Abstract
Introduction:
Direct oral anticoagulants (DOACs) have overtaken warfarin in the treatment of nonvalvular atrial fibrillation (AF) and venous thromboembolism (VTE). Limited data explore the safety of DOACs in obesity.
Methods:
This multicenter retrospective study between June 2015 and September 2019 uses the Michigan Anticoagulation Quality Improvement Initiative (MAQI2) registry to compare DOACs and warfarin across weight classes (not obese: body mass index (BMI) ⩾ 18.5 and < 30; obese: BMI ⩾ 30 and < 40; severely obese: BMI ⩾ 40). Primary outcomes include major, clinically relevant nonmajor (CRNM), and minor bleeding events per 100 patient-years. Secondary outcomes include stroke, recurrent VTE, and all-cause mortality.
Results:
DOACs were prescribed to 49% of the 4089 patients with AF and 46% of the 3162 patients with VTE. Compared to patients treated with warfarin, those treated with DOACs had a higher estimated glomerular filtration rate across BMI categories regardless of indication. In the AF population, severely obese patients treated with DOACs had more major (3.4 vs 1.8, p = 0.004), CRNM (8.6 vs 5.9, p = 0.019), and minor bleeding (11.4 vs 9.9, p = 0.001). There was no difference in stroke or all-cause mortality. In the VTE population, both CRNM (7.5 vs 6.7, p = 0.042) and minor bleeding (19.3 vs 10.5, p < 0.001) events occurred at higher rates in patients treated with DOACs. There was no difference in recurrent pulmonary embolism, stroke, or all-cause mortality.
Conclusion:
There is a higher rate of bleeding in severely obese patients with VTE and AF treated with DOACs compared to warfarin, without a difference in secondary outcomes. Further studies to compare the anticoagulant classes and understand bleeding drivers in this population are needed.
Keywords
Introduction
Direct oral anticoagulants (DOACs) have overtaken warfarin as a first-line treatment of nonvalvular atrial fibrillation (AF) and venous thromboembolism (VTE). This is due to several studies demonstrating that DOACs are safe, efficacious, and have a reduced risk of bleeding.1,2 Furthermore, DOACs use fixed dose regimens without the need for frequent lab monitoring, making them easier for patients to take and less burdensome for clinicians to monitor. Despite these many advantages for most patients with AF and VTE, the pivotal phase 3 clinical trials comparing DOACs to warfarin are limited by small sample sizes of patients with obesity.3–7 The percentage of patients with a BMI exceeding 35 ranged between 13% and 23%, and none of the trials reported the number of patients enrolled with a BMI exceeding 40. 7
Determining the efficacy of DOAC use in obesity is important because body weight influences the pharmacodynamics and pharmacokinetics of anticoagulants. This may result in lower peak concentrations, reduced drug exposure, and a shorter half-life.7,8 Obesity is also associated with changes in renal function, which is a mode of excretion for DOACs. Though warfarin dosage is tailored to the International Normalized Ratio (INR) and may be adjusted to body weight and kidney function, the fixed dose regimen of DOACs could have clinically significant implications at bodyweight extremes.
Therefore, it is important to determine the safety and efficacy of DOACs compared to warfarin across different weight categories.9,10 Post hoc subgroup analyses of the clinical trials have suggested that DOACs may be safe and efficacious for obese (BMI > 30) and extremely obese (BMI > 40) patients. 7 Additional retrospective studies have confirmed these findings. 11 However, both are again limited by population size and may not be representative of real-world populations where patients may have more morbidity and are less compliant than those included in the studies.12–14 As such, the International Society on Thrombosis and Haemostasis suggest that DOACs may be used in patients with a high BMI, but also acknowledge the need for more studies and randomized controlled trials in this group.7,15
This study aims to utilize the Michigan Anticoagulation Quality Improvement Initiative (MAQI2) registry to compare DOAC safety and efficacy to warfarin for AF and VTE across different weight classes, including obese and severely obese.
Methods
MAQI2 is a Blue Cross Blue Shield of Michigan/Blue Care funded quality improvement consortium that has been described in detail in prior publications. 16 Briefly, it is composed of seven hospital or affiliated group anticoagulation management services in the state of Michigan. Its goal is to study anticoagulation practices and outcomes to improve safety and quality of care. The MAQI2 registry was created in 2008, and patient enrollment and data abstraction began in October 2009. Patients are initiated on an anticoagulation of choice by a provider and referred to the anticoagulation service. At time of enrollment, patients are randomly selected for entry into the registry. The patients’ providers independently make all subsequent decisions about anticoagulation dosing, changes, and termination.
The registry collects variables including anticoagulant type, dose, anticoagulation indication, demographics, comorbidities, and adverse events. Each site has trained abstractors that enter patient information using an electronic data collection form. The anticoagulation services manage return visits and patient monitoring independently of registry participation. Chart reviews and data entry happen at every interaction that a patient treated with warfarin has with the service. Chart abstraction is performed every 6 months for a patient treated with a DOAC. To ensure accuracy, standardized variable definitions are used, and random chart audits are performed by the coordinating center at the University of Michigan. Data abstraction, research, and quality improvement activities have been approved by the institutional review board at the coordinating site and the other centers.
Patient criteria and outcome measures
The MAQI2 registry was utilized to identify patients taking DOAC or warfarin between June 2015 and September 2019. Two cohorts defined by the anticoagulation indication (AF or VTE) were created. Patients were characterized as not obese (BMI ⩾ 18.5 and < 30), obese (BMI ⩾ 30 and < 40), and severely obese (BMI ⩾ 40). Underweight patients with a BMI < 18.5 were excluded.
The primary outcome analyzed was bleeding rate per 100 patient-years. Major bleeding and clinically relevant nonmajor bleeding (CRNM) were defined according to the International Society on Thrombosis and Haemostasias (ISTH) guidelines.17,18 Major bleeding included fatal bleeds, symptomatic bleeds in any key body cavity (e.g., intraspinal and intraocular), or bleeds that required at least two units of packed red blood cells or caused greater than 2 g/dL drop in hemoglobin. Clinically relevant nonmajor bleeds included any bleeds that do not fit the criteria for major bleeding events but lead to a medical evaluation or intervention. Lastly, all other bleeds that did not lead to medical evaluation or intervention were characterized as minor bleeds. The secondary outcomes analyzed for patients with AF included rates of stroke, ischemic versus embolic stroke rate, and all-cause mortality. The secondary outcomes analyzed for patients with VTE included rates of stroke, ischemic versus embolic stroke, VTE, deep vein thrombosis (DVT), pulmonary embolism (PE), and all-cause mortality.
Statistical analysis
Statistical analysis was performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA) and R-64 3.3.1 (R Foundation for Statistical Computing, Vienna, Austria). Continuous variables were compared using Student’s t-tests and categorical variables were compared using chi-squared tests. A two-sided p-value less than 0.05 was considered statistically significant for each comparison. Covariates with significant differences between the groups were included in subsequent Poisson regression. Poisson regression was used to assess the differences of event rates.
Results
There are 7251 patients with at least a BMI of 18.5 included in the MAQI2 registry between June 2015 and September 2019, with 3801 (52%) prescribed warfarin and 3450 (48%) prescribed a DOAC. Of the included patients, 2654 (37%) are classified as obese and 1040 (14%) as severely obese.
Patients with atrial fibrillation (AF)
Baseline characteristics
There are 4089 patients prescribed an anticoagulant for AF (Table 1): 2085 (51%) are prescribed warfarin and 2004 (49%) are prescribed a DOAC. Apixaban (81%) is the most frequently prescribed DOAC, followed by rivaroxaban (19%) (Table 2). Of all included patients with AF, 1517 (37%) are obese and 505 (12%) are severely obese. Both not obese and obese patients have an average time in the therapeutic range (TTR) of 56%, and severely obese patients have a TTR of 58%.
Demographics and features of patients with atrial fibrillation treated with warfarin versus DOACs per BMI category.

Not obese: BMI 18.5–29; Obese: BMI 30–39; Severely obese: BMI ⩾ 40.
BMI, body mass index; DOAC, direct oral anticoagulant; eGFR, estimated glomerular filtration rate; TTR, time in therapeutic range.
DOACs used for patients with atrial fibrillation per BMI category.
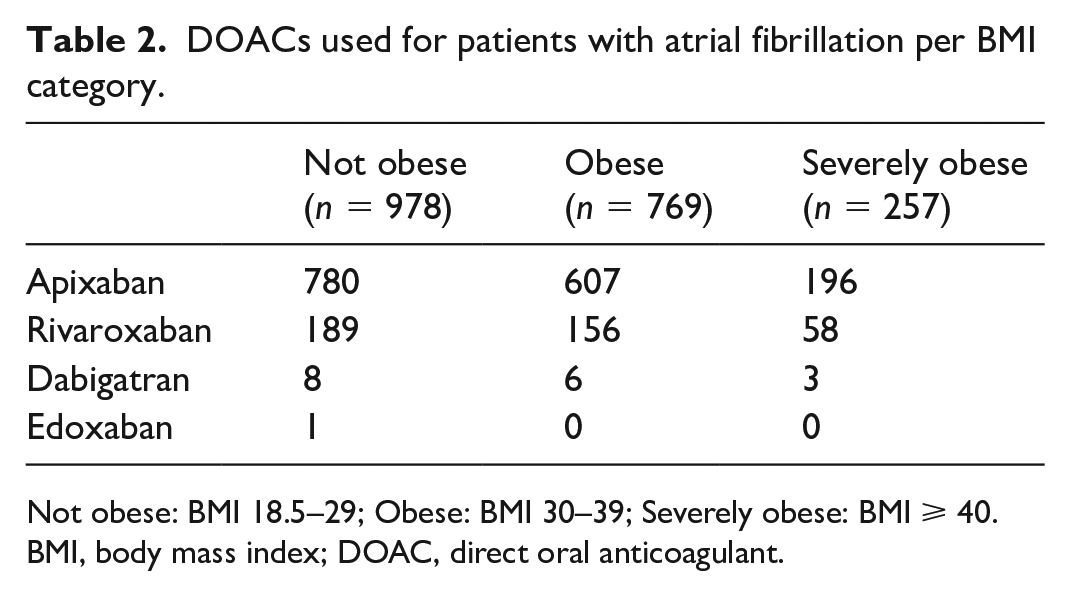
Not obese: BMI 18.5–29; Obese: BMI 30–39; Severely obese: BMI ⩾ 40.
BMI, body mass index; DOAC, direct oral anticoagulant.
Regardless of BMI category, patients treated with DOACs are older than patients treated with warfarin. Not obese (3.2 vs 2.9, p < 0.001) and obese (2.9 vs 2.7, p < 0.001) patients treated with warfarin have a higher HAS-BLED score at enrollment compared to patients treated with DOACs. Severely obese patients have similar HAS-BLED scores at time of enrollment regardless of treatment type (2.6 vs 2.6, p = 0.82). There are no significant differences in the CHA2DS2-VASc score between patients treated with warfarin or DOACs across the three BMI categories. Patients treated with DOACs have a higher mean eGFR (122.9 vs 64.3; 179.9 vs 88.6 vs 258.8 vs 127) when compared to patients treated with warfarin across all BMI categories, respectively.
Bleeding events
In the not obese category, there is no statistically significant difference observed in major bleeding events between patients treated with DOACs and warfarin (3.4 vs 3.7, p = 0.088) (Figure 1). However, not obese patients treated with DOACs exhibit a notably higher frequency of CRNM bleeding events (10.7 vs 7.0, p < 0.001) and minor bleeding events (16.0 vs 10.5, p < 0.001) compared to their warfarin-treated counterparts. In the obese category, there is no difference in major bleeding between patients treated with DOACs and warfarin (2.8 vs 4.6, p = 0.93). However, severely obese patients have more major bleeding in the group treated with DOACs (3.4 vs 1.8, p = 0.004). In both the obese and severely obese categories, there is also more CRNM bleeding (9.0 vs 7.0, p = 0.034 for obese; 8.6 vs 5.9, p = 0.019 for severely obese)) and minor bleeding (12.7 vs 11.4, p < 0.001 for obese; 11.4 vs 9.9, p = 0.001 for severely obese) in the groups treated with DOACs.
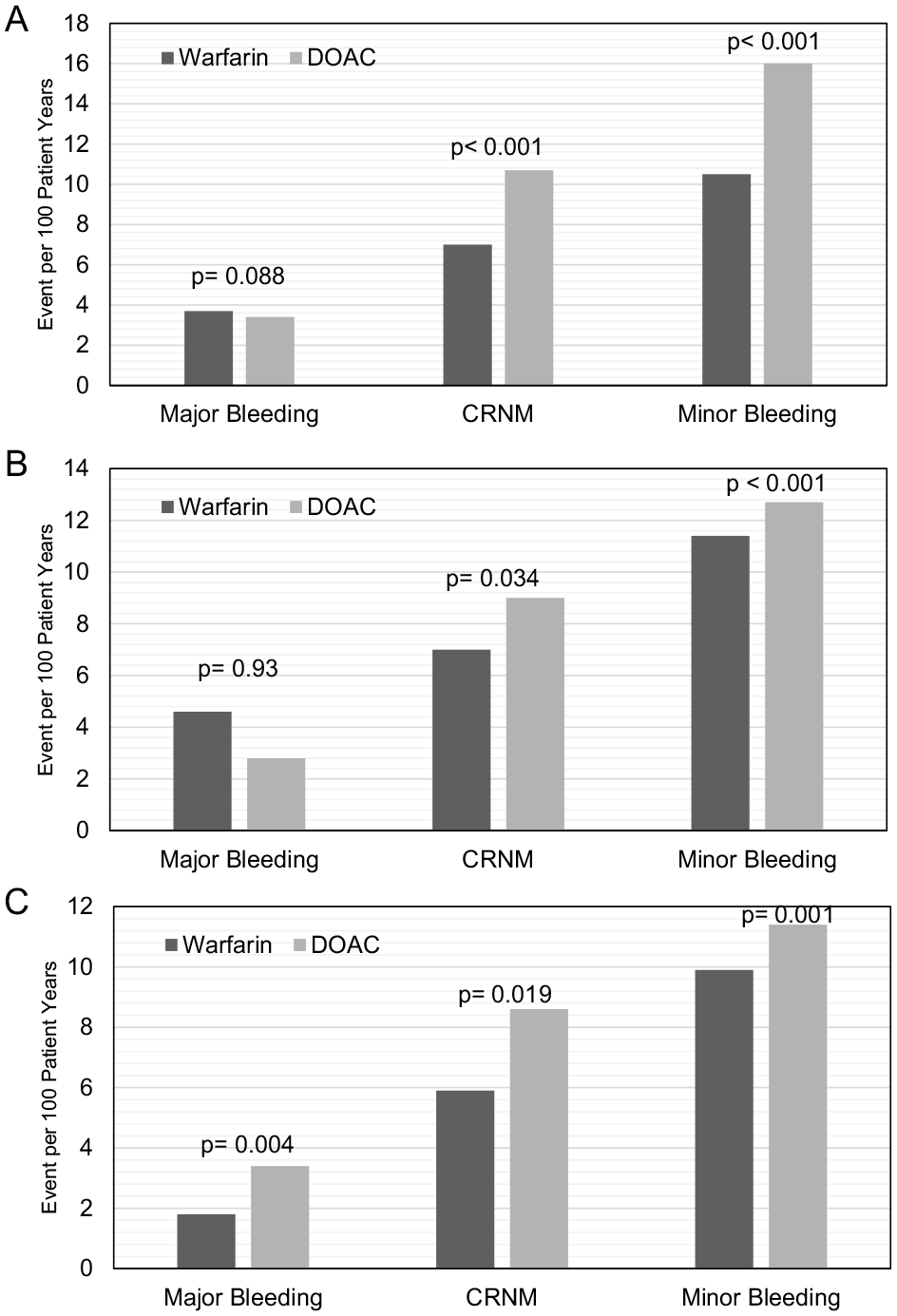
Warfarin versus direct oral anticoagulant bleeding events for patients with atrial fibrillation per BMI category.
Secondary outcomes
Figure 2 illustrates the secondary outcomes analyzed in this study across different BMI categories for patients with AF. There is a reduction of all strokes in not obese patients treated with DOACs (2.1 vs 2.2, p = 0.009), and an increase in strokes for obese patients treated with DOACs (1.7 vs 1.4, p = 0.011). Regardless of anticoagulant choice, there is no difference in all-cause mortality in these two BMI categories. Furthermore, there is no statistical difference in all stroke (2.7 vs 1.3, p = 0.086), ischemic or embolic stroke (0.2 vs 0.2, p = 0.99), or all-cause mortality (2.8 vs 4.3, p = 0.86) in the severely obese category.
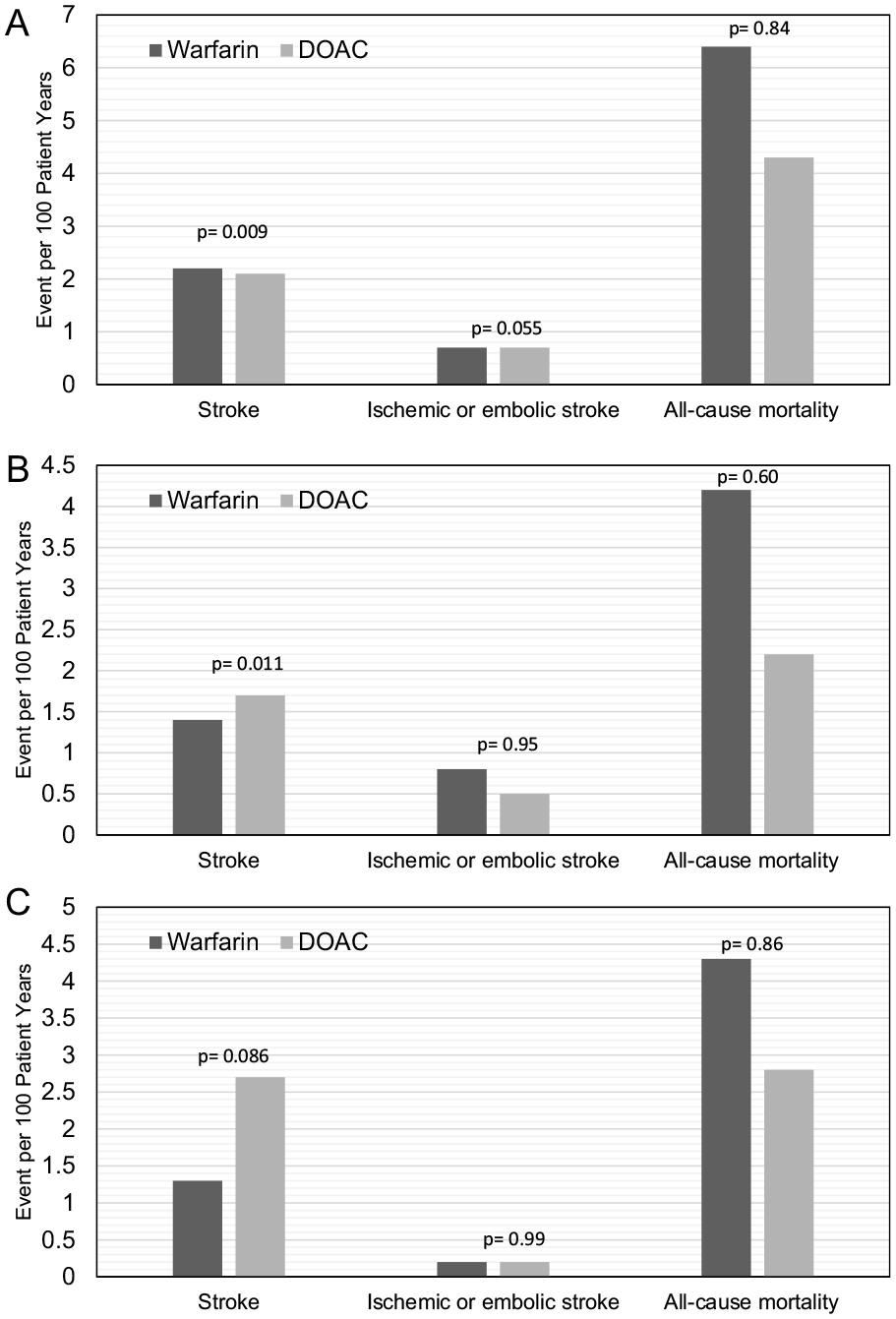
Warfarin versus direct oral anticoagulant secondary outcomes for patients with atrial fibrillation per BMI category.
Patients with VTE
Baseline characteristics
There are 3162 patients prescribed an anticoagulant for VTE (Table 3): 1716 (54%) are prescribed warfarin and 1446 (46%) are prescribed a DOAC. Apixaban (57%) is the most frequently prescribed DOAC followed by rivaroxaban (43%) (Table 4). Of all included patients with VTE, 1138 (36%) are obese and 535 (17%) are severely obese. The TTR for the not obese, obese, and severely obese categories are 53%, 56%, and 58%, respectively. Patients treated with DOACs have a lower HAS-BLED score at the time of enrollment across all BMI categories. Like patients with AF, patients with VTE treated with DOACs also have a higher eGFR compared with warfarin-treated patients across all BMI categories (150.8 vs 82.5, p < 0.001 for not obese; 206.6 vs 110.7, p < 0.001 for obese; 296.8 vs 172.1, p < 0.001 for severely obese).
Demographics and features of patients with venous thromboembolism treated with warfarin versus DOACs per BMI category.

Not obese: BMI 18.5–29; Obese: BMI 30–39; Severely obese: BMI ⩾ 40.
BMI, body mass index; DOAC, direct oral anticoagulant; eGFR, estimated glomerular filtration rate; TTR, time in therapeutic range.
DOACs used for venous thromboembolism per body mass index category.

Not obese: BMI 18.5–29; Obese: BMI 30–39; Severely obese: BMI ⩾ 40.
*6 patients did not have DOAC type data and were excluded.
BMI, body mass index; DOAC, direct oral anticoagulant.
Bleeding events
Minor bleeding is the only clinically significant difference observed between patients treated with DOACs versus warfarin in both the not obese (18.0 vs 12.1, p < 0.001) and obese (18.8 vs 12.9, p < 0.001) categories (Figure 3). However, in the severely obese category, both CRNM (7.5 vs 6.7, p = 0.042) and minor bleeding (19.3 vs 10.5, p < 0.001) events occurred at a higher rate in the patients treated with DOACs. There was no significant difference in major bleeding across all BMI categories (4.4 vs 5.7, p = 0.780 for not obese; 2.6 vs 4.1, p = 0.780 for obese; 3.6 vs 4.6, p = 0.550 for severely obese).
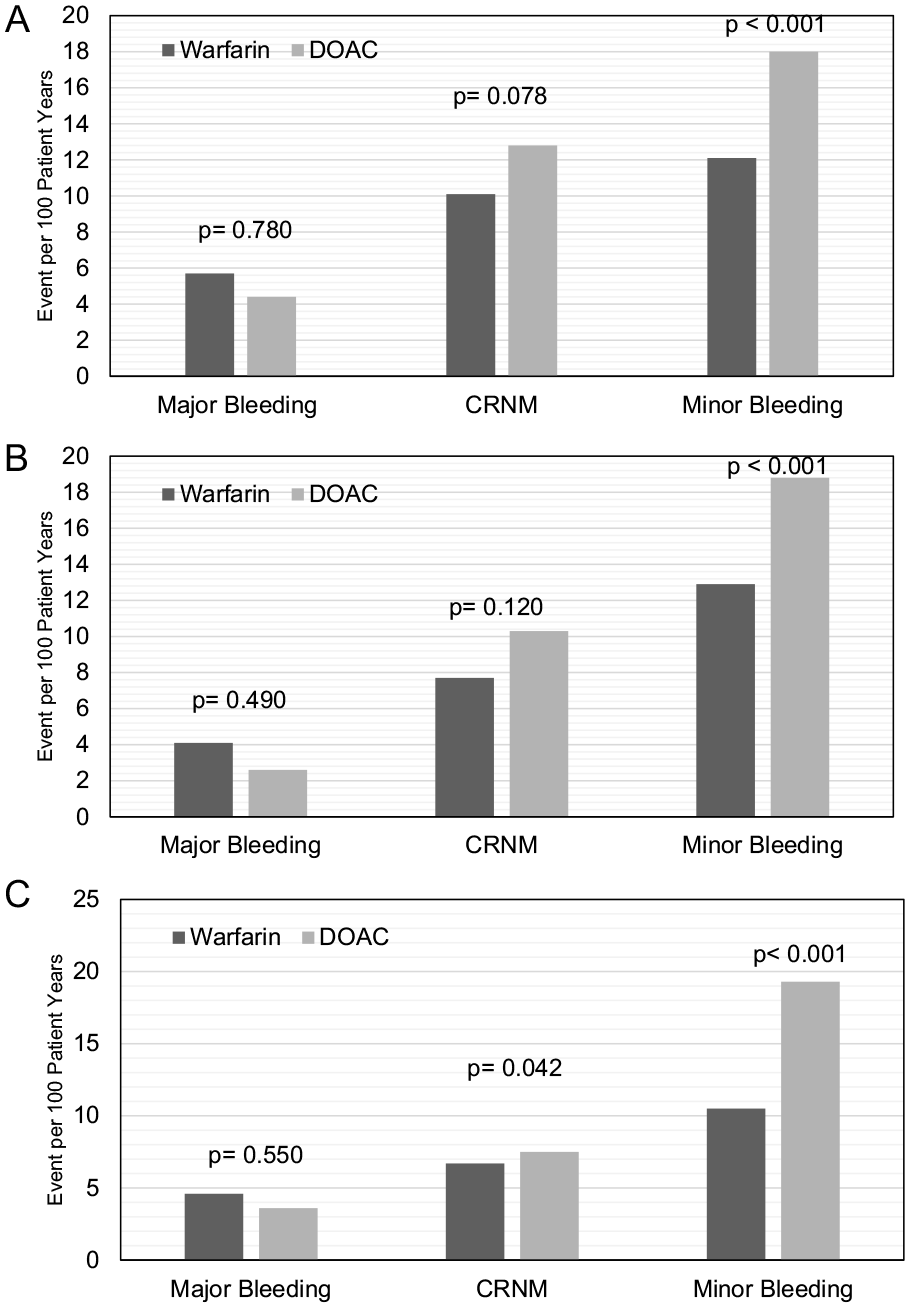
Warfarin versus direct oral anticoagulant bleeding events for patients with venous thromboembolism per BMI category.
Secondary outcomes
Figure 4 illustrates the secondary outcomes analyzed in patients with VTE across different BMI categories. Among not obese patients treated with DOACs, a significant reduction in recurrent VTE (1.8 vs 3.7, p = 0.015) and DVT (0.8 vs 2.6, p = 0.015) is observed when compared to patients treated with warfarin. This significant decrease was not observed in the obese (VTE: 2.4 vs 2.4, p > 0.99; DVT: 1.2 vs 1.9, p = 0.42) or severely obese (VTE: 1.6 vs 2.6, p = 0.42; DVT: 0.8 vs 2.0, p = 0.57) categories. In addition, irrespective of BMI category, there is no significant difference between patients treated with DOACs versus warfarin regarding recurrent PE and stroke events. In terms of all-cause mortality, not obese patients treated with DOACs had an increased all-cause mortality (5.8 vs 5.0, p = 0.044). However, there is no difference in all-cause mortality in the DOAC-treated obese (2.6 vs 3.0, p = 0.07) and severely obese (1.5 vs 2.4, p = 0.73) patients.
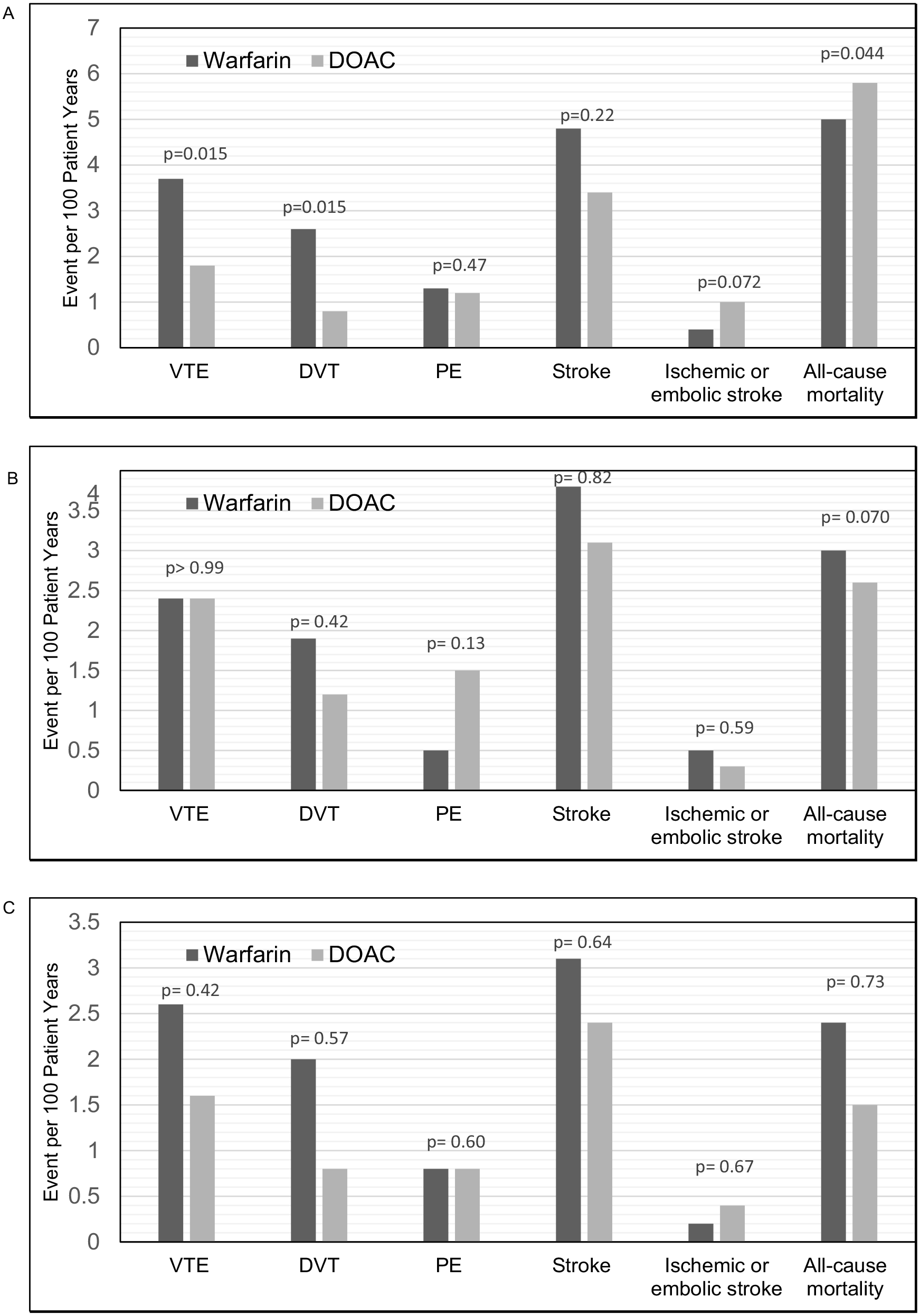
Warfarin versus direct oral anticoagulant secondary outcomes for patients with venous thromboembolism per BMI category.
Discussion
In this multicenter study involving patients treated with anticoagulants for AF, severely obese patients treated with DOACs show higher rates of major bleeding, CRNM, and minor bleeding compared to those treated with warfarin. No differences are found in rates of all-stroke, ischemic or embolic stroke, and all-cause mortality in this group. Among severely obese patients with VTE treated with DOACs, higher rates were observed only in CRNM and minor bleeding, with no differences in secondary outcomes including recurrent VTE, all stroke, and all-cause mortality.
The absence of randomized control data for DOAC utility in severely obese patients has resulted in several retrospective studies. These studies have demonstrated that DOACs are at least noninferior to warfarin in safety and efficacy when treating severely obese patients, regardless of indication. 15 However, though our study supports the noninferiority of DOAC effectiveness, it also indicates that DOACs may possess an inferior safety profile in this population. To our knowledge, this is the first retrospective study that demonstrates an increased bleeding risk in severely obese patients treated with DOACs. There are several reasons that contribute to the contrasting findings in the aforementioned studies.
First, the populations used in the post hoc and retrospective studies may not be representative of real-world populations. These studies tend to have stringent inclusion and exclusion criteria and are usually composed of healthier patients without significant comorbidities. For example, the Einstein DVT and ROCKET-AF studies excluded patients with creatinine clearance below 30 mL per minute, liver disease, high risk of bleeding, and medications that affect cytochrome P-450.4,6 Similarly the metanalysis by Almas et al. excluded patients treated with antiplatelets or certain co-morbidities like diabetes. 19 Furthermore, strict outpatient monitoring and increased compliancy of the clinical trials may not be accurately reproduced in clinical settings. The MAQI2 database is more reflective of a real-world population because it is a registry of randomly selected patients after anticoagulation is initiated. All anticoagulation management decisions are made by a referring provider, with the registry having no impact on anticoagulation method, duration, or switching agents. There is no inclusion or exclusion criteria, so patients are more representative of the general population that are being prescribed the anticoagulants. This difference between the MAQI2 registry and randomized control trials is highlighted in a prior study that determined only 40–60% of MAQI2 registry patients would qualify for the RE-LY, ROCKET-AF, and ARISTOTLE trials. 12
Another explanation is that the MAQI2 registry ensures accurate follow up and capture of bleeding or VTE events. The patients are all followed by the anticoagulation management services and ensure appropriate follow up and data entry every few months. All bleeding events are captured, even if they take place outside the hospital system or are not identified by International Classification of Diseases (ICD) codes. Other retrospective studies, such as Coons et al. and Wee et al., rely on ICD codes in their hospital system to capture bleeding or VTE events.20,21 Another weakness of using ICD codes in retrospective studies is that it cannot accurately capture changes in treatment agents. Changes or holds in anticoagulation trigger patient information in the MAQI2 registry to be updated.
In addition, it is important to consider DOAC utilization changes since the previous studies were published. After introduction to the market in 2010, DOAC prescriptions have exponentially increased. An analysis of Medicare beneficiaries revealed that DOAC utilization surged from 7.4% in 2011 to 66.8% in 2019. 22 This growth in DOAC usage is also evident in populations with higher disease burdens, such as those with chronic kidney disease or cirrhosis. Liabeuf et al. observed an upward trend in DOAC prescriptions between 2013 and 2019 in patients with moderate to severe chronic kidney disease. 23 Similarly, Simon et al. demonstrated an upward trend in DOAC use among patients with cirrhosis for AF between 2012 and 2019. 24 Considering the evolving landscape of DOAC utilization, it is plausible that the population included in the previous retrospective studies may inherently differ from the population captured by the MAQI2 registry.
Another explanation for the decrease in bleeding events in the warfarin-treated severely obese patients may be due to a subtherapeutic INR. Several studies have demonstrated that there are high rates of subtherapeutic INR in patients treated with warfarin in the clinical setting. 25 The average TTR reported in our study ranges from 53% to 58%, which is similar to those reported by the ROCKET-AF study (55%). However, the TTR is lower in our study than the mean reported in the RE-LY (64%), ARISTOTLE (66%), and ENGAGE (68%) studies.3,4,26,27 It is also important to note that there was no associated increase in recurrent VTE or stroke in patients treated with warfarin, which suggests adequate anticoagulation.
There are several limitations to this study. This was a retrospective study and may be limited by confounding factors. Propensity score matching could not be employed due to the limited number of patients in certain obesity categories and the limited number of events. We addressed the possible confounding by using empirical and/or statistically significant unbalanced variables as covariates in the regression model. Only an association between anticoagulant choice and outcome per BMI category can be identified in this study. The anticoagulant choice was determined by clinicians, and therefore the analysis may not capture all the clinical factors that might have resulted in the decision. The MAQI2 cohort and registry had no influence on management decisions made by patients’ providers. However, we do recognize that management patterns of physicians and anticoagulation service management policies may be influenced by prior MAQI2 manuscripts and quality improvement initiatives.
Conclusion
In our study population, DOAC treatment in severely obese patients was associated with higher bleeding event rates in patients with AF and VTE. There were no significant disparities in terms of recurrent VTE, stroke, or mortality compared to warfarin treatment. This finding differs from prior literature comparing DOACs to warfarin. Therefore, additional studies to compare the two anticoagulants and understand bleeding drivers in this population are needed. Meanwhile, it is important to use a shared decision-making model when considering using DOACs in the severely obese population.
Footnotes
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Geoffery Barnes reports research funding from Boston Scientific; consulting for Pfizer, Bristol-Myers Squibb (BMS), Janssen, Bayer, AstraZeneca, Sanofi, Anthos, Abbott Vascular, and Boston Scientific; and he serves on the Board of Directors for the Anticoagulation (AC) Forum. Dr Scott Kaatz reports research funding from Janssen, BMS, Osmosis Research, National Institutes of Health, and Bayer; consulting for Janssen, Pfizer, BMS, AstraZeneca, Gilead, Phase Bio, Boston Scientific, Inari, and Anthos; and Board membership for AC Forum, National Blood Clot Alliance Medical and Scientific Advisory Board, and PERT Consortium. Dr James Froelich reports research funding from Blue Cross Blue Shield of Michigan and FMD Society of America. The remaining authors have no conflicts of interest.
Funding
This study was funded by Blue Cross Blue Shield of Michigan/Blue Care.
