Abstract

Cancer survival rates are improving as a result of novel targeted and immune-based therapies. In 2020, there are more than 17 million cancer survivors in the United States (representing 5% of the total population). 1 Cancer survivors are at risk of cardiovascular disease (CVD) for a number of reasons; these include adverse cardiovascular effects of oncology treatments and common risk factors that predispose to both cancer and CVD. 2 As a result, cardiovascular care has emerged as an important consideration for patients, giving rise to ‘cardio-oncology’ as a new clinical subspecialty. While much of the early focus of this field has been on the cardiomyopathic effects of cancer therapies, cardio-oncology now encompasses various aspects of CVD, including vascular and metabolic issues. In 2019, the first American Heart Association (AHA) statement focusing on ‘vascular cardio-oncology’ was published, drawing attention to vascular medicine dimensions of the field. 3 Dedication of this issue of Vascular Medicine to vascular oncology is in line with the increasing awareness of vascular disease in cancer patients and cancer survivors.
Vascular cardio-oncology (hereafter referred to as vascular oncology) is a broad topic, as reflected by the diverse set of papers in this issue. Foremost, vascular oncology is driven by the short and long-term cardiovascular toxicity of cancer treatments, a topic reviewed by Campia et al., including a clear table. 4 Traditional chemotherapies (e.g. anthracyclines) are associated with systolic cardiac dysfunction and congestive heart failure as direct toxicities. Newer oncology therapies, many targeted, can lead to vascular perturbations, including myocardial and peripheral ischemia, thromboembolic disease, hypertension, and metabolic complications. Immune-based therapies, like immune checkpoint inhibitors (ICI), can lead to myocarditis and pericarditis but also to vasculitis, which can be fatal or debilitating. 5 Other therapies can also lead to long-term vascular toxicities. Radiation therapy, especially to the thoracic cavity, can lead to cardiac and peripheral ischemic disease that can manifest years after completion of therapy. 6 Traditional atherosclerotic risk factors contribute to long-term cardiovascular sequelae of cancer therapies. Conversely, cancer treatments may also perturb traditional atherosclerotic risk factors. For instance, androgen deprivation therapy (ADT) used in treatment for prostate cancer can cause obesity, hyperlipidemia, hyperglycemia, and hypertension. In addition, platinum-based therapies can cause metabolic disease, an issue especially important because cisplatin is the cornerstone of treatment of testicular carcinoma, a cancer type with high survival rates most prevalent in young men. 7 For novel oncology therapies, knowledge on long-term effects is often limited due to lack of follow-up. In this issue, Kondapalli et al. 8 nicely summarize the long-term effects of tyrosine kinase inhibitors that have revolutionized treatment for certain types of leukemia but are associated with long-term vascular toxicities. Given the novelty of use, long-term vascular effects associated with ICIs are completely unknown but are important considerations, given that ICIs have resulted in durable responses in previously deadly cancers such as melanoma. In addition, preclinical models where PD-1 (programmed cell death protein 1; the prototypical target of ICI) is inhibited pharmacologically or genetically, lead to significant increased atherosclerotic burden. 9
Cancer itself can lead to vascular complications. It is well known that cancer increases the risk of venous thromboembolism (VTE) and arterial thromboembolic disease, an issue that is highlighted by a number of articles in the current issue of Vascular Medicine. Cancer leads to VTE by affecting all components of Virchow’s triad: stasis, endothelial injury, and alterations in blood coagulation (hypercoagulability). These concepts are illustrated by case reports and vascular images by Banathy et al., Shah and Silver, and Wilkins et al.10 –12 Cancer therapies can also lead to increased coagulability or damage to the endothelium, further illustrating the multifaceted interaction between cancer, cancer treatment, and CVD. Therefore, indication, timing, and dosing of prophylactic anticoagulation in patients with cancer is an ongoing debate nicely summarized by an editorial by Khorana, 13 as well as a review article by Gomatou et al., 14 the latter discussing the use of prophylactic anticoagulation in the ambulatory setting in patients with lung cancer. The main reason for restraint is an increase in bleeding risk due to close relation or ingrowth of the tumor to blood vessels, a particular challenge in patients with gastric cancer and summarized by Majmudar et al. 15
Another exciting and emerging frontier in cardio-oncology is the growing appreciation that common risk factors contribute to co-occurrence of CVD and cancer. These include behavioral risk factors such as smoking and a sedentary lifestyle, as well as genetic risk factors. Intriguing recent data demonstrate that somatic genetic mutations in blood cells (termed clonal hematopoiesis of indeterminate potential (CHIP)) are associated with hematological malignancies but also CVD. 16 Further research is needed in terms of determining the causal role that CHIP plays in vascular disease, especially in patients with cancer. Finally, cardiac disease itself can trigger subsequent cancer; for instance, heart failure increases the risk of cancer propagation in preclinical models, likely as a consequence of circulating factors which stimulate tumor growth. Epidemiological data have mostly supported this observation, although more research is needed. 17
All these observations emphasize the need for further collaboration between oncology and cardiovascular physicians, including vascular medicine specialists (Figure 1). We encourage systematic evaluation and follow-up of high-risk cancer patients and cancer survivors at a dedicated cardio-oncology or vascular oncology clinic.3,18 The aims of such clinics are in line with the above-described interplay between cancer, cancer treatment, and CVD, acknowledging that these diseases can exist in the same patient. Recognizing the cardiovascular toxicities of cancer treatments, especially long-term effects, by routine follow-up of cancer survivors and mitigating cardiovascular risk factors during and after cancer treatment are important aspects of care in a cardio-oncology clinic. Before treatment, identifying patients with a high-risk of CVD can minimize additional risk due to cancer and cancer treatment. 18 During treatment, follow-up might include frequent monitoring, including cardiovascular or functional imaging, depending on specific patient variables and treatment characteristics. In some cases, lowering the dose or discontinuing treatment might be necessary both prior to treatment or after cardiovascular complications develop following treatment. At the same time, more research is needed to define specific cardiovascular monitoring in cancer survivors. Although these have been defined in the pediatric cancer survivor population, leading to a clear decline in late cardiovascular toxicity, clear recommendations are still lacking for adult cancer survivors.19,20 Current practice usually defers care to the general practitioner, especially after the first years when the follow-up for cancer recurrence by the oncologist has ended and patients are considered ‘cured’. An ‘ABCDE approach’ for cardiovascular wellness in cancer survivors has been proposed as a simple checklist for the primary care physician, but precision-based guidelines are clearly lacking, especially for the newer oncology therapies. 2
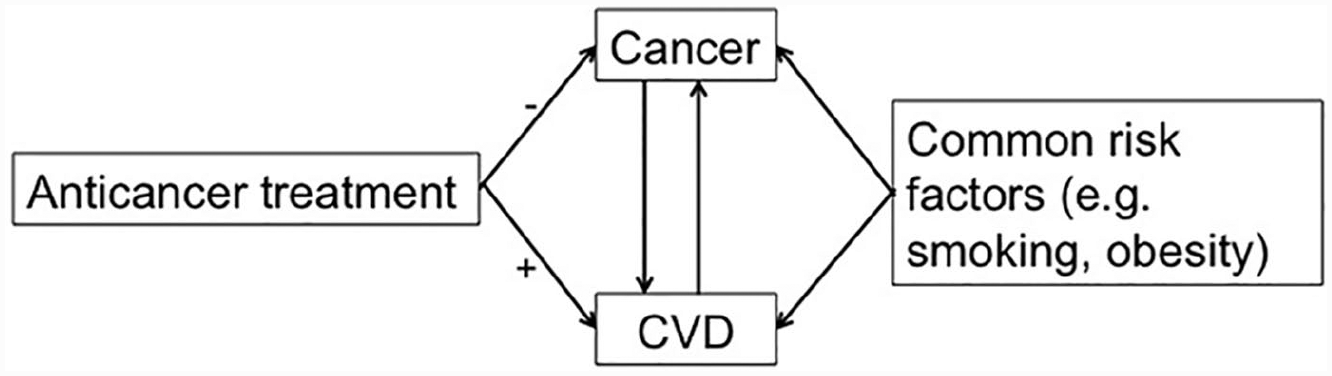
Schematic overview of the interaction between cancer, anticancer treatment and CVD.
Finally, there is a great need for research to enhance vascular care in the cardio-oncology population. Clinical data need to be consistently collected in this population to identify signals for toxicity and to better elucidate mechanisms of cardiovascular effects, especially with targeted cancer therapies. Dedicated cardio-oncology clinics enable standardized collection of clinical data that can be combined with preclinical models to better predict who will develop cardiovascular complications and to develop preventive and treatment strategies. The ultimate goal is a personalized approach for cardiovascular care for each patient, which is defined by the patient and tumor characteristics as well as oncology treatments, to help optimize the prevention of cardiovascular toxicities.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Javid Moslehi has served on advisory boards for Pfizer, Novartis, Bristol-Myers Squibb, Deciphera Pharmaceuticals, Audentes Therapeutics, Nektar Therapeutics, Takeda, Ipsen, MyoKardia, AstraZeneca, GlaxoSmithKline, Intrexon, and Regeneron. Jorie Versmissen and John Power have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Javid Moslehi is supported by National Institutes of Health grant R01 HL141466.
