Abstract
Chemotherapy, alone or in association with radiation therapy, has represented the cornerstone of cancer treatment for decades. However, in the last several years, an unprecedented progress in the understanding of cancer biology and the discovery of novel therapeutic targets have led to a paradigm shift in the management of patients with neoplastic diseases. The introduction of tyrosine kinase inhibitors, vascular endothelial growth factor pathway inhibitors, immunomodulatory agents, proteasome inhibitors, immune checkpoint inhibitors, and chimeric antigen receptor T cells, among others, has been associated with prolonged survival in many forms of cancer. A common feature of both chemotherapy and novel cancer treatments is the frequent occurrence of vascular toxicity, mainly mediated by injury to the endothelium. While the mechanisms may vary between agents, the clinical manifestations may overlap and range from hypertension, vasospastic and thrombotic arterial events (myocardial ischemia and infarction, peripheral ischemia, and limb gangrene), venous thromboembolism (deep vein thrombosis and pulmonary embolism) to capillary leak syndrome. Therefore, the effective management of patients with cancer requires a multidisciplinary team approach in which oncologist and cardiovascular medicine specialists work together to prevent, detect, and minimize acute vascular toxicity and long-term consequences of cancer therapy.
Introduction
Cancer represents one of the leading causes of adult morbidity and mortality in the United States. 1 The term cancer, which is used interchangeably in reference to any neoplastic malignancies, encompasses an extremely wide range of pathologies and clinical presentations. However, the majority of cancers share specific defining biological features, including abnormal clonal cell proliferation, loss of differentiation, altered cell metabolism, metastatic spread (the ability of neoplastic cells to detach from the site of origin, invade arteries, veins and lymphatic vessels, and circulate to distant tissues where they can home and grow), as well as the capacity to induce the formation of new vessels and to evade immune surveillance. 2 Several of the mechanisms underlying these features have been elucidated in sufficient detail to become targets for medical therapy.
The first breakthrough in the medical treatment of cancer occurred several decades ago with the introduction of various antiproliferative agents with broad cell toxicity (collectively referred to as chemotherapy), alone or in combination with radiation therapy. In addition, given the effects of hormones on cell growth in several cancers, antihormonal therapy has complemented the armamentarium of available treatments. Despite their non-specific effects, chemotherapy and radiation therapy have represented a giant step forward in the management of patients with cancer, allowing for extension of disease-free survival and palliation in a number of malignancies and, in many of them, leading to a cure. Unfortunately, acute toxicity, frequent dose-limiting side effects, and development of drug resistance by cancer cells have often represented unsurmountable challenges, leading to short-lived disease control and treatment failure. 3 Furthermore, long-term effects of cancer treatments, particularly on the cardiovascular system, have led to increased cardiometabolic risk and reduction of life expectancy in cancer survivors. 4 Indeed, the risk of developing heart failure has been reported to be as high as that of a second malignant neoplasm, with a relative risk (RR) of 15.1 (95% CI 4.8–47.9) vs 14.8 (95% CI 7.2–30.4), respectively. Also, in the same population, the risk of coronary artery disease (CAD) and stroke was remarkably increased (RR 10.4 (95% CI 4.1–25.9) and 9.3 (95% CI 4.1–21.2), respectively). 5
In the last three decades, extraordinary advances in the understanding of cancer biology and immune system function, coupled with innovations in drug development, have led to an unparalleled progress in the treatment of a large number of malignancies. Among the most important discoveries are the role of tyrosine kinases 6 and cell cycle regulatory kinases 7 in cancer cell proliferation, the elucidation of the pathways regulating cancer-induced angiogenesis, 8 and the understanding of pivotal mechanisms whereby cancer cells can evade immune recognition. 9 These discoveries have allowed the identification of multiple novel molecular targets and have ushered a new era of cancer therapy. One of the main promises of these targeted therapies is the specificity of the effect on the pathogenetic mechanisms of cancer with sparing of non-neoplastic tissues. However, the introduction of these novel agents has been associated with the occurrence of unexpected, and at times fatal, cardiovascular side effects and has uncovered previously unforeseen physiologic roles of tyrosine kinases and angiogenic pathways in vascular homeostasis.
The need to prevent and manage the adverse effects of traditional (i.e. chemotherapy, hormonal, and radiation therapy) and targeted cancer treatments on the cardiovascular system has led to the development of cardio-oncology as a novel discipline within cardiovascular medicine. Historically, the initial interest of cardio-oncology was focused on the toxic effects of cancer therapies on the myocardium. However, in recent years, the awareness of the vascular toxicity of cancer treatments has grown significantly, broadening the scope of the discipline and leading to formal recognition of the vascular impact of antineoplastic therapies. 10
This review article will outline the current understanding of the vascular effects of traditional and novel cancer treatments, with a focus on the main known mechanisms of arterial toxicity and their cardiovascular consequences. In this manuscript, the vascular effects of radiation treatment will not be reviewed. An exhaustive review on the topic has recently been published. 11
The endothelium as a target of vascular toxicity of cancer treatments
The endothelium is a single cell layer that lines the entire arterial and venous circulation. Despite its apparent structural simplicity, the endothelium is the vital controller of the exchanges between blood and the interstitial space and the master regulator of vascular homeostasis. Through highly regulated cell–cell interactions, the endothelium acts as a remarkably selective barrier between blood and the vessel wall and between blood and the capillary interstitium. In addition, by way of the finely tuned synthesis and release of multifunctional factors (the most important being the vasodilators nitric oxide (NO) and prostacyclin (PGI2), and the vasoconstrictors angiotensin II (AT-II) and endothelin 1 (ET-1)), the endothelium modulates vascular tone, thrombosis, and inflammation. In addition, endothelial cells regulate smooth muscle cell proliferation and angiogenesis. 12 In physiologic conditions, the endothelium promotes smooth muscle cell relaxation and inhibits platelet activation, thrombosis, and inflammation. However, in response to non-lethal injury, endothelial cells undergo a set of phenotypic and functional changes that tilt the balance towards vasoconstriction, platelet activation, and a prothrombotic and proinflammatory state. While endothelial dysfunction is potentially reversible, it may persist indefinitely and lead to irreversible vascular remodeling. More severe injury may lead to endothelial cell death or apoptosis, with loss of vascular integrity, severe vessel dysfunction, and thrombosis.
Several cancer treatments have been associated with vascular damage, primarily mediated by their toxic effects on endothelial cells. The clinical manifestations of endothelial injury and dysfunction reflect the vascular bed primarily involved and include the onset or worsening of hypertension,13,14 venous and arterial thrombosis, 13 myocardial interstitial extravasation, hemorrhage and edema with cardiac injury,15,16 and coronary vasospasm with myocardial ischemia.16,17
Vascular toxicity of traditional chemotherapy drugs
Alkyl-like agents
This class of drugs includes the platinum compounds cisplatin and carboplatin, which have been associated to endothelial damage and cardiovascular events. In vitro, carboplatin leads to endothelial cell activation, indicated by enhanced expression of intercellular adhesion molecule-1 (ICAM-1), tissue plasminogen activator (tPA) and plasminogen activator inhibitor (PAI), and to apoptosis. 18 In agreement with this experimental evidence, long-term survivors of testicular cancer who received cisplatin-based chemotherapy were found to have increased levels of C-reactive protein (CRP) fibrinogen, von Willebrand factor (vWF), tPA and PAI-1. 19 In addition, Numico et al. showed, in a prospective cohort of 108 patients with non-small cell lung cancer treated with cisplatin and gemcitabine, a high incidence of major vascular events at 1 year after the start of chemotherapy (22.0%, 95% CI 12.7–31.3%). 20 Unfortunately, the vascular damage induced by cisplatin-based chemotherapy may not be fully reversible and may lead to diastolic hypertension and abnormal left ventricular systolic function 21 (Figure 1).
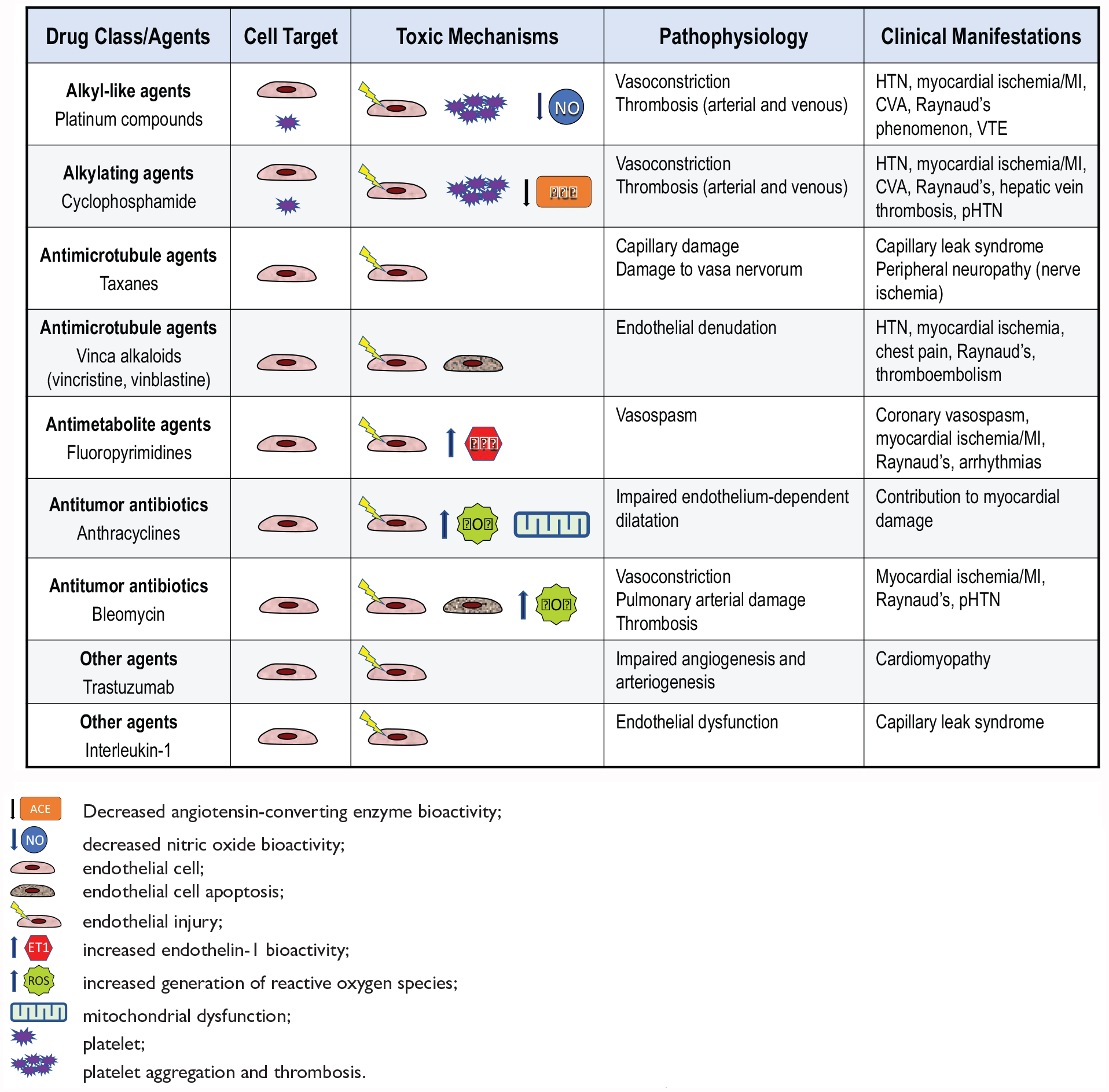
Vascular toxicity of traditional chemotherapy drugs.
Alkylating agents
Cyclophosphamide, the main drug in this group, is associated with a vascular toxicity profile similar to that observed with the platinum compounds22,23 (Figure 1).
Antimicrotubule agents
Taxanes
Drugs in this class, which includes paclitaxel and docetaxel, induce mitotic arrest, activation of apoptosis and other direct toxic effects on the endothelial cells that account for their antiangiogenic activity. 24 Antimicrotubule agents impair endothelial cell adhesion and migration, 25 and disrupt the vascular endothelial growth factor (VEGF) pathway, 26 leading to impaired endothelial NO synthase (eNOS) activation and reduced NO bioactivity. 27 Clinically, the vascular toxicity associated with taxanes includes capillary hyperpermeability with edema, 28 peripheral neuropathy from damage to the vasa nervorum, 29 and myocardial ischemia 30 (Figure 1).
Vinca alkaloids
The microtubule destabilizing drugs vincristine and vinblastine exert their cytotoxic effect mainly by inducing apoptosis. 31 In patients with testicular cancer, combination therapy including vinblastine was associated with the occurrence of Raynaud’s phenomenon and the development of hypertension. 32 Furthermore, cases of myocardial infarction have been reported 33 (Figure 1).
Antimetabolites
Antimetabolites include the fluoropyrimidine 5-fluorouracil (5-FU) and capecitabine, the oral prodrug of 5-FU. These drugs are used in various combination regimens for the treatment of gastrointestinal, breast, and head and neck cancers. Fluoropyrimidines have been associated with clinically significant cardiovascular risk, second only to anthracyclines. 34 In particular, they may induce coronary artery spasm and myocardial ischemia, even in patients with no angiographic evidence of CAD. 35 Postulated mechanisms of fluoropyrimidine-induced vasospasm include activation of protein kinase C (PKC), 36 an increased bioactivity of ET-1 37 with enhanced vasoconstriction, 38 and lethal endothelial cell damage with cytolysis, patchy intima denudation, and thrombosis 39 (Figure 1). Of note, the vasculotoxic effects of 5-FU have been shown to persist at 30 days. 40
Clinically, the incidence of coronary vasospasm and myocardial ischemia depends on the specific fluoropyrimidine used and its administration. The highest incidence of ischemic events (5.4% of patients) has been reported in patients receiving continuous intravenous infusion of high-dose 5-FU-based chemotherapy, 41 whereas lower incidence of cardiac-related events has occurred with the use of short 5-FU and leucovorin treatments (2.4% of patients). 42 Of note, 5-FU-induced cardiovascular events usually occur within 72 hours of the first cycle. 43 Capecitabine has been associated with myocardial ischemia and/or infarction in 2.9% of the patients, particularly when administered in combination with oxaliplatin and bevacizumab. 44 Coronary vasospasm and ischemia are usually reversible with interruption of fluoropyrimidine administration and treatment with vasodilators. However, fatal events have been reported, particularly after rechallenge. 43 Because of the high risk of recurrent and more severe ischemic events with fluoropyrimidine rechallenge, 45 this approach should be attempted only in patients without reasonable alternative cancer therapies. Patients should receive bolus 5-FU infusion, aspirin, vasodilators (L-type calcium channel blockers and/or nitrates) and should be monitored closely on telemetry, if possible in the coronary care unit. 46 A recent case series suggests that pretreatment with sequential administration of the calcium channel blockers nifedipine and diltiazem, and of isosorbide mononitrate (a long-acting nitrate) may allow rechallenge with bolus IV 5-FU or oral capecitabine. 47
Antitumor antibiotics
Anthracyclines
Anthracyclines are cytotoxic antibiotics used for the treatment of several hematologic and solid malignancies. Their mechanisms of action include inhibition of DNA and RNA synthesis, inhibition of topoisomerase II, and iron-mediated generation or reactive oxygen species. 48 Anthracyclines are well known for their acute and chronic dose-dependent cardiotoxicity leading to cardiomyopathy and heart failure. 49 However, animal and human studies have indicated that endothelial injury to the myocardial vasculature may also contribute to doxorubicin-mediated myocardial damage. In particular, isolated aortas from animals treated with doxorubicin exhibit abnormal endothelium-dependent relaxation in response to acetylcholine, consistent with endothelial dysfunction.50,51 In keeping with this experimental evidence, flow-mediated dilation (FMD) of the brachial artery is significantly reduced following doxorubicin treatment in women with breast cancer (Figure 1). The selective effect of anthracycline toxicity on the endothelium is confirmed by normal response to the endothelium-independent vasodilator nitroglycerin. 51 On a mechanistic standpoint, doxorubicin-mediated endothelial injury has been linked to increased intracellular levels of hydrogen peroxide leading to the activation of apoptotic signaling pathways. 52 Unfortunately, endothelial damage may not be fully reversible and may persists for several years after anthracycline treatment. In particular, Dengel et al. showed that young adult survivors who received doxorubicin as children for acute lymphoblastic leukemia have lower FMD than matched controls. 53
Bleomycin
Bleomycin is a mixture of glycopeptide antibiotics used for treatment of Hodgkin and non-Hodgkin lymphomas, head and neck cancer, and testicular cancer. Bleomycin’s main mechanism of action is damage to DNA, leading to strand breaks. Bleomycin exerts specific vascular effects by inhibiting endothelial cell proliferation and migration and promoting apoptosis, 54 and these effects appear to be mediated by generation of free radicals. 55 Clinically, bleomycin, alone or in combination with cisplatin, etoposide, or vinca alkaloids, has been associated with a threefold increased risk of secondary Raynaud’s phenomenon, 56 myocardial infarction, 57 and pulmonary hypertension. 58 Endothelial dysfunction of the digital arteries is present in patients with bleomycin-induced Raynaud’s phenomenon, 59 and endothelial injury may contribute the pathogenesis of bleomycin-induced lung toxicity 60 (Figure 1).
Other agents
Trastuzumab is a humanized monoclonal antibody against the human epidermal growth factor receptor-2 (HER2) receptor and is approved for treatment of HER2-positive breast cancer. 61 The use of trastuzumab has been associated with left ventricular (LV) systolic dysfunction, 62 particularly when administered in association with anthracyclines therapy. 61 Of interest, the HER2 receptor is also expressed in vascular endothelial cells and disruption of this signaling pathway in the arterial circulation of the myocardium may contribute to the pathogenesis of LV dysfunction 63 (Figure 1).
Interleukin-2 (IL-2) is an immune cytokine used in treatment of metastatic renal cell carcinoma and melanoma. The main vascular complication of high-dose IL-2 is vascular leak syndrome (VLS), characterized by generalized increase in capillary permeability leading to hypotension, pulmonary edema, and acute renal failure. 64
The mechanism of vascular damage in VLS is complex and includes the direct effects of IL-2 and of inflammatory cytokines such as tumor necrosis factor-α (TNF-α), and of IL-1 on endothelial cells, and direct cytotoxic effects by lymphokine-activated killer cells 65 (Figure 1).
Vascular toxicity of targeted and novel therapies
The discovery that inappropriate activation of cellular kinases is one of the fundamental biologic abnormalities in cancer that has led to a paradigm shift in our therapeutic approach. 66
The introduction of targeted tyrosine kinase inhibitors (TKI), usually small-molecule drugs that can be taken orally, has represented a giant step forward in the management of many malignancies, effectively changing their natural history. 67 Of note, the same kinases and intracellular pathways inappropriately activated in neoplastic cells also play essential functions in normal cells, including endothelial cells. 68 Therefore, inhibition of a kinase for cancer treatment may be associated with untoward effects due to interference with vital physiologic cellular functions. A prototypical example is the VEGF signaling pathway. This signaling cascade is essential for the formation of new vascular supply in growing cancers. At the same time, it plays a fundamental homeostatic role in the normal vasculature, including endothelial cell growth and survival and NO synthesis. VEGF pathway inhibition may increase blood pressure by reducing NO bioavailability and enhancing ET-dependent vasoconstriction in resistance arteries. In addition, it can cause capillary rarefaction and increased microcirculatory resistance. 69 Currently available VEGF inhibitors include monoclonal antibodies targeting circulating VEGF (bevacizumab, ramucirumab, and aflibercept) and small-molecule inhibitors targeting VEGF receptors (axitinib, cabozantinib, lenvatinib, pazopanib, regorafenib, sorafenib, sunitinib, and vandetanib). All of these agents have been associated with blood pressure increase and development of hypertension within a week. The incidence of hypertension has been reported to be as high as 57% in patients with metastatic renal cell carcinoma treated with pazopanib. 70 In addition to hypertension, VEGF inhibition has recently been associated with the occurrence of aortic dissection, 71 stroke, 72 and arterial and venous thrombosis 73 (Figure 2).
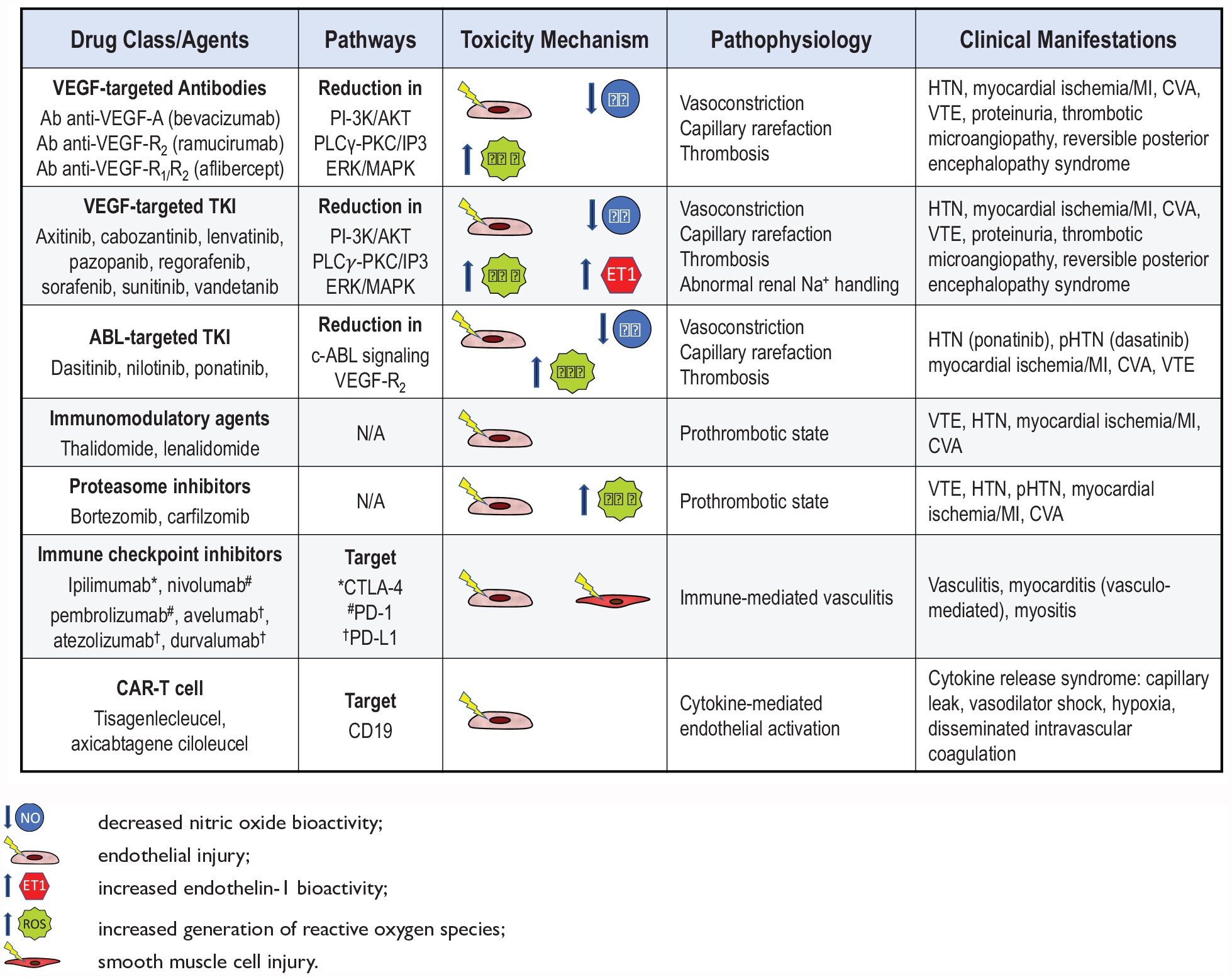
Vascular toxicity of targeted and novel therapies.
Whereas the vascular effects of VEGF inhibitors are consistent with known physiologic roles of this pathway, the occurrence of vascular toxicity with second generation (dasatinib and nilotinib) and third generation (ponatinib) Abelson murine leukemia viral oncogene homolog 1 (ABL1) kinase inhibitors has been less predictable. 74
Dasatinib treatment has been associated with the development of pulmonary arterial hypertension (PAH). In a French registry, nine patients were reported to have dasatinib-associated moderate to severe precapillary PAH. 75 Furthermore, in the 5-year follow up of the DASISION trial, which compared dasatinib with imatinib, 5% of patients assigned to dasatinib developed PAH versus 0.4% of those in the imatinib group. 76 This evidence, led the US Food and Drug Administration (FDA) to issue a warning on the cardiopulmonary risks of dasatinib, with a recommendation to evaluate patients on this agent for signs and symptoms of cardiopulmonary disease before and during treatment. 77 Of note, in the DASISION trial, dasatinib was associated with a higher risk of arterial ischemic events, but no peripheral arterial disease (PAD), at 5 years compared to imatinib (5% vs 2%, respectively). However, in a subsequent analysis of a pooled population of patients receiving dasatinib, the rates of ischemic cardiovascular events were not higher than expected after adjusting for age and sex 78 (Figure 2).
Different from dasatinib, nilotinib has been associated with an increased risk of adverse ischemic cardiovascular events, including ischemic heart disease, cerebrovascular disease, and PAD. 79 Among them, the development of severe PAD requiring revascularization and, in some cases, leading to amputation, has been particularly troublesome.80,81 The mechanisms underlying the increased risk of ischemic events with nilotinib have not been fully elucidated. However, data from the Evaluating Nilotinib Efficacy and Safety in Clinical Trials–Newly Diagnosed Patients (ENESTnd) study and other analyses indicate the occurrence of adverse metabolic effects, including increase in body mass index, and elevation of cholesterol and glucose levels.79,82 Therefore, progression of atherosclerosis has been suggested as the primary mechanism of ischemic events with nilotinib 83 (Figure 2).
Ponatinib is a next-generation TKI with activity against the highly drug-resistant ABL1 T315I mutation. Unfortunately, in the Ponatinib Ph+ Acute Lymphoblastic Leukemia (ALL) and Chronic Myeloid Leukemia (CML) Evaluation (PACE) trial, treatment with ponatinib was associated with a high incidence of hypertension (26%) as well as adverse cardiovascular, cerebrovascular, and peripheral arterial events. 84 The high incidence of cardiovascular toxicity observed in PACE at 24 months led to temporary market withdrawal of ponatinib by the FDA, together with mandatory dose reduction in the patients enrolled in the trial. Of note, ponatinib-associated cardiovascular toxicity appears to be dose-dependent, with higher risk in older patients with diabetes or history of ischemic events. 85 Interestingly, the mechanisms underlying the cardiovascular toxicity of ponatinib appear to differ from those postulated in other TKIs and represent a new form of vascular toxicity mediated by vWF-mediated platelet adhesion and a secondary microvascular angiopathy 86 (Figure 2).
Given the absence of known roles of ABL1 in the vasculature, the vascular effects during treatment with ABL1 kinase inhibitors are likely unrelated to its cancer target and suggest the occurrence of ‘off target’ effects.
In addition to targeted TKIs, a number of other novel agents have contributed to the recent progress in cancer treatment. Interestingly, these new therapies also have been associated with significant vascular side effects through multiple, and often poorly understood, mechanisms. Immunomodulators such as thalidomide and lenalidomide, which are frequently used in the treatment of multiple myeloma, are associated with a high risk of venous thromboembolism (VTE), particularly with the concomitant use of chemotherapy and dexamethasone. 87 Similarly, bortezomib and carfilzomib, proteasome inhibitors that are effective in various B-cell malignancies and multiple myeloma, have been linked to increased risk of VTE. The mechanisms underlying the prothrombotic effect of these drugs are incompletely characterized and both direct activation of the coagulation cascade secondary to endothelial injury and indirect effects through increased expression of endothelial tissue factor have been proposed. 88
An even more recent progress in cancer treatment has occurred with the introduction of immune therapies, which aim to activate and enhance natural immune defenses to target and kill neoplastic cells. 89 Among them, immune checkpoint inhibitors (ICI) have shown unprecedented efficacy in the treatment of an increasing number of malignancies. 90 A number of monoclonal antibody ICI have been introduced in clinical practice, including pembrolizumab and nivolumab (which block the programmed cell death protein 1 (PD-1)), ipilimumab (which blocks the cytotoxic lymphocyte-associated protein 4 (CTLA-4)), and avelumab, atezolizumab, and durvalumab (which block the PD-1 ligand (PD-L1)). Owing to the growing number of patients treated with ICI, our awareness of the spectrum of toxicity and side effects has rapidly grown, and an increasing number of vascular events due to small, medium, and large-vessel vasculitis have been reported.91 –93 Another form of immunotherapy that has shown remarkable promise in relapsed or refractory hematologic CD19+ B-cell malignancies is chimeric antigen receptor (CAR) modified T cells (CAR-T).94,95 This treatment involves the harvesting of T cells from the patient’s blood, genetic modification of the T cells with insertion of the CAR gene, in vitro expansion of CAR-T cells, and infusion of these cells in the patient. 96 The interaction of CAR-T cells with normal or malignant cells that express CD19 leads to CAR-T cell activation and proliferation, lysis of malignant target cells, and cytokine secretion. The latter effect represents the main mechanism underlying the cytokine release syndrome (CRS), the most significant side effect of CAR-T cell therapy. CRS is characterized by release of interferon-γ (INF-γ), soluble IL-2 receptor-α, TNF-α, IL-6, and IL-10, as well as elevation of inflammatory markers including CRP and ferritin. The initial clinical presentation of CRS includes fever, headache, rash, arthralgia, myalgia, and fatigue. In more severe cases, CRS can cause capillary leak, which can progress to life-threatening vasodilatory shock, disseminated intravascular coagulation, hypoxia, and end-organ damage. Endothelial cell activation and dysfunction, indicated by elevated levels of angiopoietin 2 and vWF, appears to be a key pathogenetic feature of CRS by amplifying the inflammatory response and causing capillary leakage, hypotension, and coagulopathy. 97
Conclusions
Progress in the understanding of cancer biology and the availability of novel therapeutic targets have led to an unprecedented advancement in the management of patients with several types of malignancies. In parallel, we have expanded our understanding of the short- and long-term cardiovascular damage of traditional cancer treatments and we are learning about new and unexpected vascular toxicity in patients receiving novel therapies. The successful management of patients with cancer requires a multidisciplinary team approach in which oncologist and cardiovascular medicine specialists work together to prevent, detect, and minimize acute vascular toxicity and long-term consequences of cancer therapy.
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
