Abstract

Keywords
Patients with chronic kidney disease (CKD) and peripheral artery disease (PAD) undergoing peripheral vascular interventions (PVI) are more likely to have major adverse events, restenosis, lower technical success rates, and higher rates of limb loss and death compared with patients with normal renal function.1,2 Drug-coated balloons (DCBs) and drug-eluting stents (DES) have emerged as newer and popular endovascular therapies with multiple randomized controlled trials demonstrating their superior patency rates compared with plain balloon angioplasty (POBA) and bare metal stents (BMS) in the treatment of femoropopliteal artery stenosis.3–9 However, patients with severe CKD (CKD stages 4 and 5) have often been excluded from PAD safety and efficacy trials of drug-coated devices. It is unknown to what degree patients with severe CKD are being offered these novel devices in real-world practice. We aimed to (1) examine the real-world use of DCB/DES among patients with and without severe CKD and (2) evaluate variability of use across operators. Understanding the real-world use of these devices in patients with severe CKD can highlight the need for future long-term surveillance data given their underrepresentation in trials.
We included all patients undergoing PVI for treatment of lesions in the femoropopliteal segment between September 2016 and December 2019 from the PVI module of the Vascular Quality Initiative (VQI) registry. The Institutional Review Board at Yale School of Medicine approved this study and waived the need for informed consent because all data were deidentified. Patients with acute limb ischemia, with no intervention performed, and those with missing creatinine information, or missing unique identifiers for centers or providers were excluded. The exposure variable of interest was severe CKD, defined as patients with CKD stage 4 or stage 5, and the comparison group were patients without severe CKD. Patients were assigned to standard CKD stages based on glomerular filtration rate (GFR) values. 10 As for treatment type, four possible primary device options were identified from each procedure: POBA, BMS placement, DCB angioplasty or DES placement. In the VQI database, the order in which devices are used in a procedure is not available. Therefore, we identified the primary treatment for each procedure based on a hierarchy of DES > DCB > BMS > POBA, as performed in prior studies. 11 Comparison groups for primary device type were defined as DCB/DES versus POBA/BMS.
We compared patient demographics, comorbidities, and lesion characteristics according to CKD group. Summary statistics were reported as frequencies for categorical variables, means with standard deviations for continuous variables, or medians and interquartile ranges for nonnormally distributed data. We then compared the percentage of interventions where DCB/DES were used as primary treatment in patients with severe CKD versus those without severe CKD. Differences between the two groups were evaluated with Cohen’s d—standardized difference for continuous variables and standardized differences for proportions for categorical and dichotomous variables, with values of d ⩽ 0.20 representing a small effect size. 12
To evaluate the variability in use of DCB/DES in patients with severe CKD by operators, we first calculated the unadjusted and adjusted median odds ratio (MOR), 13 with covariates including age, sex, race, ethnicity, primary insurance, functional status, hypertension, diabetes, coronary artery disease, congestive heart failure, preoperative anticoagulation, Trans-Atlantic Inter-Society Consensus Document (TASC) classification, Rutherford category, previous intervention on target lesion, time period after the Centers for Medicare & Medicaid Services (CMS) reimbursement change (01/2018 vs before) and after the Katsanos meta-analysis publication 14 (12/2018 vs before), and a random effect for operator and site. Significance was considered at p < 0.05. Rates of missing data were low (1.5%) for all variables except the primary device used (4.2%) and history of contralateral PVI (16%), and analyses were performed as complete cases. Analyses were performed using R statistical software, version 3.6.1 (R Project for Statistical Computing, Vienna, Austria).
After exclusions, a total of 57,753 femoropopliteal artery endovascular interventions were included. A total of 7527 patients (13.0%) had severe CKD. The mean age of the overall cohort was 69.2 ± 11 years, with 41.5% women, 75.3% White, and 6.6% Hispanic. Compared with patients without severe CKD, patients with severe CKD were slightly younger, less likely to have Medicaid/Medicare insurance, more likely to be nonsmokers, and had significantly higher rates of cardiovascular comorbidities. Patients with severe CKD were more likely to present with chronic limb-threatening ischemia (CLTI), to have a higher ankle–brachial index (ABI), and less likely to have a previously treated lesion. The rates of noncompressible ABI (> 1.3) in the study cohorts were 16.7% in the severe CKD group and 4.4% in those without severe CKD (p < 0.001, SD: 0.5). Lesion treatment lengths were similar across groups (online Supplemental Table 1).
Among patients with severe CKD undergoing femoropopliteal intervention, over a third (40.6%) were treated with DCB/DES; and in patients without severe CKD, nearly half (46.3%) were treated with DCB/DES (p < 0.001). When looking at specific devices, DCBs were more commonly used than DES and BMS in both CKD patient groups, while POBA was more often the primary treatment in patients with severe CKD. Overall, DES were used in lower rates than BMS, with the lowest rates in patients with severe CKD (online Supplemental Table 1).
In the overall population, we observed a wide variation in use of DCB/DES across operators (unadjusted physician MOR: 2.72, 95% CI: 2.57–2.88; unadjusted site MOR: 2.88, 95% CI: 2.50–3.27). When adjusted for patient and lesion characteristics, we found significant variation in DCB/DES use in patients with severe CKD across operators (adjusted MOR: 2.38, 95% CI: 2.15–2.60) and without severe CKD (adjusted MOR: 2.66, 95% CI: 2.50–2.81), implying the odds of treatment with DCB/DES for statistically identical CKD patients is two times higher when treated by one random operator compared to another random operator (Figure 1). This result also indicates that a significant portion of the variation in use of DCB/DES is explained by operator choice rather than patient or lesion characteristics.
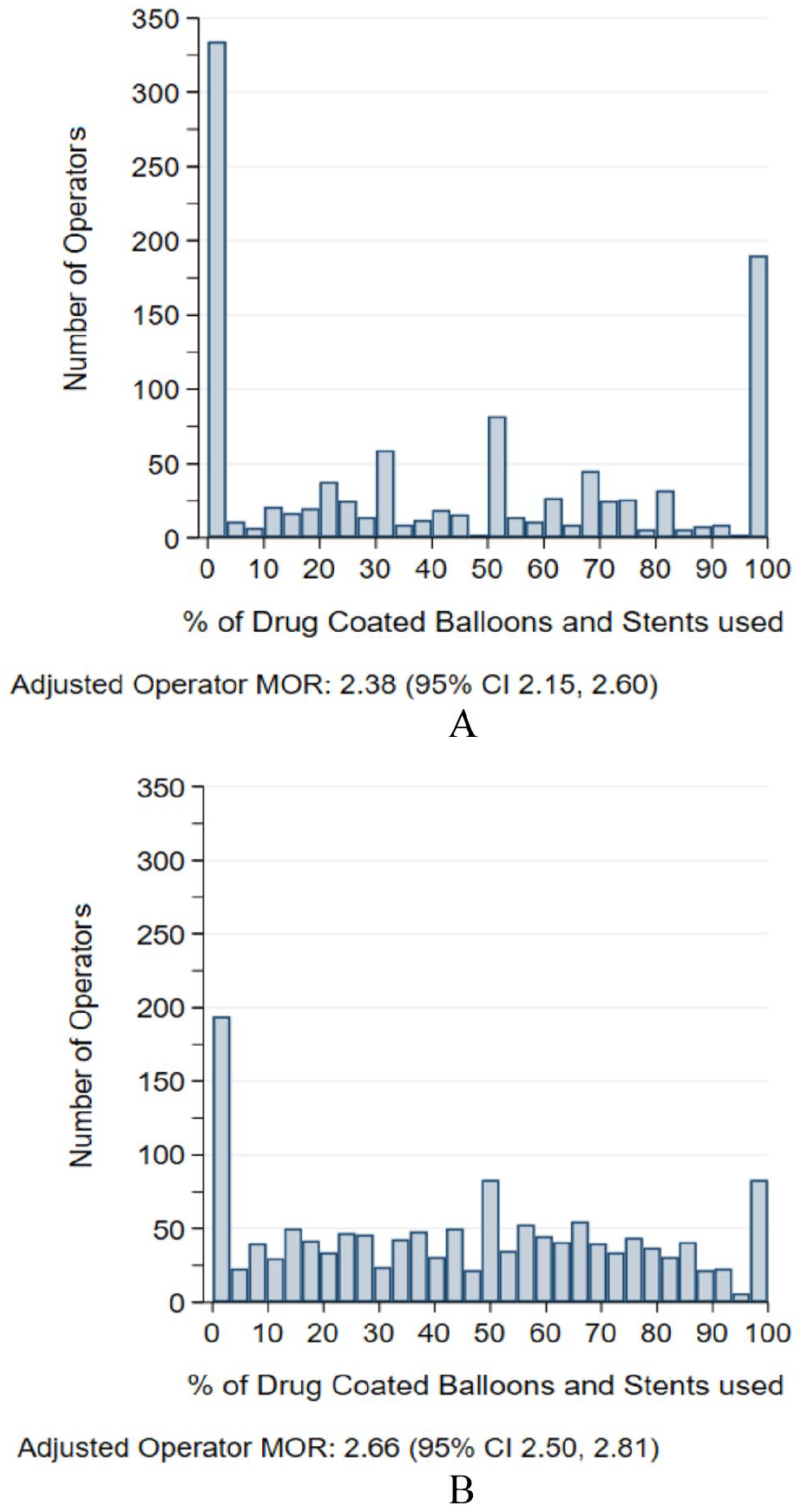
Adjusted operator variability in use of DCB/DES in femoropopliteal interventions in patients with
Using VQI registry data, this study found that among patients with severe CKD, over a third were treated with DCB/DES, despite lack of evidence showing safety or effectiveness in this population. This study showed that patients with severe CKD undergoing femoropopliteal interventions had higher rates of cardiovascular comorbidities and were more likely to present with CLTI than patients without severe CKD. We also observed a large variation in use of DCB/DES in both CKD groups, independent of patient or lesion characteristics, indicating that DCB/DES use patterns are highly driven by center and operator preferences.
To our knowledge, this is the first study to explore the use of DCB/DES in patients with severe CKD, a subgroup with more advanced PAD at presentation, lower rates of technical success, and decreased rates of limb salvage. Multiple randomized controlled trials have demonstrated the superior patency rates of drug-eluting technologies compared to POBA/BMS.3,4,7,8 However, limited data exist on patients with CKD. 15 Previous studies have shown a rapid adoption of DCB/DES to treat femoropopliteal arteries in all-comers, with nearly one-half of arteries receiving a drug-containing therapy in modern US practice. 11 In this study, we found that DCB/DES have also been increasingly adopted in the severe CKD population, despite their exclusion from pivotal clinical trials. Of note, the population from the VQI registry is predominantly White and male, which could limit the generalization of the study findings to all populations affected by PAD. Nonetheless, VQI is the largest dedicated vascular registry in the US, with over 800 contributing centers nationwide, encompassing a wide variety of institutions and practitioners. 16
We found significant variability in DCB/DES use across operators and centers. Previous studies reported a high degree of regional variability in use of DCB/DES for all patients undergoing femoropopliteal interventions, attributed to different case mix and different specialty operator mix across regions. 11 In this study, even after adjusting for patient and lesion characteristics, a wide variability in use of DCB/DES remained across operators. This implies that use of these new technologies is heavily influenced by preferences of providers and sites. Certainly, this variability could be related to operators from different specialties with distinctive treatment preferences, differential availability of devices per site, as well as the influence of cost, reimbursement, and marketing. Given the widespread and variable use of these devices despite the lack of safety and efficacy data in patients with severe CKD, long-term surveillance studies are needed in this at-risk group.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X221148521 – Supplemental material for Use of drug-coated balloons and stents in patients with femoropopliteal artery disease and severe chronic kidney disease
Supplemental material, sj-docx-1-vmj-10.1177_1358863X221148521 for Use of drug-coated balloons and stents in patients with femoropopliteal artery disease and severe chronic kidney disease by Yulanka Castro-Dominguez, Kim Smolderen, Gaelle Romain, Jeffrey Turner and Carlos Mena-Hurtado in Vascular Medicine
Footnotes
Declaration of conflicting interests
Dr Castro-Dominguez reports speaker honoraria from Medtronic and Boston Scientific. Dr Mena-Hurtado is a consultant for Abbott, Cardinal health, COOK & Optum Labs. Dr Smolderen reports unrestricted grant funding from Johnson&Johnson, Cardiva, Abbott, Merck and she is a consultant for Optum Labs, Abbott, and Cook Medical.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
