Abstract
Background:
Patients with peripheral artery disease (PAD) are at increased risk for major adverse limb and cardiac events including mortality. Developing screening tools capable of accurate PAD identification is a necessary first step for strategies of adverse outcome prevention. This study aimed to determine whether machine analysis of a resting Doppler waveform using deep neural networks can accurately identify patients with PAD.
Methods:
Consecutive patients (4/8/2015 – 12/31/2020) undergoing rest and postexercise ankle–brachial index (ABI) testing were included. Patients were randomly allocated to training, validation, and testing subsets (70%/15%/15%). Deep neural networks were trained on resting posterior tibial arterial Doppler waveforms to predict normal (> 0.9) or PAD (⩽ 0.9) using rest and postexercise ABI. A separate dataset of 151 patients who underwent testing during a period after the model had been created and validated (1/1/2021 – 3/31/2021) was used for secondary validation. Area under the receiver operating characteristic curves (AUC) were constructed to evaluate test performance.
Results:
Among 11,748 total patients, 3432 patients met study criteria: 1941 with PAD (mean age 69 ± 12 years) and 1491 without PAD (64 ± 14 years). The predictive model with highest performance identified PAD with an AUC 0.94 (CI = 0.92–0.96), sensitivity 0.83, specificity 0.88, accuracy 0.85, and positive predictive value (PPV) 0.90. Results were similar for the validation dataset: AUC 0.94 (CI = 0.91–0.98), sensitivity 0.91, specificity 0.85, accuracy 0.89, and PPV 0.89 (postexercise ABI comparison).
Conclusion:
An artificial intelligence-enabled analysis of a resting Doppler arterial waveform permits identification of PAD at a clinically relevant performance level.
Keywords
Introduction
It is estimated that 8.5 million Americans over age 40 years have peripheral artery disease (PAD), and in 25% the disease is severe. 1 Patients with PAD have an increased incidence of major adverse cardiac events (MACE), cardiovascular mortality, and all-cause mortality when compared to patients without PAD.2,3 Indeed, PAD is an independent predictor for MACE even after controlling for other traditional atherosclerotic risk variables. 4 Only 10% of patients with PAD have the classic symptoms of intermittent claudication to alert clinicians to the presence of this disease; approximately 40% do not complain of leg pain, and the remaining 50% have a variety of leg symptoms different from classic claudication.5–7 Diminished or absent pulses on physical examination lacks both sensitivity and specificity and may not be sufficient for the diagnosis of PAD. 8 Identifying patients with PAD is therefore challenging. As such, patients with PAD are often under-diagnosed and under-treated with proven guideline-endorsed therapies. 9
Noninvasive screening for PAD involves a battery of well-validated physiological measurements that include the ankle–brachial index (ABI) and arterial Doppler assessment.1,10–12 Disease severity is currently based on the ABI, which is derived by dividing the systolic pedal pressure (the higher value of the dorsalis pedis and posterior tibialis) by the brachial artery pressure (highest of the right and left brachial blood pressure). There are circumstances where the resting ABI measure lacks accuracy, including the elderly, diabetes mellitus, chronic kidney disease, arterial calcification, and isolated iliac artery disease. 1 Treadmill exercise testing with postexercise ABI assessment improves PAD detection, particularly when disease is limited to the iliac arteries. Many patients with PAD have comorbidities limiting their ability to exercise adequately, including debilitating arthritis, severe cardiopulmonary disease, gait instability, and prior limb amputation. Alternatives to exercise ABI testing for PAD evaluation would therefore be useful.
Doppler waveforms can be obtained at multiple levels of the lower extremity (common femoral, superficial femoral, popliteal, posterior tibial, and dorsalis pedis arteries) and are used (in combination with other tests) to diagnose the presence of arterial occlusive disease and approximate the location of disease within the arterial system.13,14 Standard interpretation of waveforms is based on the assessment of wave amplitude, upslope velocity, and waveform features including the presence of a dicrotic notch. However, this interpretation is subjective and requires a depth of expertise which is not widely available.
Ideally, screening tools for PAD should be affordable, accurate, reproducible, noninvasive, and technically easily to perform. Deep learning is an advanced artificial intelligence (AI) method based on artificial neural networks which produce nonlinear representation of underlying data. Herein, we sought to determine whether AI using deep neural networks can accurately identify patients with PAD based solely on Doppler waveform analysis.
Methods
Study design and patients
Consecutive patients over 18 years of age undergoing lower-extremity treadmill exercise arterial testing with continuous wave (CW) Doppler and ABI assessment at the Mayo Clinic Gonda Vascular Laboratory between April 8, 2015 and December 31, 2020 were analyzed. Subjects were excluded from the analysis if they did not undergo treadmill exercise testing for any of the following reasons: (1) non/poorly compressible vessels; (2) severe hypertension at rest (> 180/110) precluding safe exercise testing; (3) gait instability; (4) leg ulcers; or (5) lower limb amputation. Subjects were also excluded who lacked Minnesota research authorization (Figure 1). Following analysis of the primary dataset, a second patient dataset was assembled (1/1/2021 – 3/31/2021) for additional model performance validation. The study was approved by the Mayo Clinic Institutional Review Board.
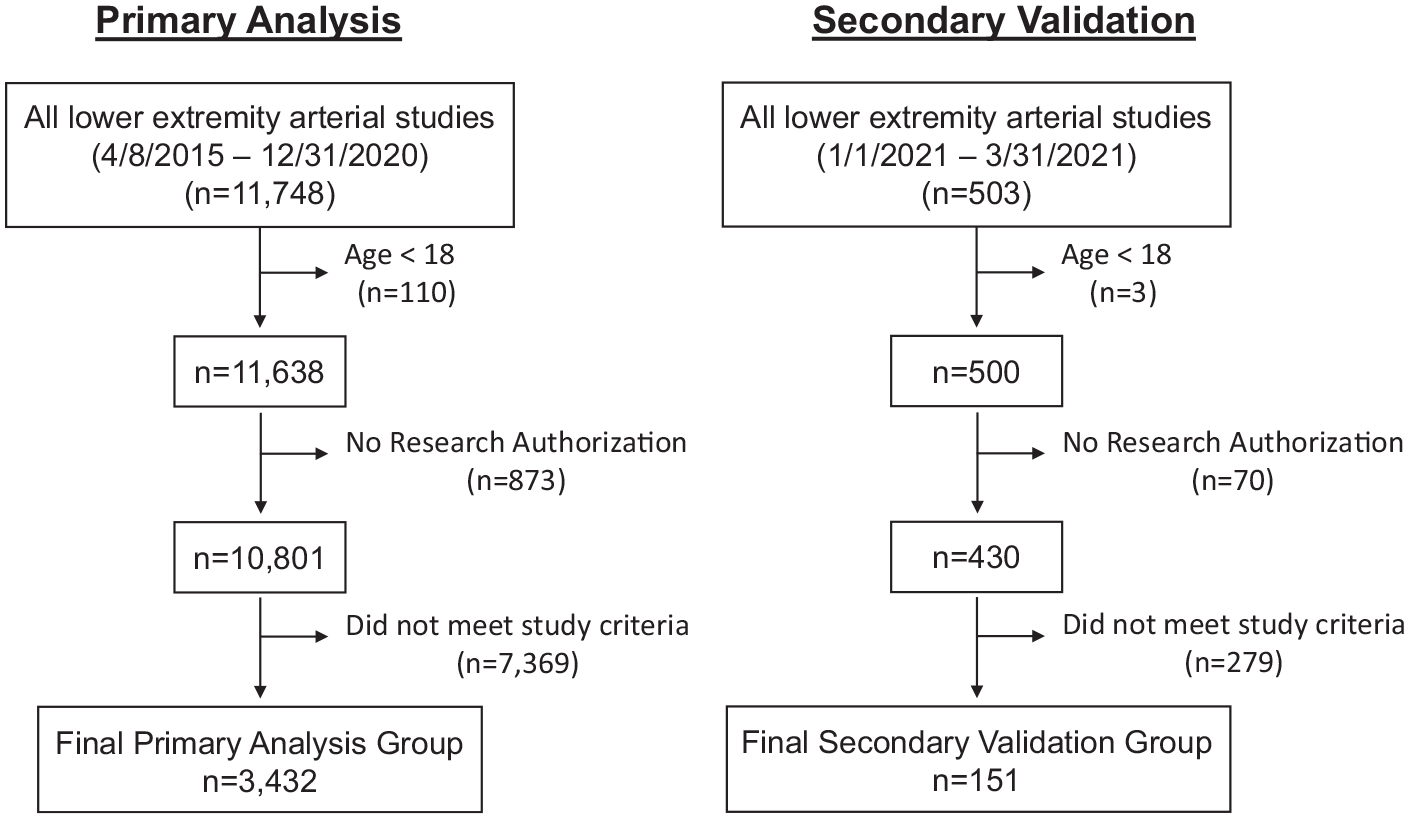
Patient selection and exclusion flow diagram. In the primary study group (2015–2020), 11,748 patients had lower-extremity arterial study performed. Of these, 110 were excluded due to age cutoff, 873 lacked Minnesota research authorization, and 7369 did not meet study criteria. Among the 503 patients in the validation cohort (2021), three patients were excluded because of age, 70 because of lacking Minnesota research authorization, and 279 who did not meet study criteria.
Clinical data were collected from a centralized digital data warehouse that contains complete records of all patients evaluated in all sites of the enterprise. The Mayo Clinic electronic medical record (EHR) data for each patient contains the details for every inpatient hospitalization, every outpatient visit regardless of the provider, every radiology examination, and all laboratory and pathology results (including autopsy reports). Patient comorbidities were defined using procedural codes and International Classification of Diseases (ICD)-9 and ICD-10 diagnosis codes found in the EHR data of patients prior to their index date.15–17 The Mayo Clinic Gonda Vascular Laboratory database contains all vascular studies performed in Rochester MN since inception and served as the source for Doppler waveforms and ABI measurements. The Vascular Laboratory study date was considered the index date for each patient. For those patients with repeat studies, the index data was used for analysis.
Lower-extremity arterial test protocol
Arterial physiology was assessed as previously described. 18 Trained technicians dedicated to the Gonda Mayo Clinic Noninvasive Vascular Laboratory performed each evaluation.
Resting study
With the patient lying supine, CW Doppler waveforms were recorded at the common femoral, superficial femoral, popliteal, posterior tibial, and dorsalis pedis arterial segments for both legs (Parks Vascular Flo-Lab System, SonovaE 2100-SX, Beaverton, OR). Once the CW Doppler signal was identified, waveform sizes were adjusted starting at a gain of 20% until the waveform fit within the monitor display and then archived electronically. ABI measurement was performed as previously described. 18
Treadmill exercise testing
Exercise ABI testing was performed as previously described. 18 The treadmill (GE Marquette Series 2000 treadmill, GE Case V6.73 computer system, General Electric Healthcare, Wauwatosa, WI) was started at a preset rate of 0.7 mph and then increased to 1, 1.5, or 2 mph according to the patient’s ability to walk in the first 30 seconds. The standard vascular exercise protocol was 2 mph, 10% grade for a 5-minute duration. While performing the exercise, the patient was monitored for leg and overall cardiovascular symptoms with brachial blood pressures and an electrocardiogram (ECG) obtained at each minute interval. Postexercise, ankle systolic blood pressures were promptly obtained using the Doppler technique. Where the ABI dropped below 1.0, a common femoral Doppler waveform was obtained and recorded. Brachial and posterior tibial or dorsalis pedis systolic pressures were repeated at 1, 3, and 5 minutes postexercise and at 5-minute intervals as indicated up to 20 minutes, or until values returned to within 0.10 of their resting ABI.
Outcome measure
The primary study outcome was the ability of the artificial intelligence (AI)-enhanced analysis of resting (preexercise) posterior tibial arterial Doppler waveform to distinguish patients with PAD from those without PAD (i.e., normal rest and postexercise lower-extremity arterial testing). This performance was mathematically assessed by the area under the receiver operating characteristic curve (AUC), sensitivity, specificity, accuracy, and positive predictive value (PPV).
Patients were characterized based on the presence or absence of PAD defined by rest and postexercise ABI values. To be characterized as normal, both the resting and postexercise ABI had to be > 0.90 in both extremities, and the postexercise common femoral arterial Doppler waveform had to be either triphasic or biphasic. If the postexercise ABI was ⩽ 0.9 in either limb or the postexercise common femoral arterial Doppler waveform in either limb was neither triphasic nor biphasic, then the patient was characterized as having PAD.
Artificial intelligence analysis
The AI analysis utilized two distinct datasets, termed primary and validation (Figure 1), both collected from the vascular laboratory described above but over two different times.19–22 All model development and initial evaluation was conducted on the primary dataset, with the validation dataset collected afterwards.
The primary dataset consisted of consecutive patients undergoing vascular laboratory testing between 4/8/2015 and 12/31/2020. The secondary validation dataset consisted of consecutive patients tested between 1/1/2021 and 3/31/2021. Included in the primary dataset and secondary validation dataset were patients with normal and abnormal resting ABI values. For patients with repeat vascular testing, only results from the index study were included. The primary dataset was divided into three subsets – training (n = 2403; 70%), validation (n = 515; 15%), and testing (n = 514; 15%) – via random sampling. These proportions were chosen to ensure an adequate number of patients with PAD in the subsets. Each patient was uniquely assigned to a single group. The validation data set was collected from an entirely different set of patients not included in the primary data set. This validation set was used to monitor performance during training and for alternative classification threshold determination, whereas the test set was used for independent evaluation.
A variety of deep neural network (DNN) architectures were explored; all trained as binary classifiers on the resting Doppler waveforms to predict normal (> 0.9) or abnormal (⩽ 0.9) rest and postexercise ABI (Figure 2; see online supplementary material for additional details). Model performance was evaluated by standard metrics such as AUC, positive and negative predictive values, specificities, and sensitivities. Prevalence-dependent quantities were evaluated at the primary dataset’s PAD prevalence of 57%. All performance metrics reported results from either the test subset of the primary dataset or the validation dataset and do not contain any observations considered during training.
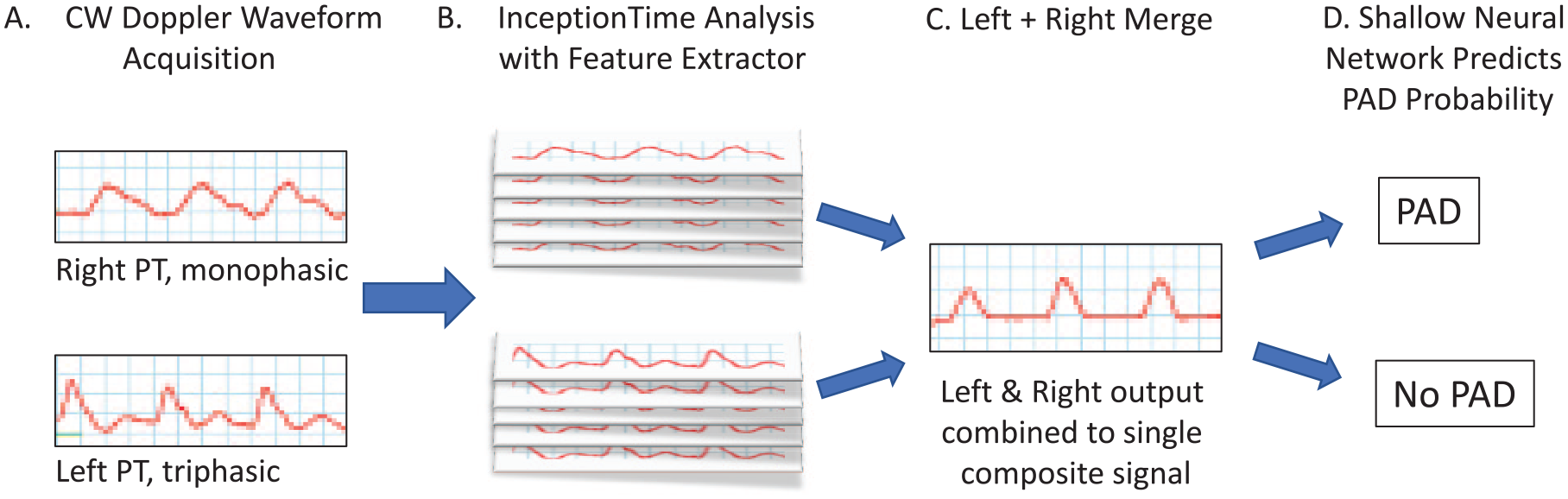
Network architecture. Resting posterior tibialis CW Doppler waveforms from the left and right legs were acquired in the vascular laboratory (
Statistical analysis
Clinical measurements and descriptive characteristics were summarized using frequencies for categorical variables and mean, SD, minimum, and maximum for continuous variables. Differences between patients with and without PAD were analyzed using the chi-squared test for categorical variables and t-test for continuous variables.
In all cases, a two-tailed p-value of less than 0.05 was considered statistically significant. Statistical analysis was done using SAS software, Version 9.4 (SAS Institute Inc.) and the R software package v3.6.2 (R Foundation for Statistical Computing). AI model development was performed using TensorFlow 2.1.0 and Python 3.7.7.
Results
Demographic characteristics
Among 3432 patients included in the primary dataset (Table 1), there were 1941 (57%) with PAD (abnormal ABI; mean age 69 ± 12 years) and 1491 (43%) without PAD (normal ABI; mean age 64 ± 14 years). A number of clinical characteristics differed significantly between patients with and without PAD, including a higher prevalence of hypertension, dyslipidemia, diabetes mellitus, and tobacco use. Patients with PAD were also more likely to have a history of cardiovascular disease, including myocardial infarction, heart failure, stroke, and obstructive pulmonary disease. Similarly, for the 151 patients in the secondary validation dataset (Table 2), significant differences between the PAD and non-PAD groups were observed.
Primary study group (2015–2020) demographics (N = 3432).
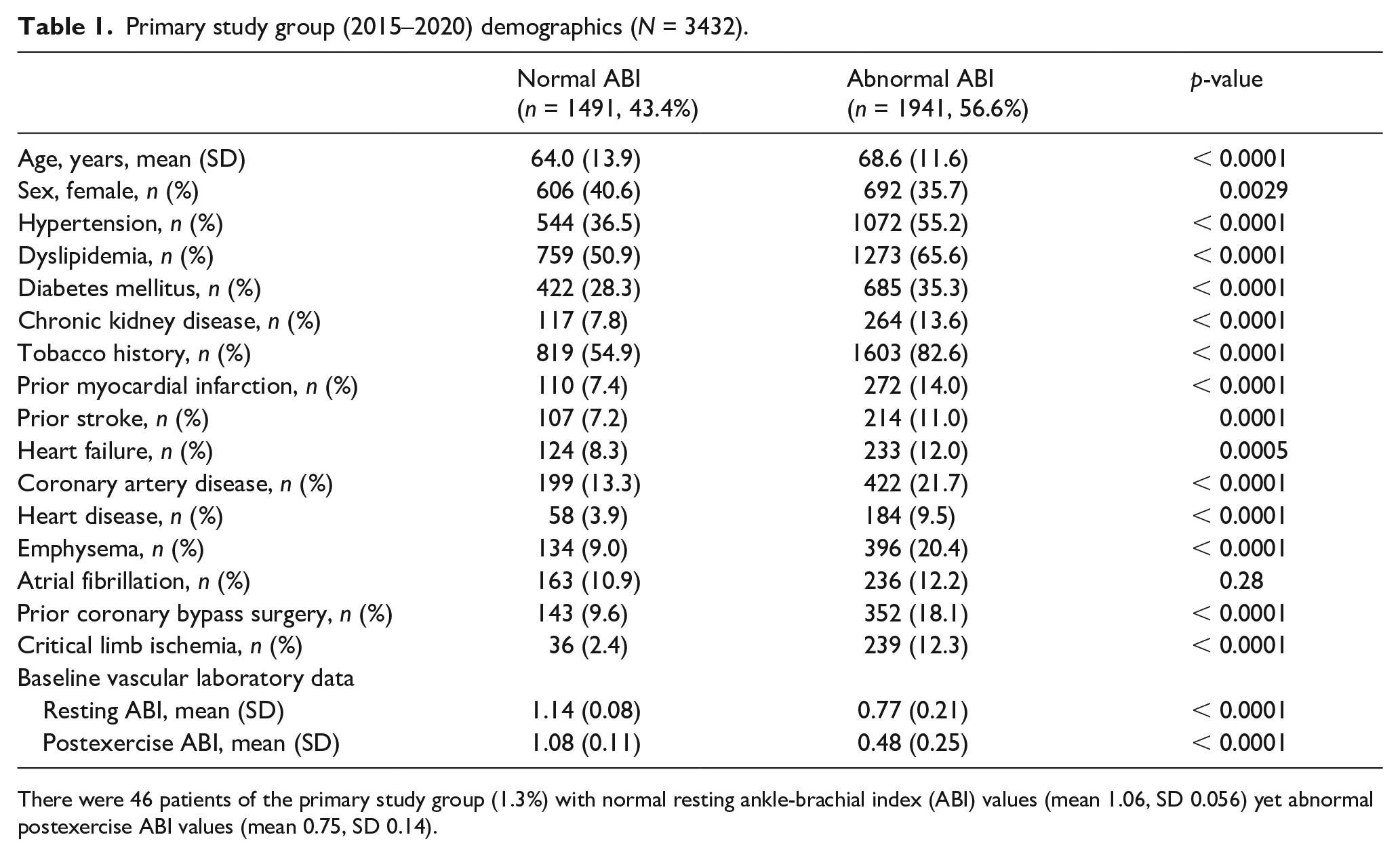
There were 46 patients of the primary study group (1.3%) with normal resting ankle-brachial index (ABI) values (mean 1.06, SD 0.056) yet abnormal postexercise ABI values (mean 0.75, SD 0.14).
Validation data set (2021) demographics (N = 151).
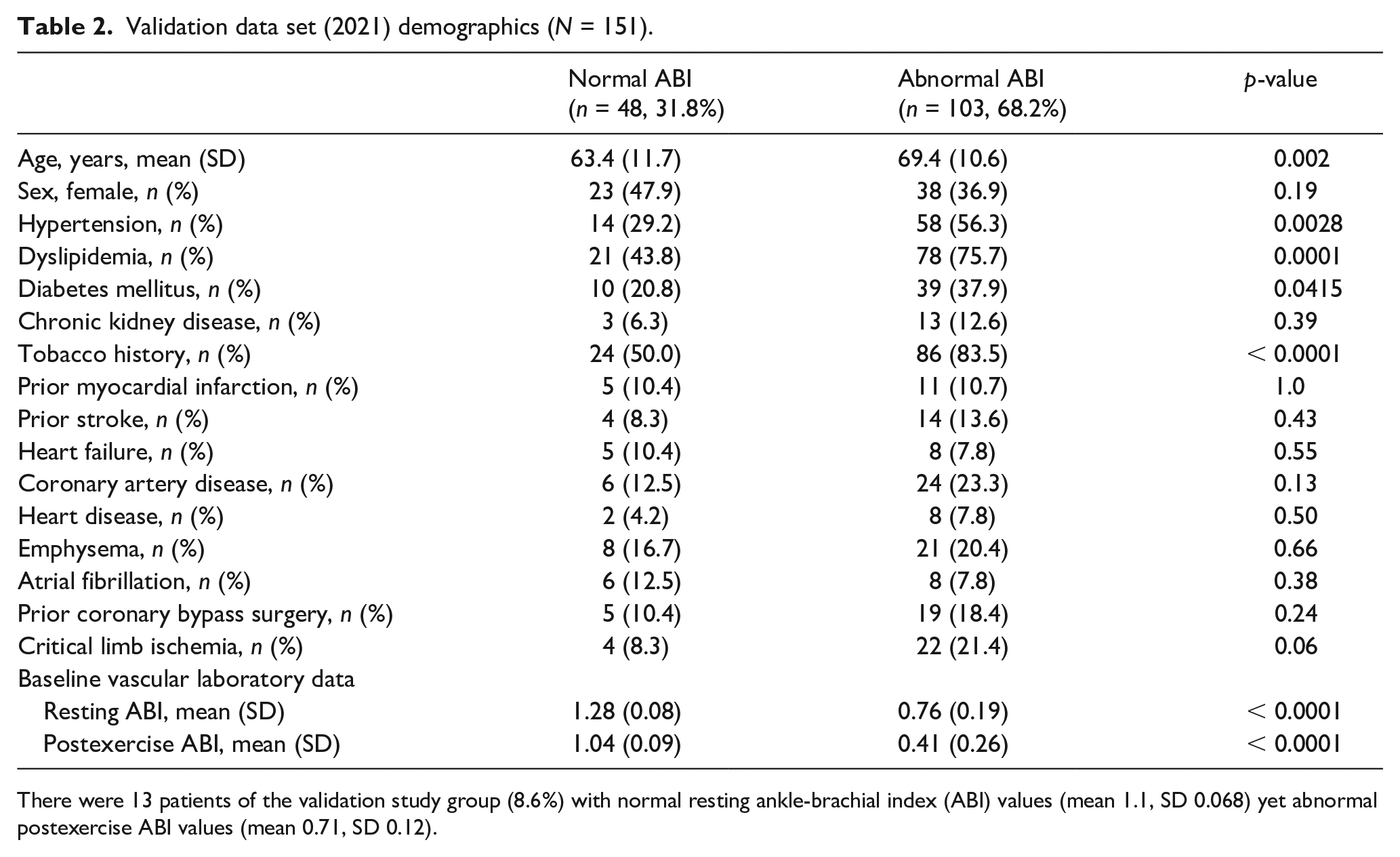
There were 13 patients of the validation study group (8.6%) with normal resting ankle-brachial index (ABI) values (mean 1.1, SD 0.068) yet abnormal postexercise ABI values (mean 0.71, SD 0.12).
Model performance
The model with the best performance (Table 3) was a novel architecture based on the InceptionTime (IT) architecture.19–21 Two inputs, one for each leg, were passed through an IT block with the final fully connected layer pruned to form a feature extractor (Figure 2). The two resulting transformed waveforms were then summed before continuing to a fully connected layer activated by a sigmoid function.
Test performance for rest and exercise ABI.
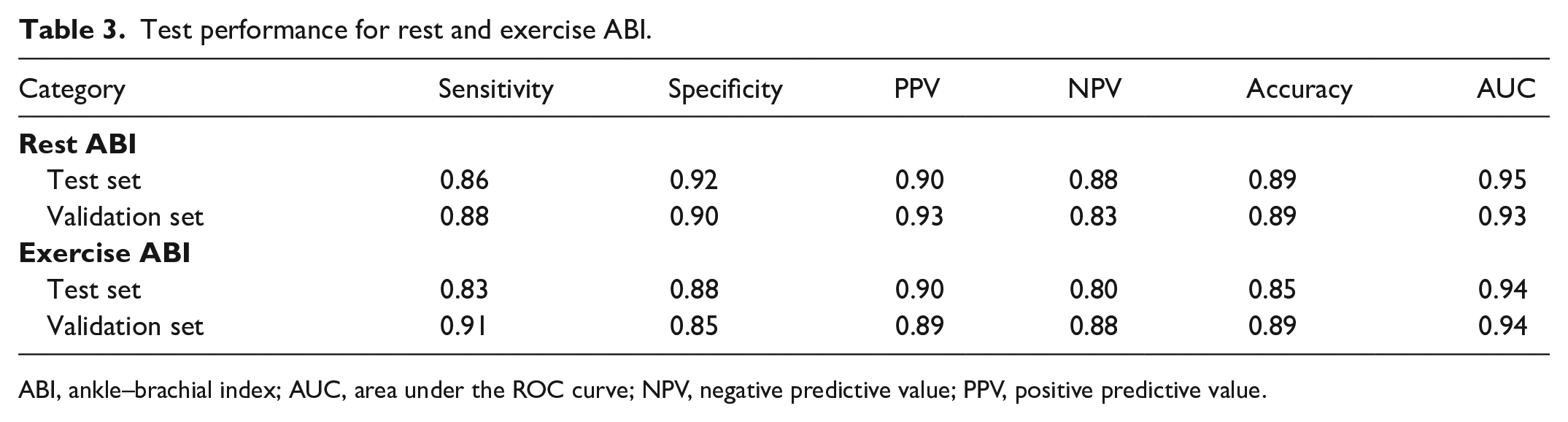
ABI, ankle–brachial index; AUC, area under the ROC curve; NPV, negative predictive value; PPV, positive predictive value.
Model training and validation were performed for both resting and postexercise ABI values. For evaluating the model on the primary dataset for resting ABI, AI interpretation of the posterior tibial Doppler signal identified PAD with an AUC 0.95, sensitivity 0.86, specificity 0.92, accuracy 0.89, PPV 0.90, and negative predictive value (NPV) 0.88 (Figure 3A and 4A; Table 3). Results were similar for the validation dataset (AUC 0.93, sensitivity 0.88, specificity 0.90, accuracy 0.89, PPV 0.93, and NPV 0.83).
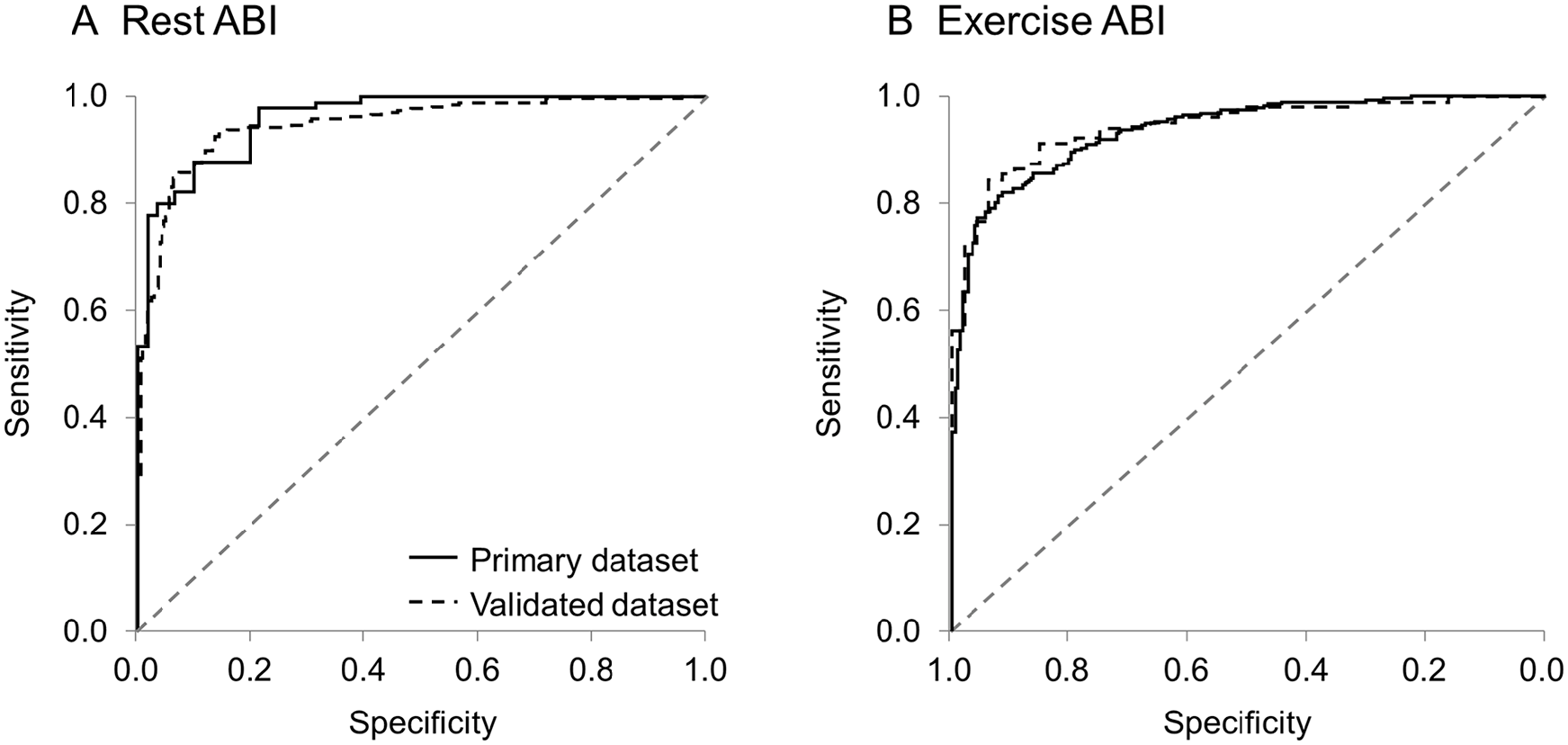
Receiver operating characteristic curves. ROC curves were generated to discriminate patients with normal and abnormal rest (
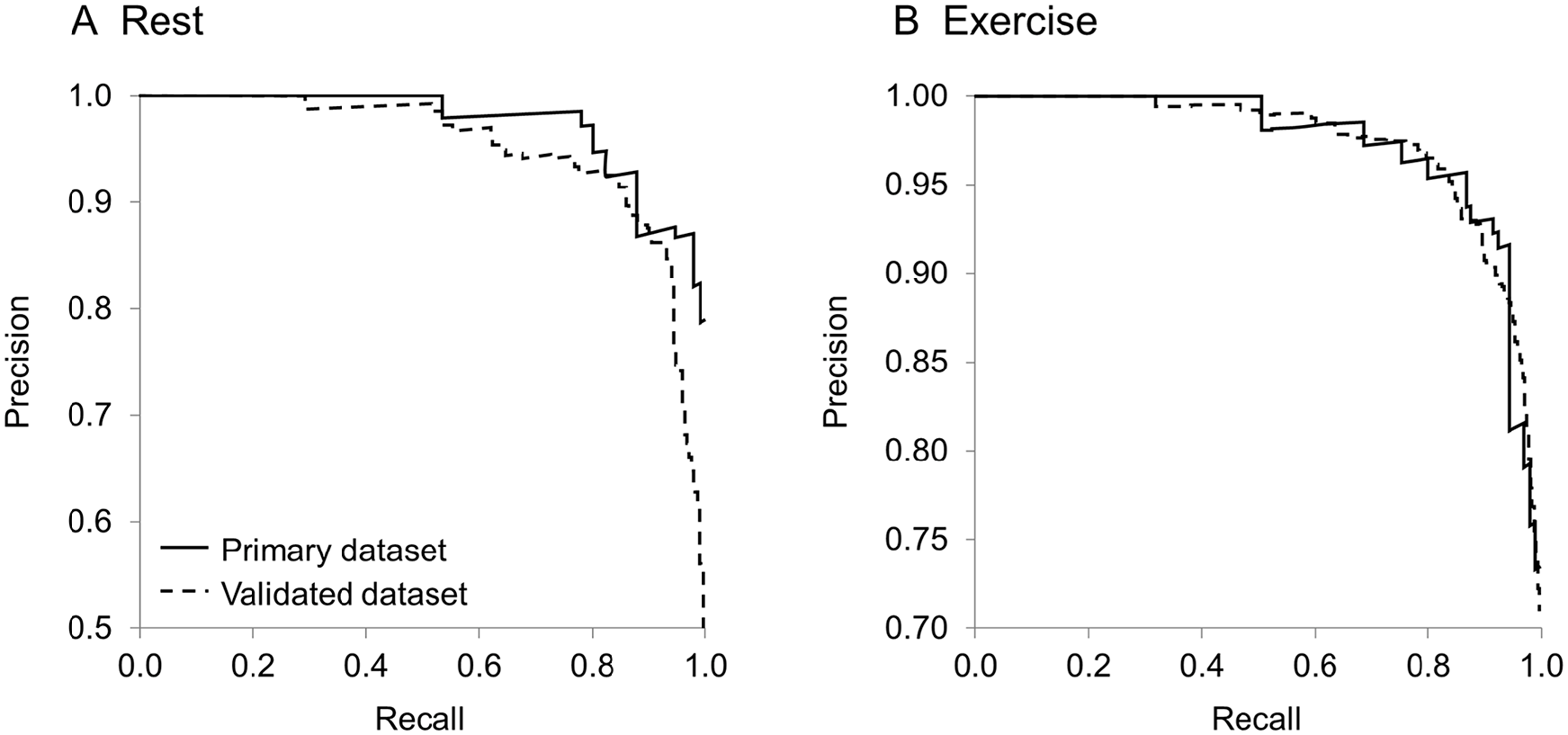
Precision recall curve. Precision recall curves were generated for rest (
For evaluating the model on the primary test set for postexercise ABI, AI interpretation of the posterior tibial Doppler signal identified PAD with an AUC 0.94, sensitivity 0.83, specificity 0.88, accuracy 0.85, PPV 0.90, and NPV 0.80. Results were similar for the validation dataset (AUC 0.94, sensitivity 0.91, specificity 0.85, accuracy 0.89, PPV 0.89, and NPV 0.88; Table 3). The test set and validation set receiver operating characteristic (ROC) curves, as well as the test set precision-recall curve for postexercise, are shown in Figure 3B and Figure 4B, respectively.
The calibration curve linearity across the full spectrum of potential PAD risk is shown in Figure 5. Examples of performance at different probability thresholds and prevalences, including the balanced threshold chosen via Youden index, as well as thresholds selected to make the test highly sensitive or highly specific, are provided (online supplementary Table 1S).
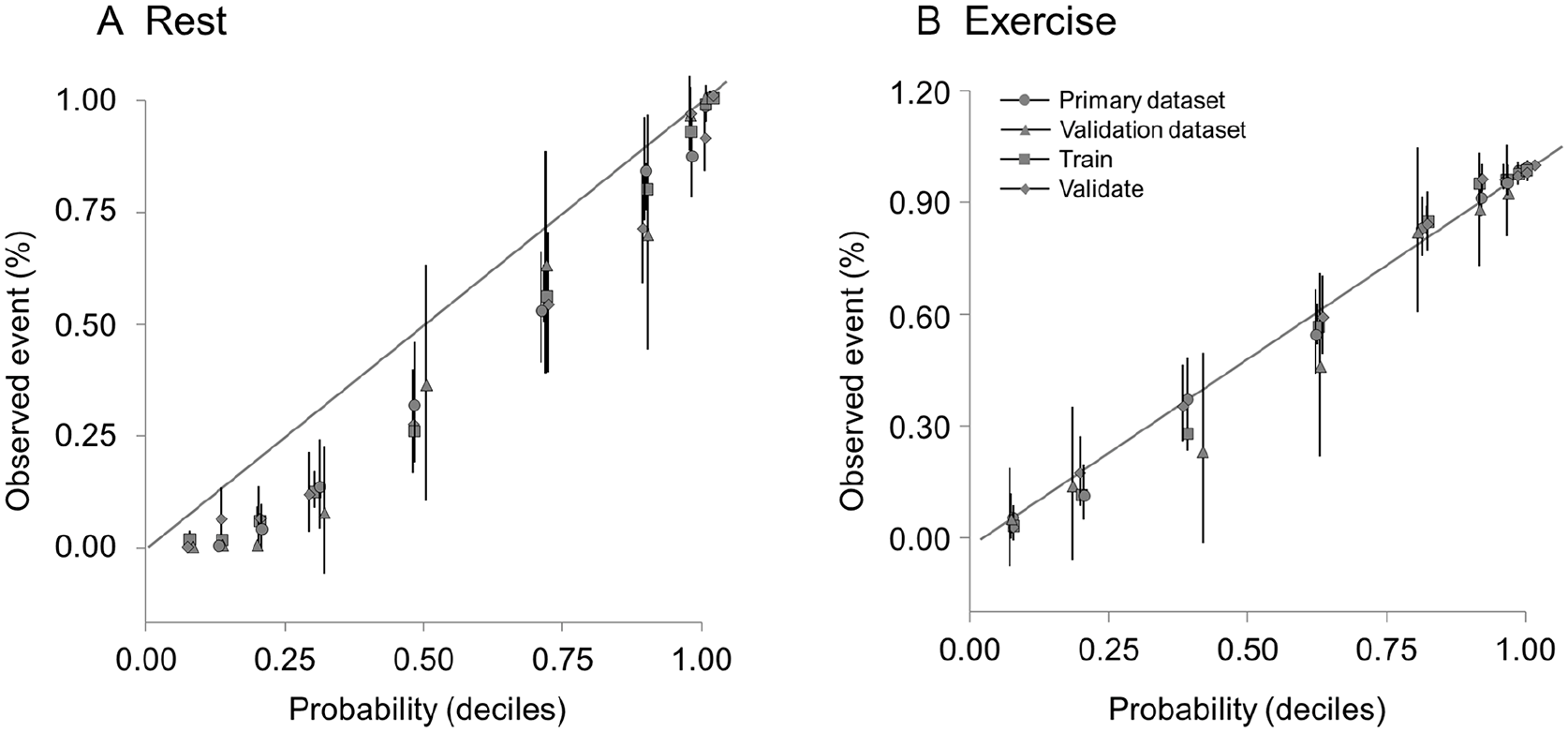
Calibration curve. Calibration compares the probability of abnormal rest (
Further subgroup analyses are provided with disease classification by AI (no PAD vs PAD) stratified by resting and postexercise ABI criteria severity. By rest ABI criteria (online supplementary Table 2S), patients misclassified by AI as ‘no PAD’ were primarily those with borderline (4.5%) or mildly reduced ABI values (1.8%). Less than 1% of patients with moderate disease and no patients with severe disease would have been missed. Conversely, the patients misclassified by AI as ‘PAD’ who actually had normal resting ABI values was much larger at 18.5%. When the same analysis was performed for exercise ABI values, there was better separation (online supplementary Table 3S). There were no patients with normal postexercise ABI values in the PAD group. Nearly 14% had mild disease, most had moderate disease (76.6%), and a lesser percentage had severe disease (9.7%). A list of signs and symptoms suggesting PAD are also provided (online supplementary Table 4S).
Subgroup analyses
The AI model’s performance was similar for several different subgroups (Table 4). Overall sensitivity was slightly lower for younger patients; those with diabetes also had a mild decline in predictive performance.
Test set performance by subgroup.
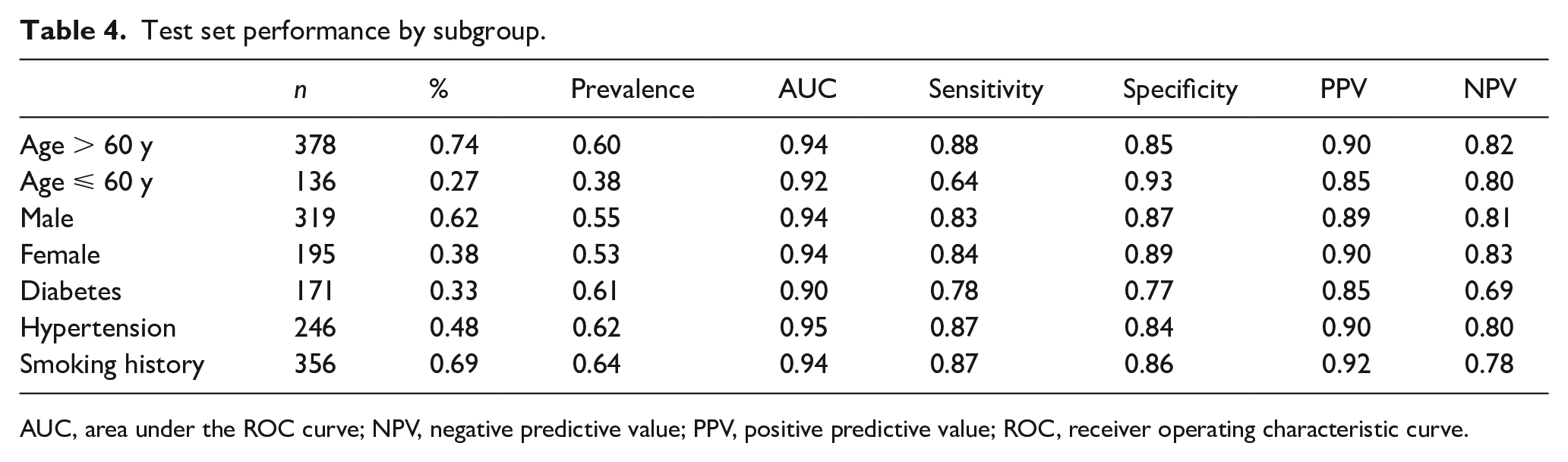
AUC, area under the ROC curve; NPV, negative predictive value; PPV, positive predictive value; ROC, receiver operating characteristic curve.
Discussion
The main results of the current study include the finding that a novel deep neural network can identify PAD with clinically relevant performance accuracy based solely on resting arterial Doppler waveform analysis. This machine analysis appears to distinguish patients with and without PAD with reasonable sensitivity, specificity, and positive and negative predictive values compared to comprehensive vascular laboratory testing including exercise treadmill testing. These findings have relevant implications for PAD screening. First, the addition of machine analysis to vascular laboratory testing might facilitate PAD identification. Small clinics without vascular expertise may be enabled to evaluate patients with leg symptoms prior to vascular center referral. This dissemination might enable more patients to be identified, at an earlier disease stage, potentially sparing future morbidity and mortality. Second, resource utilization might be conserved by enabling patient triage, such that only those high-yield patients requiring a more extensive PAD assessment are referred for specialized vascular evaluation. A comprehensive vascular laboratory requires dedicated space, equipment, and trained personnel for high-quality study performance. As such, these laboratories are resource intensive and not widely available. This triage could enhance PAD detection while conserving resource allocation. Third, patients who are unable to exercise for a variety of reasons, including debilitating cardiac, pulmonary or arthritic diseases, limb amputation or limited vascular laboratory access, could still be offered a comprehensive evaluation. For these combined reasons, machine analysis of Doppler waveforms may offer a new tool for more widespread evaluation of patients with PAD.
These findings build on laudable prior reports using neural network screening devices for the identification of PAD. Using neural network analysis of microvascular waveforms, Allen and Murray assessed the diagnostic accuracy of PAD detection with photoelectric plethysmography in 75 patients. 23 The sensitivity (93%), specificity (85%) and diagnostic accuracy (90%) were later validated on a separate cohort of 111 patients. 24 Although intriguing, photoelectric plethysmography measures are limited by interindividual differences in leg and foot landmarks, data overlap between disease and no disease, impaired accuracy in the context of bilateral lower-extremity disease, and endogenous and exogenous variables inducing vasoconstriction, such as smoking, caffeine or cold ambient temperatures.23–25 As such, the photoelectric plethysmography has enough limitations to preclude widespread utility and implementation for screening purposes. Doppler arterial tracings which are unaffected by these variables may be better suited for PAD screening. Others have successfully employed machine learning to screen for PAD by analyzing variables distinct from the vascular laboratory. 26 Ross et al. assessed machine learning to identify PAD among 1047 participants of the Gene PAD study, a prospective study to evaluate genomic factors contributing to the development of PAD. 27 Using clinical, demographic, and genomic data, machine learning outperformed logistic regression models for both the identification of PAD (AUC 0.87) and future mortality (AUC 0.76). Afzal et al. have used natural language processing analysis of clinic notes within the electronic medical record to search for patients with PAD. 28 Compared to ABI criteria, their algorithm performed well with overall accuracy (91.8%), PPV (92.9%), and specificity (92.5%). Beyond PAD, machine learning has been evaluated in other areas of vascular disease including carotid artery disease and abdominal aortic aneurysm analysis.29,30 As such, machine learning might represent a useful future adjunct for management of a broad spectrum of vascular disorders. One could envision applications for disease screening and severity tracking, medical management best practice advisor alerts, and even operative planning.
Across the globe, more than 200 million patients are impacted by PAD with an increased likelihood of morbidity, disability due to limb loss, and mortality.31–33 Identifying PAD conveys adverse prognostic implications. Myocardial infarction, stroke, limb loss, cardiovascular and all-cause mortality are all adversely impacted such that PAD has become an established independent predictor for cardiovascular mortality after controlling for other traditional risk factors. The critical first step for PAD management is the early and accurate identification of patients with this condition. Although seemingly straightforward, identifying patients with either symptomatic or asymptomatic PAD can be challenging. First, only a minority of patients with PAD present with classic symptoms of intermittent claudication. In the GetABI study of patients over 65 years of age, only 11% of patients with confirmed PAD had symptoms consistent with intermittent claudication using the World Health Organization questionnaire. 34 In the Partners study, which enrolled 1865 subjects with confirmed PAD, only 8.7% had typical claudication symptoms as assessed by the Rose criteria. 35 Furthermore, less than half of medical providers caring for these patients had recognized the PAD diagnosis at the time of study entry. Therefore, relying on a history of intermittent claudication for PAD identification has limitations. Second, demonstrating pulse deficits lacks sensitivity, specificity, and overall accuracy even among trained vascular specialists.36–41 Although pulse confirmation of the femoral artery remains reasonably accurate, correct interpretation of more distal pulses may be no more than 50%. 40 Third, resting ABI assessment carries a high sensitivity and specificity for PAD identification, but many patients may be missed if testing is limited to resting values. 41 Although adding an exercise protocol to ABI testing improves the overall accuracy of PAD ascertainment, this requires a dedicated vascular laboratory for implementation. Widespread availability of vascular laboratories across the country is lacking and many laboratories lack adequate credentials and societal accreditation. Indeed, in the VALUE (Vascular Accreditation, Location and Utilization Evaluation) Study, only 13% of facilities were accredited. 42 Furthermore, this type of exercise ABI testing would ideally include ECG monitoring to identify coexisting severe coronary disease and this practice is not universal. 18 For these combined reasons, highly accurate screening tools capable of identifying PAD would be immensely useful clinically. Early PAD identification is central to facilitate prompt and aggressive initiation of guideline-endorsed medical therapies which are known to impact both major adverse limb and cardiovascular outcomes. 9 And yet, these scientifically proven life, heart, brain, and limb-preserving therapies can only be implemented once you have identified those individuals with the greatest likelihood of benefit. This logic underscores the importance of early and accurate PAD identification. This identification requires the development of appropriate tools for this purpose.
Study limitations
This study is best understood in the context of its limitations. First, the definition of PAD is taken from industry standards and represents clinically accepted parameters using ABI and Doppler signals as outcome measures. Milder forms of early, nonflow-limiting PAD may have been missed by these criteria. Future studies assessing AI on patients with milder forms of the disease such as may be identified by computed tomography (CT) or conventional angiography with intravascular ultrasound could be informative to determine the true sensitivity of this analysis. Second, our study criteria excluded patients with prior surgical or endovascular interventions, poorly compressible vessels, severe resting hypertension, gait instability, leg ulcers and lower limb amputation. Determining whether AI assessment of PAD in the context of these important common co-conditions has the same operating characteristics would be an important next step. Third, the analysis requires a careful recording of the posterior tibial artery Doppler signal. Improper collection of this tracing by untrained personnel may limit the utility of this methodology. Future studies to assess the tolerance of such signal acquisition would be important. Fourth, limited racial diversity of our patient population may limit generalizability. Fifth, although the sensitivity, specificity, and overall accuracy of the model is good, it is not perfect. False positive results will lead to further confirmatory testing, which adds expense and further resource utilization. False negative results may lead to missed diagnoses and compromised care. As such, care process models utilizing such screening tools would ideally include an entry point of proper patient selection. In the end, all screening strategies require Bayesian logic where test performance is influenced by population prevalence of the targeted disease.
Conclusion
In conclusion, applying AI with deep neural network learning to an easy-to-perform, noninvasive ultrasound measure, may enable the Doppler signal to serve as a potential tool to screen for PAD. Further validation studies are required to assess test accuracy and reproducibility in other settings outside of a large volume academic vascular laboratory.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221094082 – Supplemental material for Artificial intelligence for the evaluation of peripheral artery disease using arterial Doppler waveforms to predict abnormal ankle-brachial index
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221094082 for Artificial intelligence for the evaluation of peripheral artery disease using arterial Doppler waveforms to predict abnormal ankle-brachial index by Robert D McBane, Dennis H Murphree, David Liedl, Francisco Lopez-Jimenez, Itzhak Zachi Attia, Adelaide Arruda-Olson, Christopher G Scott, Naresh Prodduturi, Steve E Nowakowski, Thom W Rooke, Ana I Casanegra, Waldemar E Wysokinski, Keith E Swanson, Damon E Houghton, Haraldur Bjarnason and Paul W Wennberg in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
