Abstract
The advancement of drug-coated balloons (DCB) presents an alternative nonstent method in the percutaneous treatment of atherosclerotic lesions. While the current generation of drug-eluting stents (DES) are the device of choice, especially in coronary artery disease (CAD), DCB has potential applications in the treatment of de novo lesions, in-stent restenosis (ISR), bifurcations, and in peripheral artery disease (PAD). In terms of coronary clinical experience, DCB is used most in ISR scenarios and more data are collected to support the use of DCB in de novo lesions compared to best-in-class DES. The use of DCB in bifurcation side branch treatment has demonstrated safety, and with good angiographic and clinical outcomes, but more data from randomized trials will be required to assess its clinical value. For PAD, the clinical outcomes of DCB with and without debulking devices in diseased femoropopliteal arteries and treatment of below-the-knee (BTK) vessels with DCB are discussed. Current data demonstrated conflicting long-term safety outcomes in the use of paclitaxel devices in the femoral and/or popliteal arteries, while the role of DCB in BTK disease remains uncertain due to a lack of randomized controlled trial data. In summary, this review provides an overall view of current DCB technologies and progress, followed by an update on DCB clinical data in the treatment of CAD and PAD.
Keywords
Introduction
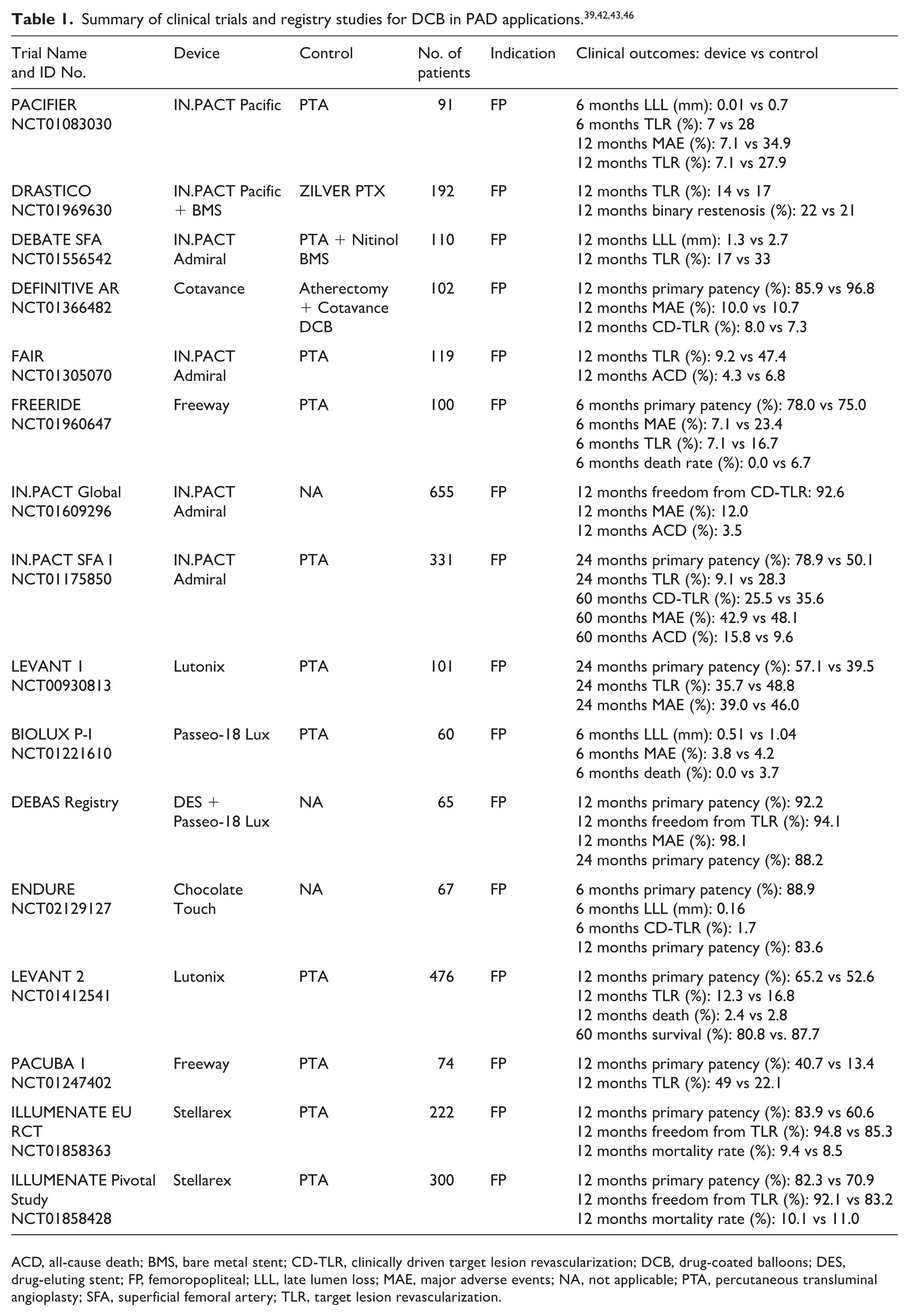
The advent of drug-eluting stents (DES) has revolutionized percutaneous coronary interventions (PCI) by reducing acute vessel closure in coronary arteries and decreasing vessel recoil in peripheral interventions, but raised concerns due to late stent thrombosis (ST), delayed healing, permanent caging, and the occurrence of in-stent restenosis (ISR). Hence, drug-coated balloons (DCB) were developed as a nonstent approach to circumvent some drawbacks of DES. The concept of DCB offers potential sustained anti-restenotic therapy without the limitations of permanent implants (Figure 1). 1 Theoretically, DCB render a more rapid and uniform drug transfer to the vessel wall. The absence of a permanent implant is expected to improve arterial healing and allows preservation and early restoration of a normal vessel function. Therefore, DCB present an alternative therapeutic option for the treatment of atherosclerotic lesions in small vessels, bifurcations, and challenging peripheral artery segments where stent implantation has proven less favorable.
The concept of drug release from DCB.
Presently, bare metal stent (BMS) and DES ISR are approved indications (with a class I, level of evidence A recommendation in the European Guidelines for Revascularization) for a DCB based strategy in coronary vessels. 2 Coronary DCB are not available in the US currently. For peripheral vessels, the Trans-Atlantic Inter-Society Consensus (TASC) II states that endovascular approach is the treatment of choice and the preferred treatment option in TASC type A and B lesions, respectively. 3 According to a 2015 international positioning document, the use of DCB is highly recommended in femoro-popliteal TASC IIA and B de novo and restenotic lesions, given that treatment is beneficial, useful, and effective based on the data derived from multiple randomized clinical trials. 4 In this review, the technological aspects, such as the choice of drug and excipient used in current and upcoming DCB platforms, will be discussed. The subsequent section will cover an overview of clinical experience with the use of DCB in coronary artery disease (CAD), which includes ISR, and de novo and bifurcated lesions. Lastly, the paper will discuss clinical data from peripheral DCB trials in peripheral artery disease (PAD), reviewing results for both femoropopliteal and below-the-knee (BTK) vessels.
The concept of drug-coated balloons (DCBs)
The therapeutic efficacy of a DCB relies extensively on the interaction between drug, excipient, and coating procedure. The interplay between these factors influences the pharmacokinetic profile, transfer efficiency, tissue retention, and subsequent clinical outcome after DCB therapy. A summary of the current and upcoming DCB can be found in online Supplemental Table 1.
Choice of anti-proliferative drug
Owing to its favorable tissue uptake by and extended retention in vessel walls, paclitaxel (PTX) has been the choice of drug used in DCB. PTX was initially coated on the first generation of coronary DES but has been replaced by limus-analogs due to cytotoxicity concerns with PTX. Clinical experience with sirolimus-eluting stents has demonstrated significantly lower restenosis and revascularization rates compared to PTX-stents in coronary vessels. 5 Between PTX-stents and everolimus-eluting stents, the latter exhibited significant reduction in composite of death or myocardial infarction (MI), ST, and target lesion revascularization (TLR). 6 This has led to the singular use of limus-analogs in the current generation of coronary DES.
Sirolimus is considered ‘safer’ but is less lipophilic and results in slower tissue absorption at the target site. However, sirolimus is relatively unstable compared to PTX and requires protection against degradation to achieve therapeutic tissue levels. Without a protective coat, sirolimus can diffuse quickly into the capillary bed and into surrounding tissues, affecting its efficacy. MagicTouch DCB (Concept Medical, FL, USA) make use of the proprietary nanoparticles with encapsulated sirolimus as a drug delivery platform (Nanolute). The company received breakthrough device designation in August 2019 from the US Food and Drug Administration (FDA) for the MagicTouch DCB for the treatment of PAD in BTK lesions. The use of nanocarriers is expected to reduce in-transit drug loss, and increase the stability and bioavailability of the drug, leading to faster uptake in tissue. The protective carriers can also enhance drug retention at the tissue site and improve therapeutic outcomes.
Choice of excipient
Ideally, a coating should be robust enough to envelope the drug uniformly and stably on the balloon surface with minimal drug loss during tracking and facilitate efficient drug transfer to the target site. The coating should have sufficient integrity without causing particulate generation and downstream embolization. Figure 2 is a summary of the different generations of DCB and their characteristics. Hydrophilic excipient, such as iopromide, was employed in the original Paccocath formulation in the first generation of DCB. Iopromide creates a high-contact molecular surface area between the drug and the vessel wall, thereby increasing the bioavailability of PTX while remaining biologically inert at the target site. 7 Subsequently, other hydrophilic excipients (e.g. urea and shellac) were investigated as DCB excipients. Hydrophilic excipients have effective drug transfer due to the polarity difference of the coating and balloon material but are often limited by a high drug wash-off rate and particulate generation. This was demonstrated in a preclinical model by Kelsch et al., whereby 25–35% of PTX was dislodged from iopromide- and urea-based DCB during tracking. 8

Comparison between different generations of DCB. The first and second generation of DCB employed paclitaxel as the anti-proliferative drug and the crystallinity of the coating played a role in the extent of particulate generation. The third generation is moving towards the use of sirolimus, which less cytotoxic.
Hydrophobic excipients result in better coating integrity and resistance to drug loss but suffer from a low drug transfer rate due to favorable affinity between the excipient and balloon material. Amphiphilic excipients, such as polysorbates, have the potential to improve drug coating integrity with minimal particulate generation, while promoting effective drug transfer and retention. Preclinical evaluation of MagicTouch’s nanocarrier/drug excipient demonstrated better inhibition of vascular smooth muscle cells in vitro and significant reduction in neointimal hyperplasia. 9
Clinical use of DCB in coronary artery disease (CAD)
Owing to the drawbacks of current platforms and inherent limitations of a DCB-only strategy for de novo coronary lesions, DCB have been used most commonly in ISR scenarios. A summary of the clinical trials and registry studies conducted for DCB in CAD settings can be found in online Supplemental Table 2.
In-stent restenosis (ISR)
Current European guidelines recommend the use of DCB in coronary ISR (class I, level of evidence A), while coronary DCB is not available and not recommended for ISR in the US. 10 The DARE-trial (multicenter randomized controlled trial [RCT], NCT01127958) compared between SeQuent Please DCB (B Braun Melsungen AG, Berlin, Germany) and Xience DES (Abbott Vascular, Chicago, IL, USA) and reported no differences in clinical endpoints, including TLR (DCB 8.8% vs DES 7.1%; p = 0.65) up to 12 months. 11 The ISAR-DESIRE-3 (randomized, open-label trial, NCT00987324) examined the use of DCB compared to PTA or DES (PTX) in 402 patients with ISR. At 3 years, the use of DCB as compared with DES has similar efficacy (risk of TLR: 0.91 to 2.33; p = 0.11) and safety with DCB superior to PTA (risk of TLR: 0.34 to 0.74; p < 0.001). 12 The recently published DAEDALUS study (meta-analysis of 10 RCTs) concluded that, at 3 years’ follow-up, the PTX-DCB arm is moderately less effective than repeat stenting with DES in terms of TLR, with no increased long-term mortality. 13 Hence, DCB was ranked as the first treatment option for ISR followed by DES for coronary ISR. 14 The use of second generation DES with thinner struts, biocompatible polymers and different limus-analog drugs may play a role in improved performance of DES, especially in small vessels. This observation was corroborated by Siontis et al. (27 trials, 5923 patients) and Kokkinidis et al. (10 studies, 2173 patients), which demonstrated that the use of second generation DES was the most effective strategy.15,16 In summary, current clinical data support the use of DCB in BMS/DES ISR as an alternative to receiving a DES stent.
De novo CAD
In de novo CAD, DES is considered the gold standard in large vessels but in the case of small vessel disease, either balloon angioplasty alone or medical therapy are considered alternatives. For trials evaluating a DCB-only approach in small coronary vessels, the conclusions were unclear as to whether this approach is beneficial compared to DES. A recent meta-analysis (19 RCTs, 5072 patients) revealed DES (first generation of sirolimus-stents followed by PTX-stents) to be the comparably more effective option than DCB based on angiographic outcomes and TLR. 17 The PEPCAD III study (multicenter randomized trial) was the only study that compared a DCB with BMS strategy against DES, reporting that the DCB with BMS treatment did not demonstrate non-inferiority compared to the DES arm. 18 Recently, Silverio et al. examined the Swedish Coronary and Angioplasty Registry (SCAAR) database (14,788 patients: 1154 with DCBs and 13,634 with n-DES) and found that the DCB arm reported a significantly higher restenosis rate compared to the newer generation DES. 19
Published data from BASKET-SMALL 2 (multicenter RCT, NCT01574534) demonstrated that DCB was non-inferior to second generation DES regarding major adverse cardiac events (MACE) up to 12 months, with similar event rates for both treatment groups in small vessels. 20 However, more clinical data, such as longer-term MACE and restenosis rates from RCTs (e.g. BASKET-SMALL 2, RESTORE SVD (NCT02946307)), will be required to establish a clear case for DCB over DES in small coronary vessels.21,22
Bifurcation lesions
Various trials and single studies have been conducted to evaluate the role of DCB in the treatment of coronary bifurcation lesions. 23 The BABILON (prospective randomized study, NCT01278186) trial compared between patients with bifurcation lesions who received main branch (MB) and side branch (SB) sequential dilation with the SeQuent Please DCB (56 patients) and patients who received DES implantation in the MB and provisional SB stenting with the T-stent technique (56 patients). The data revealed that, at 24 months, the two strategies were found to be safe, with no deaths registered, though the MACE and TLR rates were higher in the DCB group (17.3% vs 7.1%; p = 0.105, and 15.4% vs 3.6%; p = 0.045). MB restenosis was reported to be significantly higher in the DCB group (13.5% vs 1.8%; p = 0.027), but was not so in the SB restenosis (5.8% vs 3.6%, p = 0.67). It was concluded that DES performed better than DCB in medium-to-large caliber MB vessels while DCB demonstrated low and similar late lumen loss (LLL) and TLR rates to DES when used in SB. 24 On the other hand, the DEBIUT (three-arm multicenter, NCT00857441) study tested a BMS + DCB strategy for SB treatment but concluded that this approach did not show any advantage over a DES strategy, with a lower angiographic performance at 12 months and a similar rate of MACE. 25 Other studies, such as BIOLUX P-I (single-arm multicenter, NCT01221610) and DEBSIDE (nonrandomized, multicenter, NCT01485081) reported the use of DCB in SB was safe with acceptable angiographic and clinical outcomes. A review by Cortese et al. examined various studies (meta-analyses, RCT data, single-arm studies) on DCB for bifurcation lesions and concluded: 26
DES in MB should be the preferred option unless the patient is unable to receive a stent
DCB with BMS should not be considered as the first-choice option in MB
SB treatment with DCB is feasible and safe, with a good angiographic and clinical outcome.
Clinical use of DCB in peripheral artery disease (PAD)
The Society for Vascular Surgery (class I, level of evidence B) and American College of Cardiology Foundation/American Heart Association (ACCF/AHA) (class I, level of evidence A) recommended endovascular therapy for intermittent claudication in patients with significant functional or lifestyle-limiting disability when symptomatic improvement with treatment is deemed reasonable, when pharmacologic or exercise therapy, or both, have failed, and when the benefit–risk ratio is considered favorable.27,28 Owing to the unique morphology of peripheral vasculature, stent implantation in the femoropopliteal lesions did not demonstrate sustained efficacy as compared to coronary arteries. For this reason, the use of DCB has been recommended in femoropopliteal TASC IIA and B de novo and restenotic lesions. 4 The 2017 PAD European Society of Cardiology (ESC) guidelines state the consideration of DCB in short lesions (< 25 cm) with a class IIb recommendation for patients with intermittent claudication and severe chronic limb ischemia (CLI). 29 Early clinical experience with stenting in femoropopliteal segments demonstrated an increased risk of stent fracture because of the physiological torsion of the artery, with subsequent vessel wall damage and restenosis. 30 Preclinical studies have shown that metallic implants permanently overstretch the arterial wall, leading to sustained inflammation and persistent neointimal growth, which is likely responsible for the exaggerated ‘catch-up’ phenomenon observed in this vascular bed. 31 Hence, without a clear benefit of default stenting over BA, considerable interest exists in the use of DCB in PAD. Table 1 displays a summary of clinical trials and registry studies investigating DCB for the management of PAD.
ACD, all-cause death; BMS, bare metal stent; CD-TLR, clinically driven target lesion revascularization; DCB, drug-coated balloons; DES, drug-eluting stent; FP, femoropopliteal; LLL, late lumen loss; MAE, major adverse events; NA, not applicable; PTA, percutaneous transluminal angioplasty; SFA, superficial femoral artery; TLR, target lesion revascularization.
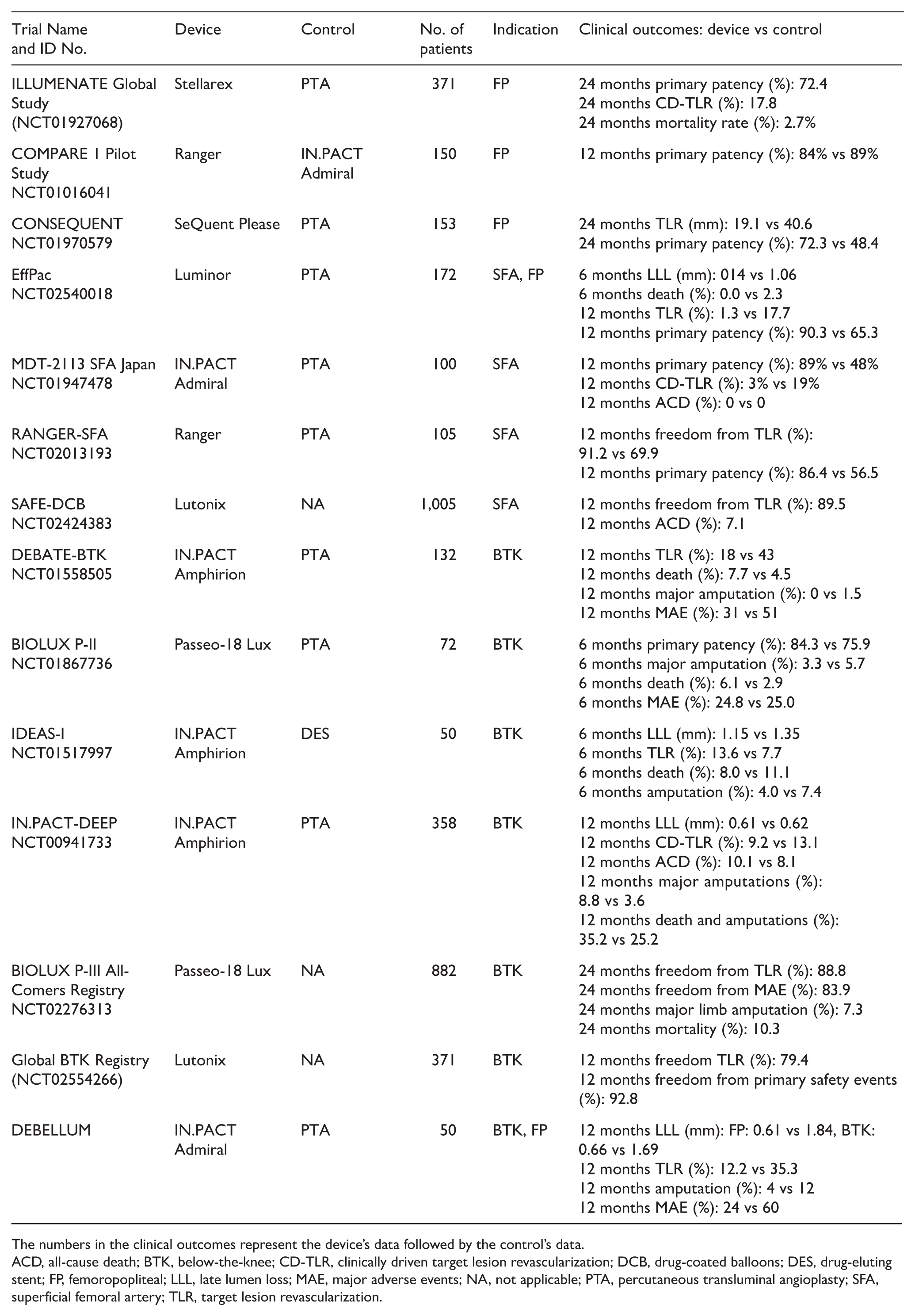
The numbers in the clinical outcomes represent the device’s data followed by the control’s data.
ACD, all-cause death; BTK, below-the-knee; CD-TLR, clinically driven target lesion revascularization; DCB, drug-coated balloons; DES, drug-eluting stent; FP, femoropopliteal; LLL, late lumen loss; MAE, major adverse events; NA, not applicable; PTA, percutaneous transluminal angioplasty; SFA, superficial femoral artery; TLR, target lesion revascularization.
Femoropopliteal arterial disease
Endovascular therapy is challenging in femoropopliteal arteries due to the high restenosis rates (20–40%) after stenting. 32 Current Society for Cardiovascular Angiography and Interventions (SCAI) guidelines recommend DCB as a standard treatment option for femoropopliteal artery disease. 33 In 2008, both the THUNDER (randomized multicenter study, NCT00156624) and FemPac (randomized study) trials demonstrated significant reduction in LLL with DCB compared to BA. 34 The meta-analysis by Cassese et al. (four RCTs, 381 patients) concluded that DCB therapy was proven to be effective both in terms of angiographic restenosis and TLR. 35 A meta-analysis (eight RCTs, 1341 patients) published recently reported that DCB strategy reduced the risk of TLR, with similar mortality at 12 months, and provided a sustained reduction in TLR without safety issues for up to 2 years compared to BA. There was a significant heterogeneity with respect to treatment effect for TLR due to the LEVANT 1 and 2 studies (multicenter RCTs, NCT00930813, NCT01412541), suggesting a differential efficacy profile associated with the DCB platform used in these trials. 36 Indeed, the DCB investigated in the LEVANT studies was the Lutonix DCB (Bard PV, Tempe, AZ, USA), which was considered to have a lower efficacy compared to other DCB. The pharmacokinetic profile of Lutonix was found to have a lower PTX tissue bioavailability compared to IN.PACT (Medtronic, Minneapolis MN, USA) and Stellarex DCB (Philips, Amsterdam, The Netherlands) after 28 days. 37 This might have attributed to lower primary patency and freedom from TLR at 1-year follow-up in the LEVANT 2 study published in 2015.38,39 More recently, the COMPARE multicenter RCT (NCT02701543) examined two DCB and showed that the Ranger DCB (Boston Scientific, Marlborough, MA, USA) was comparable with the IN.PACT DCB for the treatment of symptomatic femoropopliteal lesions. 40 Other studies, such as ILLUMENATE (multicenter RCT, NCT01858428) and CONSEQUENT (post market clinical follow-up, NCT02460042) demonstrated superior safety and effectiveness in the DCB-treated group compared to the BA-only group at longer-term follow-up (> 2 years).41,42 Other meta-analyses on the treatment of femoropopliteal arteries are consistent in their findings, demonstrating a significant reduction in TLR with the use of DCB strategy over BMS and BA groups.36,43
For femoropopliteal arteries ISR, the optimal revascularization therapy represents a matter of debate and is inconclusive due to lack of statistical power to thoroughly investigate clinical outcomes in randomized trials of DCB versus plain balloon angioplasty. Patient-level data from three randomized trials of DCB angioplasty for ISR of femoropopliteal arteries published in 2018 (263 patients, 12 months’ follow-up) showed a lower risk for TLR in the DCB arm compared to the PTA group (hazard ratio (HR) 0.25; 95% CI: 0.14–0.46; p < 0.001) and recurrent ISR (HR 0.19; 95% CI: 0.10–0.35; p < 0.001). 44 However, the DEBATE-ISR trial demonstrated that although recurrent TLR at 1 year was significantly lower in the DCB group (19.5% vs 71.8%) compared to PTA, the 3-year TLR was similar (40% vs 43%) between the two groups. Dedicated studies are required to confirm these results at long-term follow-up. 45
The use of debulking devices in association with DCB provides another treatment option for patients with restenotic or heavily calcified lesions (up to 15% of the patients). This concept combines the benefits of removing exuberant tissue or calcified plaques with atherectomy or laser with the suppression of neointimal proliferation through DCB. 46 The DEFINITIVE AR study (NCT01366482) is a prospective, multicenter pilot study involving 121 patients and compared the DCB-only therapy with atherectomy and DCB therapy in patients with calcified superficial femoral artery (SFA) lesions. Angiographic patency was 84.6% in the atherectomy and DCB arm compared to 81.6% in the DCB-only group, suggesting the potential benefit of performing debulking atherectomy before the use of DCB. 47 Data published in 2017 from various studies have also demonstrated encouraging mid-term clinical results from the use of atherectomy with DCB compared to DCB alone. 48 The study by Gandini et al. (single-center, randomized study, 448 CLI patients) concluded that combined treatment of laser debulking with DCB resulted in better outcomes (patency and TLR rates) in patients with CLI and SFA in-stent occlusion. 49
In summary, current clinical data are consistent in demonstrating significant reduction in TLR with the use of DCB strategy over BMS and BA groups, but the treatment of ISR with conventional methods remains difficult and has no clear frontline strategy. The use of debulking devices (e.g. atherectomy and laser debulking) with DCB have shown improved outcomes compared to using DCB alone. It will be important to continue to monitor the long-term safety outcomes in patients treated with PTX devices.
Atherosclerotic disease of below-the-knee (BTK) arteries
In BTK lesions, coronary DES have been found superior to BA and BMS, though the diffuse nature of atherosclerotic disease in this long vascular segment precludes the routine use of many coronary DES. The clinical performance of DCB in this area has been less conclusive. 50 Earlier experience from the single-center observational study by Schmidt et al. and randomized DEBATE-BTK trial (NCT01558505) reported superior mid-term results of using DCB compared to BA.51,52 However, results from the IN.PACT-DEEP multicenter RCT (NCT00941733) released in 2015 did not demonstrate a superiority of the IN.PACT Amphirion DCB compared to BA in terms of TLR and LLL. The IN.PACT-DEEP trial also had a higher incidence of amputation (8.8 vs 3.6%; p = 0.08) and a trend towards higher mortality in the DCB arm, leading to the withdrawal of the device from the market. 53 A 2016 meta-analysis (five trials, 641 patients) reported that DCB in BTK lesions has similar clinical efficacy and superior angiographic performance when compared with BA or DES at 1-year follow-up. The analysis showed that treatment with DCB led to relatively lower LLL, without improvement in clinical outcomes such as amputation and wound healing. The study concluded that a dedicated wound care management should be implemented for all patients with advanced-stage atherosclerotic BTK disease in order to evaluate the net clinical benefit of different revascularization strategies. 54 Data presented on the use of Lutonix DCB versus PTA in BTK lesions in a prospective, global, multicenter, single-blind RCT demonstrated no significant difference in freedom from mortality (DCB: 81% and PTA: 81%, p = 0.946), freedom from major amputation (DCB: 95.5% and PTA: 93.8%, p = 0.268) or amputation-free survival (DCB: 77.8% and PTA: 77.8%, p = 0.495). The study concluded that the Lutonix DCB versus PTA provided statistically significant efficacy outcomes at 6 months with no observed safety issues out to 3 years (presented at the Leipzig Interventional Course (LINC), January 2020 55 ). Currently, the FDA have not given market approval to the Lutonix DCB for treatment of PAD in BTK vessels. More prospective studies are warranted to clearly show the merits of DCB-based strategies in BTK lesions.
Controversy surrounding use of paclitaxel (PTX) devices
In December 2018, a meta-analysis by Katsanos et al. (28 RCTs, 4663 patients) concluded an increased risk of death (among 975 subjects from three trials, 20.1% crude risk of death at 5 years with DCB/DES treatment vs 13.4% with PTA/BMS treatment) following application of PTX balloons and stents in the femoropopliteal artery of the lower limbs, which urged further investigations. 56 This has sparked much discussion and debate over the safety of PTX devices.
The FDA conducted an internal preliminary study with their analysis of patient-level data consisting of the four pivotal RCTs (ZILVER PTX RCT, LEVANT 2, IN.PACT SFA I and II studies, and ILLUMENATE RCT) of PTX devices that supported premarket approval (PMA). In the executive summary published in June 2019, the analysis revealed an increased mortality rate in the PTX device arm at years 3 through 5 for ZILVER PTX RCT (NCT00120406), LEVANT 2 (NCT01412541), and IN.PACT SFA I and II (NCT01175850, NCT01566461) studies. 57 Since then, the FDA advised healthcare providers to consider meanwhile alternatives with patients, except in circumstances such as high restenosis risk. 58 According to the FDA’s r\evalidation of the original trial data, the 5-year mortality risk of PTX devices was 20.1% vs 13.4% in non-PTX device-treated patients (n = 975). It should be noted that the FDA did caution interpretation of these data due to limitations such as the small number of long-term cohorts.
Conflicting conclusions arose from subsequent analyses of patient-level data of certain PTX-DCB clinical trials which did not report increased mortality in the DCB arm. Recently published data examined patients who received in-hospital balloon- or stent-assisted endovascular revascularization of the iliac and lower limb arteries reimbursed under German BARMER health insurance. The 64,771 patients underwent 107,112 procedures using 23,137 PTX devices (DES or DCB). Their analysis revealed that PTX-DES was not associated with increased long-term mortality for over 11 years past application (all p > 0.057). DCB was associated with decreased long-term mortality for the first year past application (HR 0.92; p < 0.001), with insignificant correlation in the years thereafter (all p > 0.202). 59
Similarly, 3 years’ follow-up from the ILLUMENATE clinical program, which assessed mortality rates from two RCTs (n = 419 with PTX-DCB; n = 170 with BA), reported that the all-cause mortality rates were comparable between both arms; PTX exposure was unrelated to mortality (HR 1.04; 95% CI: 0.98–1.10; p = 0.23). 41 The IN.PACT Global Clinical Study (single-arm study with real-world patient population, NCT01609296) evaluated the safety and effectiveness of a DCB (IN.PACT Admiral; Medtronic) in the treatment of patients with atherosclerotic femoropopliteal disease, including de novo ISR in the SFA or the entire length of the popliteal artery. The investigators reported a high patency and a low clinically driven TLR at 12 months, confirming the safety and effectiveness of DCB in complex femoropopliteal lesions. 60 The recently published data in 2018 from the IN.PACT Global Clinical Study confirmed the positive findings for up to 2 years. 61 Patient-level data from the Lutonix clinical program (LEVANT 1, LEVANT 2, LEVANT Japan) demonstrated no significant differences in mortality rates between the PTX-DCB and BA arms. The 5-year HR was 1.01 (95% CI: 0.68–1.52) in the aggregated LEVANT trials. The investigators concluded that the lack of dose–response relationships or clustering of causes of death argues against a causal relationship between PTX and mortality. 62 The Society for Vascular Surgery Vascular Quality Initiative has also reported similar mortality between the PTX and non-PTX device groups (8376 patients in total) in the treatment of femoral-popliteal occlusive disease at 1-year follow-up. 63
These data shed light on the possible limitation of Katsanos’ analysis 56 , such as the studies included were not designed or powered for long-term analysis of mortality and the high rate of patients lost to follow-up, and lack of access to patient data.
Conclusion
The current generation of coronary DES with thin struts and superior performance remains the device of choice in the treatment of de novo lesions, but emerging data (BASKET-SMALL 2, RESTORE SVD) show that DCB may have a role for small vessel indications. The development of DCB with improved safety profile and clinical performance has led to favorable outcomes in the treatment of coronary ISR. For coronary ISR, DCB clinical development is centered on comparing long-term results with the current generation of DES, as well as improving vessel preparation before DCB treatment. Finally, use of DCB in side branch treatment of bifurcation lesions also has been shown to be feasible, with satisfactory angiographic and clinical outcomes.
Initial clinical experience with the use of DCB in femoropopliteal disease showed benefits of the treatment in terms of efficacy but a recent analysis has shed some uncertainty and questions over the long-term safety profile of PTX devices in peripheral vessels. The correlation between the use of PTX-DCB and increased long-term mortality signal in peripheral vessels is not well understood and the underlying pathomechanisms remain elusive. 64 Lastly, opposite effects have been found in a systematic meta-analysis of coronary PTX-DCB RCTs, showing that PTX-DCB resulted in a significantly lower mortality and cardiac mortality compared to control at 3 years.
Research and development work in newer generations of DCB are focused on improving the robustness of the drug coating to decrease drug loss during tracking, facilitate efficient transfer to the vessel wall, and improve uptake of the drug without causing embolization. Limus-analogs, such as sirolimus (which will require novel carriers), may help to mitigate any cytotoxicity of paclitaxel. This newer generation of DCB can potentially overcome the shortcomings of current DCB, but more clinical studies are needed to ascertain the relative efficacy and safety of these novel technologies compared to current DCB and DES.
Supplemental Material
Supplemental_Material_final – Supplemental material for Drug-coated balloons: Technical and clinical progress
Supplemental material, Supplemental_Material_final for Drug-coated balloons: Technical and clinical progress by Huiying Ang, Tobias R Koppara, Salvatore Cassese, Jaryl Ng, Michael Joner and Nicolas Foin in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr M Joner has served as a consultant for OrbusNeich and Biotronik. Dr N Foin holds an appointment with Philips. All other authors have reported that they have no relationships relevant to the contents of this paper to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.