Abstract
Introduction
Intermittent claudication (IC) is a common manifestation of peripheral artery disease (PAD) and is a significant detriment to quality of life (QoL) of affected individuals. In addition to arterial obstruction leading to demand-ischemia in the muscles of the lower extremity, endothelial dysfunction, inflammation, and changes in skeletal muscle metabolism also likely contribute to the limb symptoms in IC. 1 Recent studies have also suggested a role for arterial thrombosis in the progression of PAD. 2 According to the recent American Heart Association/American College of Cardiology guidelines, patients with IC should be managed with single antiplatelet therapy, either aspirin or clopidogrel, but mainly for the reduction in risk of major cardiovascular events such as cardiovascular death, nonfatal myocardial infarction (MI), or stroke.3,4 However, the direct benefit of antiplatelet therapy such as aspirin or clopidogrel on walking performance is variable.4,5
The antithrombotic agent vorapaxar, however, represented another therapeutic option in patients with IC and PAD. As an inhibitor of protease-activated receptor-1 (PAR1), vorapaxar primarily blocks thrombin-mediated platelet activation. 6 However, PAR1 is also expressed on other vascular wall cells, such as smooth muscle cells (SMCs), macrophages, and endothelial cells in addition to platelets. Physiologic studies in patients with IC have demonstrated that calf muscle pain is not only a manifestation of inadequate muscle perfusion, but also ischemia-reperfusion injury that generates inflammatory reactive oxygen species. 7 The pleotropic effects secondary to PAR1 inhibition in vascular wall cells may improve leg muscle pain associated with claudication by inhibiting pathways involved in inflammation, endothelial dysfunction, and even angiogenesis, thus potentially targeting the other etiologies for muscle pain besides transient exercise-induced hypoperfusion.1,6,8 Moreover, clinical studies suggested improved limb outcomes with vorapaxar in patients with PAD.9,10 These findings go hand-in-hand with evidence supporting the role of thrombosis, rather than progressive atherosclerosis, in the arterial occlusions that are associated with PAD. 2
We therefore conducted a randomized placebo-controlled trial to test the hypothesis that the addition of vorapaxar to daily background antiplatelet therapy in patients with established PAD and IC would lead to an improvement in symptom status.
Methods
The Excellence in Peripheral Artery Disease Thrombin Receptor Antagonist Intervention in Claudication Evaluation (XLPAD-TRACE) trial was a phase 4 multicenter, randomized, double-blind, placebo-controlled study of patients with IC (Figure 1). A study executive committee with seven voting members was responsible for the leadership and oversight of the study. An independent Data and Safety Monitoring Board (DSMB) was established to assess the safety data, critical efficacy endpoints, and provide recommendations to the study executive committee. A clinical events committee (CEC) served as a group of independent experts responsible for the adjudication of prespecified events as they occurred during the study. The data coordinating center was at the Dallas VA Medical Center. A total of 12 institutions participated in the study. The study protocol was reviewed and approved by all the relevant Institutional Review Boards at the participating institutions. The study was registered at
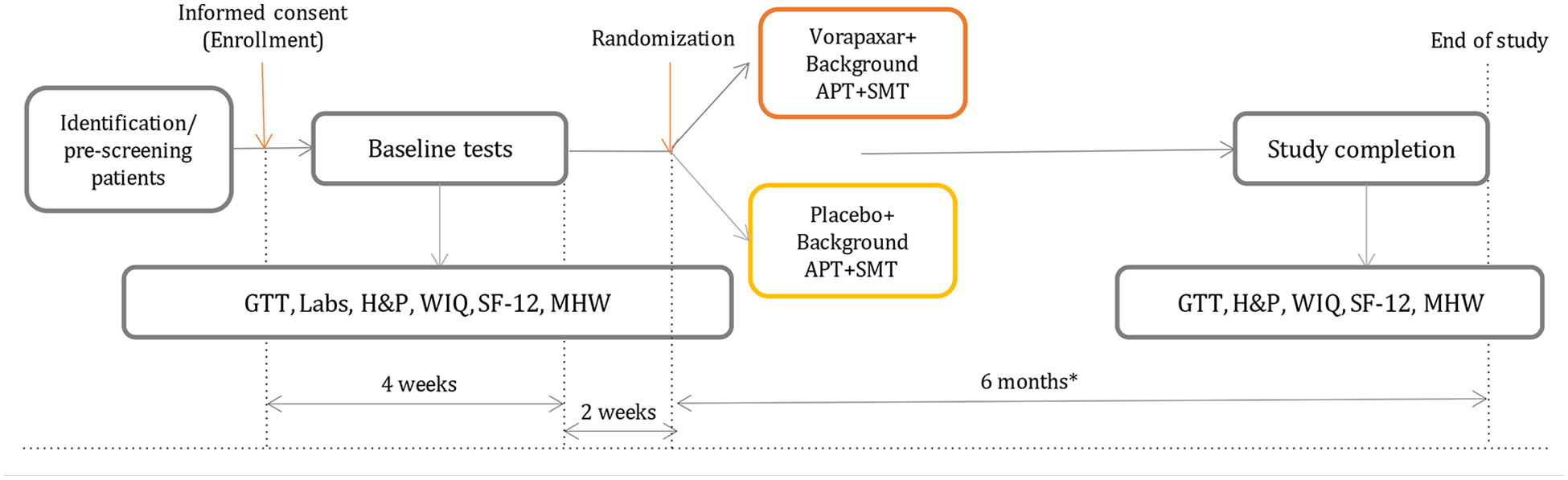
Study scheme. Study duration was 6 months. A 90-day phone call was made to reinforce the home walking program by reviewing walking diaries, assess compliance with study and clinically indicated medications, and determine study related adverse or severe adverse events.
Subjects
All subjects provided written informed consent. Prescreening criteria, for the identification of eligible patients with IC, are listed in online Supplemental Table 1. PAD was defined by an ankle–brachial index (ABI) < 0.8 at rest or ⩾ 20% decrease in ABI with exercise and one of the following criteria in the affected limb: (1) prior surgical and/or endovascular intervention or (2) known presence of ⩾ 70% stenosis documented on angiogram, computed tomography (CT) scan, magnetic resonance imaging (MRI), or duplex. Inclusion and exclusion criteria are listed in Table 1 and online Supplemental Table 1. Notable exclusion criteria included previous stroke or transient ischemic attack, warfarin or other anticoagulant use within 14 days, and use of ticagrelor, clopidogrel, prasugrel, or ticlopidine within 7 days. All enrolled patients participated in a 2-week run-in phase to assess compliance with the home-based walking protocol. Participants were instructed to walk at home for 15 minutes a day at a pace of at least 20 steps/minute, at least three times a week. Their home walking routine was captured in a self-reported journal. Baseline data included laboratory values, ABI, completion of the 12-Item Short-Form Survey (SF-12), and a graded treadmill test (GTT), on which peak walking time (PWT) and claudication onset time (COT) were recorded. The GTT protocol consisted of walking on a treadmill at 2.0 miles/hour at gradually increasing inclines (online Supplemental Table 2) that advanced by 2% grade every 2 minutes. All patients were required to have background antiplatelet therapy (aspirin) and standard medical therapy (SMT), which included any one or more of the following drugs: angiotensin-converting enzyme inhibitor (ACEI), angiotensin receptor blocker (ARB), lipid-lowering therapy, and beta-blocker therapy.
Inclusion and exclusion criteria.
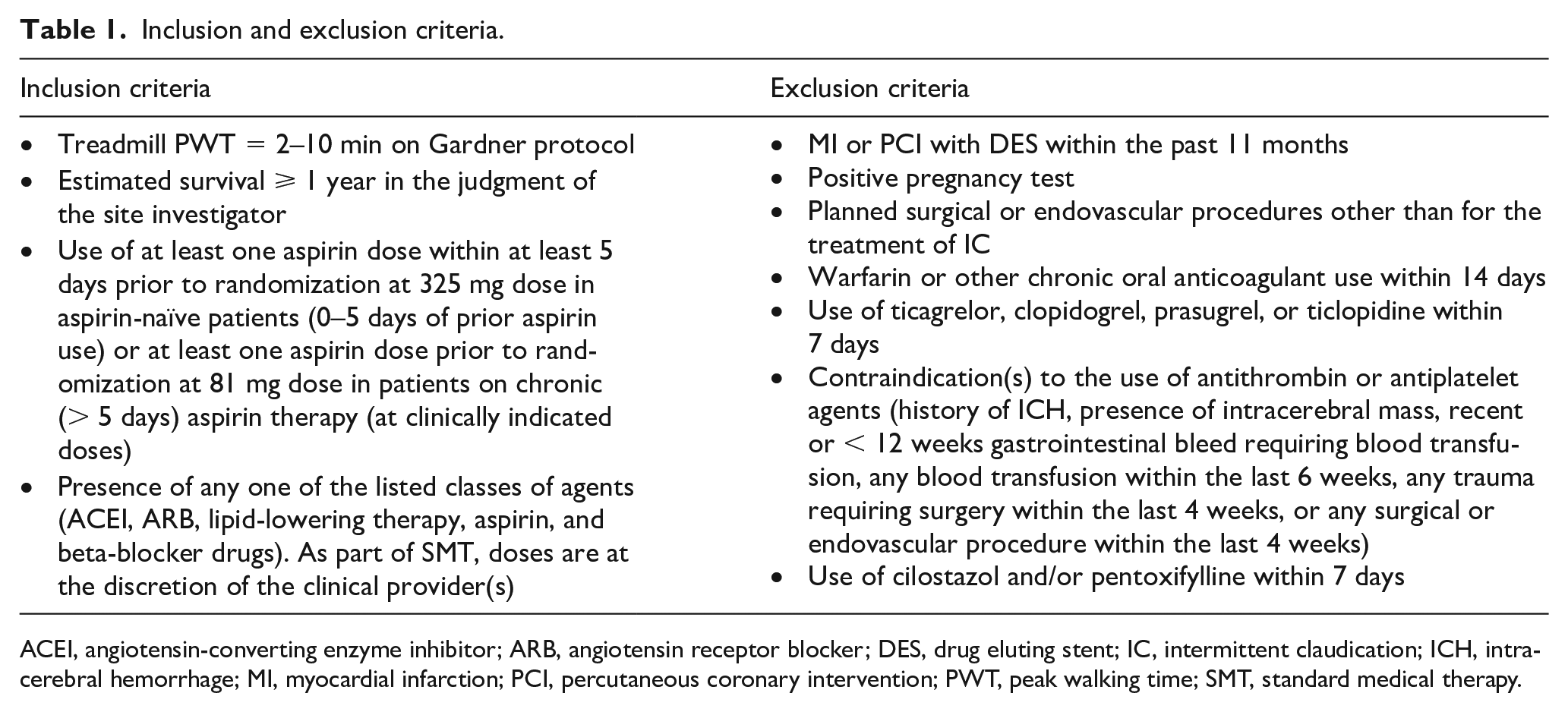
ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; DES, drug eluting stent; IC, intermittent claudication; ICH, intracerebral hemorrhage; MI, myocardial infarction; PCI, percutaneous coronary intervention; PWT, peak walking time; SMT, standard medical therapy.
Randomization
Once patients had completed the 2-week run-in phase, they were randomized 1:1 to either the treatment arm (vorapaxar 2.08 mg daily) or the matched-placebo arm.
Exercise training and monitoring
All patients were instructed to continue the home walking regimen as demonstrated in the run-in phase. After 90 days (3 months), all patients received a follow-up phone call to ensure compliance with the home walking program, study medications, and completion of walking journal entries.
Outcome measurements
Walking performance was assessed by GTT at baseline and after 6 months. PWT and COT were recorded. QoL measurements were assessed by the SF-12, which was administered at baseline and after 6 months.
The primary endpoint was the change in PWT on the GTT from baseline to 6 months between participants enrolled in the treatment and control arms of the study. Secondary endpoints were to assess (1) the change in COT during the GTT and (2) the change in SF-12 results from baseline to 6 months between participants in the treatment and control arms. Tertiary endpoints were to identify the first occurrence of (1) clinically indicated lower extremity endovascular or surgical revascularization, (2) all-cause death, MI, or ischemic stroke, and (3) severe bleeding defined according to the Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries (GUSTO) during the entire study duration postrandomization in participants in the treatment and control arms of the study.
Statistical analyses
The initial planned sample size was 200 patients. Estimated baseline PWT was 5 minutes, and a 25% increase in the PWT was considered to be clinically significant.11,12 Based on an estimated 15% increase in PWT in the control group and at least a 40% increase in PWT in the vorapaxar group, with a SD of 3.2 minutes, 160 patients would be needed to detect a difference with 80.08% power at the two-sided 0.05 significance level. Accounting for a 20% loss to follow-up at 6 months, a total of 200 patients were planned for enrollment in the study. The study ended prematurely when the sponsor unexpectedly declared bankruptcy.
Descriptive data were summarized as means ± SD or frequency (with percentages). The differences between the active and control groups were analyzed with a t-test or Wilcoxon rank sum test for continuous variables and χ2 test or Fisher’s exact test for categorical variables. For the endpoints of PWT and COT, t-tests were conducted to evaluate the differences between the active and control groups in mean change from baseline to 6 months. Responses to the SF-12 were used to calculate a mental component score (MCS) and physical component score (PCS) using online software (orthotoolkit.com/sf-12). For both the MCS and PCS scores, t-tests were conducted to evaluate the differences between the active and control groups in mean change from baseline to 6 months. All statistical analyses were two-sided and p < 0.05 was considered statistically significant. Statistical analyses were performed in R, version 3.6.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Overall, 128 patients were enrolled, and 102 were randomized. The study was terminated early by the industry sponsor when the sponsor filed for bankruptcy. Eleven patients were lost to follow-up or withdrawn, and four patients were noncompliant in taking their study medications. One patient died; the death certificate reported that he had passed away in his sleep. Sixty-six patients completed all study procedures, and their data were analyzed. Baseline demographics for the two study groups are shown in Table 2. All patients who completed the study also completed the 3-month follow-up. However, only 27 of the 66 patients (41%) who completed the study returned walking journals.
Baseline demographic and clinical data.
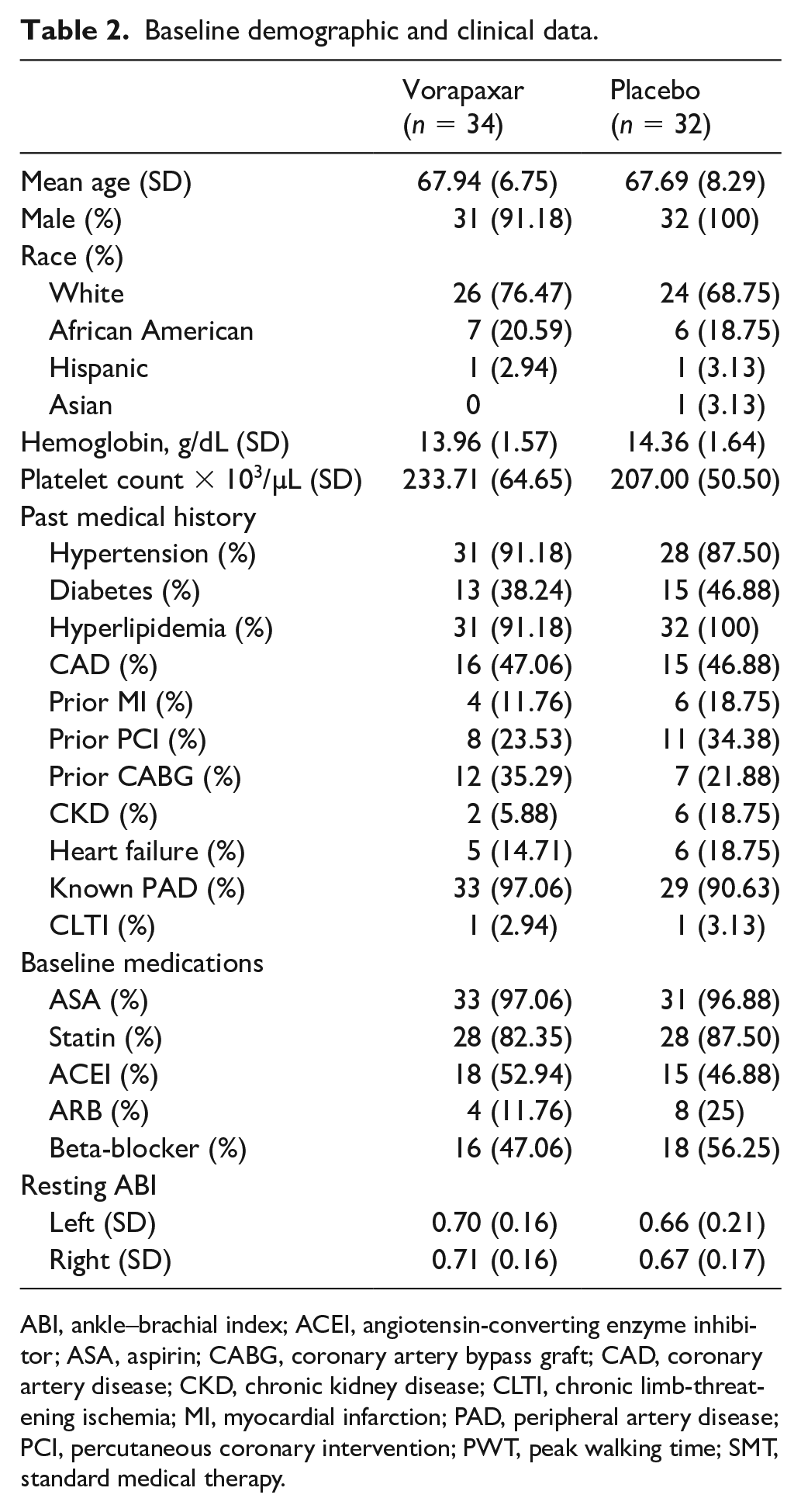
ABI, ankle–brachial index; ACEI, angiotensin-converting enzyme inhibitor; ASA, aspirin; CABG, coronary artery bypass graft; CAD, coronary artery disease; CKD, chronic kidney disease; CLTI, chronic limb-threatening ischemia; MI, myocardial infarction; PAD, peripheral artery disease; PCI, percutaneous coronary intervention; PWT, peak walking time; SMT, standard medical therapy.
After 6 months, PWT as measured on GTT was not significantly changed in either the treatment or the placebo groups (change in PWT: 0.03 ± 1.94 minutes in the vorapaxar group; 0.23 ± 1.48 minutes in the placebo group, p = 0.56; Figure 2). COT increased in both groups: 2.11 ± 1.73 to 2.35 ± 2.03 minutes in the vorapaxar group; 2.06 ± 1.34 to 2.34 ± 1.99 minutes in the placebo group. However, the change in COT (0.24 ± 2.05 minutes in the vorapaxar group and 0.28 ± 1.47 in the placebo group) was not significantly different (p = 0.95). There was no significant change in ABI between baseline and 6 months in either group.
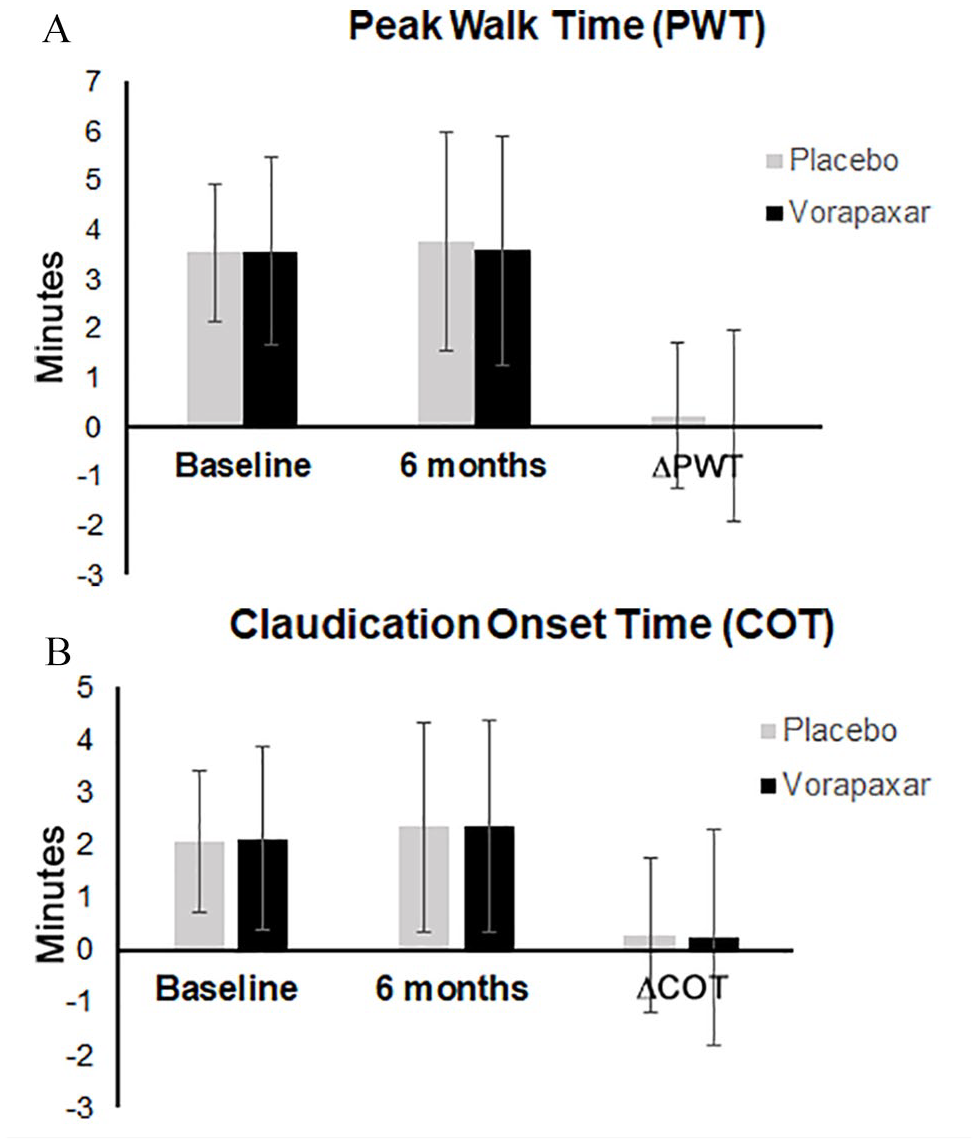
Results (mean ± SD) of the graded treadmill test.
Results of QoL measurements, as reflected by the SF-12, are depicted in Figure 3. After 6 months, there was no significant difference between the placebo and vorapaxar groups when assessing change in PCS (p = 0.63) or MCS (p = 0.32).
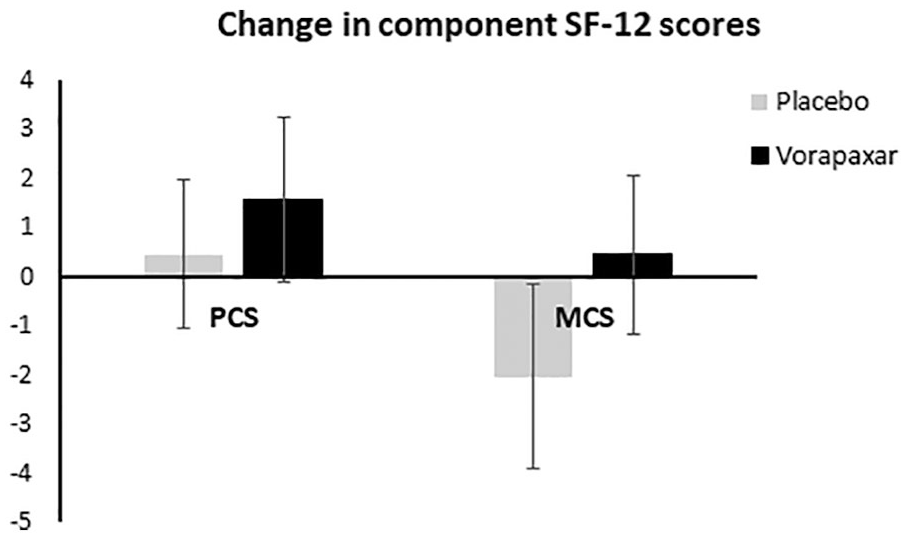
Change in the physical component score and mental component score components of the SF-12 survey in the placebo and vorapaxar groups (mean ± SD).
Adverse events and serious adverse events were assessed in all 102 randomized patients, and there were no significant differences between the placebo and the vorapaxar groups. In terms of limb events, one patient in the vorapaxar group reported worsening claudication. In the placebo group, one patient underwent percutaneous lower extremity balloon angioplasty, and one patient underwent minor amputation (left 4th toe). One patient died in the vorapaxar group, four patients experienced chest pain or unstable angina (three in the vorapaxar group, one in the placebo group), and one patient suffered a stroke in the placebo group. There were no cases of severe bleeding as per criteria in the GUSTO trial; however, there were three patients (two in the placebo group, one in the vorapaxar group) who had mild or moderate bleeding events (retroperitoneal hematoma, anemia, melena).
Discussion
The current study found that vorapaxar, in addition to aspirin and standard medical therapy for PAD, did not affect walking performance after 6 months in patients with IC. Although target enrollment was not reached, a power analysis based on the results from the 66 patients with complete data found that over 2000 patients would be required to detect a significant difference in the primary endpoint, PWT. Therefore, even had the study been completed as planned, it was unlikely that the findings would be significantly changed. The study found that there was no benefit of vorapaxar; however, there was also no increased risks. Specifically, there was no difference in bleeding events between the vorapaxar and the placebo group. Although previous clinical trials of vorapaxar did report an increased rate of bleeding, the majority of patients in those studies were on triple therapy (aspirin, clopidogrel, and vorapaxar).13,14 In contrast, patients screened for the current study were excluded if they were taking a P2Y12 inhibitor.
For patients with PAD, antithrombotics are often recommended to decrease risk of coronary or cerebrovascular events. 15 Recently, a subanalysis of the COMPASS trial showed that the combination of low-dose rivaroxaban and aspirin in patients with PAD decreased the risk of major adverse cardiovascular events (MACE) and major adverse limb events (MALE), including amputation. 16 Of the subjects enrolled in the stable PAD substudy of the COMPASS trial, 45% had a history of IC. 16 Even in this cohort of patients, a significant portion of which had nonlimb-threatening PAD, there was a benefit associated with low-dose rivaroxaban in decreasing the rate of future MALE, including amputation. However, there are, as yet, no data suggesting a role for antithrombotics to improve walking performance in patients with IC.
Randomized clinical trials of medications to improve walking performance in patients with IC are rare. Many patients with IC also have coronary artery disease and are managed with antiplatelet therapy, antihypertensives, and lipid-lowering agents to decrease their risks of coronary events, thus confounding the effect of medications on claudication. Furthermore, it is well known that individuals with IC have a 1–3% risk of major amputation over 5 years, but a 10–15% risk of death, most often from cardiovascular causes. 17 Thus, is it not surprising that medical therapies in patients with IC are targeted to decrease the risk of cardiovascular death.
IC, however, is a significant detriment to QoL, and identifying a pharmacologic therapy that improves walking performance and QoL is pertinent and timely. Currently, cilostazol, a phosphodiesterase type 3 inhibitor, is the only medication that is recommended to treat leg symptoms in patients with IC. 3 Notably, the last major randomized studies demonstrating the benefits of cilostazol were published over 20 years ago.18,19 Furthermore, cilostazol is contra-indicated in patients with heart failure, and many patients do not tolerate the side effects of cilostazol, thus limiting the widespread clinical use of this medication. Pentoxifylline, a rheological modifier that improves the deformability of red blood cells, has been shown to be significantly less effective than cilostazol, and is not recommended for patients with IC.3,20
Study limitations
There were several limitations to this study. First, the study was terminated prematurely when the industry sponsor filed for bankruptcy. Despite this, 102 patients in over 12 centers were randomized and 66 patients completed the study. The absolute number of patients enrolled compares favorably with the only other recent placebo-controlled trial in patients with IC, which investigated ramipril versus placebo. 21 In that study, 160 patients were screened with ultimately 33 enrolled and 29 completing the study. Both our current study and the previously reported studies, all of which comprise small numbers of patients, highlight the challenges of conducting clinical trials in patients with IC, which include biases on the part of both the physician and the patient, wide disparities in patient anatomy, symptomatology, and motivation to walk. 22
Second, the study was limited by the known challenges associated with unsupervised home exercise therapy. 23 Similar to the aspirin versus clopidogrel study, study participants were carefully screened through treadmill tests and a 2-week lead-in phase to demonstrate compliance with a home walking regimen. Despite this, compliance with home walking therapy was presumably poor; only 27 of 66 patients (41%) returned walking journals. Previous studies that have demonstrated the effectiveness of home-based walking programs 11 have also included intensive structured monitoring and feedback, neither of which were included in this current study. Finally, although baseline assessments were obtained prior to randomization, outcomes assessment was not blinded, which may have introduced some bias.
Conclusion
Completion of a randomized placebo-controlled trial of pharmacologic interventions to improve walking in patients with IC remains a challenging yet important goal. This study demonstrated that there is no benefit of vorapaxar on walking performance in patients with IC, despite promising aspects of the mechanism of action of the PAR1 inhibitor. Improved understanding of cellular and molecular mechanisms affected by novel pharmacologic discoveries or already existing medications should drive future clinical trials of medications that ultimately may improve walking and QoL in patients with IC.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X211042082 – Supplemental material for No benefit of vorapaxar on walking performance in patients with intermittent claudication
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X211042082 for No benefit of vorapaxar on walking performance in patients with intermittent claudication by Shirling Tsai, Yulun Liu, Mohamad Amer Alaiti, Jorge Antonio Gutierrez, Emmanouil S Brilakis and Subhash Banerjee in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X211042082 – Supplemental material for No benefit of vorapaxar on walking performance in patients with intermittent claudication
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X211042082 for No benefit of vorapaxar on walking performance in patients with intermittent claudication by Shirling Tsai, Yulun Liu, Mohamad Amer Alaiti, Jorge Antonio Gutierrez, Emmanouil S Brilakis and Subhash Banerjee in Vascular Medicine
Footnotes
Acknowledgements
This material is the result of work supported with resources and the use of facilities at the VA North Texas Health Care Systems. The contents do not represent the views of the US Department of Veterans Affairs or the United States Government. We would like to acknowledge all of the participating site investigators and study coordinators who contributed to this work.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Shirling Tsai: none. Yulun Liu: none. Mohamad Amer Alaiti: none. Jorge Antonio Gutierrez: none. Emmanouil S Brilakis: consulting/speaker honoraria from Abbott Vascular, Amgen, Biotronik, Boston Scientific, Cardiovascular Innovations Foundation (Board of Directors), ControlRad, CSI, Ebix, Elsevier, GE Healthcare, InfraRedx, Medtronic, Siemens, and Teleflex; research support from Regeneron and Siemens; owner: Hippocrates LLC; shareholder: MHI Ventures. Subhash Banerjee: consulting honoraria from Medtronic, Cordis, AngioSafe and Kaneka; research grants (institutional) from Boston Scientific Corporation, and Chiesi.
Funding
This work was supported by a research grant from Merck & Co., Inc. and Aralez Pharmaceuticals R&D, Inc. All data analysis and manuscript preparation was performed independently of the sponsors.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
