Abstract
The aim of this study was to evaluate subclinical atherosclerosis and related factors in young type 1 diabetes (T1D) patients and healthy peers. Carotid intima-media thickness (cIMT) and anthropometric/laboratorial data were obtained for 83 T1D patients (mean age 19.5 ± 4.0 years, disease duration 9.8 ± 4.8 years) and for 36 matched healthy subjects. Considering all the participants as one group, male sex (p = 0.008), weight (p = 0.016) and T1D (p < 0.001) were positively associated with a higher cIMT. High-density lipoprotein (HDL) (p = 0.036) was negatively associated with cIMT in T1D. In the male T1D patients, HDL ≤47.5 mg/dL had a sensitivity of 87.5% and specificity of 57% (p = 0.035) in detecting those belonging to a higher cIMT tercile. In conclusion, weight and T1D were associated with increased cIMT. HDL levels ≤47.5 mg/dL were related to a higher cIMT in male T1D patients.
Keywords
Introduction
In the type 1 diabetes (T1D) population, the atherosclerotic process begins in childhood and continues silently over a long period of time before the occurrence of clinical events. 1 However, some characteristics and risk factors that may initiate and accelerate this process remain unclear in this population, 2 and a normal lipid profile and an increased high-density lipoprotein (HDL) cholesterol level may not always protect against cardiovascular disease (CVD). 3
The carotid intima-media thickness (cIMT) is a reliable surrogate marker of atherosclerosis. 4 However, studies on the risk factors related to cIMT in T1D have yielded conflicting results, as the majority of them included young individuals not attaining their glycaemic goals. 5 We previously described that glycaemic control might influence the prevalence of CVD risk factors 6 and the behaviour of dyslipidaemia in this population. 7
In this study, we aimed to evaluate cIMT and its related factors in a population of young T1D patients with suitable glycaemic control and to compare these patients to matched healthy control peers.
Subjects and methods
This cross-sectional study involved 83 T1D patients 8 and 36 healthy control subjects matched for age, height and body mass index (BMI). Exclusion criteria were the use of statins, current or past smokers and glycosylated haemoglobin (A1C) above 9.0%.
The patients were recruited consecutively during their regular visits as outpatients at the Diabetes Center of Federal University of Sao Paulo (Sao Paulo, Brazil). The healthy controls were medical students from this University. Written informed consent was obtained from either the participants or their legal guardians. The local ethics committee approved the study design (02030/07).
Study design
The subjects were evaluated at 8:00 a.m. following a 12-h overnight fast. Anthropometric and blood pressure measurements were recorded, and blood samples were collected. A flexible tape was positioned midway between the lowest rib and the superior border of the iliac crest to measure the waist circumference (WC). The systolic blood pressure (SBP) and diastolic blood pressure (DBP) were considered in the normal range if less than the 90th percentile for age and gender. 9
Blood and urine chemistry
The plasma glucose levels and cholesterol and triglycerides levels were determined using glucose hexokinase II and enzymatic methods (Bayer Advia 1650 analyser, serial number: CA176503-764), respectively. A1C was measured by high-performance liquid chromatography (HPLC; Tosoh G7, HLC-723; normal value (nv) = 4.0%–6.0%). Ferritin and C-reactive protein (CRP) were determined using chemiluminescent (Access®, Beckman Coulter, USA) and immunoturbidimetric assays (Olympus AU640, USA), respectively. Tumour necrosis factor-alpha (TNF-α) was measured by an immunoenzymatic assay (TNF-α EASIA, no. KAP1751), and microalbuminuria was determined using an immunoturbidimetric assay [AER (albumin excretion rate) > 20 µg/min].
cIMT
Carotid ultrasonographic studies were performed by two radiologists blinded to the study group. The studies were performed using a high-resolution ultrasound scanner (Voluson 730 Pro; GE Medical Systems, Kretztechnik GmBH and Co. OHG, Austria) with B-mode imaging according to the standardised protocol. 4 The measurements of the left and right common carotid arteries were recorded 1 cm from the carotid bulb. In total, three measurements were obtained for the right and left carotid arteries, and the mean value of these six points was calculated for each patient. The intra-assay coefficient of variation (CV) was 4.9%.
Statistics
The data were expressed as the mean ± standard deviation (SD). An analysis for the categorical variables between the groups was performed using a chi-square test; a Fisher exact test was performed in the event that an expected value was less than 5. For the continuous variables, statistical analyses between the groups were performed using a Student’s t-test, and Pearson’s correlations were performed for continuous variables associated with cIMT. Multivariate linear regression analyses were performed to identify variables independently related to cIMT. The Jonckheere–Terpstra test was performed to verify the association between HDL and cIMT terciles in both sexes with T1D. A receiver operating characteristic (ROC) curve analysis was performed to identify the HDL level associated with a greater cIMT in T1D men (p < 0.05).
Results
A total of 83 T1D patients and 36 healthy, matched control individuals participated in this study. The characteristics of the participants are shown in Table 1.
Clinical, anthropometric and metabolic characteristics of the study groups.
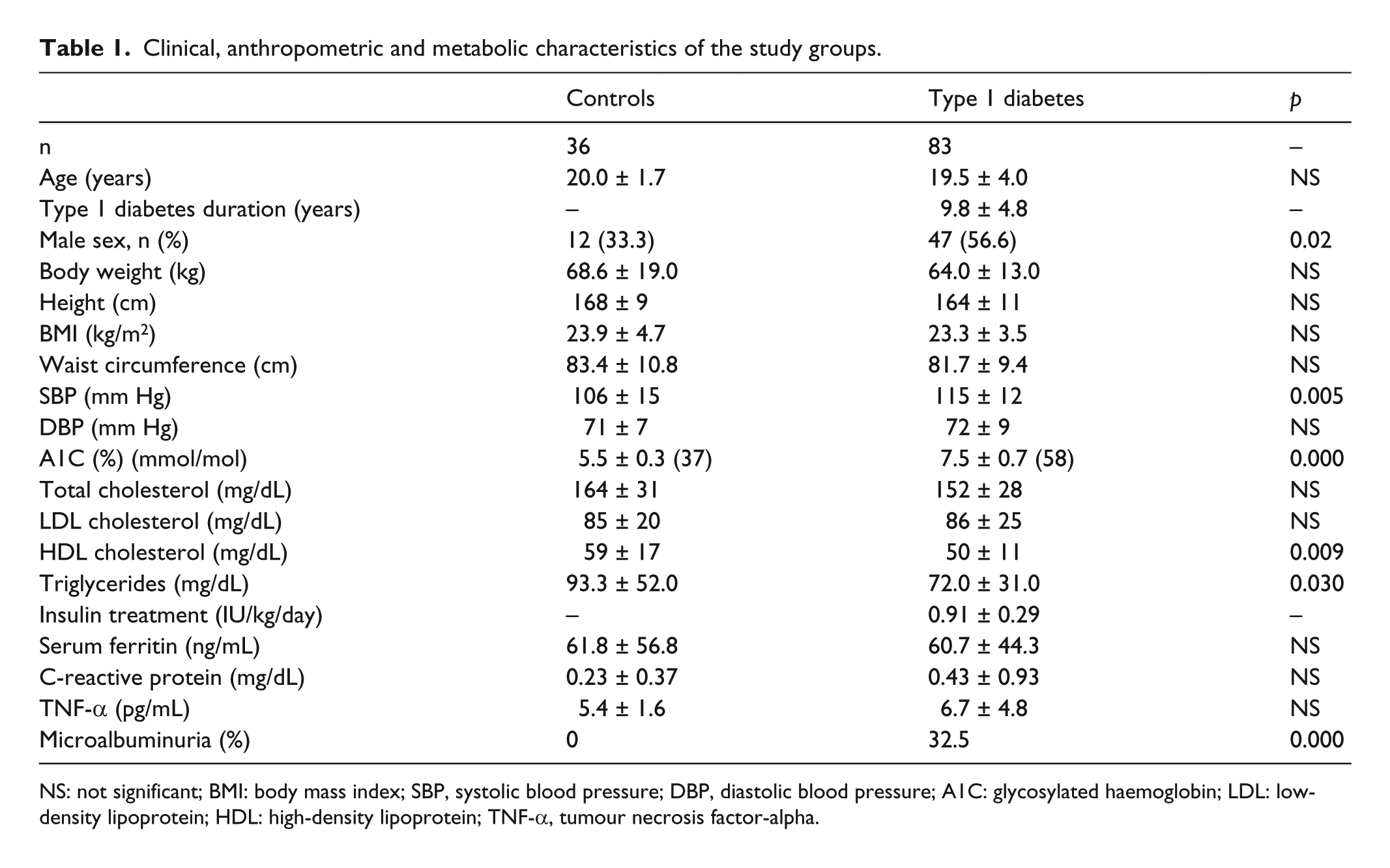
NS: not significant; BMI: body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; A1C: glycosylated haemoglobin; LDL: low-density lipoprotein; HDL: high-density lipoprotein; TNF-α, tumour necrosis factor-alpha.
Overall cIMT
According to the analysis of all the participants as one group, cIMT was greater in men (0.46 ± 0.10 vs 0.41 ± 0.06 mm; p = 0.004) and in the T1D patients (0.46 ± 0.09 vs 0.39 ± 0.04; p < 0.001). Based on the multivariate analysis, the male sex (β = 0.163, p = 0.008), weight (β = 0.001, p = 0.016) and T1D (β = 0.070, p < 0.001) were positively related to cIMT, whereas HDL levels were negatively associated with cIMT only in men (β = −0.002, p = 0.014). Of note, no association between cIMT and inflammatory markers (TNF-α, CRP, ferritin) was found (data not shown).
cIMT in the healthy control group
In the control group, weight and BMI (r = 0.519, p = 0.001) were positively related to cIMT (r = 0.433, p = 0.008). Furthermore, BMI (β = 0.005, p < 0.001) remained positively associated with cIMT in the multivariate analysis.
cIMT in the T1D patients
In the T1D patients, cIMT was increased in those whose disease duration was longer than 6 years (0.47 ± 0.09 vs 0.43 ± 0.05 mm; p = 0.017) and in men (0.48 ± 0.10 vs 0.43 ± 0.07 mm; p = 0.02). No difference in cIMT was found with regard to the presence of microalbuminuria, and there was no relationship between cIMT and inflammatory markers (TNF-α, CRP, ferritin; data not shown).
After adjusting for sex, weight (β = 0.001, p = 0.022) and HDL (β = −0.001, p = 0.036) were positively associated with cIMT according to the multivariate analysis.
By analysing terciles of cIMT in both sexes to identify the HDL levels associated with each cIMT sex tercile, we found that the male T1D patients who belonged to the greater tercile of cIMT had lower HDL serum levels (HDL for each tercile of cIMT: 52.0 ± 11.9, 46.2 ± 8.2 and 44.8 ± 10.0 mg/dL; p = 0.044, respectively). In contrast, we did not identify any difference of HDL among the terciles of cIMT in women (data not shown).
Based on an ROC curve to identify the HDL levels related to a high probability of a young T1D man to have a cIMT ≥0.58 mm (median of the third tercile of cIMT in male T1D patients), we found that HDL ≤47.5 mg/dL had a sensitivity of 87.5% and a specificity of 57.0%, with a power of 70%, in identifying a man with T1D and a thicker cIMT (≥0.58 mm) [p = 0.035, 95% confidence interval (CI): 0.592–0.874] (Figure 1).
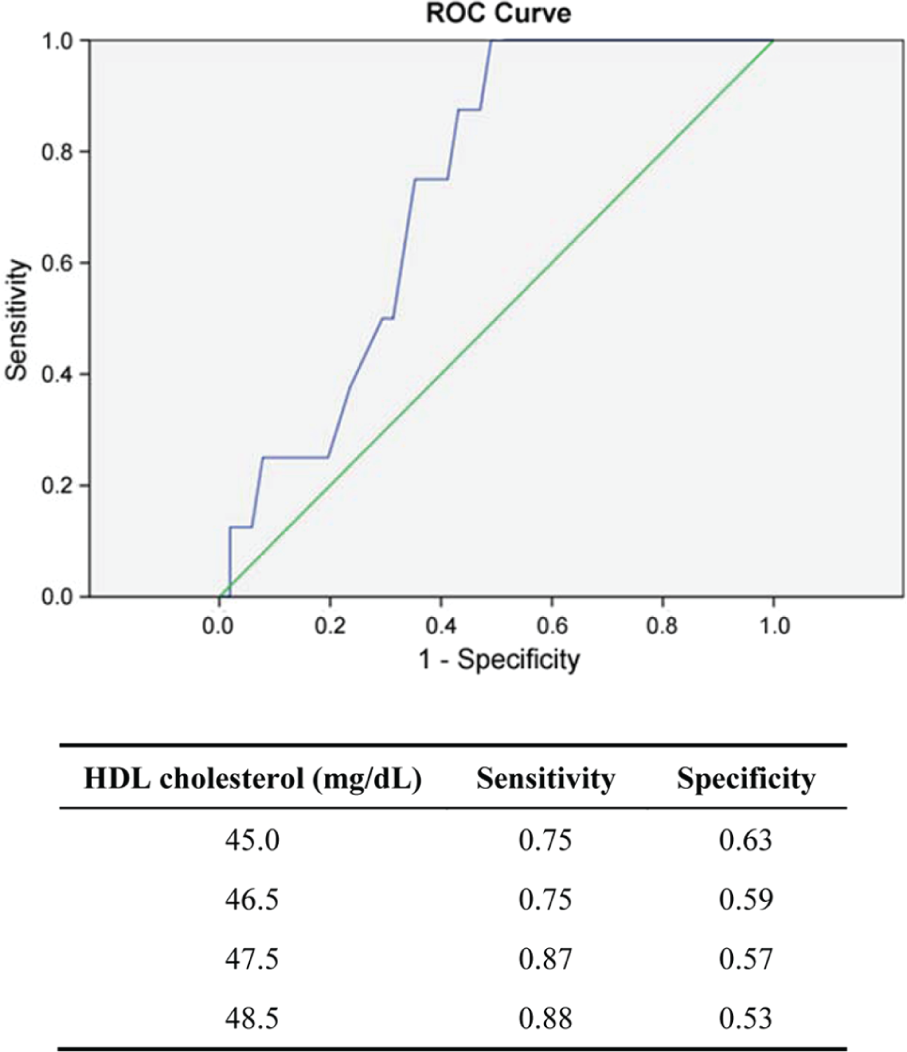
ROC curve to identify the HDL levels related to a value of cIMT ≥0.58 mm (median of the third tercile) in male T1D patients.
Discussion
Here, we demonstrate that the male sex, weight and T1D were positively associated with a greater cIMT in a group of young individuals. Weight was the most common variable that was positively related to cIMT in both healthy and T1D patients, and HDL was the main lipid fraction related (negatively) to cIMT in the T1D group. Furthermore, the HDL levels ≤47.5 mg/dL were related to a higher risk of a young T1D man to have a thicker cIMT.
Our T1D patients showed a mean cIMT (0.46 ± 0.09 mm) similar to that found in a Mexican study (0.46 ± 0.04 mm). 10 Nevertheless, in comparison to another study of obese adolescents without diabetes, 11 we found that our T1D population had a lower cIMT. Therefore, we may suggest that both obesity and hyperglycaemia are involved in the pathogenesis of subclinical atherosclerosis in young populations. Interestingly, weight was a common factor associated with cIMT in both our T1D and healthy groups, even though our participants had a normal mean BMI.
In our T1D patients, lower HDL levels were related to a higher risk of a T1D patient belonging to the third tercile of cIMT, though only in the male group. The protective effect of HDL on CVD in T1D patients is not well understood, 2 and although a linear inverse association between HDL and CVD incidence occurs in T1D men, this pattern appears to be disturbed in women. 3
The HDL value that can be considered normal or altered in young T1D populations has been well discussed. 3 In this study, we found that HDL serum levels ≤47.5 mg/dL in the T1D men were related to a cIMT ≥0.58 mm. This value of cIMT was significantly higher than the 90th percentile of our sex-age normal control group (0.49 mm) and may correspond to an increase in approximately 10 years in the biologic vascular ageing of this population. 12
We conclude that weight and T1D are associated with increased cIMT in our population. HDL levels ≤47.5 mg/dL were associated with a higher cIMT in the T1D patients. These findings may help us to identify the HDL values associated with weight control that should be considered as influential in reducing the atherosclerotic process in T1D patients.
Footnotes
Declaration of conflicting interests
The authors do not have any conflict of interest to declare.
Funding
This study was supported by the CAPES-PROEX institution of support for research, Brasilia, Brazil.
