Abstract
Peripheral artery disease (PAD) and diabetes mellitus are two overwhelming health problems associated with major cardiovascular (CV) and limb events, in addition to increased mortality, despite advances in medical therapies including statins and renin-angiotensin system inhibitors. Sodium-glucose cotransporter 2 inhibitors (SGLT2i) and glucagon-like peptide 1-receptor agonists (GLP1-RA) are two new antihyperglycemic drug classes that have been associated with a significant reduction of major adverse cardiovascular events (MACE) in patients with type 2 diabetes (T2D) and CV risk. Whereas most studies had enrolled patients with T2D and concurrent CV disease (CVD), patients with PAD were obviously underrepresented. Furthermore, there was a signal of increased risk of amputation in one of the main trials with canagliflozin. We aim to provide a general review of the current literature and summarize societal guideline recommendations addressing the role of SGLT2i and GLP1-RA drugs in patients with CVD focusing on the PAD population when data are available. Endpoints of interest were MACE and, when available, major adverse limb events (MALE).
Introduction
Cardiovascular disease (CVD) continues to be the leading cause of morbidity and mortality in patients with type 2 diabetes (T2D) despite many advances in evidence-based therapies during the past two decades. 1 Therefore, the development of treatment strategies to improve CV outcomes in this vulnerable population remains very significant. 2 Compared to a conservative approach, intensive glucose control in patients with T2D has consistently demonstrated improvement in microvascular outcomes. However, no significant demonstrable difference in CV outcomes has been observed, even when improvements in hemoglobin A1c (HbA1c) were achieved.3,4 The opportunities for improving clinical outcomes in patients with T2D and established or at risk for CVD have recently expanded.
Peripheral artery disease (PAD) affects more than 8.5 million Americans, out of whom approximately one-third have concurrent diabetes mellitus (DM).5,6 The prevalence of PAD may be underestimated in the diabetic population due to the asymptomatic nature of concurrent diabetic neuropathy. 5 Out of patients with prediabetes alone, 20% have an abnormal ankle–brachial index (ABI) compared with only 7% of patients with normal glucose levels. 7 DM increases the incidence of limb ischemia and amputation in patients with PAD and is the second most powerful risk factor for PAD after cigarette smoking, with more infrapopliteal involvement.8–10 Mechanisms of elevated risk include increased inflammation, endothelial dysfunction, augmented vasoconstriction, and thrombosis. 5
Multiple sodium-glucose cotransporter 2 inhibitors (SGLT2i) and glucagon-like peptide 1-receptor agonist (GLP1-RA) drugs have shown significant reduction in major adverse CV events (MACE; described as a composite of CV death, nonfatal myocardial infarction (MI), and nonfatal ischemic stroke) in patients with T2D and established or at risk for CVD. 11 Some SGLT2i drugs have also shown a substantial reduction in hospitalization for heart failure (HHF) and progression of diabetic kidney disease (DKD), even in nondiabetic patients. Although the exact mechanisms of CV and renal benefits are still unclear, they are beyond glycemic control and may be related to additional mechanisms of action related to each class. 12 Although multiple CV outcome trials for both SGLT2i and to less extent GLP1-RA drugs have included patients with PAD, this population was underrepresented.
We aim to provide a general review of the available literature and summarize the current guideline recommendations in support of SGLT2i and GLP1-RA drugs in patients with CV risk focusing on the PAD population when available. We performed a narrative review of the existing evidence on the effectiveness and safety of novel antihyperglycemic therapies in patients with PAD. Public, PubMed databases were searched using the terms ‘SGLT2i and PAD’, ‘SGLT2i and CV risk’, ‘GLP1-RA and PAD’ or ‘GLP1-RA and CV risk’ for all full-text articles on humans only written in English between January 1, 2000 and August 15, 2022. Search results yielded 306 articles. Articles with no distinct CV risk evaluation or no direct treatment with SGLT2i or GLP1-RA drugs were not discussed. Studies were divided into two groups: SGLT2i and GLP1-RA. Endpoints of interest were MACE, and when available, major adverse limb events (MALE) including acute limb ischemia and/or amputation.
Evidence
Sodium-glucose cotransporter 2 inhibitor (SGLT2i)
Mechanism of action
SGLT2i drugs act by blocking the sodium-glucose cotransporter in the proximal tubule of the nephron, leading to glucosuria and natriuria. SGLT2 receptor is responsible for reabsorbing roughly 90% of glucose from the luminal to intracellular space using sodium gradient with the remaining glucose reabsorbed by the SGLT1 receptor. 13 Increased expression of these receptors on maladaptive kidneys of diabetic patients 14 is associated with a 20% increase in glucose reabsorption. 15 By promoting glucosuria, these drugs lower fasting plasma glucose and HbA1c levels without significant risk for hypoglycemia or the need for insulin use. Lowering blood pressure, 3 reducing weight, and lowering blood lipid and uric acid levels are among other effects. 16 All of these actions, in addition to reducing arterial stiffness, epicardial fat, and cardiac fibrosis, may explain the positive CV outcomes. Although the exact explanation for the CV benefits of these drugs has not been fully understood, several proposed final mechanisms include reductions in preload and afterload through diuresis, alterations in myocardial metabolism, and prevention of myocardial fibrosis, among others. 17 Currently, eight SGLT2i drugs (canagliflozin, dapagliflozin, empagliflozin, ertugliflozin, luseogliflozin, ipragliflozin, sotagliflozin, and tofogliflozin) have been approved globally for the treatment of T2D, either as monotherapy or in combination with other hypoglycemic drugs. Dapagliflozin, canagliflozin, and empagliflozin have been approved in Europe, the USA, and other parts of the world, whereas ipragliflozin, tofogliflozin, and luseogliflozin have been approved only in Japan. 18
Reduction in major cardiovascular events
Based on outcomes from multiple trials, SGLT2i drugs have emerged as a very promising antidiabetic class with remarkable CV and renal benefits, in addition to reduced all-cause mortality (Tables 1 and 3).
Summary of randomized control trials that tested the efficacy of different SGLT2i drugs for various primary outcomes.
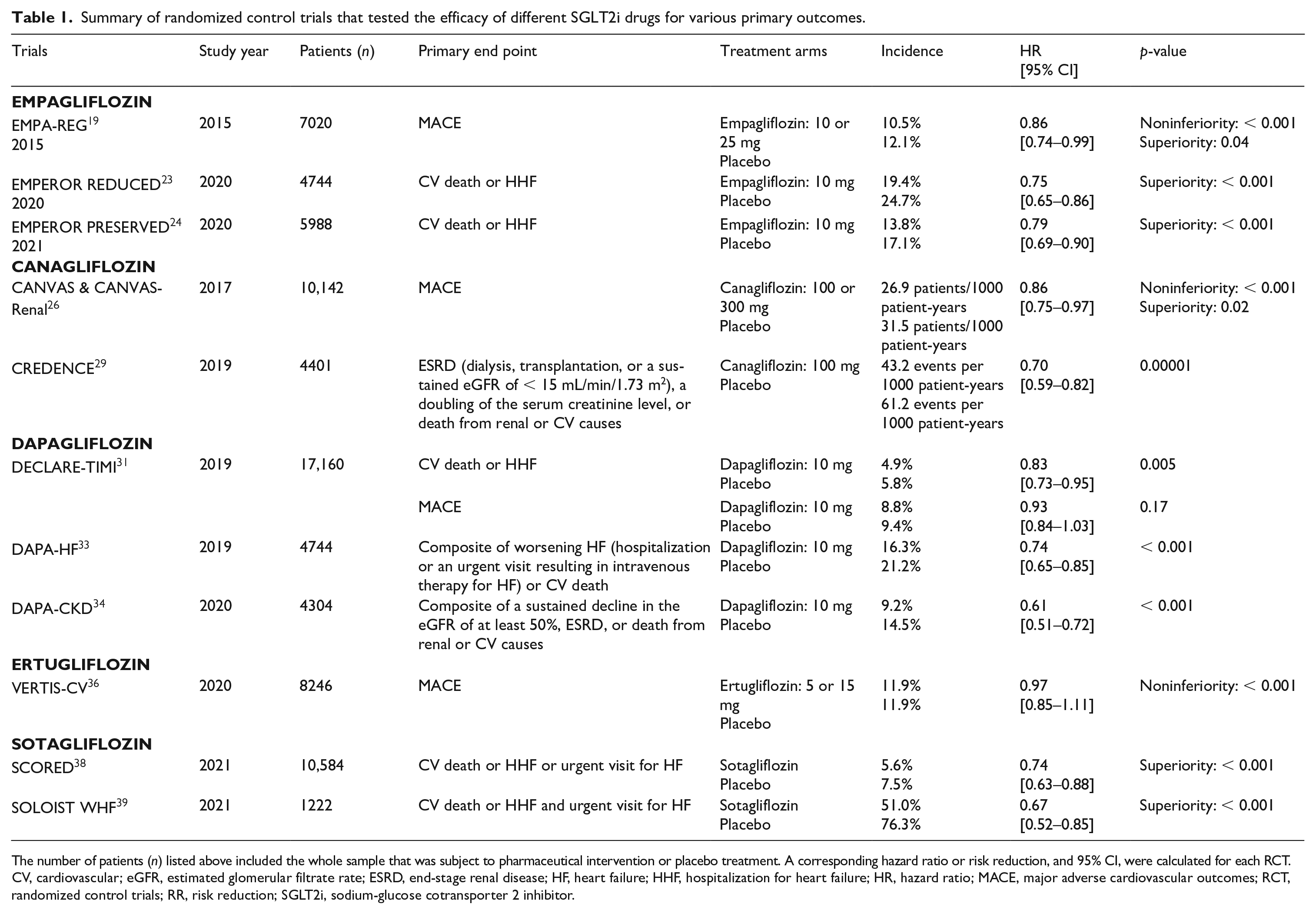
The number of patients (n) listed above included the whole sample that was subject to pharmaceutical intervention or placebo treatment. A corresponding hazard ratio or risk reduction, and 95% CI, were calculated for each RCT.
CV, cardiovascular; eGFR, estimated glomerular filtrate rate; ESRD, end-stage renal disease; HF, heart failure; HHF, hospitalization for heart failure; HR, hazard ratio; MACE, major adverse cardiovascular outcomes; RCT, randomized control trials; RR, risk reduction; SGLT2i, sodium-glucose cotransporter 2 inhibitor.
Summary of randomized control trials that tested the efficacy of different GLP1-RA drugs for various primary outcomes.
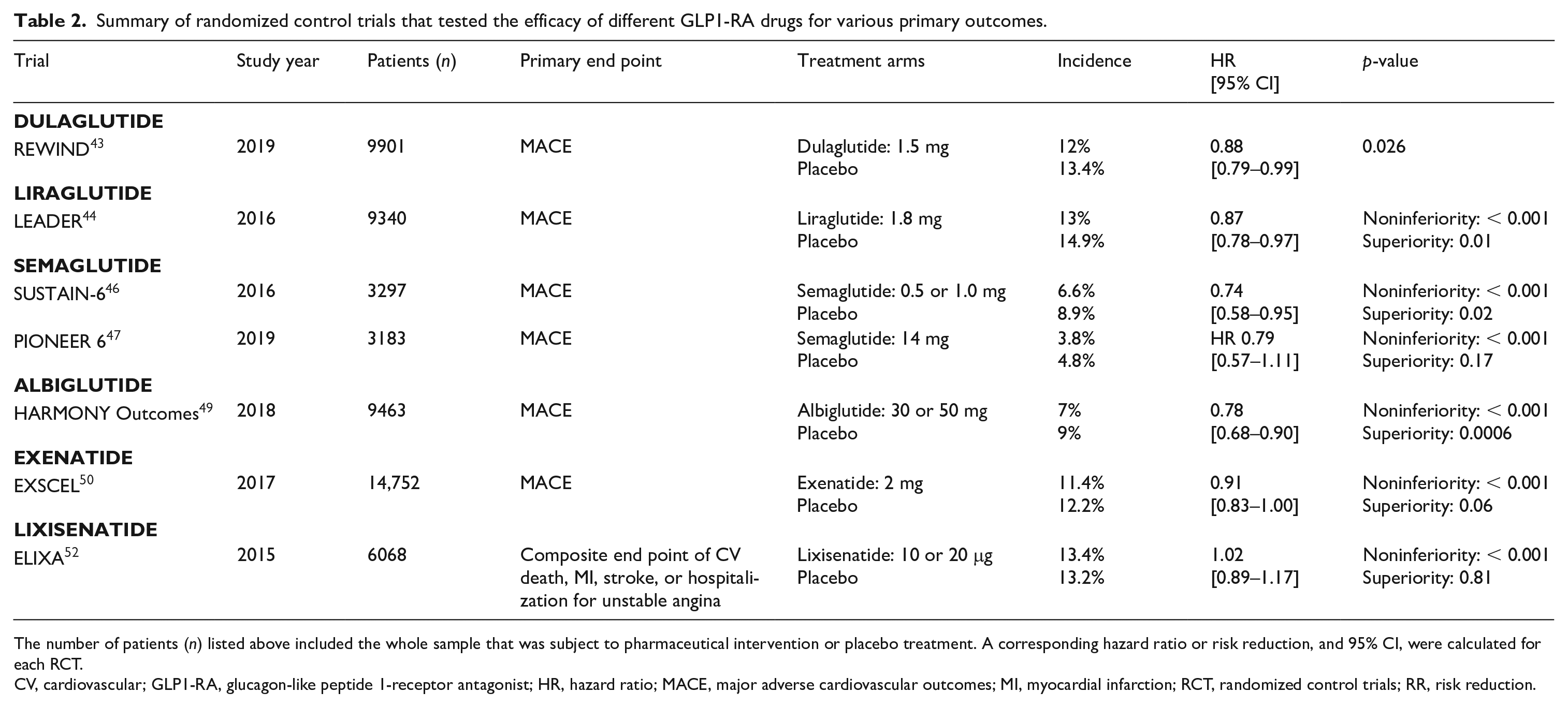
The number of patients (n) listed above included the whole sample that was subject to pharmaceutical intervention or placebo treatment. A corresponding hazard ratio or risk reduction, and 95% CI, were calculated for each RCT.
CV, cardiovascular; GLP1-RA, glucagon-like peptide 1-receptor antagonist; HR, hazard ratio; MACE, major adverse cardiovascular outcomes; MI, myocardial infarction; RCT, randomized control trials; RR, risk reduction.
Summary of the randomized control trials or post hoc analyses that tested the efficacy of different SGLT2i or GLP1-RA drugs for various primary outcomes.
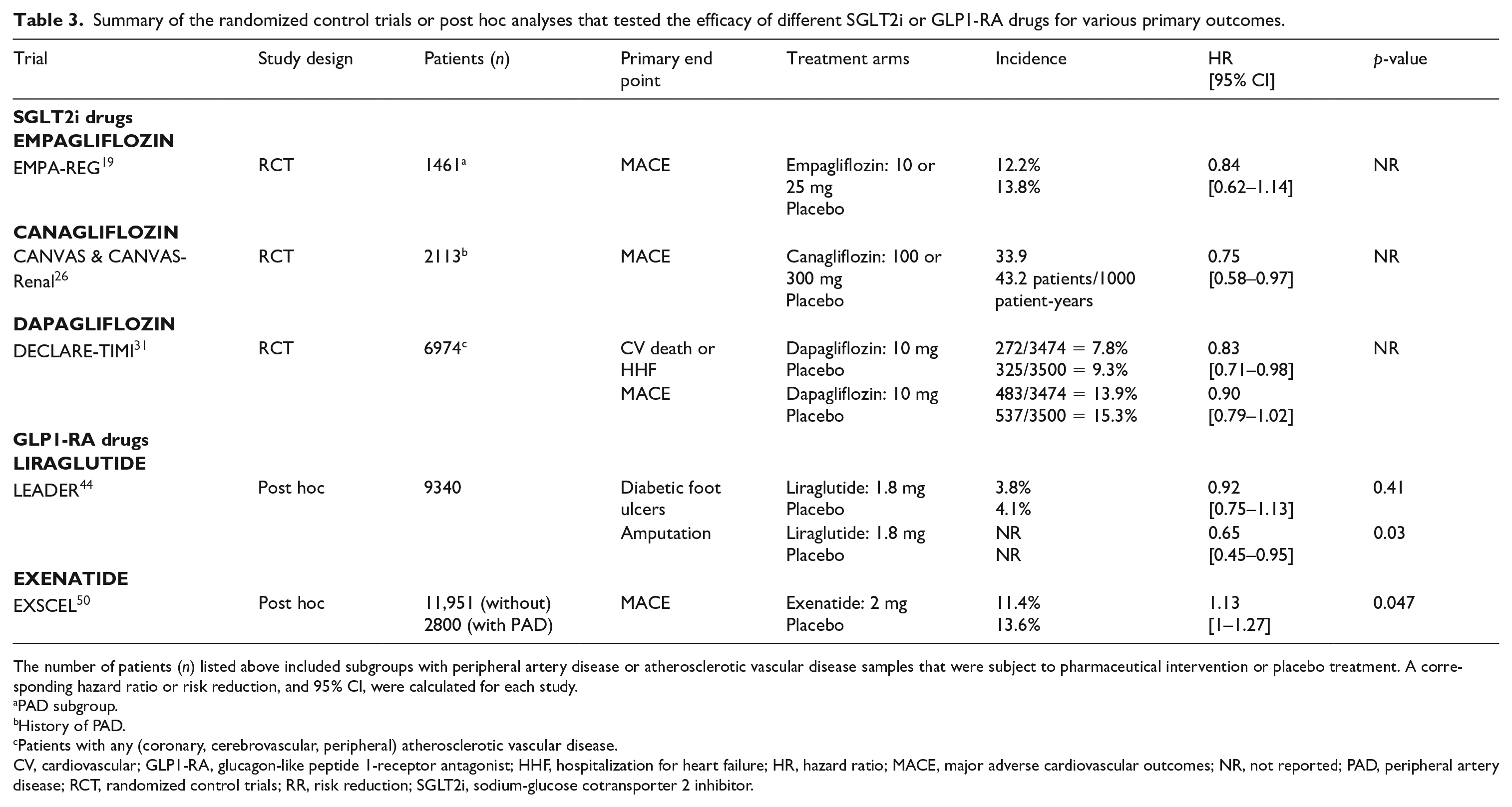
The number of patients (n) listed above included subgroups with peripheral artery disease or atherosclerotic vascular disease samples that were subject to pharmaceutical intervention or placebo treatment. A corresponding hazard ratio or risk reduction, and 95% CI, were calculated for each study.
PAD subgroup.
History of PAD.
Patients with any (coronary, cerebrovascular, peripheral) atherosclerotic vascular disease.
CV, cardiovascular; GLP1-RA, glucagon-like peptide 1-receptor antagonist; HHF, hospitalization for heart failure; HR, hazard ratio; MACE, major adverse cardiovascular outcomes; NR, not reported; PAD, peripheral artery disease; RCT, randomized control trials; RR, risk reduction; SGLT2i, sodium-glucose cotransporter 2 inhibitor.
Empagliflozin
In the Empagliflozin CV Outcome Event Trial in Type 2 Diabetes Mellitus Patients (EMPA-REG OUTCOME) trial, compared to placebo, empagliflozin was associated with lower rates of the composite MACE (HR, 0.86; 95.02% CI, 0.74 to 0.99; p = 0.04 for superiority), CV mortality (3.7% vs 5.9%; 38% relative risk reduction), HHF (2.7% and 4.1%; 35% relative risk reduction), and death from any cause (5.7% and 8.3%; 32% relative risk reduction).
The difference between the two groups occurred early in the trial, and the reduction of primary CV composite was consistent for both empagliflozin doses. Furthermore, The EMPA-REG OUTCOME trial showed no significant difference in lower-limb amputation risk between the two groups. 19
Based on a subanalysis of the EMPA-REG OUTCOME trial, 1461 patients (20.8%) had PAD at baseline (982 treated with empagliflozin, 479 treated with placebo). Although empagliflozin did not lead to significant improvement of three-point MACE (HR, 0.84; 95% CI, 0.62 to 1.14), it was associated with significant reduction of CV death (HR, 0.57; 95% CI, 0.37 to 0.88), all-cause mortality (HR, 0.62; 95% CI, 0.44 to 0.88), HHF (HR, 0.56; 95% CI, 0.35 to 0.92), and incidence of or worsening nephropathy (HR, 0.54; 95% CI, 0.41 to 0.71). Furthermore, among patients with PAD, there was a numerically lower risk for limb amputation that occurred in 5.5% with empagliflozin versus 6.3% with placebo (HR, 0.84; 95% CI, 0.54 to 1.32). There was no significant difference in adverse events between patients with or without PAD. The reduction in CV death with empagliflozin in patients with T2D and PAD translates to a number needed to treat of 29 patients over 3.1 years to prevent one event. 20 Large-scale safety pooled analysis of phase I–III clinical trials revealed the same low rate of lower-limb amputations in patients with T2D treated with empagliflozin and placebo of 1.1%. 21 This was consistent with postmarketing surveillance analysis of empagliflozin in Japanese patients with T2D. 22
Both EMPEROR-Reduced and -Preserved trials showed a significant risk reduction of CV death and HHF in patients with reduced (⩽ 40%) (HR, 0.75; 95% CI, 0.65 to 0.86; p < 0.001) and preserved (> 40%) (HR, 0.79 ; 95% CI, 0.69 to 0.90; p < 0.001) ejection fraction (EF) in adult patients with heart failure (HF) and elevated pro-brain natriuretic peptide (pro-BNP).23,24 Empagliflozin is currently approved by the US Food & Drug Association (FDA) to reduce the risk of CV death in patients with T2D and established CVD, to reduce the risk of CV death and HHF in patients with HF (regardless of EF%), and in addition, to control blood sugar along with diet and exercise in patients with T2D. 25
Canagliflozin
The combined Canagliflozin Cardiovascular Assessment (CANVAS) and CANVAS-Renal (CANVAS-R) Studies demonstrated a significant reduction in the primary endpoint of MACE compared to placebo (HR, 0.86; 95% CI, 0.75 to 0.97; p < 0.001 for noninferiority and p = 0.02 for superiority) but with no significant difference in CV death. The risk of lower-extremity amputation (primarily at the level of toe or metatarsal), was almost doubled with canagliflozin versus placebo (6.3 vs 3.4 participants per 1000 patient-years: HR, 1.97; CI, 1.41 to 2.75). A total of 187 participants underwent amputation during follow-up, with a total of 290 amputation events; 123 participants experienced a single amputation event with 71% of the affected participants having minor amputation at the level of the toe or metatarsal. The highest absolute risk of amputation occurred among patients with a history of amputation or PAD, but the relative risk of amputation with canagliflozin as compared with placebo was similar across these subgroups. 26
A subanalysis of the CANVAS Program demonstrated an increased risk of amputation (mainly minor) with canagliflozin compared to placebo. Anticipated risk factors for amputation included prior history of amputation, PAD, and neuropathy, but no specific etiological mechanism or at-risk subgroup for canagliflozin was identified. 27
The FDA had previously issued a black box warning of lower-limb amputations associated with the use of canagliflozin, which was later removed. 28
The primary end point (a composite of end-stage renal disease (ESRD), a doubling of the serum creatinine level, or death from renal or CV causes) of the Canagliflozin and Renal Outcomes in T2D and Nephropathy (CREDENCE) trial was lower in the canagliflozin group than in the placebo group, with event rates of 43.2 and 61.2 per 1000 patient-years, respectively (HR, 0.70; 95% CI, 0.59 to 0.82; p = 0.00001). The canagliflozin group also had a lower risk of MACE (HR, 0.80; 95% CI, 0.67 to 0.95; p = 0.01) and HHF (HR, 0.61; 95% CI, 0.47 to 0.80; p < 0.001). Although the trial was not powered to investigate the amputation rate, it was reported in 12.3 and 11.2 per 1000 patient-years (HR, 1.11; 95% CI, 0.79 to 1.56) in the canagliflozin and placebo group, respectively. 29
A pooled analysis of CANVAS and CREDENCE trials aimed to assess the effects of canagliflozin in patients with and without PAD. Of the total 14,543 participants from both trials, 3159 (21.7%) had PAD at baseline. Canagliflozin reduced MACE (HR, 0.76; 95% CI, 0.62 to 0.92) in patients with PAD, with similar relative benefits for other CV and renal outcomes in participants with or without PAD at baseline (all p-interaction > 0.268). No increase in the relative risk of extended MALE was observed with canagliflozin, regardless of the PAD status (p-interaction > 0.864). Furthermore, the absolute benefits of canagliflozin were greater in those with PAD. 30
Canagliflozin is now FDA-approved as an adjunct to diet and exercise to improve glycemic control in T2D, reduce the risk of MACE in adults with T2D and established CVD, and reduce the risk of ESRD, doubling serum creatinine, CV death, and HHF in adults with T2D and diabetic nephropathy with albuminuria. 28
Dapagliflozin
Although the Dapagliflozin on the Incidence of Cardiovascular Events-Thrombolysis In Myocardial Infarction-58 (DECLARE-TIMI 58) trial 31 showed no significant risk reduction of MACE (HR, 0.93; 95% CI, 0.84 to 1.03; p = 0.17) with dapagliflozin versus placebo, it did result in a lower rate of CV death or HHF (4.9% vs 5.8%; HR, 0.83; 95% CI, 0.73 to 0.95; p = 0.005), which reflected a lower rate of HHF (HR, 0.73; 95% CI, 0.61 to 0.88), but no difference in CV death per se (HR, 0.98; 95% CI, 0.82 to 1.17). A renal event occurred in 4.3% in the dapagliflozin group and in 5.6% in the placebo group (HR, 0.76; 95% CI, 0.67 to 0.87), and death from any cause occurred in 6.2% and 6.6%, respectively (HR, 0.93; 95% CI, 0.82 to 1.04).
Based on a focused collection of events, there was no evidence of a higher risk of amputations with dapagliflozin versus placebo. Of the 17,160 patients enrolled in this trial, 1025 (6%) had PAD. In a prespecified subanalysis, patients with PAD had higher risk of MACE, CV death, HHF, and renal events compared with those without, regardless of being on dapagliflozin or placebo. The absolute risk reduction with dapagliflozin was greater among patients with PAD compared to those without, at 1.4% versus 0.9% for CV death or HHF, and 2.1% versus 1.3% for renal events. However, the relative risk reduction with dapagliflozin in patients with PAD versus without was consistent for CV death or HHF (HR 0.86 vs 0.82, p-interaction = 0.79) and renal events (HR 0.78 vs 0.76, p-interaction = 0.84). No significant difference in limb events was observed between dapagliflozin and placebo: 3.37% versus 3.16% for limb ischemia (HR, 1.07; 95% CI, 0.90 to 1.26; p = 0.45) and 1.43% vs 1.32% for amputation (HR, 1.09; 95% CI, 0.84 to 1.40; p = 0.53). Furthermore, there were no significant interactions by history of PAD versus not. 32
The Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction (DAPA-HF) trial 33 revealed significant risk reduction of the primary outcome of worsening HF (hospitalization or an urgent visit resulting in intravenous therapy for HF) or CV death with dapagliflozin (16.3%) than placebo (21.2%) (HR, 0.74; 95% CI, 0.65 to 0.85; p < 0.001). There was also significant risk reduction of worsening HF (HR, 0.70; 95% CI, 0.59 to 0.83), CV death (HR, 0.82; 95% CI, 0.69 to 0.98), and death from any cause (HR, 0.83; 95% CI, 0.71 to 0.97). Findings were consistent in patients with or without T2D. There was no significant difference in amputation risk.
The Dapagliflozin in Patients with Chronic Kidney Disease (DAPA-CKD) trial’s 34 primary outcome of a composite of a sustained decline in the eGFR of at least 50%, ESRD, or death from renal or CV causes, was significantly lower (9.2%) with dapagliflozin versus placebo (14.5%) (HR, 0.61; 95% CI, 0.51 to 0.72; p < 0.001). Death from renal causes was also significantly reduced (HR, 0.56; 95% CI, 0.45 to 0.68; p < 0.001). The effects of dapagliflozin were consistent in patients with and without T2D. There was no significant difference in amputation risk between the two groups.
In a pool-analysis of safety data from Phase IIb/III clinical trials that assessed amputation risk with dapagliflozin, lower-limb amputation was reported in eight (0.1%) and seven (0.2%) patients treated with dapagliflozin and placebo, respectively. Risk factors for amputation in both groups were neuropathy, CVD, dyslipidemia, and nephropathy. The onset of events in the dapagliflozin group occurred after 150 days, which was similar to the control group. 35
Dapagliflozin is now FDA approved as an adjunct to diet and exercise to improve glycemic control in adults with T2D, to reduce the risk of HHF in adults with T2D and either established or multiple CV risk factors, to reduce the risk of CV death and HHF in adults with heart failure and reduced ejection fraction (HFrEF) (NYHA class II–IV), and reduce the risk of sustained eGFR decline, ESRD, CV death, and HHF in adults with CKD at risk of progression.
Ertugliflozin
The Cardiovascular Outcomes Following Ertugliflozin Treatment in Patients with Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease (VERTIS-CV) trial 36 revealed no significant risk reduction of MACE with ertugliflozin versus placebo (HR, 0.97; 95.6% CI, 0.85 to 1.11; p < 0.001 for noninferiority). The differences in CV death or HHF were not significant either (HR, 0.88; 95.8% CI, 0.75 to 1.03; p = 0.11 for superiority). Amputations were performed in 54 patients (2.0%) who received the 5-mg dose of ertugliflozin and in 57 patients (2.1%) who received the 15-mg dose, as compared with 45 patients (1.6%) who received placebo.
Sotagliflozin
Sotagliflozin is a dual SGLT2 and 1 inhibitor that has also been shown to have beneficial cardiorenal effects. 37
The Effect of Sotagliflozin on Cardiovascular and Renal Events in Patients with Type 2 Diabetes and Moderate Renal Impairment Who Are at Cardiovascular Risk (SCORED) trial’s 38 primary endpoint, which was changed during the trial to the total number of CV death, HHF, and urgent visits for HF, was significantly lower with sotagliflozin versus placebo (5.6% vs 7.5%; HR, 0.74; 95%; CI, 0.63 to 0.88; p < 0.001). The original endpoint of MACE and co-primary endpoint of CV death or HHF were also lower with sotagliflozin: HR, 0.84; 95% CI, 0.72 to 0.99 and HR, 0.77; 95% CI, 0.66 to 0.91, respectively. The trial ended early owing to a loss of funding.
The Effect of Sotagliflozin on Cardiovascular Events in Patients with Type 2 Diabetes Post Worsening Heart Failure (SOLOIST-WHF) trial 39 revealed a significantly lower primary endpoint of CV death and hospitalizations and urgent visits for HF in the sotagliflozin group than with placebo (51.0% vs 76.3%; HR, 0.67; 95% CI, 0.52 to 0.85; p < 0.001). Adverse events leading to amputations occurred in four versus one patient (0.7% vs 0.2%) in the sotagliflozin and placebo groups, respectively. This trial also ended early because of loss of funding from the sponsor.
Glucagon-like peptide 1-receptor agonists (GLP1-RAs)
Mechanism of action
GLP-1 is secreted directly in response to food intake from L-cells in the distal ileum 40 ; then becomes inactivated after 2–3 minutes by the dipeptidyl peptidase-4 (DPP-4) enzyme. 41 GLP-1 production is reduced in patients with T2D. Following administration of a GLP1-RA, stimulated secretion of supraphysiological concentration of GLP-1 reduces glucose by increasing glucose-dependent insulin secretion, decreasing glucagon secretion, and delaying gastric emptying, which in turn reduces postprandial hyperglycemia. 11 GLP1-RAs also have beneficial CV effects including weight and blood pressure reduction, natriuresis, and triglyceride level reduction in addition to anti-inflammatory properties. 11 As a result, GLP1-RAs have atherosclerotic and antithrombotic effects. 42 There are six GLP1-RA subcutaneous (SC) injectable agents that are currently available including short-acting (short-acting exenatide, lixisenatide), intermediate-acting (liraglutide), and long-acting (long-acting exenatide, dulaglutide, albiglutide, and semaglutide). An oral formulation of semaglutide has been later introduced to the US market.
Reduction in atherosclerotic cardiovascular disease
Several trials have evaluated these agents for their CV benefits. These trials compared dulaglutide (REWIND), liraglutide (LEADER), semaglutide (SUSTAIN-6), oral semaglutide (PIONEER 6), albiglutide (HARMONY), long-acting exenatide (EXSCEL), and lixisenatide (ELIXA) to placebo as add on to standard diabetic care for follow up at 2–4 years (Tables 2 and 3).
To date, liraglutide, semaglutide SC, and dulaglutide are the only agents approved by the FDA to reduce the risk of MACE in adults with T2D and established CVD, with dulaglutide being the only agent also approved for CVD reduction in patients without established atherosclerotic cardiovascular disease (ASCVD). 27
Dulaglutide
The Researching CV Events with a Weekly Incretin in Diabetes (REWIND) trial 43 randomized 9901 patients with T2D and previous CV events or risk, to either weekly SC dulaglutide (1.5 mg) or placebo. Dulaglutide reduced the risk of three-point MACE significantly (HR, 0.88; 95% CI, 0.79 to 0.99; p = 0.026). These results were consistent across the subgroups of patients with and without known CV events and were driven by significant risk reduction of stroke (HR, 0.76; 95% CI, 0.62 to 0.94). Though there was no significant risk reduction in CV death, gastrointestinal adverse events were significantly higher (p < 0.0001).
Liraglutide
The LEADER (Liraglutide Effect and Action in Diabetes: Evaluation of CV Outcome Results) trial 44 randomized 9340 patients with T2D and high CV risk (81% had established CVD) to receive liraglutide or placebo. The primary outcome of MACE occurred significantly lower with liraglutide (13.0%) than placebo (14.9%) (HR, 0.87; 95% CI, 0.78 to 0.97; p < 0.001 for noninferiority; p = 0.01 for superiority). In addition, CV death was also lower with liraglutide (4.7%) than placebo (6.0%) (HR, 0.78; 95% CI, 0.66 to 0.93; p = 0.007). There was no difference in fatal, nonfatal, and silent MI or HHF.
Based on a post hoc analysis of the LEADER trial, during a median of 3.8 years’ follow-up, similar proportions of patients reported at least one episode of diabetic foot ulcer (DFU) in the liraglutide and placebo groups (176/4668) versus 4.1% (191/4672), respectively (HR, 0.92; 95% CI, 0.75, 1.13; p = 0.41). Analysis of DFU-related complications demonstrated a significant reduction in amputations with liraglutide versus placebo (HR, 0.65; 95% CI, 0.45, 0.95; p = 0.03). However, no differences were found for foot infections, involvement of underlying structures, or peripheral revascularization in the main analysis. 45
Liraglutide and PAD (STARDUST) is an ongoing open-label, two-arm randomized control trial, aimed at evaluating the effects of liraglutide on peripheral perfusion, as compared with the aggressive treatment of cardio-metabolic risk factors, in people with T2D and PAD. The estimated study completion is June 30, 2022.
Semaglutide
The Trial to Evaluate CV and Other Long-Term Outcomes with Semaglutide in Subjects with T2D (SUSTAIN-6) 46 enrolled 3297 patients using the same trial inclusion criteria and the same primary composite endpoint as LEADER. Semaglutide reduced the three-point MACE (HR, 0.74; 95% CI, 0.58 to 0.95; p < 0.001 for noninferiority), mostly driven by reduction of nonfatal stroke (HR, 0.61; 95% CI, 0.38 to 0.99; p = 0.04). No reduction in all-cause mortality, CV mortality, or HHF was observed with semaglutide compared to placebo.
The Peptide Innovation for Early Diabetes Treatment (PIONEER 6) trial, 47 enrolled 3183 patients with established CV or CKD to receive oral semaglutide or placebo. There was no significant difference in MACE, which occurred in 3.8% in the oral semaglutide and 4.8% in the placebo group (HR, 0.79; 95% CI, 0.57 to 1.11; p < 0.001 for noninferiority).
A Research Study to Compare Semaglutide Against Placebo in People With PAD and T2D (STRIDE) is an ongoing trial to determine if weekly injectable semaglutide affects walking ability compared with placebo in people with PAD and T2D. The estimated study completion date is August 17, 2023. In a post hoc analysis of LEADER and SUSTAIN 6 trials, 1184/9340 (12.7%) and 460/3297 (14.0%) patients had PAD at baseline, respectively. In both studies, patients with PAD had around 35% greater risk of MACE irrespective of treatment, compared to those without (LEADER: HR, 1.36; 95% CI, 1.17 to 1.58; SUSTAIN 6: HR, 1.33; 95% CI, 0.94 to 1.83). The benefit of MACE risk reduction was consistent in patients with PAD (liraglutide: HR, 0.77; 95% CI, 0.58 to 1.01; semaglutide: HR, 0.61; 95% CI, 0.33 to 1.13) and in those without (liraglutide: HR, 0.89; 95% CI, 0.79 to 1.00; semaglutide: HR, 0.77; 95% CI, 0.58 to 1.01; p-interaction = 0.34 for liraglutide and 0.49 for semaglutide). However, absolute risk reductions for MACE with both medications were greater in patients with PAD (liraglutide: 4.13%-point, 95% CI, −0.15 to 8.42; semaglutide: 4.63%-point, 95% CI, −0.58 to 9.84) versus without (liraglutide: 1.42%-point, 95% CI, −0.03 to 2.87; semaglutide: 1.90%-point, 95% CI, 0.00 to 3.80). 48
Albiglutide
The Albiglutide and CV outcomes in patients with T2D and cardiovascular disease (HARMONY Outcomes) trial randomized 9463 patients with T2D and CVD to receive weekly SC albiglutide or placebo. In a median follow-up period of 1.5 years, albiglutide was found to be superior for the primary composite endpoint (MACE) compared with placebo (HR, 0.78; 95% CI, 0.68–0.90; p = 0.0006). The risk of fatal or nonfatal MI and the expanded composite outcome of CV death, MI, stroke, or urgent coronary revascularization for unstable angina were lower with albiglutide, but rates of CV death, fatal or nonfatal stroke, all-cause mortality, and the composite of CV death or HHF were similar compared to placebo. 49 The drug was withdrawn from the market due to economic reasons.
Exenatide
The Exenatide Study of Cardiovascular Event Lowering (EXSCEL) trial randomized 14,752 patients with T2D with or without CVD to receive weekly SC exenatide (2 mg) or a matching placebo for a median follow-up of 3.2 years. Exenatide was found to be noninferior to placebo for the primary outcome of composite MACE (HR, 0.91; 95% CI, 0.83 to 1.00; p < 0.001 for noninferiority and p = 0.06 for superiority). 50
Based on a post hoc analysis, 2800 (19%) of the EXSCEL patients had PAD and were less likely to be treated with optimal secondary prevention therapy for CVD. Patients with PAD had higher unadjusted and adjusted rates of MACE compared with patients without (13.6% vs 11.4%, respectively) (adjusted HR, 1.13; 95% CI, 1.00 to 1.27; p = 0.047). Furthermore, patients with PAD had higher all-cause mortality (adjusted HR, 1.38; 95% CI, 1.20 to 1.60; p < 0.001) and more frequent lower-extremity amputation (adjusted HR, 5.48; 95% CI, 4.16 to 7.22; p < 0.001). Patients treated with exenatide, or placebo, had similar rates of MACE and amputation, regardless of PAD status. Patients with PAD had similar adjusted rates of MACE, higher rates of CV and all-cause mortality, and higher adverse lower-limb events compared to patients without. There was no difference in MACE or amputation rates with exenatide versus placebo regardless of PAD status; however, exenatide was associated with lower all-cause mortality in patients with T2D and PAD. 51
Lixisenatide
The Evaluation of Lixisenatide in Acute Coronary Syndrome (ELIXA) trial 52 randomized 6068 patients with T2D and a history of acute coronary syndrome within 180 days to receive lixisenatide or placebo for a follow-up period of 2.1 years. Although lixisenatide was found to be noninferior, it failed to demonstrate a significant reduction in four-point MACE composite outcome (CV death, nonfatal MI, nonfatal stroke, or hospitalization for unstable angina) (HR, 1.02; 95% CI, 0.89 to 1.17; p = 0.81). There were no significant differences in the rate of HHF (HR, 0.96; 95% CI, 0.75 to 1.23) or the rate of death (HR, 0.94; 95% CI, 0.78 to 1.13).
Discussion
Sodium-glucose cotransporter 2 inhibitor (SGLT2i)
Although there was a concern about a possible increased risk of amputation reported with canagliflozin versus placebo in the CANVAS trial, this was not confirmed based on a pooled analysis of CANVAS and CREDENCE trials. Furthermore, such correlation was not reported in the trials with other SGLT2i agents.
Based on a pooled analysis of data from CANVAS and CREDENCE trials, participant characteristics exhibited multiple statistical differences between the two studies, reflecting the large sample sizes, but trial populations were mostly similar. Key exceptions were CVD history at baseline (65.6% in the CANVAS Program vs 50.4% in CREDENCE) and CKD at baseline (17.5% nephropathy and 20.1% eGFR < 60 mL/min/1.73 m2 in the CANVAS Program vs 100.0% nephropathy and 59.8% eGFR < 60 mL/min/1.73 m2 in CREDENCE). In the CANVAS Program, 2.3% of patients had a history of amputation at trial entry versus 5.3% in CREDENCE. Some attributed the discrepancy in amputation risk between CANVAS and CREDENCE trials due to the differences between the two populations. Key differences include the exclusion of patients who had a history of atraumatic amputations from the CREDENCE study, the proportion of patients with CVD history at baseline, and the update in CREDENCE trial protocol to minimize the adverse effect of canagliflozin on amputation (130 participants (3%) permanently discontinued the study drug for reasons related to amputation risk: 20 due to perceived risk and 110 as a result of a lower-limb adverse event). However, the analysis found no increase in amputation risk in the canagliflozin group compared to placebo before or after the protocol update (HR, 1.03; 95% CI, 0.70 to 1.54 vs HR, 1.34; 95% CI, 0.69 to 2.66). The authors concluded that none of these differences could have explained the discrepancy in amputation risk between the two trials. 53
There have been different proposed mechanisms behind the increased risk of amputation. Glycosuria-induced osmotic diuresis and hypovolemia that worsen the hypo-perfusion to distal extremities have been proposed as a potential mechanism for increased risk of lower-limb amputation with SGLT2i drugs, especially canagliflozin. 54 This was supported by the SURDIAGENE prospective observational cohort study that demonstrated a significant increase in lower-limb events, especially lower-limb amputation in diabetic patients treated with diuretics. The incidence of lower-limb events was 1.80 per 100 patient-years in diuretic users versus 1.00 in nonusers (p < 0.001). 55 A meta-analysis of randomized control trials of SGLT2i drugs in patients with T2D showed a positive association between SGLT2i-induced blood pressure and weight reduction and the risk of lower-limb events including amputation and PAD. 56 However, a cohort study using data from the Clinical Practice Research Datalink GOLD (2013–2019) demonstrated using SGLT2i drugs in 51,847 patients, with or without signs of hypovolemia, was not associated with an increased risk of lower-limb amputation. 57
Six observational studies using the Truven MarketScan health claims database from the USA were conducted to investigate the effect of SGLT2i drugs use on amputation risk. The results vary from no difference in amputation risk with canagliflozin compared with non-SGLT2i glucose-lowering agents in three studies58–60 and lower amputation risk with SGLT2i drugs (where canagliflozin therapy comprised 70% of SGLT2i use) compared with sulfonylurea agents, but not compared to DPP-4 inhibitors in two studies61,62 and only one study that showed a higher amputation risk with SGLT2i drugs compared with DPP-4 inhibitors, but not compared with sulfonylurea agents or non-metformin, non-SGLT2i glucose-lowering agents. 63
Another observational study using the US Department of Defense Health System showed that patients newly started on SGLT2i drugs had a nearly twofold increased risk of lower-limb amputation compared to non-SGLT2i antidiabetic agents (HR, 1.99; 95% CI, 1.12 to 3.51), with most of the amputations occurring with canagliflozin. 64 The same findings were noted in another observational study using the Swedish/Danish National Register, which found increased amputation incidence with SGLT2i drugs compared with GLP1-RA (HR, 2.48; 95% CI, 1.14 to 5.40). 65
In a large multicenter observational study, using administrative data from seven Canadian provinces and the UK, there was no evidence of an association between SGLT2i use and incidence of below-knee amputation compared with DPP-4 inhibitor use in patients with T2D (HR, 0.88; 95% CI, 0.71 to 1.09). Furthermore, no association has been noticed with specific SGLT2i drugs, including canagliflozin, compared with DPP-4 inhibitors. 66
Similarly, a retrospective analysis from the Taiwan National Health Insurance Database found no significant difference in lower-limb amputation between SGLT2i drugs compared to DPP-4 inhibitors, GLP1-RA, and other older medications. 67
This heterogeneity of results among previous observational studies may be explained by the differences in studies’ population, methods, comparator drugs, extent of amputation recorded, follow-up duration, and inclusion or exclusion of patients with a history of amputations. 66
Until strong data are available, SGLT2i drugs and specially canagliflozin may need to be avoided in patients with high risk for amputation (previous amputation, ischemic ulcers, or neuropathy).
Glucagon-like peptide 1-receptor agonists (GLP1-RAs)
Previous trials have not exclusively addressed PAD or lower-limb complication as a primary or secondary endpoint. Although a post hoc analysis of the LEADER trial 44 demonstrated a reduction in amputations with liraglutide versus placebo, there is a significant lack of evidence regarding the effect of GLP1-RAs on MALE in the literature.
A real-world study has evaluated the role of GLP1-RA in clinical practice, which showed a favorable effect of this class in 9772 patients with T2D. Use of GLP1-RAs was associated with lower rates of death, cerebrovascular disease and ischemic stroke, peripheral vascular disease, and lower-limb complications compared with other standards of care therapies. 68 The same was seen with liraglutide compared to DPP-4 inhibitors in a Scandinavian population-based study. 69 A retrospective analysis conducted using the Taiwan National Health Insurance database has shown that GLP1-RAs were associated with significantly lower risks of MALE when compared with DPP-4 inhibitors. MALE was defined as PAD, critical limb ischemia, percutaneous transluminal angioplasty, or peripheral bypass for PAD. This was mainly driven by the reduction of the amputation rate. Moreover, GLP1-RAs were also associated with lower risks of MACE and death from any cause. 70
Based on a network meta-analysis involving 764 trials including 421,346 patients, authors suggested utilizing SGLT2i or GLP1-RA drugs in patients with T2D and CV risk since both drug classes have been proven to reduce the risk of all-cause and CV mortality in addition to MI. However, benefits vary across different CV risks, high versus low, which require risk-stratified management. Whereas some SGLT2i drugs were associated with reduced mortality and HHF, other drugs from the GLP1-RA class were associated with lower nonfatal stroke risk. 71
With both classes showing CV benefits, independently from metformin, 72 there is a push towards using these agents in patients with elevated ASCVD risk ahead of metformin. However, risks and benefits, side effects, and costs need to be discussed with patients before drug initiation. 73 Standardized implementation of these recommendations is urgently required since these agents continue to be under-prescribed, reaching only 6% for SGLT2i drugs and 3% for GLP1-RAs. Based on this study, metformin and DPP-4 inhibitors were the most commonly used drug classes despite the lack of CV benefit. 74
Current societal guideline recommendations
Emerging evidence of CV benefits of the antihyperglycemic therapies has surely had its impact on the management of diabetic patients and, for the first time, the selection of antidiabetic is not solely dependent on HbA1c levels. A multidisciplinary approach, involving cardiologists and diabetologists, is required for better care and outcomes in patients with T2D and CV risk. None of the current societal guidelines made specific recommendations primarily for patients with PAD (Table 4).
Summary of societal guideline recommendations for antiglycemic therapies.

CKD, chronic kidney disease; CV, cardiovascular; CVD, cardiovascular disease; DKD, diabetic kidney disease; DM, diabetes mellitus; eASCVD, established atherosclerotic cardiovascular disease; eGFR, estimated glomerular filtration rate; GLP1-RA, glucagon-like peptide 1-receptor agonist; HF, heart failure; HFmrEF, heart failure with mildly reduced ejection fraction; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; KD, kidney disease ; SGLT2i, sodium-glucose cotransporter 2 inhibitor; T2D, type 2 diabetes.
2021 American Diabetic Association Standards
Among patients with T2D with established ASCVD or indicators of high-risk, established kidney disease (KD), or HF, a SGLT2i or GLP1-RA with demonstrated CV benefit is recommended as part of the glucose-lowering regimen independent of A1c, and in consideration of patient-specific factors. In patients with T2D, a GLP1-RA is preferred to insulin when possible. In patients with HFrEF, SGLT2i with proven benefit in this population should be considered. In patients with CKD and albuminuria, a SGLT2i with primary evidence of reducing CKD progression should be considered. GLP1-RA with proven CV benefit is an alternative if SGLT2i drugs are not tolerated. 75
2019 American College of Cardiology (ACC)/American Heart Association (AHA) Guideline on the Primary Prevention of Cardiovascular Disease
The American Heart Association (AHA) recommends screening for ABI in asymptomatic diabetic patients, age 50–64 years or < 50 years, with at least one additional risk factor for atherosclerosis if results would impact the management plan. For patients with diabetes and established ASCVD (e.g., PAD), an HbA1c goal of < 7 should be considered. Less stringent goals may be accepted in older patients with multiple comorbidities.
For adults with T2D and additional ASCVD risk factors who require glucose-lowering therapy despite initial lifestyle modifications and metformin, it may be reasonable to initiate an SGLT2i or a GLP1-RA to improve glycemic control and reduce CV risk. 76
2020 ACC Expert Consensus Decision Pathway on Novel Therapies for Cardiovascular Risk Reduction in Patients with Type 2 Diabetes
In patients with T2D and ASCVD, recommend starting SGLT2i or GLP1-RA with CV benefit depending on patient-specific factors and comorbidities. Patients with concurrent DKD and/or HF consider a SGLT2i. Alternatively, a GLP1-RA can be considered for eGFR < 30 mL/min/1.73 m2. At hospital discharge (with close outpatient follow-up) after admission for an ASCVD, consider an SGLT2i or GLP1-RA, and an SGLT2i in case of HFrEF. 11
2019 European Society of Cardiology Guidelines on DM and CVD
Empagliflozin, canagliflozin, dapagliflozin, liraglutide, semaglutide, or dulaglutide are recommended over metformin as monotherapy in patients with T2D and CVD, or at very high/high CV risk, to reduce CV events. Furthermore, empagliflozin or liraglutide are recommended in patients with T2D and CVD, or at very high/high CV risk, to reduce the risk of death. 77
More recent position statements from the ESC provided an evidence-based summary supporting the use of SGLT2i in patients with HF, T2D with CV risk, and CKD. 78 In patients with HFrEF (with or without T2D), dapagliflozin or empagliflozin reduced the risk of CV death and HHF.
Although empagliflozin and canagliflozin reduced the risk of MACE in patients with T2D at high/very high risk of CVD, empagliflozin was also associated with reduced CV death in this population.
In patients with T2D at high/very high risk of CVD, empagliflozin, canagliflozin, or dapagliflozin reduced the risk of CV death or HHF. However, ertugliflozin was only associated with a reduced risk of HHF. In patients with T2D, the risk of KD progression is reduced by empagliflozin, canagliflozin, or dapagliflozin.
In patients with T2D and proteinuric diabetic KD, progression to ESRD is reduced by canagliflozin or dapagliflozin. In patients with proteinuric CKD, with or without T2D, the risk of KD progression is reduced by dapagliflozin. Furthermore, in patients with T2D and CKD, the risk of CV death or HHF is reduced by canagliflozin, dapagliflozin, or sotagliflozin.
2022 ACC/AHA/Heart Failure Society of America (HFSA) Guideline for the Management of Heart Failure
The new classification of HF is as follows: HFrEF: LVEF (left ventricular ejection fraction) ⩽ 40%; HFimpEF (HF with improved EF): previous LVEF ⩽ 40% and follow-up measurement of LVEF > 40%; HFmrEF (HF with mildly reduced EF): LVEF 41–49%; HFpEF (heart failure with preserved ejection fraction): LVEF ⩾ 50%. New recommendations were made for the use of SGLT2i in HF. In symptomatic patients with chronic HFrEF, SGLT2i is recommended to reduce hospitalization and CV mortality, regardless of the presence of T2D (Class of Recommendation 1a). SGLT2i can also be beneficial in patients with HFmrEF and HFpEF (Class of Recommendation 2a). 79
Though the 2016 AHA/ACC Guideline on The Management of Patients with Lower Extremity PAD did not refer to the use of SGLT2i or GLP1-RA, we expect the next update to include new recommendations regarding the use of these drugs in this high-risk patient population. 80
Figure 1 provides an evidence-based algorithm summarizing current recommendations to reduce CV risk in patients with T2D and CV risk such as PAD.
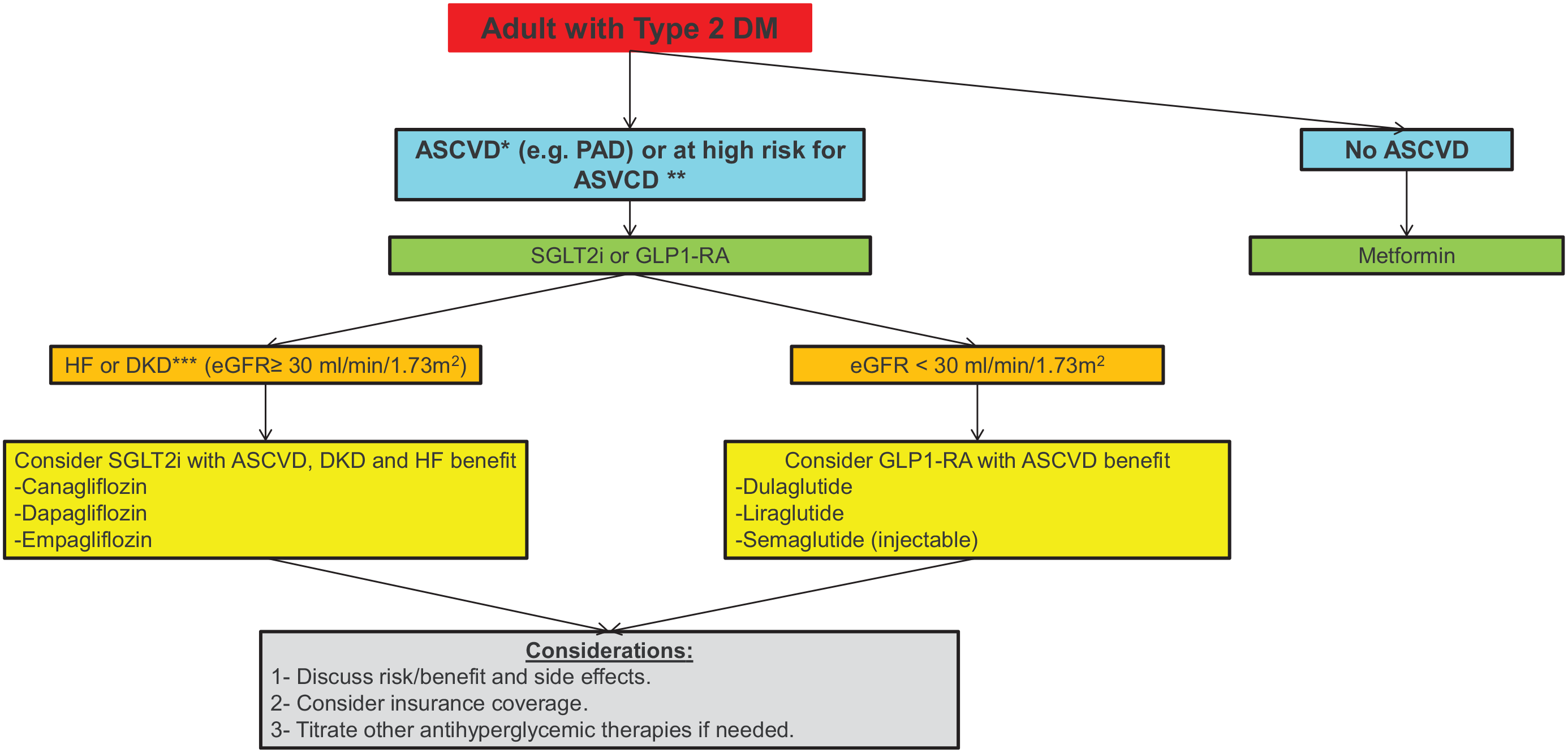
Evidence-based algorithm summarizing current recommendations to reduce CV risk in patients with T2D and CV risk such as PAD
Study limitations
This general review aims to summarize the current evidence regarding the role of SGLT2i and GLP1-RA drugs in reducing major CV and limb events in patients with or at risk for CVD, focusing on patients with PAD. However, none of the trials we reviewed exclusively enrolled patients with PAD. Furthermore, patients with PAD were underrepresented in most of these trials. Although the majority of trials considered MACE, HHF, and renal events as primary outcomes, MALE were not prespecified targets. Overall, the data were very limited to make any specific or strong recommendations to favor either drug class in patients with PAD.
Conclusions
Peripheral artery disease and T2D are two prevalent major health problems that have been associated with increased morbidity and mortality. These patients have continued to be at a significantly increased risk of CV complications and death despite major advances in medical therapy during the last two decades. SGLT2i and GLP1-RA are two promising drug classes that have shown a significant reduction of MACE in patients with T2D and CVD. Though some of the SGLT2i drugs have shown a significant reduction in CV mortality, HHF, and renal outcome, few GLP1-RA drugs have shown risk reduction of ischemic stroke. Certainly, the benefit of these therapeutics may extend to include patients with PAD. However, there is a significant lack of evidence regarding this population at this time, mainly based on post hoc analyses or real-world studies. Fortunately, additional research is underway to study the benefit exclusively in this population. Although there was a signal of increased amputation risk in the CANVAS trial with canagliflozin, a post hoc analysis of the LEADER trial showed a lower risk with liraglutide. Based on our review, we cannot provide strong recommendations in favor of one drug class in patients with PAD. However, until strong evidence is available, patients with PAD should be offered GLP1-RA if they have concurrent CKD (eGFR < 30 mL/min/1.73 m2) and other ASCVD with high amputation risk (previous amputation, ischemic ulcers, or neuropathy), and SGLT2i if they have concurrent HF and/or DKD.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
