Abstract

Although surgical endarterectomy is the first-line therapy for common femoral artery (CFA) lesions in patients with peripheral artery disease (PAD), 1 endovascular therapy (EVT) has been conditionally chosen in clinical practice. However, the clinical outcomes of drug-coated balloon (DCB) versus noncoated balloon (NCB) treatment of CFA lesions have not been systematically studied. Therefore, we conducted the CELERY (Common fEmoraL artERY intervention) study.
A total of 224 consecutive patients with symptomatic PAD underwent primarily EVT for CFA lesions between April 2016 and June 2019 at four cardiovascular centers in Japan. After excluding the patients treated with stents (18 patients), the remaining 206 patients were analyzed. The current study was performed in accordance with the Declaration of Helsinki and was approved by the ethics committee of Kansai Rosai Hospital (reference number 20D031g).
The primary outcome measure was the 1-year primary patency rate. A loss of primary patency was defined as a peak systolic velocity ratio > 2.4 according to duplex ultrasound, or the recurrence of stenosis ⩾ 50% of the arterial diameter, as determined by angiography. In the patient who underwent common femoral surgical endarterectomy for restenotic lesions after failed DCB angioplasty, we conducted histological evaluation of the removed plaque to assess the vascular response of DCB for neointimal inhibition.
Data are presented as the mean ± SD for continuous variables and as percentages for discrete variables. Cumulative primary patency rate was estimated using the Kaplan–Meier method. Independent associations were explored using a multivariate Cox proportional hazards model. Statistical significance was considered at p < 0.05. Statistical analyses were performed using SPSS software version 24.0 J (IBM Corp., Armonk, NY, USA).
Baseline characteristics are summarized in the online Supplemental table. The 1-year primary patency rate was 57.5% (48.1% to 66.9%) in the DCB group and 63.3% (59.4% to 67.2%) in the NCB group, with no significant intergroup difference (p = 0.59; Figure 1A). In Figures 1B and 1C, a representative case subsequently undergoing surgical endarterectomy after failed DCB angioplasty shows that remarkable neointimal proliferation (red asterisk) on the luminal side of a calcified plaque (black arrow) with a crack (red arrow) was histopathologically evaluated. Multivariate analysis revealed that bilateral calcification, presence of a calcified nodule, and the Medina 1-1-1 bifurcation lesions were significantly associated with loss of primary patency; the adjusted hazard ratios [95% CIs] were 1.77 [1.05–2.98] (p = 0.032), 2.99 [1.56–5.70] (p = 0.001), and 1.94 [1.13–3.34] (p = 0.016), respectively.
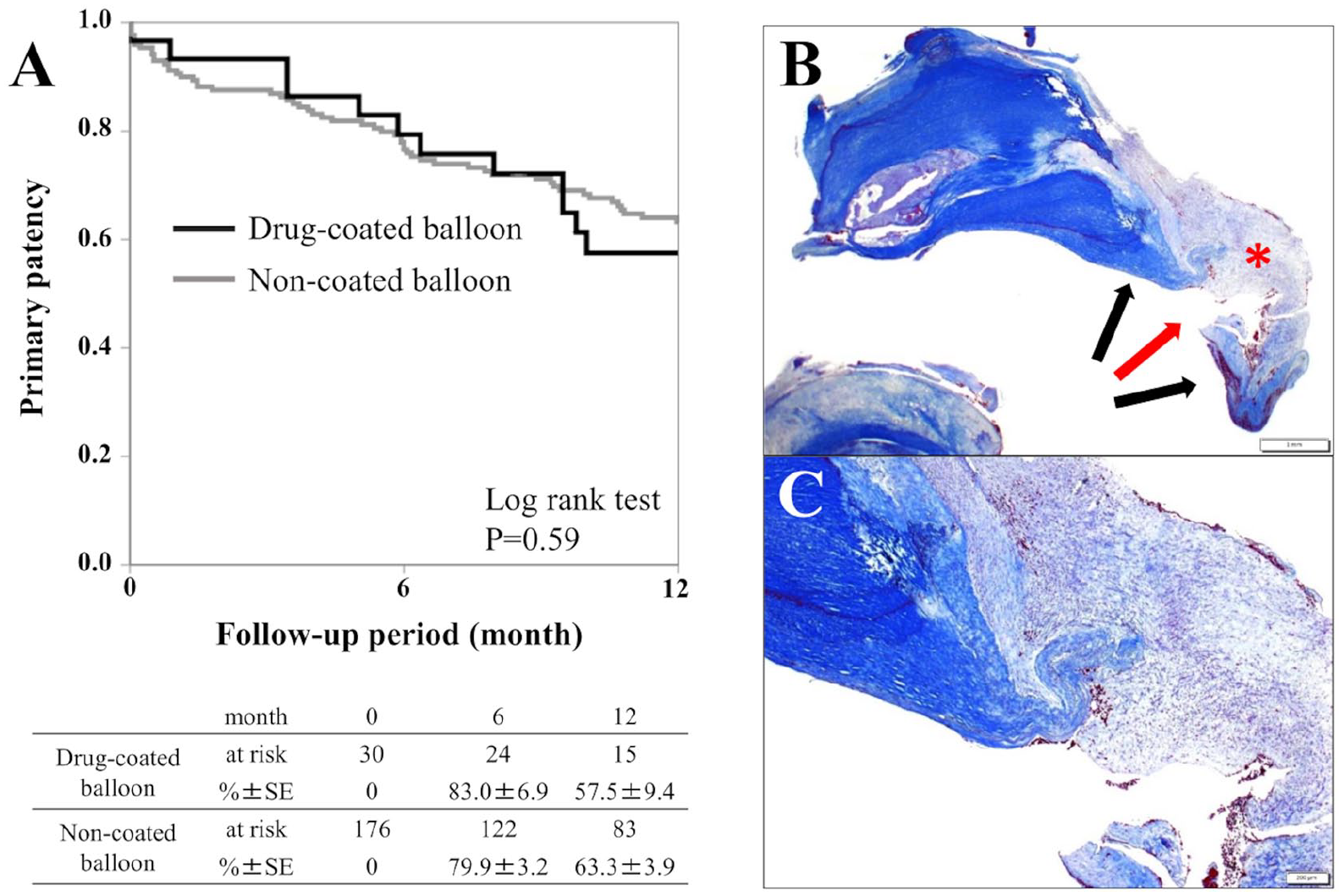
The 1-year primary patency rate after endovascular therapy for CFA lesions, and a representative case with histological evaluation of calcified plaque in surgical endarterectomy for restenotic lesions after DCB angioplasty. (A) The 1-year primary patency rate (95% CI) was 57.5% (48.1% to 66.9%) in the DCB group and 63.3% (59.4% to 67.2%) in the NCB group, with no significant intergroup difference (p = 0.59). (B) Histological evaluation revealed neointimal proliferation (red asterisk) on the luminal side of a calcified plaque (black arrows) with a crack (red arrow). (C) This is a magnified view of Figure 1B.
The current study revealed that 1-year primary patency was not significantly different between DCB and NCB in real-world CFA practice. Although the results of the TECCO (Endovascular Versus Open Repair of the Common Femoral Artery) trial, a sole randomized trial comparing surgical endarterectomy with stenting for CFA, reported comparable primary patency rates in both treatments, 2 a previous study revealed DCB angioplasty showed a significantly lower primary patency rate compared with endarterectomy (75.0% vs 96.7% at 1 year, respectively). 3
Potential reasons for the ineffectiveness of DCB for CFA lesions may include (1) early recoil of the lesions due to insufficient expansion and (2) suboptimal paclitaxel uptake by the vessel wall due to calcified lesions. Regarding insufficient expansion, 58% of cases in this study were accompanied by bilateral calcification. Restenosis would be attributed to vessel recoil due to bilateral calcification after balloon dilatation. Furthermore, Fanelli et al. reported that the efficacy of DCB in femoropopliteal lesions was attenuated in lesions with full circumferential calcification due to poor uptake of paclitaxel. 4 The concomitant use of debulking or plaque modification, which is not available in Japan, followed by DCB may improve patency.
The current study has several limitations. First, this was a retrospective analysis and conducted with the limited devices available in Japan. For instance, advanced vessel preparation techniques, including atherectomy and intravascular lithotripsy, were not available for treatment of the CFA in Japan during the study period. Second, the number of patients in the DCB group was relatively small. Finally, balloon sizing in most cases was determined by angiography. The utilization of intravascular imaging to determine the appropriate balloon sizing would improve the outcome of DCB.
In conclusion, both 1-year primary patency after balloon-based treatment with or without an antirestenotic drug for CFA was suboptimal without the availability of advanced strategies for vessel preparation. Although stenting might appear reasonable and an attractive alternative to therapy, surgical endarterectomy should remain the preferred therapy for CFA disease.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X221080485 – Supplemental material for Similar one-year primary patency rates of common femoral artery angioplasty alone when performed utilizing drug-coated versus noncoated balloons for the treatment of peripheral artery disease
Supplemental material, sj-docx-1-vmj-10.1177_1358863X221080485 for Similar one-year primary patency rates of common femoral artery angioplasty alone when performed utilizing drug-coated versus noncoated balloons for the treatment of peripheral artery disease by Yosuke Hata, Osamu Iida, Tomonori Katsuki, Yoshimitsu Soga, Masashi Fukunaga, Daizo Kawasaki, Masahiko Fujihara, Mitsuyoshi Takahara, Katsumi Inoue and Toshiaki Mano in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
