Abstract
The incidence of depression has been rising rapidly, and depression has been recognized as one of the world’s leading causes of disability. More recently, depression has been associated with an increased risk of symptomatic atherosclerotic disease as well as worse perioperative outcomes in patients with cardiovascular disease. Additionally, recent studies have demonstrated an association between depression and peripheral artery disease (PAD), which has been estimated to affect more than 200 million people worldwide. These studies have identified that depression is associated with poor functional and surgical outcomes in patients with PAD. Although the directionality and specific mechanisms underlying this association have yet to be clearly defined, several biologic and behavioral risk factors have been identified to play a role in this relationship. These factors include tobacco use, physical inactivity, medical non-adherence, endothelial and coagulation dysfunction, and dysregulation of the hypothalamic-pituitary-adrenal axis, autonomic system, and immune system. In this article, we review these potential mechanisms and the current evidence linking depression and PAD, as well as future directions for research and interventional strategies. Understanding and elucidating this relationship may assist in preventing the development of PAD and may improve the care that patients with PAD and comorbid depression receive.
The University of Virginia School of Medicine designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Credit™ per article. Participants should claim only the credit commensurate with the extent of their participation in the activity. Learners are expected to read this article, along with any references and supporting material as appropriate, and complete the online post-test questions with an 80% pass rate to receive credit. CME credit may be claimed immediately after completing the quiz. This activity expires 2 years after the publication date, on
The faculty, staff and planning committee of the University of Virginia Office of Continuing Medical Education have no financial affiliations to disclose.
The CME planning committee disclosed the following:
Heather Gornik has disclosed research support from AstraZeneca and CVR Global, intellectual property rights from Summit Doppler Systems, Inc., and intellectual property rights and stock/ownership from FlexLife Health (proceeds donated). Aditya Sharma has disclosed research support from National Institute of Health Sciences, AstraZeneca, Biomet Biologics, and Portola Pharmaceuticals. Valerie Clark has no financial affiliations to disclose.
The authors have disclosed receipt of financial support for the research, authorship, and/or publication of this article. Please refer to the Funding section at the end of this article for a complete list of funding.
Introduction
The 1-year prevalence of major depressive disorder in the general population in the United States (US) is 7%, with a lifetime prevalence as high as 16%. 1 Depression has been recognized as one of the world’s leading causes of disability 2 and is projected to become the second leading cause of disability adjusted life years by 2030. 3 Over 15 million people with cardiovascular disease (CVD) in the US have comorbid depression, corresponding to a prevalence as high as 45%.4,5 Depression has been linked to an increased incidence of CVD, and has been shown to be associated with physical impairment, cardiac mortality, and all-cause mortality in patients with CVD.6–8 As such, based on a large body of evidence, the American Heart Association (AHA) recently recommended the inclusion of depression as a risk factor for adverse medical outcomes in patients with acute coronary syndromes (ACS). 9
The presence of depression or depressive symptoms has been demonstrated to predict the incidence of coronary artery disease (CAD) in healthy patients, 10 secondary events in patients with known coronary heart disease (CHD),11 –14 and adverse outcomes in patients undergoing coronary artery bypass graft. 15 Moreover, psychosocial factors have been identified as stronger risk factors for incident myocardial infarction (MI) than traditional cardiovascular risk factors such as smoking, hypertension, diabetes mellitus, dyslipidemia, or obesity. 16 However, depression was only officially recognized as a risk factor for CHD in the 2010 Global Burden of Disease Study. 17 In addition to CHD, depression has also been associated with the incidence of heart failure 18 and stroke, 19 as well as adverse outcomes in the same populations19–21 independent of traditional cardiovascular risk factors.
Peripheral artery disease (PAD) is characterized by atherosclerotic narrowing of the peripheral arteries and is a marker for systemic atherosclerosis. Furthermore, PAD and CAD are commonly comorbid and share several common risk factors, 22 including smoking, hypertension, diabetes mellitus, dyslipidemia, or obesity. PAD has been independently associated with impaired quality of life, 23 limb loss, 24 increased risk of cardiovascular events 25 and increased mortality. 26 The prevalence of depression in patients with pre-existing PAD is similar or greater in magnitude than that in patients with CAD. 27 Given the established link between depression and CVD, and the shared underlying pathogenesis and risk factors between PAD and CAD, clinicians have begun to investigate the link between depression and PAD. This narrative review addresses the current evidence and potential mechanisms linking PAD and depression, as well as implications for intervention and research.
Evidence linking PAD and depression
Among patients with PAD, the prevalence of depression ranges from 16% 28 to 46%. 29 PAD is associated with impaired quality of life, 30 which may increase the incidence of depression. 31 In addition, a history of depression or a new diagnosis of depression have been shown to significantly increase the rate of incident PAD diagnoses (hazard ration (HR): 1.24, 95% CI: 1.18–1.30), adjusted for age, sex, socioeconomic status, comorbidities, and patient characteristics. 32 When compared with a low level of depressive symptoms, moderate and high levels of depressive symptoms were associated with greater incidence of PAD. 33 In a prospective cohort study of patients with PAD, individuals who were depressed at baseline had more significant impairments in PAD symptoms compared to patients without depression at baseline. 29 Moreover, among patients with PAD, patients with comorbid depression at baseline were persistently depressed throughout the follow-up period at 6, 12, and 18 months; whereas patients without depression at baseline did not develop any new depressive symptoms during follow-up. 29 In a cohort of patients with PAD and depressive symptoms at baseline, 70.3% and 66.3% had persistence of symptoms at follow-ups of 1 and 2 years, respectively. 34 In comparison, in the subgroup of patients with PAD and no depressive symptoms at baseline, only 14% and 9% developed new depressive symptoms at 1 and 2-year follow-ups, respectively. 34
Although the majority of studies have focused on male patients, several studies have suggested that women with PAD may also be at an increased risk of developing depression. Grenon et al. reported that depression was significantly more common in women with PAD compared to women without PAD (odds ratio (OR): 3.83, 95% CI: 1.33–11.0). 35 Additionally, young women with PAD have been shown to be significantly more likely to suffer from depression 36 compared to women without PAD. Several studies have investigated sex-based differences in the association between PAD and depression. 37 When compared to men with PAD, women with PAD have been reported to have lower physical and mental health as measured by the Short Form-12. 38 In a study by Smolderen et al., women younger than 65 years of age had a fourfold greater odds of significant depressive symptoms compared to men older than 65 years of age, which remained significant in multivariate models at 6-month follow-up (OR: 4.1, 95% CI: 2.0–8.4). 39 The PORTRAIT study demonstrated that women with PAD had a higher prevalence of depression and poorer functional capacity, as assessed by the Peripheral Artery Questionnaire, 40 when compared to men with PAD, despite similar ankle–brachial indices. 41 In addition, a recent meta-analysis of patients with PAD has further confirmed that women are more likely than men to have depression. 27 Lastly, women seem to have shouldered a greater portion of the rising PAD morbidity and mortality over the past 20 years compared to men, 42 suggesting that the burden of depression in women with PAD is likely to continue rising.
Patients with pre-existing PAD have also been reported to be more likely to develop depressive symptoms. McDermott et al. reported that patients with PAD were more likely to develop depression (HR: 1.57, 95% CI: 1.12–2.21) after a mean follow-up of 2.7±1.2 years, although this was no longer significant after adjusting for walking ability. 43 In a similar study, Ruo et al. reported that new depressive symptoms were associated with worse functional outcomes in patients with PAD, 34 which likely contribute to the progression of PAD. Hence, results of these observational studies have revealed bi-directional associations between these comorbid diseases, and studies of biologic and behavioral risk factors have further supported this bi-directionality.
Mechanisms linking depression and atherosclerosis
A substantial body of literature has explored the mechanisms linking depression and CVD, which contributes to our current understanding of the relationship between depression and PAD. Despite the overlap between the biologic and behavioral risk factors of atherosclerosis, we will address each separately below (Figure 1). It is important to note that literature specific to PAD is limited and understanding the relationship between PAD and depression requires applying knowledge of the mechanisms that link depression and CVD.
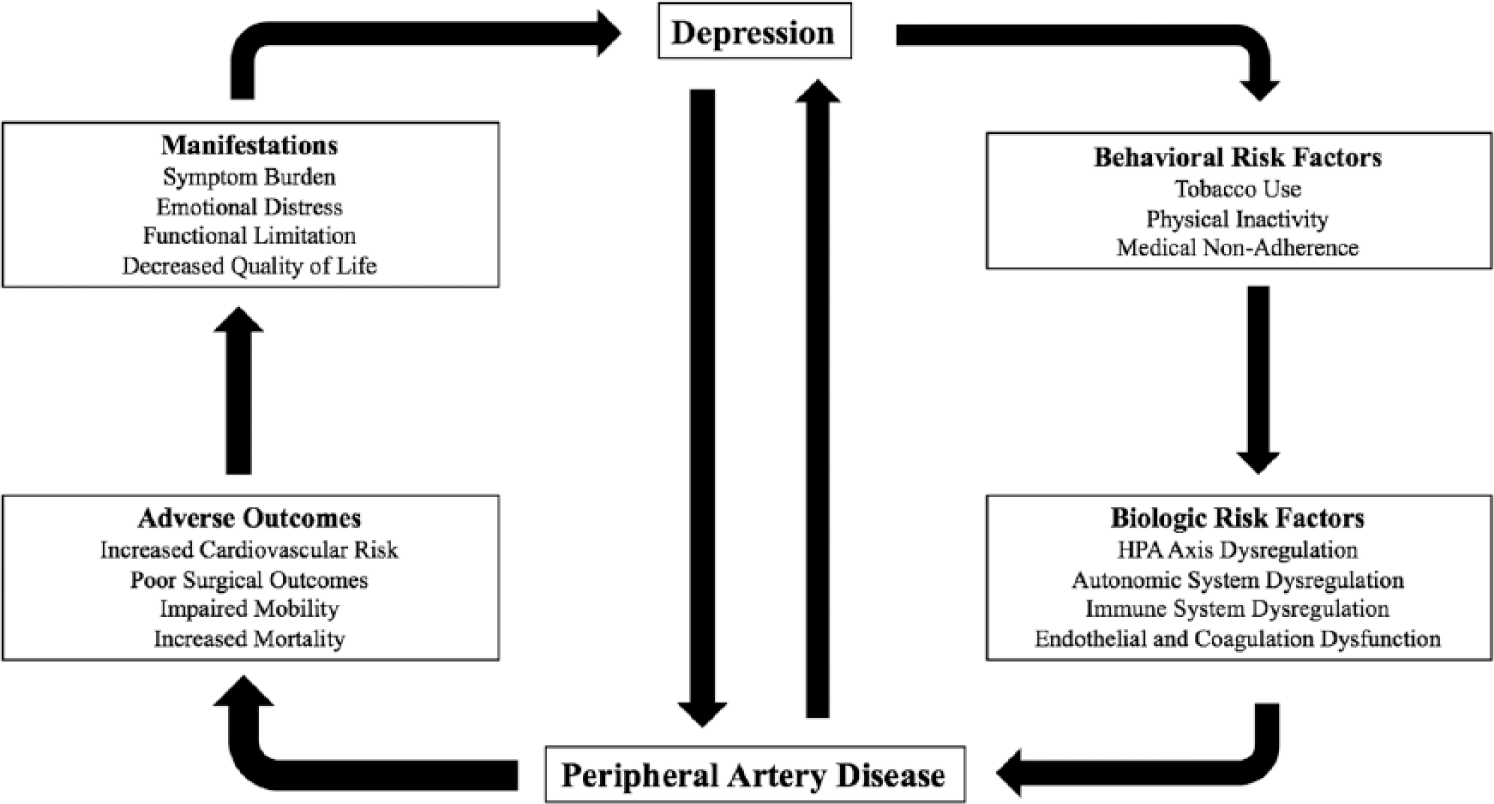
Proposed relationship between depression and peripheral artery disease.
Biologic risk factors
An individual’s allostatic load has been described as the long-term cumulative effect of one’s physiologic response to stress. It has been hypothesized that a combination of genetic factors, life stressors, and traumatic life events contribute to several physiologic responses, including hypothalamic-pituitary-adrenal (HPA) axis dysregulation, autonomic system dysregulation, immune system dysregulation, and endothelial and coagulation dysfunction. Over time, these physiologic responses may increase one’s risk of CVD, 44 including PAD. Depression may mediate similar physiologic changes that increase susceptibility to CVD. 45
HPA axis dysregulation
The HPA axis plays a large role in the stress response, and its end products alter human physiology. 46 Dysregulation of the HPA axis, as seen in depression, has been hypothesized to contribute to the development and progression of atherosclerosis. 47 In patients with pre-existing CHD, increased urinary cortisol levels were observed in patients with current depression and a history of depression. 48 Although there is substantial evidence supporting cortisol’s role in the development of atherosclerosis, 46 there are limited data studying HPA axis dysregulation specifically in patients with PAD.
Autonomic system dysregulation
Dysregulation of the autonomic nervous system, as measured by urinary norepinephrine levels, has also been reported in patients with depression.49,50 In addition, patients with depression have been shown to have reduced heart rate variability (HRV), favoring tachycardia, another sign of dysregulation of the autonomic system. 51 Reduced HRV is associated with adverse cardiovascular outcomes after MI, 52 such as the incidence of arrhythmias and sudden death. 53 Although there are no data analyzing the effects of depression on HRV specifically in patients with PAD, impaired HRV has also been reported among patients with PAD.54,55
Immune system dysregulation
A robust association between depressive symptoms and immune system dysregulation has been identified in a diverse range of patient populations. 56 Several inflammatory markers, including C-reactive protein (CRP),57,58 interleukin-6,59,60 gamma interferon, 56 and tumor necrosis factor-α, 56 have been shown to be elevated in patients with depression. In addition, patients with depression, intense feelings of anger, or excessive hostility have been shown to have elevated levels of complement (C3 and C4). 61 Depressive symptoms have also been associated with elevated markers of fibrosis, 62 which mediate chronic atherosclerosis. Despite these results, it remains unclear whether increased systemic inflammation leads to worse depressive symptoms, or whether depressive symptoms lead to increased systemic inflammation,63 –65 and both in fact may be true. However, the causal pathway from inflammation to atherosclerosis has been well-established.66–68 Interestingly, patients with CVD and comorbid depression have higher levels of serum inflammatory markers, suggesting that inflammation may partially mediate the increased risk of CVD progression seen in patients with comorbid depression.69 –71
Endothelial and coagulation dysfunction
Depression has been implicated with impaired endothelial function and platelet activity, both of which are believed to contribute to the development of atherosclerosis and PAD. Patients with CHD and comorbid depression were shown to have higher levels of platelet factor 4 and β-thromboglobulin, mediators in the thrombosis pathway, when compared to CHD patients without depression. 72 Patients with depression may also have increased platelet activation, mediated by elevated serum serotonin levels.73 –75 Abnormal serotonin metabolism in both the brain and circulating platelets of patients with depressive symptoms may result in pathologic triggers to platelet activation, degranulation, and aggregation, all contributing to progression of atherosclerosis and thrombosis.6,76,77 Platelet enzymatic activity of nitric oxide synthase has been shown to be impaired in patients with depression, which may further lead to endothelial dysfunction 78 as reduced platelet nitric oxide promotes platelet aggregation,6,79 which may therefore lead to thrombosis and the development of atherosclerosis.
In addition to coagulation dysfunction, endothelial dysfunction has been recognized as a key contributing mechanism to the progression of atherosclerosis. 80 Local endothelial dysfunction, as measured by directed flow-mediated vasodilation (FMD), has been reported in symptomatic and atherosclerotic vessels of patients with PAD. 81 In addition, impaired FMD predicts adverse cardiovascular outcomes and is inversely related to cardiovascular risk.82,83 Endothelial dysfunction, including impaired FMD, has also been shown to be independently associated with depression and post-traumatic stress disorder (PTSD) in multiple studies,84 –88 and a meta-analysis of cross-sectional and retrospective studies further supports the association between depressive symptoms and impaired FMD. 89 However, the relationship between endothelial dysfunction and depression specifically in patients with PAD has yet to be explored.
Behavioral risk factors
Behavioral risk factors, including tobacco use, physical inactivity, and medical non-adherence, have been shown to contribute significantly to the development of atherosclerosis, PAD, and poor cardiovascular outcomes.
Tobacco use
It is well-established that patients with depression are more likely to smoke, 90 which is independently associated with the development of atherosclerosis, PAD, and adverse cardiovascular events. 91 In patients with depression, smoking has been recognized as one of the most significant drivers of the development and progression of atherosclerosis.92,93 In patients with known CAD, smoking has also been shown to be independently associated with depressive symptoms and mortality. 94
Physical inactivity
Depression has been linked to sedentary lifestyles95,96 and physical inactivity. Similar to tobacco use, physical inactivity plays a significant role in the development of both atherosclerosis in patients with depression,92,93 and mortality in patients with pre-existing CAD. 94 In a prospective study of outpatients with CAD, the risk of cardiovascular events was largely mediated by physical inactivity. 97 Similarly, in a prospective study of patients with depression who recently experienced an acute MI, patients who did not exercise suffered more than a twofold increase in fatal events when compared to patients who exercised regularly. 98 Both depressive symptoms and physical inactivity have been shown to be independently associated with increased cardiovascular mortality.99,100 Physical exercise has also been documented to decrease levels of inflammatory markers in human serum,101,102 providing evidence for the interrelationships between biologic and behavioral risk factors. In addition to its benefits for physical health, exercise is also associated with improved well-being, mood, and mental health. 103 Conversely, PAD symptoms such as claudication directly impair patients’ ability to ambulate pain-free, leading to a decline in physical function 104 which further contributes to both depressive symptoms and PAD progression. However, physical inactivity only partially explains the relationship between depression and poor cardiovascular outcomes. 105
Medical non-adherence
Patients with depression are less likely to follow recommended behavior and lifestyle changes as well as medical treatment recommendations. 106 In patients with CAD, patients who reported non-adherence to medications had elevated Patient Health Questionnaire-9 (PHQ-9) scores when compared to patients reporting adherence to their medications. 107 In the same study, non-adherence to medications was associated with a greater than twofold increased rate of adverse cardiovascular events. 107 Patients with depression and CAD were reported to have lower levels of potentially PAD-protective omega-3 polyunsaturated fatty acids (PUFAs) 108 when compared to sex-matched controls without depression, which may be at least partially attributed to poor adherence to physician recommended dietary habits. 109 Deficiencies in omega-3 PUFA intake may also contribute to elevated levels of inflammation noted in patients with depression, 110 again providing evidence for the co-relationships between biologic and behavioral risk factors.
Despite all of the evidence highlighting the role of these behavioral risk factors in the development of atherosclerosis, PAD, and poor cardiovascular outcomes, it appears that they likely only explain a portion of the relationship.35,111
Impact of depression on PAD outcomes
These is a growing body of evidence linking depressive symptoms and depression to poor outcomes in PAD (Table 1). Depression has been associated with impaired physical function as objectively measured by maximum walking distance (MWD) and pain-free walking distance (PFWD). 29 Patients with PAD and depressive symptoms were shown to have significantly shorter MWD than patients without depressive symptoms, 29 a shorter 6-minute walk distance, slower usual-pace velocity, slower fast-pace walking velocity, and reduced scores in the short physical performance battery.34,112
Studies analyzing the impact of depression and depressive symptoms on PAD outcomes.
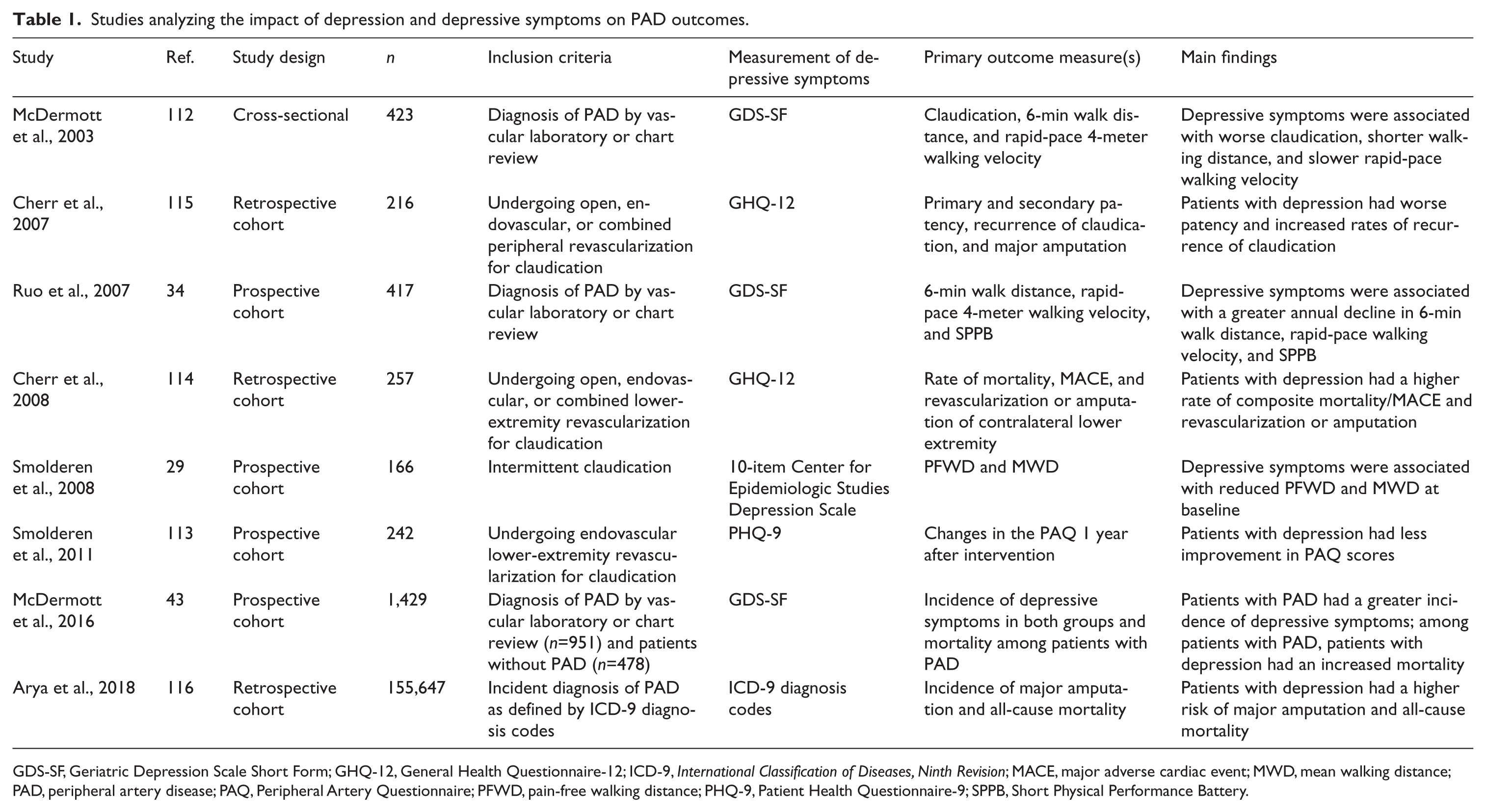
GDS-SF, Geriatric Depression Scale Short Form; GHQ-12, General Health Questionnaire-12; ICD-9, International Classification of Diseases, Ninth Revision; MACE, major adverse cardiac event; MWD, mean walking distance; PAD, peripheral artery disease; PAQ, Peripheral Artery Questionnaire; PFWD, pain-free walking distance; PHQ-9, Patient Health Questionnaire-9; SPPB, Short Physical Performance Battery.
Recently, depression has been identified as a risk factor for mortality as well as poor surgical outcomes in patients with PAD. In a prospective study of patients with PAD, depression was significantly associated with increased all-cause mortality (HR: 1.57, 95% CI: 1.12–2.21) and cardiovascular mortality (HR: 1.80, 95% CI: 1.03–3.13). 43 For patients undergoing lower-extremity open or endovascular interventions, patients with comorbid depression had significantly reduced reported health status, 113 as well as an increased risk of the composite endpoint of death and major adverse cardiac events (HR: 2.05, 95% CI: 1.16–2.86), adverse cardiac events (HR: 1.75, 95% CI: 1.03–2.98), and contralateral PAD (HR: 2.20, 95% CI: 1.22–3.96). 114 Furthermore, among those receiving lower-extremity revascularization, patients with PAD and depression had worse primary and secondary patency rates at 2 years, 115 along with increased progression of contralateral PAD. 114
In a recent study of Veterans, Arya et al. reported that depression independently increased the risk of major amputation (HR: 1.13, 95% CI: 1.07–1.19) and all-cause mortality (HR: 1.17, 95% CI: 1.14–1.20) in patients with PAD. 116 In a separate study of patients who underwent lower extremity amputations for complications related to diabetes mellitus or PAD, there was a 16% prevalence of suicidal ideation at 1 year, which was significantly associated with lower levels of mobility and activities of daily living independence. 28
Implications for intervention and research
Depression is a chronic and disabling condition that carries a tremendous economic burden to society, and increases the risk of adverse outcomes in patients with PAD. PAD is a disease of systemic inflammation which significantly impairs quality of life and mobility, both of which likely increase the risk of developing depressive symptoms. Since vascular specialists commonly care for patients with PAD, it is critical that they be aware of the impact that PAD and CVD may have on mental health. It is also essential that vascular specialists be able to recognize patients exhibiting signs or symptoms of depression and refer them to mental health professionals for evaluation and treatment. Additionally, vascular specialists should recognize the importance of depression as a potential risk factor, alongside smoking, hyperlipidemia, and obesity, for PAD.
The AHA has recognized the importance of depression as a significant risk factor in patients with CAD. 9 The AHA has further recognized the heterogeneity in study designs and measurements of depression and depressive symptoms, and they have recommended standardizing depression reporting standards. 117 Lastly, the AHA has also recommended that physicians increase screening and counseling regarding treatment options for depression in patients with CAD (Figure 2). 117 Given the clinical implications of the effects of depression on cardiac-specific and all-cause and mortality in cardiovascular patients, the AHA made recommendations for physicians to consider screening for depression and provide guidance on treatment options in this statement. Although there are currently no tools recommended for depression screening among patients with PAD, the AHA recommends utilizing the PHQ-9 to screen for depression among patients with CAD. The PHQ-9 is a nine-item questionnaire that assess for the presence and severity of depressive symptoms as well as for suicidal ideation. A score ⩾10 is concerning for major depression and warrants a referral to a mental health professional. The presence of suicidal ideation should always prompt a referral to a mental health professional or immediately to an emergency department (Figure 2). The PHQ-9 can be completed within minutes and has a sensitivity of 88% and specificity of 88% for major depression. 118
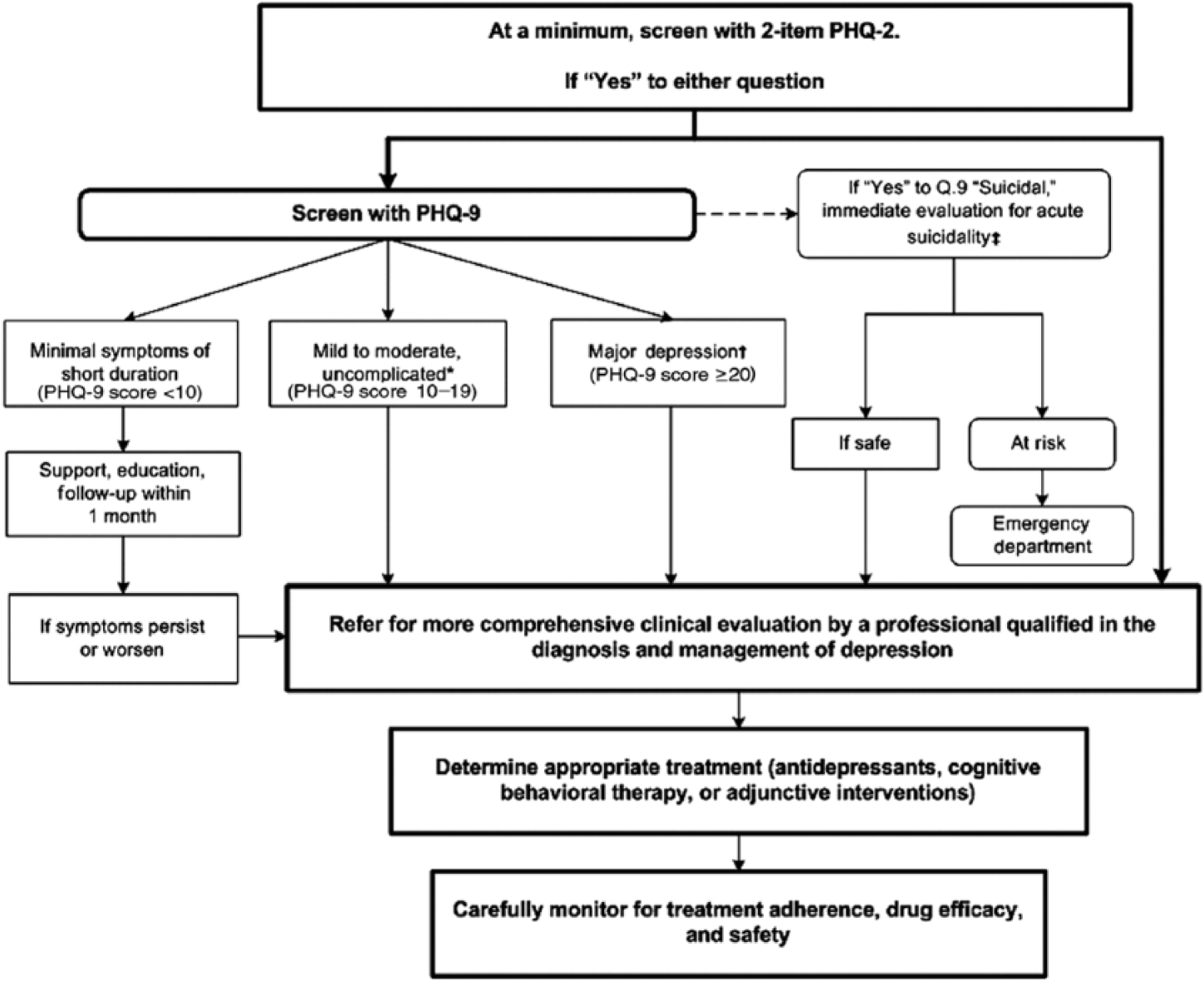
The American Heart Association’s recommendations for screening for depression in patients with coronary heart disease.
There is currently no consensus on depression screening tools or guidelines for patients with PAD. Similarly, no guidelines exist for optimal operative or medical management for patients with PAD and depression. However, as evidence linking depression to increases in morbidity and mortality in patients with PAD grows, it is critical for clinicians to continue to consider the impact of mental well-being on their patients’ outcomes. Patients with PAD and comorbid depression are a high-risk group of vascular surgery patients; hence, physicians should recognize that depression may be associated with poorer outcomes in this subgroup. It is also important to note that making the diagnosis of major depressive disorder is more complex than simply screening for depressive symptoms and includes meeting several criteria, as outlined in the Diagnostic and Statistical Manual of Mental Disorders-5 (DSM-5). The DSM-5 also specifies that in order to make a diagnosis of major depressive disorder, symptoms should not be attributable to another disease. PAD has been associated with social, occupational, and physical impairment, which therefore may lead to an overestimation of the prevalence of depression in this population. Referral to the appropriate mental health professional, or to a primary care physician who may be able to provide this referral, is the best course of action if there is concern for depression or depressive symptoms.
There currently exists no strong evidence that treating depression with antidepressants improves survival in patients with CVD. 7 Indeed, a Cochrane Review concluded that behavioral therapy and pharmacological interventions as a standard of care for the treatment of depression provided a modest benefit in depression scores; however, no benefit was shown in cardiovascular morbidity or mortality.119,120 However, a recent study has shown that elderly patients with comorbid chronic depression and CAD who adhered to medical treatment for their depression had lower events of CAD-related hospitalizations. 121 Additionally, Arya et al. reported that among Veterans with PAD, antidepressant use was associated with a lower risk of major amputation when compared to patients not taking antidepressants. 116 However, several antidepressants have been reported to increase cardiovascular risk, highlighting the importance of careful selection of antidepressant medications in patients with PAD and CVD. 122
Mindfulness is a psychological process of self-awareness of current experiences, feelings, and emotions. 123 Mindfulness techniques include several forms of directed meditation including breathing exercises and thoughtful reflection, among others. Although mindfulness techniques have been shown to improve depressive symptoms and quality of life among patients with CVD, 124 to our knowledge this technique has not yet been reportedly used in patients with PAD and nor has the impact of outcomes in this population been investigated. Other non-pharmacological techniques for the treatment of depression and depressive symptoms have been reported to include cognitive behavioral therapy and other forms of psychotherapy. However, the effects of these treatments on cardiovascular outcomes and among patients with PAD remain unclear. A recent meta-analysis summarized randomized clinical trials that assessed the effects of several non-pharmacological psychological interventions on outcomes among patients with CAD. 125 They report that low-quality evidence identified a 21% reduction in cardiac mortality (RR: 0.79, 95% CI: 0.63−0.98) and improved depressive symptoms (standardized mean difference: −0.27, 95% CI: −0.39 to −0.15). However, studies included in this meta-analysis were generally of poor quality, included a wide range of psychological interventions, were not focused exclusively on patients with depression, and were not specific to patients with PAD. Given these findings, further research on managing cardiovascular risk, PAD, and comorbid depression is warranted. 126 Future work should investigate optimal operative management in patients with depression and PAD, and whether treating depression in these patients results in improved clinical and patient-centered outcomes.
As reported earlier, PAD and depression have several overlapping risk factors, which, if addressed, could lead to improvement in both conditions. Supervised vascular rehabilitation programs that focus on increasing medical compliance and addressing behavioral risk factors appear promising in addressing both PAD and depressive symptoms.127,128 Exercise is known to improve outcomes in PAD 129 as well as depression. 130 Previous investigators have also demonstrated promising results utilizing mobile technology to address behavior change 131 and cardiovascular risk factors,132,133 and randomized clinical trials assessing these interventions are underway. 134 It is likely that results from these studies and future studies can also be applied to patients with PAD and comorbid depression.
Conclusion
Current evidence strongly suggests a relationship between depression and PAD, and depression has been linked to poor outcomes in patients with PAD. Depression and PAD are growing public health concerns, and the evaluation of depression in PAD has the potential to enhance individualized vascular care. Despite the increasing body of evidence suggesting an association between depression and PAD, the specific biologic and behavioral risk factors linking these two diseases remain unclear, and only a limited number of studies have focused specifically on depression in PAD cohorts. More research is required to understand the complex interrelationship between depression and PAD.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this project was supported by the National Center for Advancing Translational Sciences, National Institutes of Health, through UCSF-CTSI Grant Number TL1 TR001871 with additional student research support from the Society for Vascular Surgery Student Research Fellowship Award and the American Heart Association Student Scholarship (JLR). Furthermore, this work was supported by start-up funds from the University of California San Francisco and the Northern California Institute for Research and Education, by Award Number KL2RR024130 from the National Center for Research Resources, Award Number 1K23HL122446-01 from the National Institutes of Health/National Heart, Lung, and Blood Institute, and a Society for Vascular Surgery Seed Grant and Career Development Award (SMG). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health. The funding organizations were not involved in the design and conduct of the study, collection, management, analysis, and interpretation of the data, or preparation, review or approval of the manuscript.
