Abstract
Introduction:
Low high-density lipoprotein (HDL)-cholesterol is frequent in patients with peripheral artery disease (PAD) and also in type 2 diabetes mellitus (T2DM), the major risk factor for PAD. The transfer of cholesterol from the other lipoproteins to HDL is an important aspect of HDL metabolism and function, and may contribute to atherogenic mechanisms that lead to PAD development.
Objective:
The aim of this study was to investigate the status of cholesterol transfers in patients with PAD without or with T2DM.
Methods:
Patients with PAD (n = 19), with PAD and T2DM (PAD + DM, n = 19), and healthy controls (n = 20), all paired for age, sex, and BMI were studied. Transfer of both forms of cholesterol, unesterified (UC) and esterified (EC), was performed by incubating plasma with a donor nanoemulsion containing radioactive UC and EC, followed by chemical precipitation and HDL radioactive counting.
Results:
Low-density lipoprotein (LDL)-cholesterol and triglycerides were similar in the three groups. Compared to controls, HDL-C was lower in PAD + DM (p < 0.05), but not in PAD. Transfer of UC was lower in PAD + DM than in PAD and controls (4.18 ± 1.17%, 5.13 ± 1.44%, 6.59 ± 1.25%, respectively, p < 0.001). EC transfer tended to be lower in PAD + DM than in controls (2.96 ± 0.60 vs 4.12 ± 0.89%, p = 0.05). Concentrations of cholesteryl ester transfer protein (CETP) and lecithin-cholesterol acyltransferase (LCAT), both involved in HDL metabolism, were not different among the three groups.
Conclusion:
Deficient cholesterol transfer to HDL may play a role in PAD pathogenesis. Since UC transfer to HDL was lower in PAD + DM compared to PAD alone, it is possible that defective HDL metabolism may contribute to the higher PAD incidence in patients with T2DM.Keywords
Keywords
Introduction
Peripheral artery disease (PAD) is an atherosclerotic obstructive disease that occurs in 12% of the world adult population. 1 Type 2 diabetes mellitus (T2DM) is the most prominent PAD risk factor. 2 The overall prevalence of the main PAD manifestations, namely intermittent claudication and critical limb ischemia, is 3–10% and increases up to 15–20% in patients over 70 years of age. 1 Patients with PAD have a high risk of obstructive atherosclerotic disease in other artery beds: 30–50% incidence of coronary artery disease (CAD) and 15–25% of carotid stenosis.1–3
In PAD, hypertriglyceridemia and high low-density lipoprotein cholesterol (LDL-C) are not typically predominant, but low high-density lipoprotein cholesterol (HDL-C) is frequent. 2 HDL has several antiatherogenic functions, such as the role in cholesterol esterification, which stabilizes the cholesterol plasma pool, and in cholesterol reverse transport, whereby cholesterol is extracted from the cells of peripheral tissues and transported to the liver for excretion in the bile. 4 Cholesterol esterification is catalyzed by lecithin-cholesterol acyltransferase (LCAT), an enzyme associated to the HDL fraction, using apolipoprotein (apo) A-I, the main apo in HDL, as co-factor. 5
Nascent HDL is produced in the liver and intestine as discoid particles composed of apo A-I, phospholipids, and unesterified cholesterol. 6 These particles are progressively transformed into a spherical shape by the transfer of lipids to HDL and cholesterol esterification. Lipid transfers to and from HDL are crucial for HDL metabolism and atheroprotective functions. 7 Transfer of lipids is facilitated by transfer proteins such as cholesteryl ester transfer protein (CETP) and phospholipid transfer protein (PLTP). 8 Therefore, transfer of cholesterol to HDL, wherein the esterification process chiefly occurs, is essential for plasma cholesterol homeostasis.
An in vitro assay to assess the simultaneous transfer of unesterified and esterified cholesterol, phospholipids, and triglycerides from a donor lipid nanoemulsion to HDL was described and validated.8,9 Transfers of unesterified and esterified cholesterol were found diminished in patients with premature CAD 10 and in patients with CAD and T2DM. 11
Although HDL-C is one of the most important predictors of cardiovascular disease, systematic tests of the HDL functional and metabolic aspects may add much more to the overall comprehension of the HDL impact on atherogenesis. Since T2DM has a large impact, as PAD risk factor and low HDL-C accompanied by HDL defective function is a typical trait of T2DM, it is important to tell apart the differences of PAD with T2DM from PAD alone. Thus, the aim of this study was to examine the cholesterol transfers to HDL in conjunction with related biomarkers in patients with PAD with and without T2DM.
Methods
Study volunteers
Volunteers with PAD alone (PAD group; n = 19, 11 men, 68 ± 11 years) and with PAD and T2DM (PAD + DM group; n = 19, 13 men, 69 ± 9 years) were selected for the study from the Outpatient Clinics of the Gaspar Viana Hospital in the city of Belém, Brazil, where they were being followed. A group of healthy volunteers, without PAD or T2DM, was also included (control group; n = 20, 11 men, 68 ± 7 years). The three groups were prospectively selected for the study, as paired for sex, age, and body mass index (BMI).
Diagnosis of PAD had been previously established based on clinical history, physical examination, and ankle/brachial index (ABI). Normal ABI ranges 0.91–1.40 and lower, ⩽ 0.9 values, are considered abnormal. All the study patients had severe PAD according to the Rutherford classification, 12 namely categories 3, 4, and 5. Exclusion criteria were ABI > 0.9, type 1 diabetes mellitus, presence of chronic degenerative diseases, malignant or benign neoplasms, chronic alcoholism, renal failure (creatinine plasma concentrations above reference values), liver failure (total bilirubin and fractions, aspartate aminotransferase activity, alanine amino transferase, gamma glutamyl transferase, and alkaline phosphatase above reference values), hypothyroidism or hyperthyroidism, previous history of hemorrhagic stroke, recent bleeding, thrombocytopenia, gastric ulcer, and recent surgery and spinal puncture.
Diagnosis of T2DM was based on the American Diabetes Association criteria. 13 Patients who were using statins or other lipid-lowering drugs had this treatment discontinued for at least 45 days before they were studied to allow wash-out of the statin effects on plasma lipid metabolism.
The study was approved by the Ethics Committees of the University of São Paulo Medical School Hospital and of the Gaspar Viana Hospital, conforming to the guidelines set out in the Declaration of Helsinki, and all participants signed provided informed consent.
Serum biochemistry
Blood samples were collected after 12 hours of fasting. Glucose, triglycerides, and total cholesterol were determined by commercial enzymatic colorimetric methods (Dimension RxL; Siemens Healthcare, Newark, NJ, USA). HDL-C was measured by the same method used for total cholesterol after lipoprotein precipitation with magnesium phosphotungstate. LDL-C was estimated by the Friedewald formula. 14 Non-HDL-C was determined by subtracting the total cholesterol minus HDL-C. Apo A-I and apo B were determined by immunonephelometry (BN II and BN ProSpec System; Siemens Healthcare).
In vitro assay of cholesterol transfers to HDL
The radioactively labeled lipid donor nanoemulsion was prepared from a lipid mixture, as previously described by Maranhão et al. 15 The in vitro assay of lipid transfer from the lipid nanoemulsion to HDL was previously described by Lo Prete et al. 9 An aliquot of 200 µL of the plasma was incubated with 50 µL of the nanoemulsion labeled with 14 C-unesterified cholesterol and 3 H-cholesteryl oleate, under agitation, for 1 hour at 37°C. After incubation, 250 µL dextran sulfate/MgCl2 was added as a precipitation reagent for the apo B-containing lipoproteins. The mixture was shaken for 30 seconds and centrifuged for 10 minutes at 3000 rpm. Aliquots of 250 µL of the supernatant, containing the HDL fraction, were added in vials containing 5 mL of scintillation solution (PerkinElmer Inc., Waltham, MA, USA) and the radioactivity was measured in a liquid scintillation analyzer (Packard BioScience, Meriden, CT, USA). Radioactivity was then measured with a Packard 1600 TR model Liquid Scintillation Analyzer (PerkinElmer, Inc.). The transfer of unesterified and esterified cholesterol from the nanoemulsion to the HDL fraction was expressed as a percentage of the total incubated radioactivity as determined in a plasma sample without the addition of the precipitation reagent.
Determination of the plasma concentration of CETP and LCAT
CETP and LCAT concentrations were determined by a commercial two-step sandwich method of enzyme-linked immunosorbent assay (ELISA) following the instructions of the manufacturer (ALPCO Diagnostics, Salem, NH, USA).
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, version 20.0 (IBM Corp., Armonk, NY, USA). A Shapiro–Wilk test was performed to evaluate Gaussian distribution. Gaussian data were analyzed using the ANOVA test, followed by Bonferroni correction for multiple analyses. Non-Gaussian parameters were analyzed using the Kruskal–Wallis test, followed by the test for multiple comparisons of Dunn. The Fisher’s exact test was used to analyze percentages. Multiple linear regression analysis was used to determine the independent contribution of variables to lipid transfers. The model was based on variables that had significant association with lipid transfers on univariate analyses, besides age, sex, BMI, and PAD presence. In all analyses, parameters were considered significantly different when p < 0.05. The results are expressed as mean ± SD.
Results
Table 1 shows the physical and clinical characteristics of the study participants and use of medications. There were no statistically significant differences among the study groups when comparing sex, age, BMI, and time of PAD diagnosis. About 26% of patients used metformin, but glyburide, glimepiride, and insulin were also medications used by the PAD + DM patients.
Physical and clinical characteristics and plasma biochemistry data of the study participants.
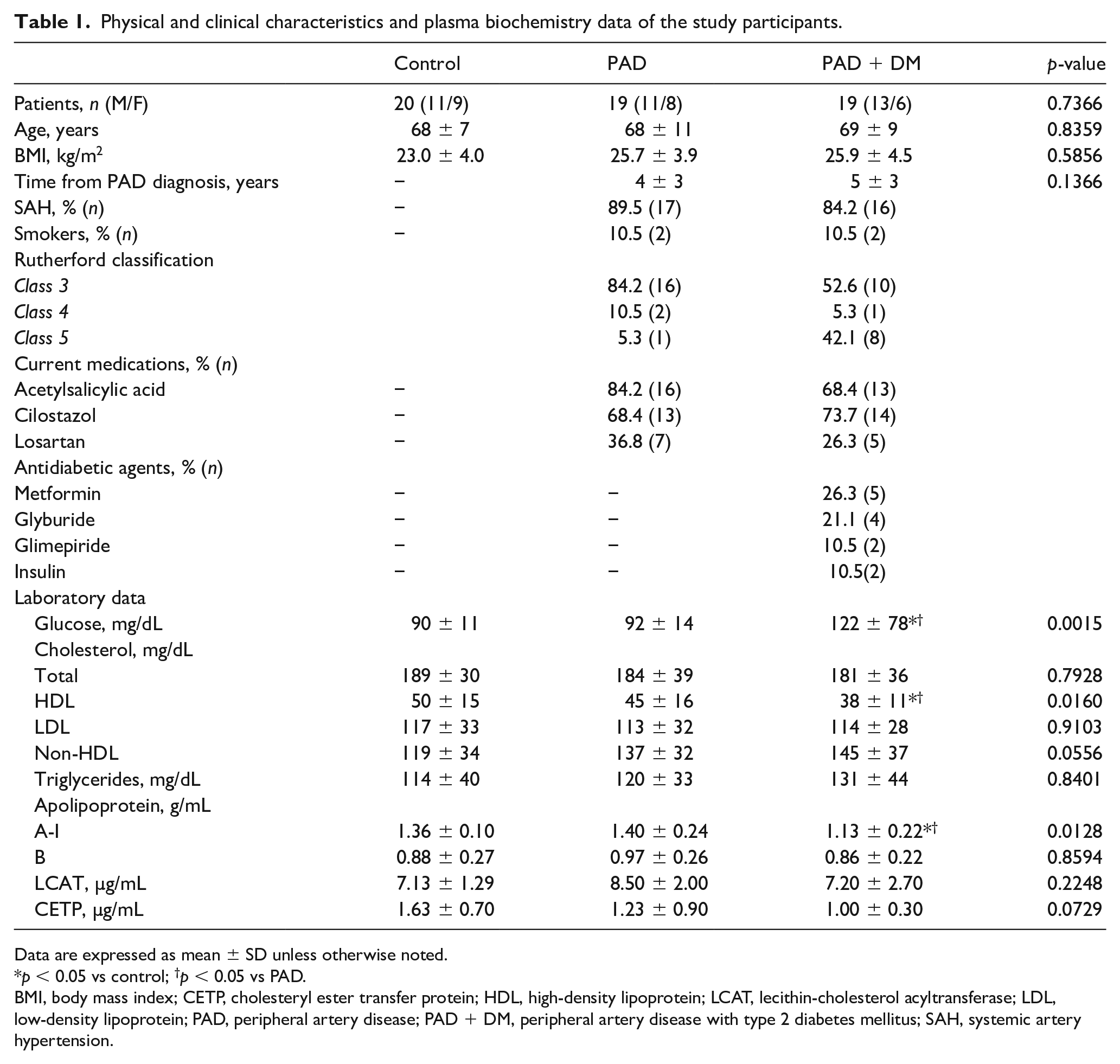
Data are expressed as mean ± SD unless otherwise noted.
p < 0.05 vs control; †p < 0.05 vs PAD.
BMI, body mass index; CETP, cholesteryl ester transfer protein; HDL, high-density lipoprotein; LCAT, lecithin-cholesterol acyltransferase; LDL, low-density lipoprotein; PAD, peripheral artery disease; PAD + DM, peripheral artery disease with type 2 diabetes mellitus; SAH, systemic artery hypertension.
Blood biochemistry parameters of the three groups are shown in Table 1. As expected, PAD + DM had the highest glycemic levels (p < 0.05). Triglycerides and LDL-C were equal in the three groups. The PAD + DM group had lower HDL-C than PAD and controls (p < 0.05). Lower concentrations of apo A-I were found in PAD + DM compared to PAD and control (p < 0.05), but apo B was equal in the three groups. CETP and LCAT were also equal.
Figure 1 shows the rates of transfer of the two cholesterol forms from the donor nanoemulsion to the HDL fraction. The transfer of unesterified cholesterol was lower in the two groups of patients with PAD than in controls (p < 0.001) but in PAD + DM was even lower than in PAD (p < 0.05). Esterified cholesterol transfer was lower in PAD + DM than in controls (p < 0.001), but in PAD there was no difference from the controls.
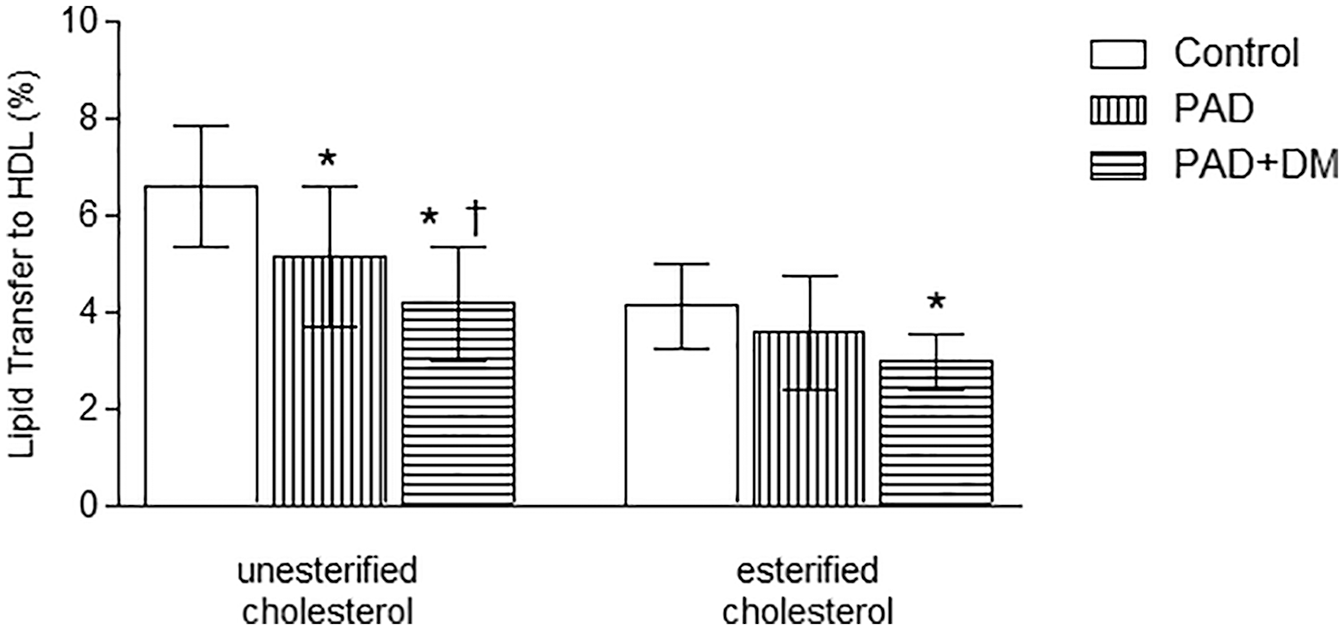
Transfer of unesterified and esterified cholesterol to high-density lipoprotein in the study groups (mean ± SD).
Table 2 shows the results of the correlation study performed by bivariate analysis in the study groups. The transfer rates of the two cholesterol forms and the concentrations of CETP and LCAT were correlated with various biochemical parameters and BMI. Positive correlations were found between esterified cholesterol transfer and CETP (p < 0.01) and apo A-I (p < 0.05) concentration, unesterified cholesterol transfer and HDL-C (p < 0.01) and apo A-I (p < 0.01), and between LCAT concentration and BMI (p < 0.01), and CETP and LDL-C (p < 0.01).
Correlations between lipid transfers to high-density lipoprotein, serum concentrations of CETP and LCAT, and lipid parameters.
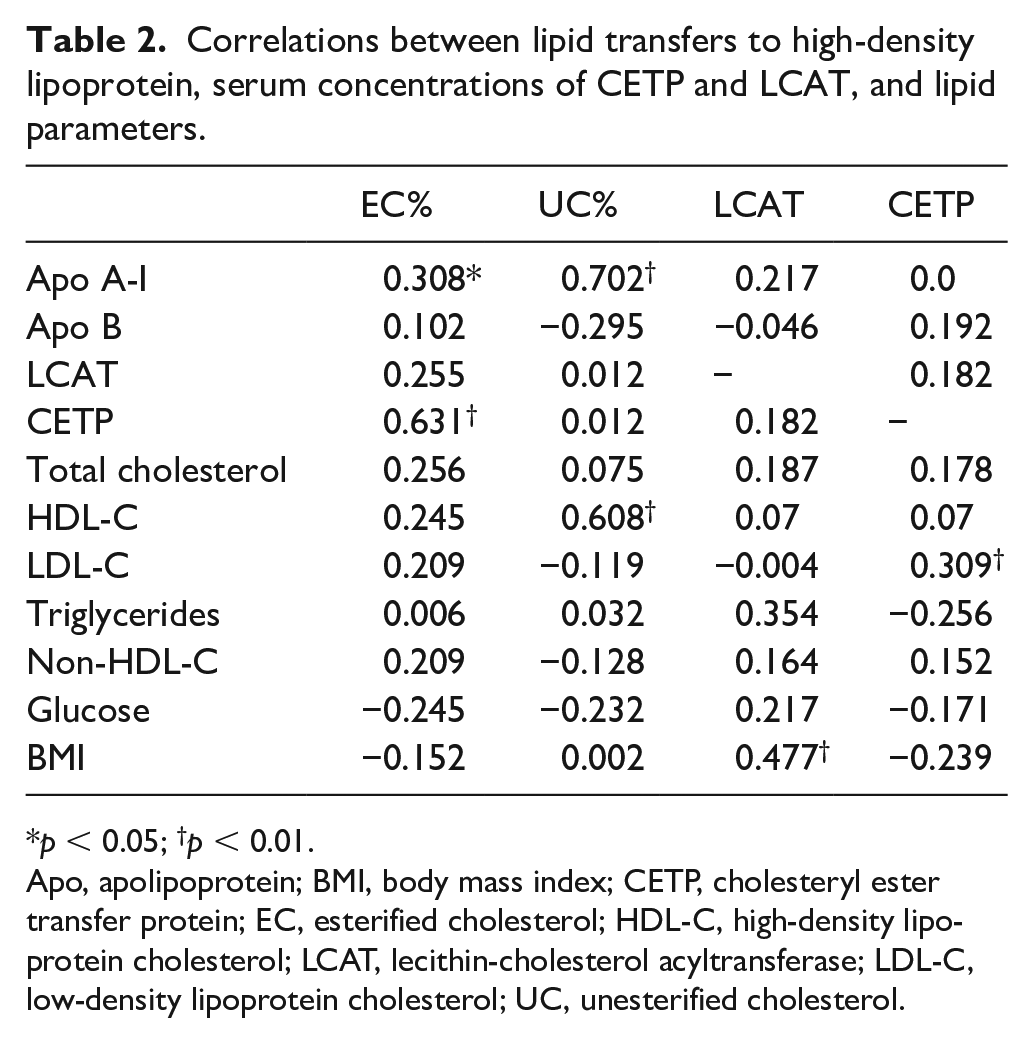
p < 0.05; †p < 0.01.
Apo, apolipoprotein; BMI, body mass index; CETP, cholesteryl ester transfer protein; EC, esterified cholesterol; HDL-C, high-density lipoprotein cholesterol; LCAT, lecithin-cholesterol acyltransferase; LDL-C, low-density lipoprotein cholesterol; UC, unesterified cholesterol.
Regarding the multiple linear regression analysis performed with the study data (Table 3), esterified cholesterol transfer was independently associated with CETP (β = 0.99, p < 0.001) and the presence of T2DM (β = −1.00, p = 0.007), but not with PAD (β = 0.17, p = 0.583). The transfer of unesterified cholesterol was independently associated with HDL-C (β = 0.076, p < 0.001) and glucose levels (β = 0.0163, p = 0.037) but not with PAD (β = −0.71, p = 0.091).
Correlations between lipid transfer to high-density lipoprotein, serum CETP concentrations, and presence of diabetes.
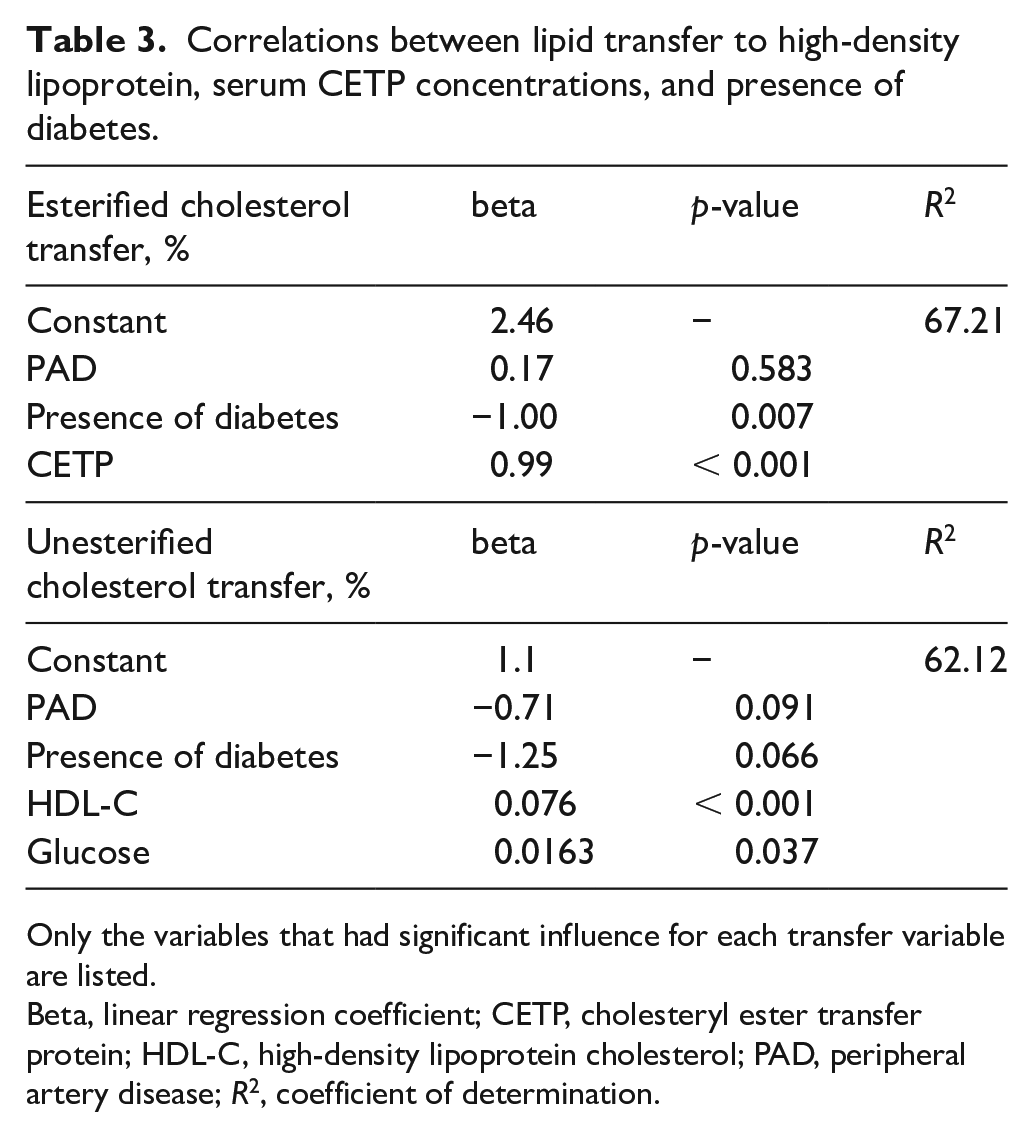
Only the variables that had significant influence for each transfer variable are listed.
Beta, linear regression coefficient; CETP, cholesteryl ester transfer protein; HDL-C, high-density lipoprotein cholesterol; PAD, peripheral artery disease; R2, coefficient of determination.
Discussion
In this study, it was shown that in both PAD and PAD + DM groups, the biomarkers related to the metabolism of pro-atherogenic apo B-containing lipoproteins, namely LDL-C and triglycerides, were equal to the controls. Regarding the biomarkers of the HDL metabolism, HDL-C and apo A-I, the main HDL apolipoprotein, were lower in the PAD + DM than control groups, but not in the PAD group. In fact, data on plasma lipids reported in the literature also suggest that LDL-C has weak association with PAD, and the association with triglyceridemia is still weaker.16,17 On the other hand, low HDL-C seems more consistently associated with PAD presence and stands as a PAD predisposing factor. 2 Therefore, although obtained from a small sample size, our results are in line with those previous studies that highlight the different weights of the traditional lipid risk factors for development of atherosclerotic disease in different artery beds. Compared with coronary ischemic disease or cerebral ischemic disease, it seems that plasma lipids bear relatively less importance as PAD biomarkers. 2
In this context, it is noteworthy that the transfer of both forms of cholesterol, unesterified and esterified, was lower than in controls in both PAD and PAD + DM groups, as observed in the in vitro assay. Similarly to the transfer of cholesterol from cells to HDL that prevents cholesterol cellular accumulation, 18 transfer of cholesterol from the apo B-containing lipoproteins to HDL can also be protective against atherogenesis. 8 In this regard, association of lower rates of unesterified and esterified cholesterol transfers to HDL in patients with precocious CAD and in patients with DM with CAD was previously found by Maranhão et al. 10 and Sprandel et al., 11 using the same in vitro lipid transfer assay.
Consequent to the impact of insulin resistance on the action of lipoprotein lipase and on other steps of lipid metabolism, the main feature of the dyslipidemia of T2DM consists in hypertriglyceridemia and lowering of HDL-C, with less interference on LDL-C levels. 19 In this study, lower HDL-C was present in PAD + DM, but statistically higher triglyceride levels were not encountered. Of note is the fact that LDL and HDL fractions were quantified by routine cholesterol serum biochemistry, and not by fast protein liquid chromatography that yields more precision in the quantification of plasma lipoprotein fractions.
Another important aspect of the current results was the finding that the in vitro transfer of unesterified and esterified cholesterol to HDL was still lower in PAD + DM than in PAD. In fact, in the study by Sprandel et al., 11 the transfer of both unesterified and esterified cholesterol was diminished in patients with T2DM and CAD compared with those without CAD. Thus, it can be hypothesized whether the markedly diminished transfers of the two cholesterol forms could be a factor of aggravation of the atherosclerosis process in peripheral arteries.
LCAT concentration was equal among the three groups, but CETP showed a nonsignificant trend for lower concentration when PAD + DM values were compared to controls. In case CETP concentration was indeed lower in that group, this could at least in part account for the lower transfer of esterified cholesterol observed in PAD + DM.
This study highlights the importance of exploring the association of HDL functional and metabolic aspects with the presence of diseases. In this regard, whereas HDL-C was not different between PAD and controls, the cholesterol transfer rates were lower, indicating a possible deficiency in the lipoprotein capacity of promoting the homeostasis of the cholesterol plasma pool.
Among the results of the correlation analysis, two positive correlations seem particularly meaningful. The positive correlation between CETP concentration and the transfer of esterified cholesterol was expected due to the well-known function of CETP in boosting this transfer. The positive correlation between the transfer of unesterified cholesterol and HDL-C can be explained by the fact that increased transfer of cholesterol to the HDL fraction leads to an increase in HDL-C.
The increase in unesterified cholesterol transfer to the HDL fraction, where it is esterified by LCAT action, tends to increase HDL-C, which could have accounted for this positive correlation. Nonetheless, it is also possible that the higher concentrations of HDL have determined the increased transfers to HDL, by mass action law.
Limitation
As a limitation of the study, in patients with T2DM, ABI is not as reliable as the toe/brachial index (TBI) for critical ischemia grading; ABI values may have been overestimated due to frequent artery calcification that occurs in some patients with T2DM. Unfortunately, TBI was not available for our analysis.
Conclusion
Low transfer of unesterified cholesterol to HDL was associated with the presence of PAD. Among the patients with PAD, it was relevant that those with PAD and T2DM had an unesterified cholesterol transfer rate still lower than those with PAD alone. This finding could be related to PAD pathophysiology in T2DM, and might suggest the hypothesis that a reduction of cholesterol transfer to HDL favors the development of PAD in patients with T2DM.
Footnotes
Acknowledgements
The authors are thankful to Josefa MHS Lima, (Laboratório de Metabolismo e Lípides, Instituto do Coração do Hospital das Clínicas da USP [InCor-HCFMUSP]) for her help with the experiments.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the São Paulo Research Foundation (FAPESP, grant 2014/03742-0, Brazil), National Council for Scientific and Technological Development (CNPq, grant 431290/2016-4, Brasilia, Brazil), and the National Institute of Science and Technology Complex Fluids (INCT-FCx, Brazil). Dr Maranhão has an A-1 Research Carrier Award from CNPq.
