Abstract
Catheter-directed thrombolysis (CDT) and systemic thrombolysis (ST) are used to treat intermediate/high-risk pulmonary embolism (PE) in the absence of comparative safety and effectiveness data. We utilized a large administrative database to perform a comparative safety and effectiveness analysis of catheter-directed versus systemic thrombolysis. From the Optum® Clinformatics® Data Mart private-payer insurance claims database, we identified 100,744 patients hospitalized with PE between 2004 and 2014. We extracted demographic characteristics, high-risk PE features, components of the Elixhauser Comorbidity Index, and outcomes including intracranial hemorrhage (ICH), all-cause bleeding, and mortality among all patients receiving CDT and ST. We used propensity score methods to compare outcomes between matched cohorts adjusted for observed confounders. A total of 1915 patients (1.9%) received either CDT (n = 632) or ST (n = 1283). Patients in the CDT group had fewer high-risk features including less shock (5.4 vs 11.1%; p < 0.001) and cardiac arrest (6.8 vs 11.0%; p = 0.004). In 1:1 propensity-matched groups, ICH rates were 1.9% in both the CDT and ST groups (p = 1.0). All-cause bleeding was higher in the CDT group (15.9 vs 8.7%; p < 0.001), while in-hospital mortality was lower (6.5 vs 10.0%; p = 0.02). Among a nationally representative cohort of patients with PE at higher risk for mortality, CDT was associated with similar ICH rates, increased all-cause bleeding, and lower short and intermediate-term mortality when compared with ST. The competing risks and benefits of CDT in real-world practice suggest the need for large-scale randomized clinical trials with appropriate comparator arms.
Introduction
Pulmonary embolism (PE) accounts for nearly 100,000 deaths in the US each year and is the third leading cause of cardiovascular death after myocardial infarction and stroke.1–3 Approximately 25% of patients presenting with sudden death are found to have had a PE on autopsy. 4 Catheter-directed thrombolysis (CDT) and systemic thrombolysis (ST) have been used to treat intermediate and high-risk PE, although the evidence supporting the safety and effectiveness of CDT is sparse. The use of both CDT and ST has been increasing nationwide, but both therapies remain underutilized even in higher-risk cohorts.5,6 The underutilization is at least in part due to lack of data, and also related to the lack of widespread availability of these treatments, which may also stem from a poor database.
Only a few small studies of CDT safety/effectiveness appear in the literature, and these studies were not powered to evaluate clinical outcomes including intracranial hemorrhage, major bleeding, and mortality.7–10 Previous observational studies utilizing publicly available datasets attempting to examine the comparative safety/effectiveness of CDT have been limited by lack of detailed procedural coding data for CDT.11,12 As such, there is a pressing need for evidence in regards to the safety profile of CDT prior to its widespread use for PE. 13 In this study, we aim to describe the comparative safety and clinical effectiveness of CDT and ST in a real-world population of patients with acute PE identified from a large administrative claims database.
Methods
Data source
We conducted a retrospective analysis of administrative claims data from the Optum® Clinformatics® Data Mart national commercial insurance claims database. This database contains privately insured and Medicare Advantage patients across the United States, but with heavy representation from the South Atlantic and Midwestern regions. Medical and hospitalization claims used include International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnostic codes, ICD-9-CM procedural codes, Current Procedural Terminology (CPT®) codes, and diagnosis-related groups (DRGs). The study protocol was deemed exempt by the University of Pennsylvania Institutional Review Board and the need for informed consent was waived as all data were de-identified.
Study population
We identified all patients hospitalized with a PE between January 1, 2004 and December 31, 2014 by searching the database for hospitalization claims with a PE ICD-9-CM code (415.1X, 634.6X, 635.6X, 636.6X, 637.6X, 638.6, 673.2X).14,15 To identify patients who received CDT, we linked hospital claims to provider claims and then searched this cohort for patients who had a noncoronary transcatheter thrombolytic infusion using CPT codes (37201, 37211). We identified the cohort of patients receiving ST as patients without a CDT code, but with an ICD-9-CM procedural code for infusion of a thrombolytic agent (99.10). Any patient in the CDT or ST cohort who had a primary ICD-9-CM code for a nonhemorrhagic stroke (433.X, 434.X) was excluded. Any patient with deep vein thrombosis (DVT) as a primary diagnosis (453.4X) who was in the CDT group was also excluded (Figure 1). Subjects with a myocardial infarction (MI) ICD-9-CM code were not excluded as PE may be associated with cardiac biomarker release that is coded as MI; however, the vast majority of patients in the final analysis did not have an MI coded during the index hospitalization (7.2%). Patients with multiple PE admissions with ST or CDT were included more than once if the PE admissions were at least 2 years apart. If multiple PE admissions were within 2 years, only the initial PE admission was considered in this study due to uncertainty that these patients’ subsequent PE admissions were unrelated to the index hospitalization.
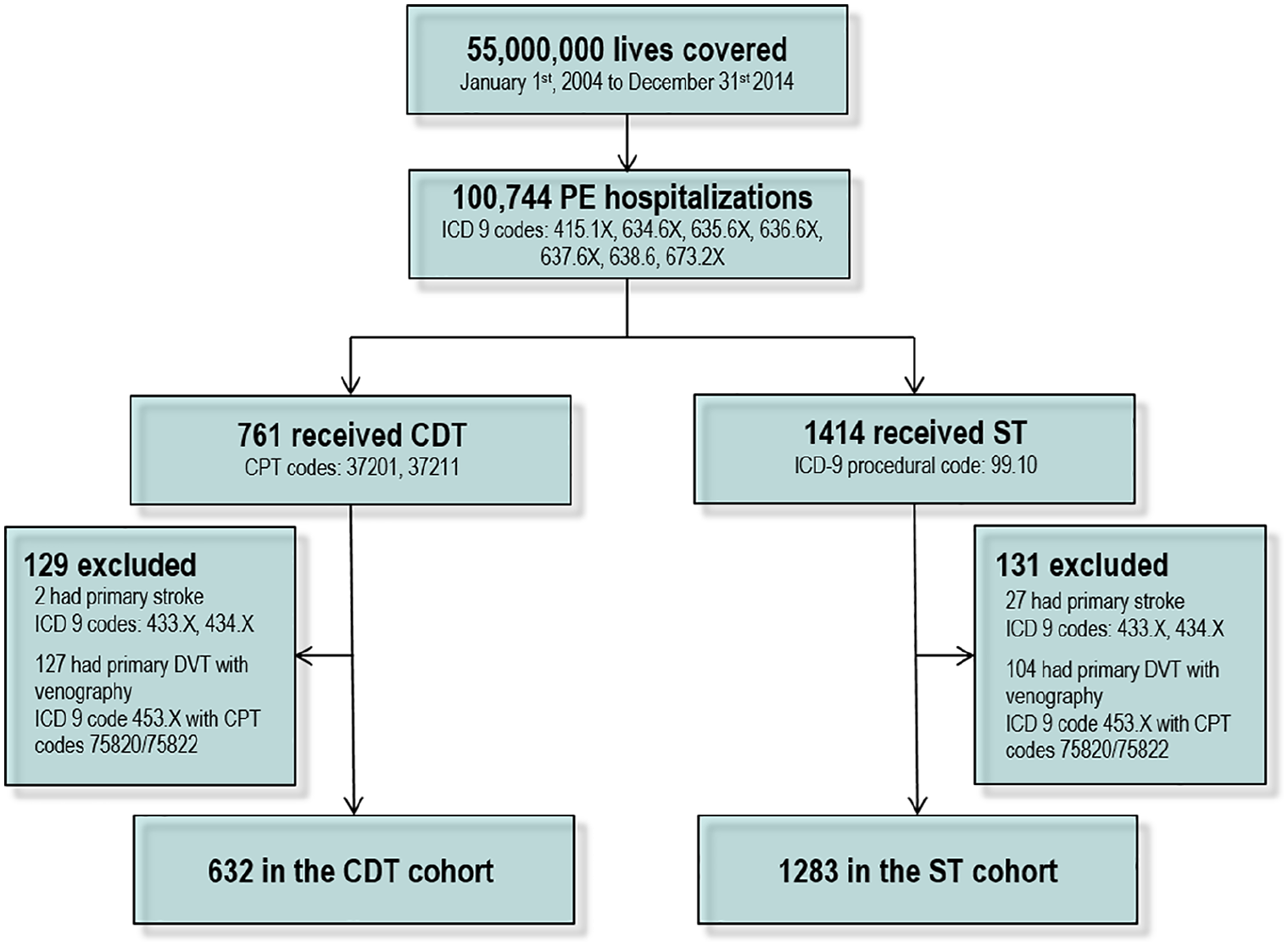
Cohort generation methodology.
We obtained each patient’s demographic characteristics, components of the Elixhauser Comorbidity Index, prior surgical procedures, and presence/absence of high-risk PE features. Demographic variables included age, sex, and geographic region. Comorbidities were assessed by searching all claims over the prior 12 months for components of the Elixhauser Comorbidity Index based on ICD-9-CM coding, 16 and a composite comorbidity score was then calculated. 17 Prior surgical procedures were obtained by searching for surgical DRGs within 3 months prior to the index hospitalization (DRGs related to PE were excluded). Saddle PE location was assessed by searching the index hospitalization for ICD-9-CM codes for saddle PE (415.13). Higher-risk PE features were ascertained by searching the index hospitalization for ICD-9-CM codes including shock (785.5), cardiac arrest (427.5), ventilator dependence (V46.1), and vasopressor use (00.17). Critical care management of this PE cohort was assessed using CPT codes for critical care services (99291).
Outcomes
The primary outcome measures were intracranial hemorrhage (430.X–432.X) 18 during the index hospitalization, in-hospital bleeding, and in-hospital mortality. In-hospital bleeding was defined by ICD-9-CM codes for major bleeding events using previously published definitions. 19 In-hospital mortality was assessed by using a discharge status of deceased. Secondary endpoints included 30-day mortality and 1-year mortality. These were assessed by using the Social Security Death Index Death Master File as well as hospital discharge status. The discharge disposition of the patient was also assessed by using the discharge status field.
Analysis
In the primary unadjusted analyses, categorical variables were compared using chi-squared testing and continuous variables were compared using t-tests or analysis of variance (ANOVA). Because patients were not randomly assigned to treatment groups, we used propensity score matching to adjust for known covariates. Using logistic regression, we calculated a propensity score based on age, sex, saddle PE, cardiac arrest, shock, extracorporeal membrane oxygenation (ECMO), prior surgery, stroke, and each of the Elixhauser comorbidities. We then performed 1:1 greedy nearest neighbor matching with a 0.25 SD caliper size using the propensity scores. Matches were made without replacement. All analyses were conducted using Stata version 14.1 (StataCorp, College Station, TX, USA) and SAS version 9.3 (SAS Institute, Cary, NC, USA).
Results
Population and baseline characteristics
Between January 1, 2004 and December 31, 2014, there were 100,744 PE admissions identified in 86,050 unique patients with PE. Of these admissions, 25% involved critical care billing. A total of 1915 patients (1.9%) received either CDT (n = 632) or ST (n = 1283) (Figure 1). The mean age was 57.2 ± 16.2 years in the CDT group and 57.4 ± 16.3 years in the ST group. In the CDT group, 54.4% were male, while in the ST group, 50.9% were male. A history of PE and a history of DVT were more prevalent in those receiving CDT than in the ST group. The Elixhauser comorbidities were similar between the two groups (Table 1).
Comparison of clinical characteristics in patients receiving CDT versus ST.
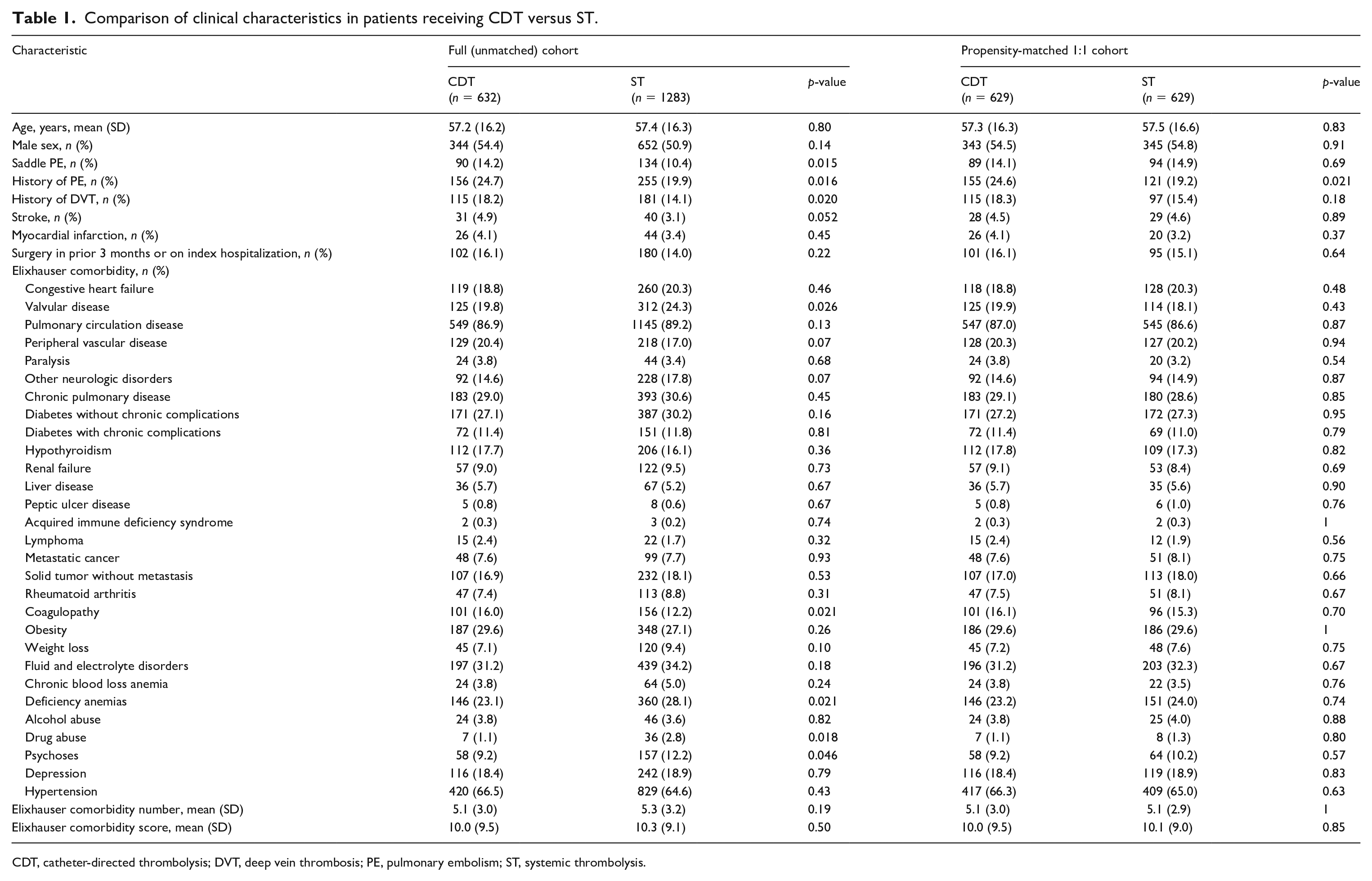
CDT, catheter-directed thrombolysis; DVT, deep vein thrombosis; PE, pulmonary embolism; ST, systemic thrombolysis.
Unadjusted analysis
Patients in the CDT group had significantly fewer higher-risk features than the patients in the ST group. Patients in the CDT group had lower rates of shock (5.4% vs 11.1%; p < 0.001) and cardiac arrest (6.8% vs 11.0%; p = 0.004) than those treated with ST. Extracorporeal membrane oxygenation use was uncommon in both groups (0.5% vs 0.5%; p = 0.98) (Table 2). Other high-risk PE markers, such as ventilator dependence and vasopressor use, were not commonly coded. There was a nonsignificantly higher number of intracranial hemorrhage (ICH) in the CDT group than the ST group (2.4% vs 1.4%; p = 0.12). Bleeding was significantly more common in the CDT group (15.8% vs 10.7%; p < 0.001). Mortality was lower in the CDT group during the index hospitalization (6.6% vs 12.5%; p < 0.001), at 30 days (8.1% vs 14.7%; p < 0.001), and at 1 year (11.7% vs 18.6%; p < 0.001) (Table 3).
Higher-risk PE features in patients receiving CDT versus ST.
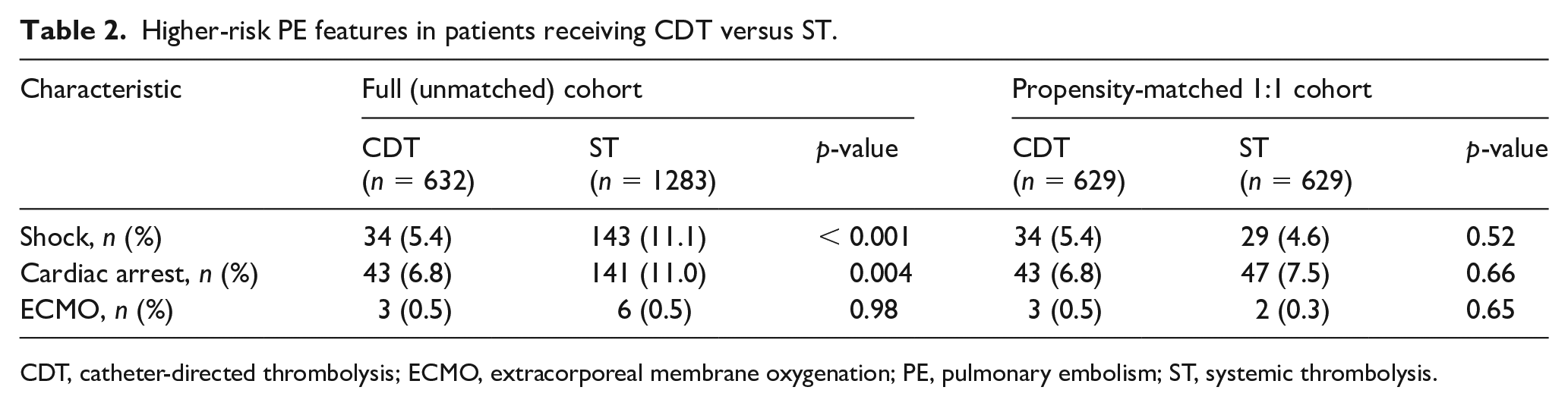
CDT, catheter-directed thrombolysis; ECMO, extracorporeal membrane oxygenation; PE, pulmonary embolism; ST, systemic thrombolysis.
Outcomes in patients receiving CDT versus ST.
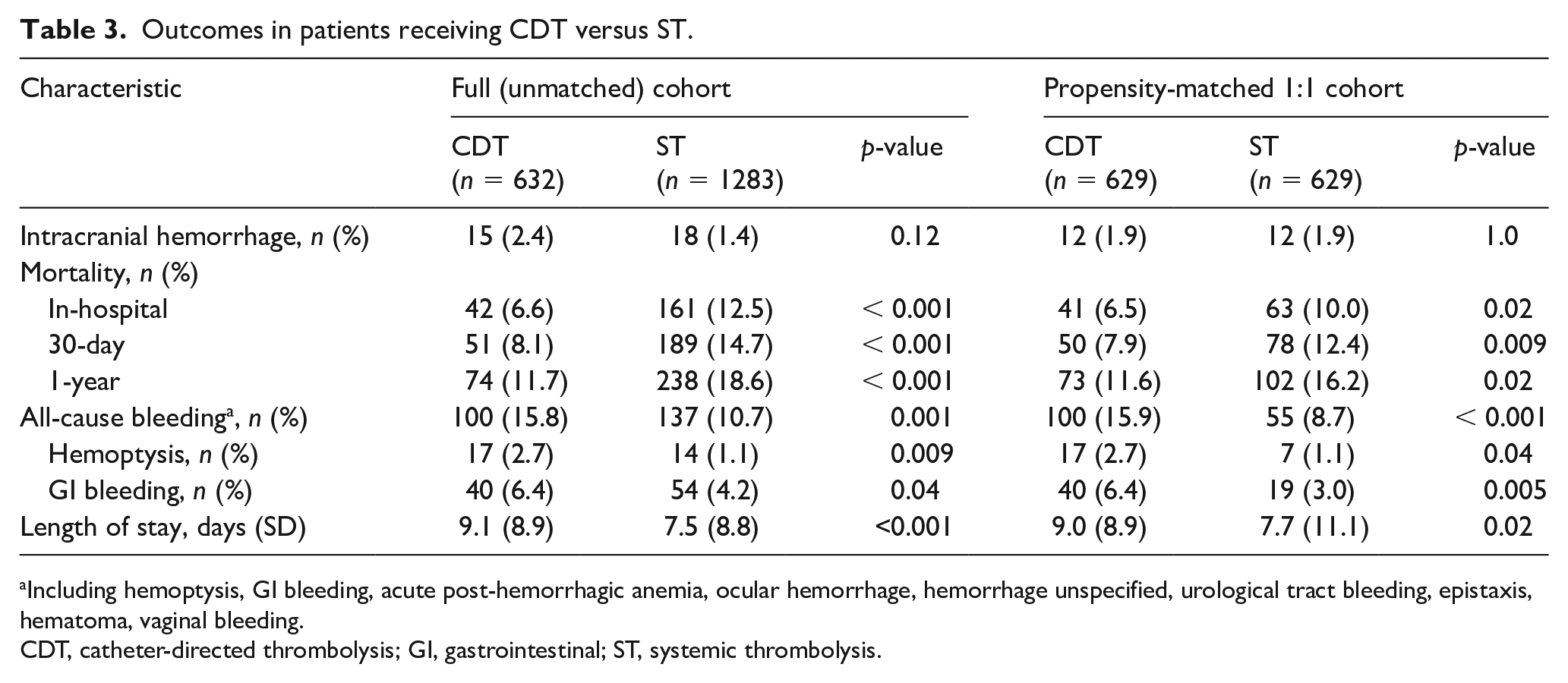
Including hemoptysis, GI bleeding, acute post-hemorrhagic anemia, ocular hemorrhage, hemorrhage unspecified, urological tract bleeding, epistaxis, hematoma, vaginal bleeding.
CDT, catheter-directed thrombolysis; GI, gastrointestinal; ST, systemic thrombolysis.
Propensity-matched analysis
Propensity score 1:1 matching yielded 629 patients who received CDT matched to 629 patients who received ST. The propensity score-matched cohorts appeared well balanced for measured covariates (Table 1). The rate of intracranial hemorrhage was 1.9% in both groups (p = 1.0). Bleeding was significantly more common in the CDT group than in the ST group (15.9% vs 8.7%; p < 0.001). Mortality was lower in the CDT group during the index hospitalization (6.5% vs 10.0%; p = 0.02), at 30 days (7.9% vs 12.4%; p = 0.009), and at 1 year (11.6% vs 16.2%; p = 0.02) (Table 3).
The length of stay was significantly longer in the CDT group (9.0 ± 8.9 vs 7.7 ± 11.1; p = 0.02). The discharge status of patients in the two arms were similar (p = 0.25): discharged home without services (CDT vs ST: 64.9% vs 68.7%), home with services (5.7% vs 5.9%), transferred to another hospital (4.8% vs 5.1%), nursing facility/rehab (10.3% vs 6.0%), and hospice (1.3% vs 1.4%).
Discussion
Out of the 1915 patients who received CDT or ST for acute PE, we found, in propensity score-matched analyses, that 1.9% of patients in each group experienced ICH. There was significantly more all-cause bleeding in patients who received CDT as compared to those who received ST, but CDT was associated with reduced short and intermediate-term mortality. This study expands the current understanding of CDT use for PE and its implications in real-world practice. While prior observational studies have attempted to compare CDT to ST, these studies have not had access to CPT codes and have utilized invasive pulmonary angiography as a surrogate for CDT, which may not accurately capture actual CDT use.11,12 CDT can be performed without invasive pulmonary angiography and the performance of invasive pulmonary angiography does not guarantee use of CDT. This study represents by far the largest comparative safety and effectiveness analysis of CDT and ST in patients with PE using more accurate CPT coding rather than ICD-9-CM codes alone.
Despite an elevated mortality in this population, the use of both CDT and ST was relatively uncommon. Of the 100,744 PE admissions, over 25,000 hospitalizations involved critical care billing, but fewer than 2000 of these cases were treated with CDT or ST. This finding is concordant with a prior study reporting only a minority of patients with intermediate or high-risk PEs received thrombolytic therapy. 20
Although over 1700 patients have been randomized in various trials of ST for acute PE, 21 only one small randomized clinical trial has been conducted comparing CDT to an alternative therapy. 8 This study randomized 59 patients with intermediate-risk PE to CDT versus anticoagulation alone, and no ICH were reported. There have been five additional studies prospectively examining CDT for PE, four single arm studies and one randomized dose-finding trial. In these six studies, there were a total of five ICH out of 556 enrolled patients. Overall, the 1.9% adjusted ICH rate identified in the current real-world analysis is higher than the 0.7% adjusted rate identified in six studies of carefully selected patients who were enrolled in prospective clinical trials.7,9,22–25
The ICH rate of 1.9% reported in the ST population here is similar to that reported in several prior observational and randomized studies of ST.21,26–29 The similar incidence of ICH between ST and CDT, coupled with a higher rate of all-cause bleeding in patients receiving CDT in our study, suggest that CDT may not necessarily be a safer modality of thrombolysis compared to ST. The safety of a lower total dose of thrombolytics in CDT compared to ST may be offset by a longer infusion time as well as the invasive nature of the former procedure. Thus, further research is required on the doses and duration of both CDT and ST.
In both unadjusted and propensity score-matched analyses, in-hospital, 30-day, and 1-year mortality rates were higher in those patients receiving ST. There are theoretical benefits to CDT that could contribute to this – most notably the administration of thrombolytic directly into the pulmonary circulation leading to greater efficacy in clot resolution. 30 Additionally, CDT may be more commonly used in higher volume, more experienced centers, which may have more favorable outcomes, and skew the mortality data in favor of CDT.
There has been little study characterizing granular clinical differences between patients with acute PE selected for ST versus CDT. The current study attempted to account for multiple administrative codes denoting acute illness severity including ventilator dependence, shock, vasopressor use, and ICU stay to compare balanced groups in our propensity-matched analyses. However, a true comparison of efficacy can only be obtained through well-powered randomized data. Our study does imply that during the period of this analysis, aggressive therapies were largely reserved for patients at the higher end of the PE risk continuum.
Limitations
Our study has a number of limitations. First, it was retrospective in nature and baseline characteristics, procedural data, and outcomes were all ascertained from administrative rather than clinical data. However, as we noted above, we were able to utilize more detailed CPT and ICD-9-CM procedural codes as compared to prior studies on this topic. This coding does not allow the ascertainment of dosing of thrombolytics or infusion times. Next, owing to the limitations of administrative datasets, characterizing each patient as high or intermediate risk by consensus guideline criteria was not possible.31,32 However, our data likely reflect a higher-risk population because the 30-day mortality in this study (12.5%) is consistent with prior population studies of patients with PE between the upper end of the intermediate-risk spectrum and the high-risk spectrum.33,34
Regarding the secondary outcome of all-cause bleeding, it is possible that some of the bleeding events in the CDT arm were an indication for CDT, rather than an adverse outcome of the procedure. Importantly, although we propensity score-matched our CDT and ST groups, there still exists the possibility of residual confounding due to unobserved factors not accounted for in our propensity score. This may be an issue given the dramatic baseline differences in measured factors associated with higher-risk PE status among those patients treated with ST. Additional unmeasured confounders may have also increased the severity of illness in the ST group. We also could not account for the likelihood that the sickest patients, when treated urgently, are more likely to receive ST than CDT due to the former often being the only urgently available therapy. All these factors would have biased the results towards increased mortality and increased ICH with ST. Additionally, center expertise may be a factor in choosing ST and CDT, which is not accounted for in the present study. Finally, the study covered a 10-year period ending in 2014 with techniques used for catheter-directed lysis and doses of thrombolytics experiencing an ongoing evolution since that time.
Conclusion
Among a nationally representative cohort of patients with PE, CDT was associated with similar ICH rates, increased all-cause bleeding, and lower short and intermediate-term mortality when compared with ST. This analysis examined patients with PE at a higher risk of mortality and the results should not be extrapolated to patients with a lower risk of mortality. In patients with PE at higher risk for mortality, the competing risks and benefits of CDT in real-world practice suggest the need for large-scale randomized clinical trials with appropriate comparator arms.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Bram J Geller – none to report. Srinath Adusumalli –none to report. Steven C Pugliese – none to report. Sameed Ahmed M Khatana – none to report. Ashwin Nathan – none to report. Ido Weinberg – scientific advisory board at Novate Medical. Michael R Jaff – employee at Boston Scientific Corporation, consultant at Glide Healthcare, equity shareholder at Embolitech. Taisei Kobayashi – none to report. Jeremy A Mazurek – none to report. Sameer Khandhar – none to report. Lin Yang – none to report. Peter W Groeneveld – none to report. Jay S Giri – board of directors at the PERT Consortium, a 501c3 not-for-profit organization; advisory boards for AstraZeneca and Philips Medical; research funds to the Institution from St Jude Medical and Recor Medical.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was conducted while Srinath Adusumalli was supported by the Ruth L. Kirschstein National Research Service Award (5T32HL007843) from the National Heart, Lung, and Blood Institute.
