232) provided data on average days to discharge. The median (IQR) days to discharge was 6 (5.4–10.3) among these patients.
Quality of evidence
The quality of the data collected and resulting conclusions was determined to be of low confidence for all outcomes of interest since they are derived from noncomparative studies with at least moderate risk of bias and heterogeneity in approaches to MAT.
Discussion
Thrombolytic therapy is effective in reducing PE-related mortality and composite all-cause death, with odds ratios [95% CI] of 0.29 [0.14–0.60] and 0.59 [0.36–0.96], respectively.33,34 Although effective, up to 47% of patients with acute PE have contraindications to use of thrombolytic therapy, and a Nationwide Inpatient Sample database study showed that only 30% (21,390 of 72,230) of all unstable PE patients received thrombolytic therapy.35,36 Percutaneous mechanical thrombectomy (PMT), consisting of CDF, RT, and MAT, are alternative endovascular strategies to treat acute high- and intermediate-high-risk PEs.
In our analysis of MAT, technical and clinical success proportion was 97.1% and 90.8%, respectively, across 14 studies. Kuo et al. performed a meta-analysis evaluating studies including patients with acute PE undergoing use of low-profile devices (⩽ 10 F), CDF and/or aspiration of PE including RT, and intraclot thrombolytic injection if a local drug was infused. In their analysis of 35 studies (594 patients), the pooled clinical success percentage was 86% [95% CI: 82.1%, 90.2%], comparable to the current study. 37 The slightly higher proportion found in the current study may be attributable to differences in patient population (Kuo’s study included only patients with high-risk PE), thrombectomy devices utilized, and perhaps most importantly, the definition of clinical success used in the analysis.
In this study, clinical success was defined as stabilization of hemodynamic parameters, resolution of hypoxia, and survival to hospital discharge. Although this may be an appropriate definition for high-risk PE, it may not be suitable for intermediate-risk PE. Clinical success has more recently been based on surrogate marker of improvement of RV strain by echocardiography or computed tomography angiography (CTA) 24–48 hours after the procedure. 38 Most studies (9/14, 64%) did not report this outcome measure, and thus a more general definition of clinical success as survival to hospital discharge was used. This may possibly overestimate true clinical success since using a particular time frame, specific hemodynamic criteria, or imaging criteria, rather than survival to hospital would be more restrictive.
It is imperative that future studies stay up to date on reporting standards and describe other measures of clinical success, including long-term outcome measures such as persistent RV dysfunction, functional capacity limitation, development of chronic thromboembolic pulmonary hypertension, and quality of life assessment. Interventional societies should also consider revisiting the definition and reporting standards of technical success, since current literature relies on the older definition of technical success from the 2003 SIR reporting standards. 13 Since none of the studies in this analysis included a nonintervention control group, it is difficult to ascertain if clinical improvement observed was truly related to thrombectomy intervention. Future studies with a control group are required to study clinical improvement.
In our analysis, 158 patients had high-risk PE and 358 patients had intermediate-risk PE. There was significant heterogeneity overall in our analysis and this is at least partially related to the mixed cohort of PE treated. We found that percent of high-risk PE was a significant predictor of in-hospital mortality. Although this finding may seem intuitive, the co-variable of percent high-risk PE was generated as part of this study and there is ecologic bias introduced when this is used in analysis as an independent variable. For similar reasons, mean age is also subject to ecologic bias. Future studies should focus on identifying which patients will most benefit from this intervention.
In our analysis, the pooled percentage of major bleeding was 0.5%, with three reported cases of ICH in 516 patients. This perhaps is one of the most important benefits of MAT techniques compared to thrombolytic therapies, particularly in patients with risk factors for bleeding. A meta-analysis by Chatterjee et al. demonstrated a proportion of major bleeding of 9.2% in patients undergoing predominantly systemic thrombolytic therapy.33,34 In their meta-analysis of 860 patients who underwent CDT, Bloomer et al. reported ICH in 0.35% of patients and a major complication proportion of 4.65%. 39 Overall, major complication occurred, including both major bleeding and major nonbleeding, in 2.7% of patients (14/516) in our study, underscoring the comparable safety of MAT techniques.
The improvement in mean RL/LV ratio from 1.44 to 1.04 and Miller index from 20.6 ± 3.2 to 14.1 ± 3.4 in our analysis was similar to the SEATTLE II trial where the RV/LV ratio decreased from 1.55 to 1.13 (p < 0.0001) and Miller index score decreased from 22.5 to 15.8 (p < 0.0001). 40
Importantly, our analysis is on a mixed cohort of patients where 10/14 (71.4%) studies did not use any form of thrombolysis, whereas the remaining four studies used some combination of IP- and PP-thrombolysis in a patient-specific manner. The studies did not present specific criteria for determining which patients received thrombolysis, which adds a confounding factor in analyzing the data. Although the current study did not find a significant difference in in-hospital mortality, major bleeding, or technical or clinic success between studies that employed any form of thrombolysis and studies which did not use thrombolytics, it would be incorrect to draw any conclusions from this as our analysis was not performed at a patient level. There may well be differences among the two cohorts that are not accounted for in our study-level analysis. Future studies should specifically study the benefits of IP- and PP-thrombolysis at a patient level, whether a RCT or a case–control study.
Study limitations
There are significant limitations in this study, some of which are inherent to the systematic review process. There was heterogeneity in clinical, physiologic, and angiographic data collected and reported. For example, many studies did not report PAPs, RV/LV ratio, or length of stay. None of the studies are RCTs and, rather, consist of retrospective or single-arm prospective studies. There are limitations in definition of clinical success as described above. Small study effects were not evaluated in this study. A significant number of these studies are funded through industry. All these factors can increase risk of confounding, or selection, or publication bias. Additionally, long-term clinical follow up and patient level data were not available.
Conclusion
This systematic review and meta-analysis evaluation of mechanical aspiration thrombectomy (alone or with concomitant CDT) calculated point estimate proportions of in-hospital mortality, major-bleeding, and clinical and technical success, which are comparable to existing studies for other thrombectomy, fragmentation, and CDT techniques. This review highlights the significant heterogeneity in the current data, which limits any conclusions that can be drawn from the literature. High-quality RCTs with consistent parameters and outcomes measures are paramount in determining the appropriate treatment for patients with PE.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221124681 – Supplemental material for Mechanical aspiration thrombectomy for the treatment of pulmonary embolism: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221124681 for Mechanical aspiration thrombectomy for the treatment of pulmonary embolism: A systematic review and meta-analysis by Vishnu M Chandra, Minhaj S Khaja, Marc C Kryger, Akhilesh K Sista, Luke R Wilkins, John F Angle and Aditya M Sharma in Vascular Medicine
Footnotes
Declaration of conflicting interests
Minhaj S Khaja has received speaking honoraria from Penumbra, Inc.; Boston Scientific, Inc., and Medtronic, Inc. Additionally, Dr Khaja has institutional grant funding from the Society of Interventional Radiology Foundation and Boston Scientific, Inc. Akhilesh K Sista has a research grant administered through New York University (NYU) from Penumbra, Inc., research support administered through NYU from Thrombolex, Inc. and Vascular Medcure and is a U34 award recipient (National Heart, Lung, and Blood Institute). Aditya M Sharma has received speaking honoraria from Boston Scientific, Inc. and has institutional grant funding from Vascular Medcure. The remaining authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
