Abstract

Paclitaxel-coated devices were frequently used for the treatment of femoropopliteal peripheral artery disease (PAD) after the first FDA-approved device came to market at the end of 2012. 1 Since then, a series of paclitaxel drug-eluting stents (DES) and drug-coated balloons (DCB) have been developed and are currently approved for the treatment of femoropopliteal PAD.1,2 Paclitaxel, a chemotherapeutic agent, prevents neointimal hyperplasia resulting in improved patency and freedom from reintervention when compared with bare metal stents (BMS) and plain balloon angioplasty (POBA).2,3 Despite superior efficacy outcomes, recent events have taken place that may have impacted paclitaxel devices’ widespread use.
A first development was the termination of additional payment for DCBs in January of 2018, effectively equalizing payment to the reimbursement level of POBA. Second, in December of 2018, a meta-analysis of randomized controlled trials comparing survival after treatment of the femoropopliteal segment with paclitaxel-coated versus uncoated devices was published and demonstrated a statistically significant increased risk of death at 2 and 5 years postintervention. 4 It is unknown how the real-world use of drug-coated devices has evolved over time, and to what degree it has decreased after the occurrence of these two pivotal external events.
The Vascular Quality Initiative (VQI) National Database was queried for endovascular femoropopliteal artery interventions performed between 09/2016 and 12/2019 as September of 2016 was the first month DCB and DES were recorded in VQI. Patients with acute limb ischemia and those undergoing open surgery were excluded. This study was approved by the Yale Institutional Review Board.
The analysis was done at the artery level to account for multiple lesions treated in a single patient. Treatment of each artery was identified in a hierarchical manner with DCB > DES > BMS > POBA. Two time variables were created to represent the expiration in ambulatory payment classification and the publishing of the Katsanos meta-analysis. These dates were entered in a logistic regression model predicting use versus no use of drug-coated devices (DCB and DES combined). The model was adjusted for patient and lesion characteristics including age, sex, race, ethnicity, primary insurance, hypertension, diabetes mellitus, coronary artery disease, congestive heart failure, chronic kidney disease, preoperative anticoagulation, functional status, treated length, Rutherford category, preoperative ankle–brachial index (ABI), previous intervention on target lesion, and previous intervention on contralateral limb.
A total of 62,054 arterial interventions were analyzed among 47,965 unique patients. DES 8.8% (n = 5488), DCB 34.4% (n = 21,352), BMS 27.1% (n = 16,787), and POBA 25.5% (n = 15,832). The mean age was 69.2 ± 11.0 years, 41.5% female, 24.9% non-White, and 6.5% Hispanic. We observed an initial increase in DCB/DES use, which continued to rise despite the ambulatory payment expiration through the end of 2018 when the Katsanos meta-analysis was published (adjusted OR = 1.13, 95% CI 1.05–1.21) (Figure 1A). After this point, DCB and DES use decreased significantly from 50.1% of all interventions in January 2019 to 25.1% of interventions in June of 2019 (adjusted OR = 0.27, 95% CI 0.25–0.30). The individual breakdown of device usage combinations over time is presented in Figure 1B.
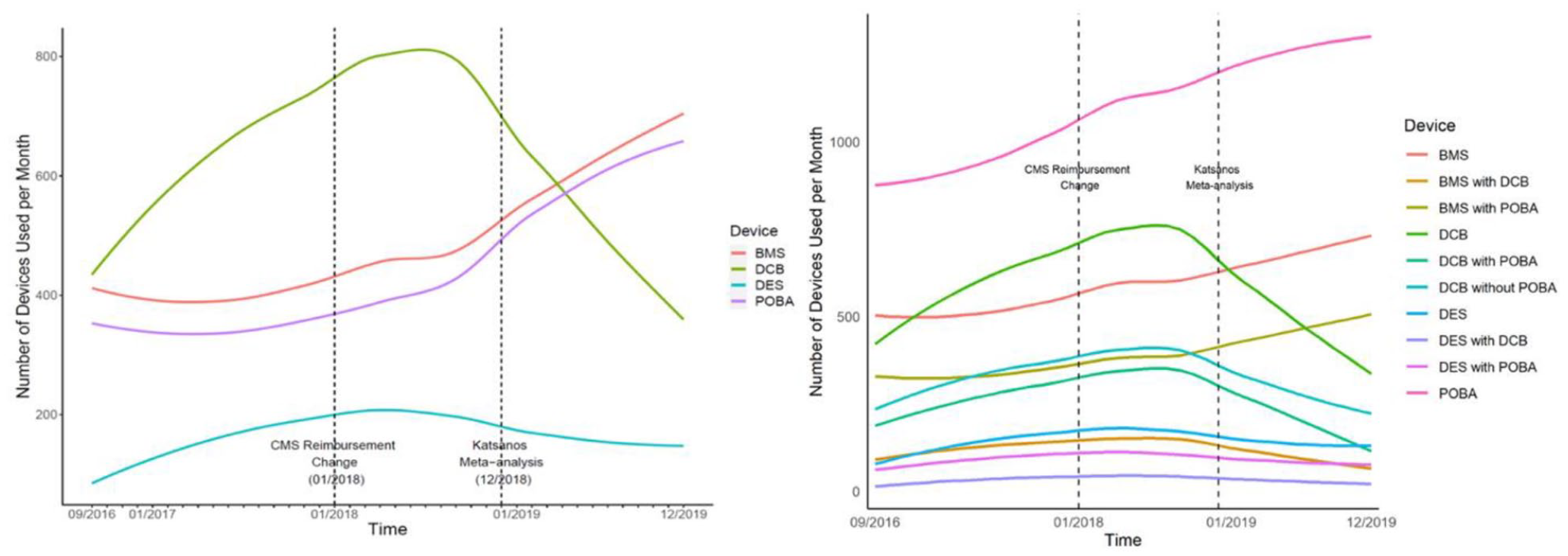
Trends of device use by month for superficial femoral and popliteal artery lesions by
DCB and DES use were not influenced by the decrease in the reimbursement rate for DCB as device use significantly increased over this period, but there was a steep decrease in use when safety concerns came to light. The Katsanos analysis safety concerns, however, have not been reproduced when outcomes were analyzed at the patient level for both DCB and DES, with recent randomized controlled trial evidence further confirming their safety. 5 With widespread safety concerns preceding sharp declines in the usage of new drug-coated device technology, there is an urgent need for robust long-term outcomes surveillance data regarding the safety and efficacy of drug-coated devices across a diversity of patient profiles. It is unclear how much these concerns are still impacting treatment practices today, as we restricted our period of observation to December 2019 as the COVID-19 pandemic created an anomaly in the way patients were receiving care, including peripheral vascular interventions. Going forward, contemporary surveillance data for PAD devices are, however, very much needed as it can help tailor the most favorable benefit–risk ratio of drug-coated devices to patient-level factors and angiographic-level factors.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Mena-Hurtado is a consultant for Abbott, Cardinal Health, Cook Medical, Medtronic, and OptumLabs. Dr Smolderen reports unrestricted grant funding from Johnson & Johnson, Cardiva Medical, Merck, and Abbott and she is a consultant for Abbott and OptumLabs. All other authors have no conflicts of interest to report.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
