Abstract

Keywords
Clinical evidence regarding combination anticoagulant–antiplatelet therapy has largely focused on atrial fibrillation (AF) as the primary indication for anticoagulation. Expert recommendations from the American College of Cardiology (ACC) discourage the use of so-called ‘triple therapy’, that is, anticoagulation and dual antiplatelet therapy (aspirin and a P2Y12 inhibitor), in patients on preexisting antiplatelet therapy who develop a new venous thromboembolism (VTE). 1 However, this guidance is extrapolated from evidence in AF without robust data in the VTE population, which has distinct demographics, comorbidities, and time-varying risk of recurrent thrombosis. 2 To address this gap, we explored a multicenter cohort of patients initiated on warfarin therapy for management of VTE. Our study aimed to evaluate the frequency and outcomes of antiplatelet therapy in addition to warfarin for patients with VTE.
We retrospectively analyzed adult patients in the Blue Cross Blue Shield-sponsored Michigan Anticoagulation Quality Improvement Initiative (MAQI 2 ) registry who were newly initiated on warfarin therapy for VTE and entered in the registry between April 1, 2010 and October 31, 2020.3,4 The University of Michigan institutional review board approved the study. Patients with a history of atrial fibrillation/flutter, heart valve replacement, and/or antiphospholipid antibody syndrome were excluded. Patient data were abstracted from the time of enrollment through the earliest of either the first adverse event, discontinuation of warfarin, or last follow-up before October 31, 2020. Patient characteristics were assessed at enrollment, while adverse events were assessed at each follow-up visit. Adjusted associations between number of antiplatelets and event outcomes were examined using Cox proportional hazards.
Of the 2918 patients initiating warfarin for treatment of VTE, 1333 (45.7%) were men with a mean (SD) age of 61.0 (16.4) years. During the study period, 820 patients (28.1%) received warfarin plus one or more antiplatelet medications, compared to 2098 (71.9%) who received warfarin monotherapy (online Supplementary Table 1). Patients on combination warfarin and antiplatelet therapy had a higher incidence of documented coronary artery disease, prior myocardial infarction, and coronary revascularization. Compared to the warfarin monotherapy group, the combination warfarin–antiplatelet group had a higher average RIETE bleed score (2.5 vs 2.0, p < 0.001).5,6 Aspirin monotherapy was the most common antiplatelet regimen (83.4%), followed by dual antiplatelet therapy (11.0%) and P2Y12 inhibitor monotherapy (5.1%).
The warfarin monotherapy reference group experienced 3.3 thrombotic events per 100-patient-years, compared to warfarin plus single antiplatelet (2.7 per 100-patient-years, p = 0.190) or warfarin plus dual antiplatelet (2.2 per 100-patient-years, p = 0.773; Figure 1). The warfarin monotherapy reference group experienced 20.0 overall bleeding events per 100-patient-years, compared to warfarin plus single antiplatelet (24.7 per 100-patient-years, p = 0.013) or warfarin plus dual antiplatelet (58.9 per 100-patient-years, p < 0.001; Figure 1).
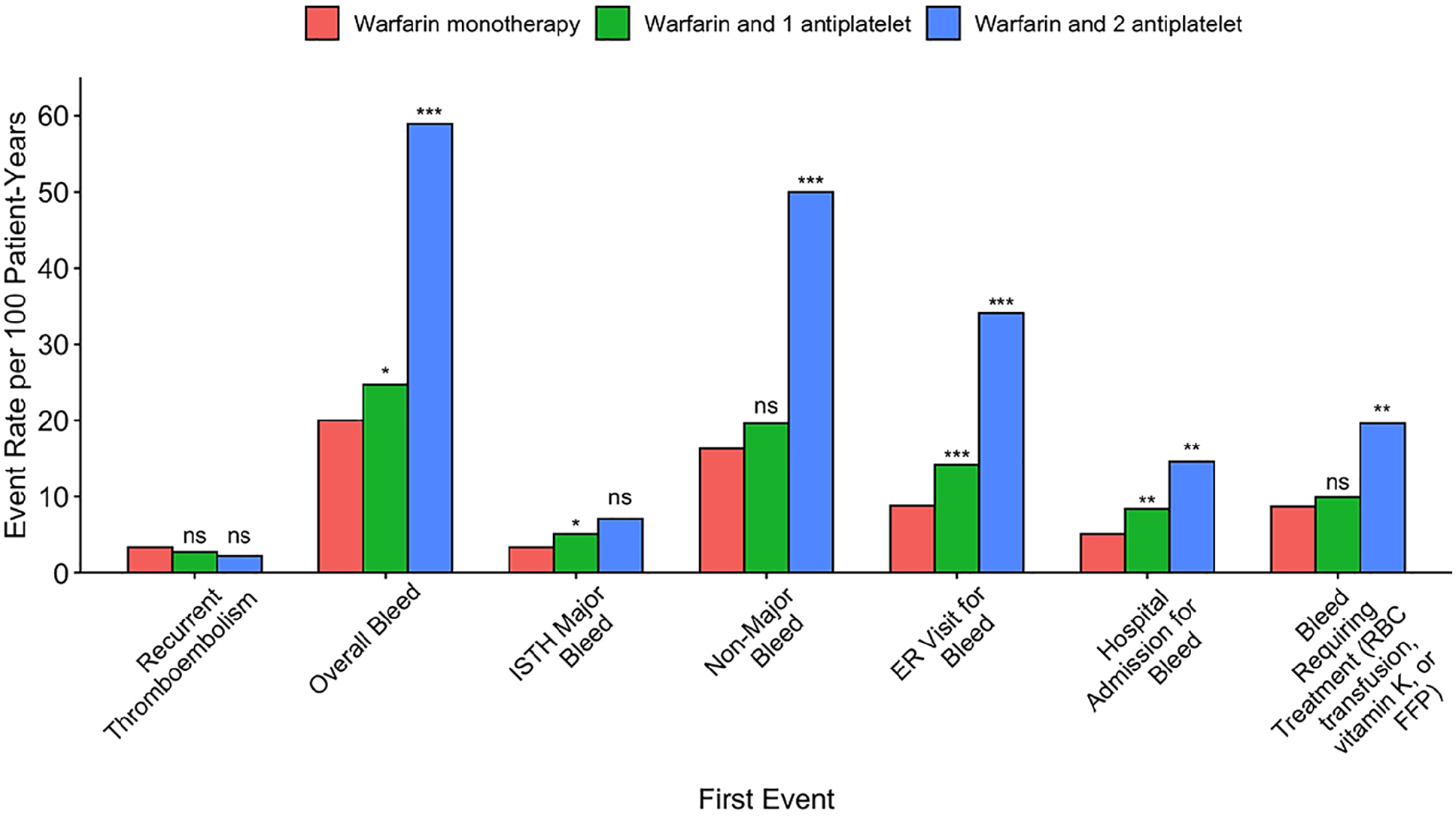
Rates of thrombotic, bleeding, and associated events in patients with VTE with no, one, or two antiplatelet medications in addition to warfarin.
After controlling for age, sex, race, provoked status of VTE, and RIETE score, use of one antiplatelet medication (hazard ratio (HR) 1.21; 95% CI, 1.02–1.43) and use of two antiplatelet medications (HR 2.37; 95% CI, 1.65–3.39) were associated with increased overall bleeding. When examining major bleeding as defined by the International Society on Thrombosis and Haemostasis as the outcome variable, 7 use of one antiplatelet medication (HR 1.37; 95% CI, 0.93–2.02) and use of two antiplatelet medications (HR 1.58; 95% CI, 0.57–4.36) were not significant. With regards to nonmajor bleeding, use of one antiplatelet medication (HR 1.20; 95% CI, 0.99–1.45) was not significant whereas use of two antiplatelet medications was significant (HR 2.53; 95% CI, 1.71–3.75).
In this large, registry-based cohort study of patients followed by anticoagulation clinics while receiving warfarin for VTE, 28.1% of patients received either one or two additional antiplatelet medications. Compared to warfarin monotherapy, combination warfarin–antiplatelet therapy in patients with VTE was associated with increased bleeding and similar rates of thrombosis. This differential bleeding risk is largely driven by nonmajor bleeds.
Our investigation is one of a few dedicated VTE studies examining antithrombotic management in an unselected, practice-based setting. These results provide additional evidence to support the recent 2021 ACC guidelines discouraging ‘triple therapy’ regimens in patients with VTE and atherosclerotic cardiovascular disease, due to unnecessary bleeding risk. 1 Moreover, antiplatelet therapy has been identified as one of the few potentially modifiable bleeding risk factors in patients with VTE. 6 Our findings are also consistent with research demonstrating a significantly higher rate of nonmajor bleeding, in particular among patients taking combination aspirin–direct oral anticoagulant (DOAC) therapy compared to DOAC monotherapy. 8
Limitations of this study include those inherent to registry data, including the potential for unmeasured confounding. It is unknown how these findings generalize to patients receiving DOACs, though these results are consistent with a meta-analysis of six randomized trials that demonstrated an increased risk of bleeding and similar risk of thrombosis in patients with VTE treated with combination DOAC–antiplatelet therapy. 9
In conclusion, use of combination anticoagulant–antiplatelet therapy among patients with VTE is common and associated with higher risk of bleeding. Prospective studies to validate the safety and efficacy of antiplatelet reduction for patients receiving anticoagulation for VTE would strengthen expert management recommendations.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221089333 – Supplemental material for Association of adding antiplatelet therapy to warfarin for management of venous thromboembolism with bleeding and other adverse events
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221089333 for Association of adding antiplatelet therapy to warfarin for management of venous thromboembolism with bleeding and other adverse events by Melinda Song, Brian Haymart, Xiaowen Kong, Mona Ali, Scott Kaatz, Jay Kozlowski, Gregory Krol, Jordan Schaefer, James B Froehlich and Geoffrey D Barnes in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SK serves as a consultant for Janssen, Pfizer, Bristol Myers Squibb, PhaseBio, and Gilead. JBF serves as a consultant for Janssen, Merck, and Novartis. GDB serves as a consultant for Janssen, Pfizer, Bristol Myers Squibb, and Acelis Connected Health. All other authors have no conflicts of interest to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
