Abstract

Cardiovascular (CVD) mortality rates in the United States (US) have substantially decreased in the past two decades. 1 Between 2000 and 2011, there was a steady decline in CVD mortality by 3–4% per year; however, recently there has been a deceleration in this decline. 2 Patients with cancer are at increased risk of venous thromboembolism and one of the highest risks for CVD mortality in cancer is pulmonary embolism (PE). Recent studies in Europe have documented a potential rise in PE mortality. 3 Trends and disparities of PE mortality in specific cancers are incompletely characterized. 4 We sought to investigate the patterns and trends of fatal PE in patients with cancer in the US over the past two decades.
This was a retrospective cohort study utilizing the multiple cause of death files maintained by the National Center for Health Statistics.5,6 We sought to identify the change in PE mortality between 1999 and 2020 by specific cancer diagnosis. 7 As this was a publicly available dataset, approval from the review board was not required. Our primary outcome was PE age-adjusted mortality rate (AAMR; standardized to the 2000 US Census data) in patients with concomitant cancer as a cause of death (overall cancers, and subtypes: gastrointestinal [GI], thoracic/lung, breast, prostate, and hematologic). To calculate AAMR, we selected the category of disease ‘Pulmonary embolism’ (International Classification of Disease, Tenth Revision [ICD-10] code I26) as the underlying cause of death. We then modified the population by selecting the additional diagnosis code for the population of interest; for example: ‘Malignant neoplasms’ (ICD-10: C00–C97), GI (ICD-10: C15–C26), lung (ICD-10: C30–C39), breast (ICD-10: C50), prostate (ICD-10: 61), and hematologic (ICD-10: C81–96). We additionally analyzed the AAMR for Black and White individuals overall and stratified by Hispanic versus nonHispanic ethnicity. We calculated the ratio of AAMR for Black compared to White individuals. We also evaluated the trends in overall CVD mortality (ICD-10: I00–I99) for comparative purposes. Linear regression analyses were used to evaluate mortality trends.
Overall, 13,917,133 patients dying from cancer were analyzed during the study period. Cardiovascular AAMR decreased by 18% (822 to 677 per million persons, p < 0.001) in individuals with cancer, whereas AAMR for PE in cancer increased by 43% (from 27 to 39 per million persons, p < 0.001). This trend was observed in cancer subtypes including GI (6.7 to 9.9 per million persons, ∆47%, p < 0.001), lung (6.9 to 9.8 per million persons, ∆43%, p < 0.001), breast (2.4 to 3.3 per million persons, ∆37%, p = 0.004), but not prostate (p = 0.28) and hematologic (p = 0.53) (Figure 1A). Black individuals had a 50% higher rate of AAMR from PE compared to White individuals (57.8 vs 38.1 per million persons). This disparity remained consistent across all malignancies: GI (56%, 15.1 vs 9.7 per million persons), lung (35%, 13.1 vs 9.7 per million persons), breast (82%, 5.7 vs 3.1 per million persons), prostate (171%, 5.0 vs 1.8 per million persons), and hematologic (29%, 4.3 vs 3.4 per million persons) (Figure 1B). Hispanics had the highest rates of AAMR PE compared with nonHispanics with lung cancer (6.2 from 4.8 per million persons, 36%) and breast cancer (5.0 from 4.4 per million persons, 30%) (online Supplementary Figure 1).
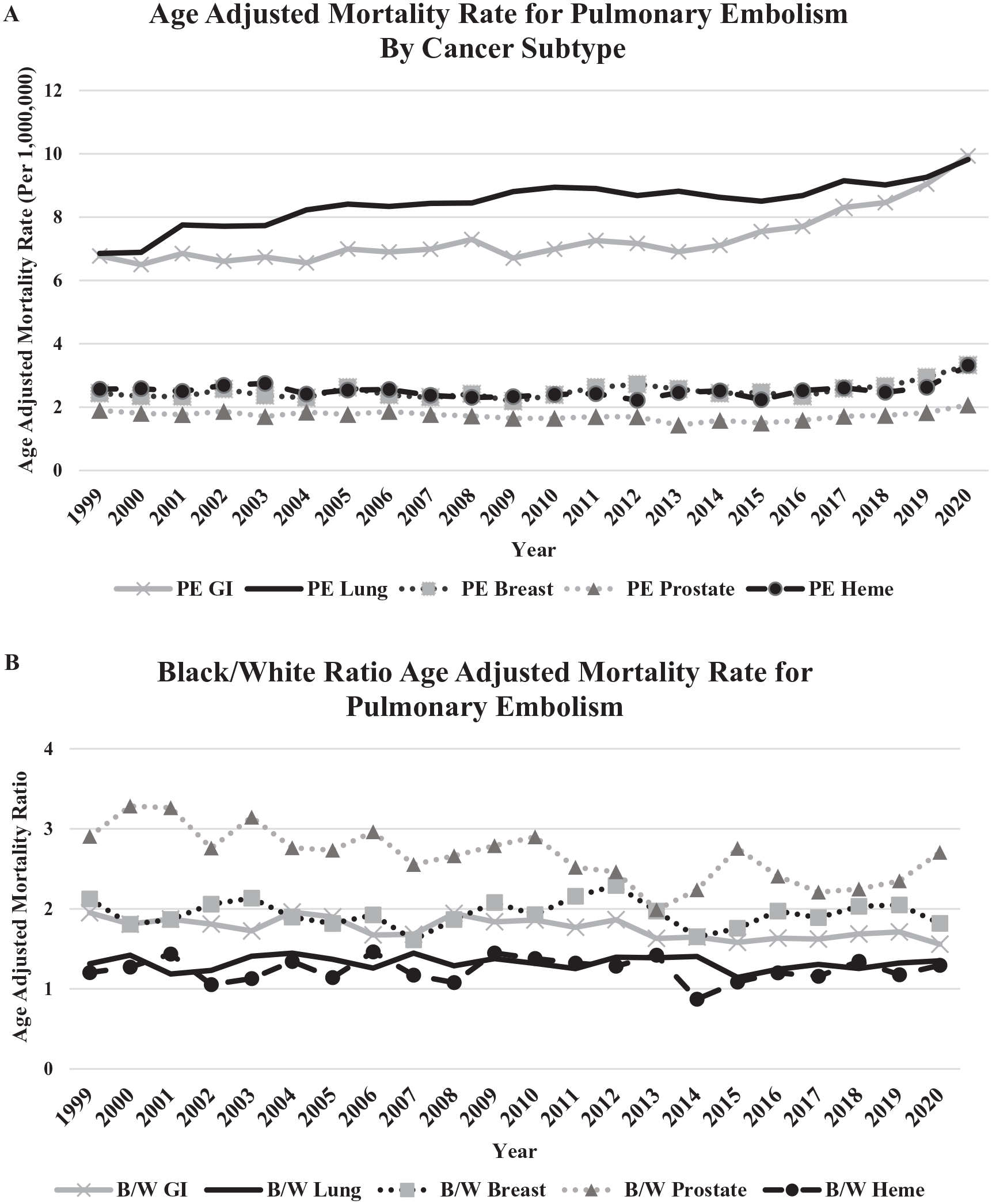
This retrospective national database analysis demonstrated AAMR for PE in cancer increased by 43% and there are persistent and significant racial disparities in these rates, with Black individuals experiencing 29–171% higher rates without significant change over time. Understanding the burden of PE mortality is essential for identifying and developing risk stratification tools and treatment algorithms to improve outcomes. Currently, cancer-specific VTE risk assessment tools may underestimate the risk of thrombosis in Black and Hispanic patients. 8 Mechanisms underpinning these findings remain speculative, but are likely multifactorial. An increased recognition and improved testing modalities may play a role in the growing AAMR. Furthermore, the new biologic and immune checkpoint inhibitors may increase risk in oncologic patients directly or indirectly by prolonging lives of patients with cancer.
Improved recognition of the healthcare disparities of this devastating prothrombotic state is imperative to reduce the critical morbidity and mortality in Black and Hispanic patients with cancer. Previous literature has shown patients with PE have higher severity of disease in minority populations compared to White patients, 9 and Black patients had worse outcomes despite adjusting for severity of disease. 10 Reasons behind these disparities are not fully known, but likely involve structural racism in healthcare access and prescription of anticoagulation. 11
This study is limited by lack of granularity and cause of death misclassification. Despite limitations, this study provides sufficient scope to examine the temporal and demographic patterns of PE and represents a complementary and substantial contribution to the existing PE and cancer mortality literature to encourage future studies.
In conclusion, this retrospective review of the multiple cause of death data in the US suggest a continued rise in PE AAMR in the past two decades in the US, and shows significant persistent racial disparities between Black/Hispanic and White populations in PE mortality. Future research must address improving health equity and eradicating disparities in the treatment of PE.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221103626 – Supplemental material for Trends and disparities in pulmonary embolism mortality among patients with cancer
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221103626 for Trends and disparities in pulmonary embolism mortality among patients with cancer by Scott E Janus, Tarek Chami, Mohamad Karnib, Haytham Mously, Jamal Hajjari, Anshul K Badhwar, Taha Al-Juhaishi, Jun Li, Mehdi H Shishehbor and Sadeer G Al-Kindi in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Li is on the advisory board for Inari Medical and Boston Scientific, and is a consultant for Abbott Vascular and Philips. Dr Shishehbor is a consultant for Abbott Vascular, Medtronic, Terumo, Philips, and Boston Scientific. The remaining authors have no disclosures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
